2020-02-08 14:59:48
Aimee K. Clarke+, Hon E. Ho+, James A. Rossi-Ashton+, Richard J. K. Taylor, and William P. Unsworth
1.Introduction
Indole synthesis has been extensively reviewed previously, however, a comprehensive review focusing specifically on silver-catalyzed approaches has not been reported before. The indole core is a key structural component in many natural products and pharmaceuticals and serves as a fundamental building block in organic synthesis.[1] The synthesis of indole scaffolds has therefore been the focus of much research and a myriad of methods to construct indole rings have been devel- oped over the years.[2] Classical methods include the Fischer indole[3] and Bartoli syntheses, which are widely and routinely used by the synthetic community.[4] Nonetheless, limitations as- sociated with these classical procedures mean that establishing novel strategies to prepare indoles is still important and con- tinues to be actively pursued.
Many indole syntheses make use of alkyne activation ap- proaches, typically involving coordination of a metal catalyst to the alkyne to activate it towards cyclization.[5] Silver, a member of the “coinage metal” family, can be readily obtained in the form of silver(I) salts with a variety of different counter- ions. These salts, which have a d10 electronic configuration at silver, are well-established as being good s-/p-Lewis acids and are recognized as being powerful catalysts in alkyne activa- tion.[6] In addition to their ability to interact with p-systems to promote useful reactivity, the use of silver in organic transfor- mations has important economic benefits relative to other more expensive transition metals such as gold, palladium and platinum.[5,6]
Indole synthesis has been extensively reviewed previously,[2] however, a comprehensive review focusing specifically on silver-catalyzed approaches has not been reported before. As the use of silver catalysis in heterocycle synthesis is becoming more prevalent,[5] a review of this topic in the context of indole synthesis is timely. To the best of our knowledge, this Minireview summarizes all silver-catalyzed indole syntheses to date, with a cut-off period of papers published before January 2019. Note that whilst we believe that all publications that fea- ture examples within the remit of this review are discussed, we have not reproduced all of the individual examples from these studies.
Many indole syntheses utilize silver in mixed catalytic sys- tems (e.g. mixed gold/silver systems),[5] but this Minieview is limited to examples in which the silver species have been shown to be competent at catalyzing the reaction without the influence of another metal species. The review is organized in chronological order and is divided based on the type of reac- tion used to construct the indole core, starting with the most commonly used hydroamination pathway, before moving on to other methods. Mechanisms are included and described in more detail whenever they deviate from the generally accept- ed hydroamination mechanism.
2.Hydroamination Strategies
Alkyne hydroamination[7] is by far the most common synthetic strategy used for silver-catalyzed indole syntheses. In such re- actions, anilines 1 substituted with an alkyne at their 2-position are treated with a silver(I) species which acts as a p-acid to ac- tivate the alkyne towards attack from a pendant aniline nitro- gen via a 5-endo-dig cyclization mode (3!4). Protodemetalla- tion then liberates the silver(I) species (meaning that the reac- tions can be catalytic in the silver species) and deprotonation completes the synthesis of the indole product 2 (Scheme 1).
To the best of our knowledge, the earliest example of the silver-catalyzed hydroamination strategy being used to prepare indole derivatives was reported by Rutjes and co-workers in 2004.[8] This group described the transition metal-catalyzed cyclization of o-alkynylanilines to access indole 2-propargylgly- cine (isotryptophan) derivatives. o-Alkynylpropargylglycine ani- lines 5 and 6 were prepared using Sonogashira cross-coupling between o-iodoanilines and enantiopure propargylic glycine precursors. The use of 10 mol% AgOTf in MeCN at reflux for 20 h furnished the isotryptophan products 7 and 8 in good yields. By comparison, the use of a PdII catalytic system result- ed in formation of the undesired cyclization product 9, which was not observed when AgOTf was employed (Scheme 2).
In 2007, Li et al. reported a gold and silver co-catalyzed double hydroamination of o-alkynylanilines with terminal al- kynes to access N-vinyl indole derivatives.[9] During the catalyst screening process, the separate use of both 5 mol% AgOTf and 5 mol % AgBF4 at 60 8C for 2 h under neat conditions gave the N-vinyl indole product 12 in 62 % and 59 % yields, respec- tively (Scheme 3). Although a silver(I) species can facilitate the cascade hydroamination process alone, it was later revealed that the combination of 5 mol % of AuCl3/AgOTf at RT was more efficient and hence was the main focus of the study.
In 2009, Liu et al. reported a gold and silver co-catalyzed mi- crowave-assisted intramolecular hydroamination of o-alkynyla- mides to construct N1-carbamide indole derivatives.[10] Although the combination of AuI/AgI in aqueous media using microwave irradiation at 150 8C was chosen as the optimal re- action conditions, using 10 mol % AgOTf or Ag2CO3 alone dis- played catalytic activity to afford the cyclized indole product 14 in 23 % and 75 % yields, respectively (Scheme 4 A). It was also found that the reaction conditions were exclusive to o-ter- minal alkynes as no reaction was observed when 2-substituted o-alkynylcarbamides 15 and 16 were used as substrates (Scheme 4 B).
