Title: A randomized, placebo-controlled trial of cenicriviroc for treatment of nonalcoholic steatohepatitis with fibrosis.
Journal: Hepatology (Baltimore, Md.) 20180501
Title: Isolation and characterization of human immunodeficiency virus type 1 resistant to the small-molecule CCR5 antagonist TAK-652.
Journal: Antimicrobial agents and chemotherapy 20070201
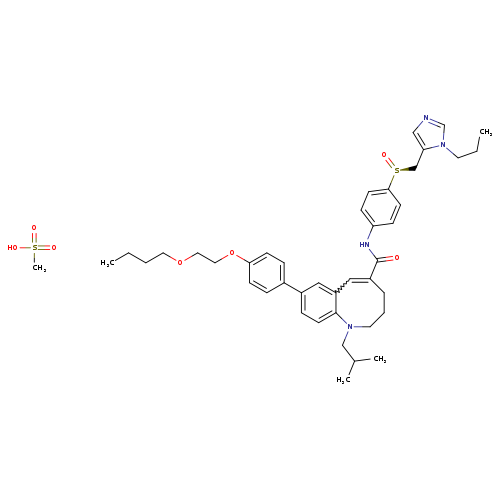
Title: Highly potent and orally active CCR5 antagonists as anti-HIV-1 agents: synthesis and biological activities of 1-benzazocine derivatives containing a sulfoxide moiety.
Journal: Journal of medicinal chemistry 20060323
Title: TAK-652 inhibits CCR5-mediated human immunodeficiency virus type 1 infection in vitro and has favorable pharmacokinetics in humans.
Journal: Antimicrobial agents and chemotherapy 20051101
Title: Lefebvre E, et al. Antifibrotic Effects of the Dual CCR2/CCR5 Antagonist Cenicriviroc in Animal Models of Liver and Kidney Fibrosis. PLoS One. 2016 Jun 27;11(6):e0158156.
Title: Visseaux B, et al. Cenicriviroc, a Novel CCR5 (R5) and CCR2 Antagonist, Shows In Vitro Activity against R5 Tropic HIV-2 Clinical Isolates. PLoS One. 2015 Aug 6;10(8):e0134904.
Title: Lalezari J, et al. Safety, efficacy, and pharmacokinetics of TBR-652, a CCR5/CCR2 antagonist, in HIV-1-infected, treatment-experienced, CCR5 antagonist-naive subjects. J Acquir Immune Defic Syndr. 2011 Jun 1;57(2):118-25.
Title: Baba M, et al. TAK-652 inhibits CCR5-mediated human immunodeficiency virus type 1 infection in vitro and has favorable pharmacokinetics in humans. Antimicrob Agents Chemother. 2005 Nov;49(11):4584-91.