In 2009, Ding et al. reported a silver-catalyzed hydroamina- tion process using (o-alkynylphenyl)guanidines 17 to access N- carboximidamide or N-carboximidoate indole derivatives 18 (Scheme 5).[11] By using 5 mol% AgNO3 at RT and MeCN as the solvent, guanidines 17 were found to selectively undergo 5- endo-dig cyclization to afford a range of indole derivatives 18 in good yields. The authors also conducted a comparison study between AgI and other commonly used p-acids such as PdII and CuI salts. It was reported that the reaction using a AgNO3 catalyst was the most effective, proceeding efficiently and in high yield; meanwhile, the analogous reactions using both PdII and CuI catalytic systems were incomplete, even after extended reaction times. Overall, this silver(I)-catalyzed cycliza- tion provides access to N-carboximidamide or N-carboximi- doate indole-2-phenyl derivatives under simple and mild reac- tion conditions.
In 2010, Oh et al. reported a silver(I)-catalyzed cascade pro- cess based on the reaction of o-alkynylformidates 19 and acti- vated methylene compounds 20 to synthesize 3-vinyl indole derivatives 21 (Scheme 6).[12] Typically, these reactions were performed using 5 mol% AgOTf in toluene at 808C for 12 h, enabling a range of 3-vinyl indoles 21 to be prepared in mod- erate to good yields.
The authors suggested a plausible mechanism for this trans- formation, involving an interesting 3-alkenyl migration process (Scheme 7). First, coordination of silver(I) to the alkyne facilitates enolate addition into imine 22 to form 23. This is fol- lowed by p-acid activation of the alkyne by silver(I) to induce a 5-endo-dig cyclization to form the indole core. 1,3-Alkenyl mi- gration is then proposed to occur via a silver-carbene inter- mediate 26, which is followed by rapid protodemetallation under acidic conditions to furnish the 3-vinyl indole product 21. Note that similar migration patterns have also been report- ed by using other transition metals such as PdII, PtII, and AuIII.[5, 13]
The 1,3-alkenyl migration mechanism shown in Scheme 7 was supported by a series of control experiments. For example, when o-alkynylenamine 28 was subjected to the standard reac- tion conditions, only the hydroamination product 29 was iso- lated in 55 % yield (Scheme 8). This suggested that fast proto- demetallation was competing with the 1,3-alkenyl migration pathway in some instances.
In 2010 Chan et al. described a system for the synthesis of indoles via gold-catalyzed cycloisomerization reactions.[14] During this investigation, as a control experiment, 1,3-diphen- yl-1-(2-(tosylamino)phenyl)prop-2-yn-1-ol 30 was treated with 5 mol% AgOTf, which yielded the corresponding indenyl-fused indole 31 in 16 % yield, alongside the alcohol-tethered indole 32 in 48 % yield (Scheme 9). Although it was proven that AgOTf could facilitate indole formation, a gold-catalyzed method was shown to be more efficient and was the main focus of this investigation.
Two years later, Chan et al. developed a silver-catalyzed tandem heterocyclization/alkynylation process using propargyl- ic 1,4-diols 33 to generate o-alkynyl indoles 34, liberating two molecules of water as the sole by-products (Scheme 10).
This was the first reported indole synthesis that introduced alkyne moieties at the 2-position of the indole ring without re- lying on traditional cross-coupling methods. A variety of tosyl- protected o-alkynyl indoles 34, bearing additional substituents in the 3-, 5- and 6-positions, were generated in good to excel- lent yields employing AgOTf as the catalyst. Interestingly, the reaction proceeds well in the absence of a group in the R1 po- sition, which leads to the formation of 3H-indole products; this is particularly noteworthy as these products cannot be formed using traditional cross-coupling approaches. The authors sug- gested that the silver catalyst activates the C@OH bonds in the diol substrates, rather than the alkyne moiety directly, and that this subsequently triggers cyclization/hydroamination.
In 2012, Van der Eycken et al. reported the microwave-assist- ed syntheses of pyrazino-quinazolines and indolyl-pyrazinones from alkyne-tethered pyrazinones using either silver or gold catalysis.[16] Treatment of alkyne-tethered pyrazinone 35 with AgOTf, using conventional heating, resulted in the synthesis of indole 36 in 18 % yield, alongside quinazoline product 37 in 75 % yield (Scheme 11). Ag(I) was found to be the superior cat- alyst for the formation of the quinazoline products, but AuCl was in fact identified as the optimum catalyst for formation of the indole products.
In 2012, Tang et al. reported a silver-catalyzed process for the synthesis of bis(indolyl)methanes 40 from o-alkynylanilines 38 and aryl aldehydes 39 (Scheme 12).[17] Their simple one-pot procedure was performed in the presence of 5 mol % AgNO3 in DMSO at 808C for 12 h. A wide range of o-alkynylanilines 38 and aryl aldehydes 39 were tolerated in this process, providing access to the corresponding bis(indolyl)methanes 40 in moder- ate to excellent yields. Based on previously reported mecha- nisms, the authors suggested that these reactions proceed via a hydroamination pathway in which the silver catalyst activates both alkyne and aldehyde starting materials.
In 2013, Liu et al. reported the synthesis of (3-indolyl)stan- nanes 42 via a silver-catalyzed cyclization/stannylation cascade process.[18] Starting from a series of o-alkynylanilines 41 and re- acting with 5 mol% AgSbF6 and two equivalents of 2-tributyl- stannylfuran, a wide range of N1-protected-(3-indolyl)stan- nanes 42 were synthesized (Scheme 13 A). The procedure was shown to tolerate both electron-donating and electron-with- drawing groups on the alkyne phenyl ring and substituents at the 4-position of the parent aniline ring. It was found that the presence of an electron-withdrawing protecting group is es- sential to the success of the reaction, as the non-stannylated 3H-indole product was isolated when a N-methyl aniline start- ing material was tested. It was also found that indoles bearing electron-withdrawing protecting groups other than sulfonyl were unstable during purification via column chromatography. The authors showcased the utility of the 3-stannylated indole products 42 by performing a series of elaboration reactions. To probe the reaction mechanism, 3H-indole was subjected to the optimized reaction conditions and no stannylated product was observed, which indicated that the stannylation did not occur via C@H functionalization of the indole product but instead through a silver-tin transmetallation process as shown in Scheme 13 B. In this mechanism, the silver is proposed to have a dual role; activating the alkyne towards attack from the amino group via the silver-coordinated alkyne 43 whilst also catalyzing the destannylation of 2-tributylstannylfuran through a transmetallation protodemetallation pathway, thus liberating
Bu3Sn+ which goes on to react with the 3-indolyl silver(I) intermediate.
In 2014, McNulty et al. reported the synthesis and applica- tion of a series of homogenous silver(I) acetate complexes as catalysts in intramolecular hydroamination reactions using N- protected o-alkynylanilines 46.[19] A range of N-protected in- doles 48 were formed in high yields with the process per- formed at room temperature in the absence of base or other additives (Scheme 14). The authors conducted a catalyst screen of heterogeneous silver(I) salts at 100 8C, which furnished indole products from their corresponding o-alkynylanilines. It was found that those salts with weakly coordinating anions were ineffective in the intramolecular hydroamination. Next, they screened a series of homogeneous ligated silver species and found catalyst 47 to be the most efficient at catalyzing indole formation. Further investigations revealed that the reac- tions could be performed at room temperature and that polar solvents were required, with DMF being optimal. A wide range of N-tosylindoles 48 were then accessed in high yields and the procedure was shown to tolerate sensitive functionalities such as a TMS-functionalized alkyne.
In 2014, Song and You et al. reported a one-pot silver-catalyzed cyclization/fluorination cascade to deliver structurally di- verse fluorinated indole derivatives 50 from o-alkynylanilines 49 (Scheme 15).[20] Overall, four different indolenine, indoline and indole derivatives were accessed in good yields by altera- tion of the reaction conditions. A limited range of 3-fluorinated indoles 50 were synthesized as it was found that an electron- withdrawing group on the alkynyl phenyl ring was required for a successful reaction. The reaction was shown to tolerate both unprotected and N-methyl protected aniline starting materials. A tentative general mechanism was proposed, whereby silver(I) activates the alkynyl aniline triple bond towards attack by the nitrogen, forming the corresponding 3H-indole upon protode- metallation, at which point the mechanism for each derivative varies according to the reaction conditions. In the case of the 3-fluorinated indole products 50, the 3H-indole is simply mon- ofluorinated by the electrophilic SelectfluorS reagent, forming the corresponding 3-fluorinated indole.
In 2014, Rang and Fan et al. reported the synthesis of N-pro- tected 4-acetonylindoles 53 from o-alkynylanilines 51 and silyl enol ethers 52 (Scheme 16).[21] Their two-step, one-pot proce- dure involves a hypervalent iodide mediated oxidative dearomatisation and silver-catalyzed cyclization and includes an in- teresting conjugate addition step of a silyl enol ether (mecha- nism discussed later, see Scheme 17). Iodosylbenzene was iden- tified as the optimum oxidant due to the absence of acidic contaminants that were produced when using other hyperva- lent iodine sources. Although the acidic species did not impede the oxidation step, they were found to be harmful to the subsequent silyl enol ether reaction. To avoid decomposi- tion of the silyl enol ether, methanol from the iodosylbenzene step was removed under reduced pressure prior to addition of the silyl enol ethers. The catalytic activity of a variety of metal salts was investigated, and AgOTf was found to be the opti- mum salt for catalyzing the process. Gold(I) salts gave reduced yields of the 4-acetonylindole products 53 relative to that of AgOTf, whereas other metal salts, such as BiIII, InIII, ZnII, CuII, AuIII, PdII, or PtII gave rise to the 4-methoxyindole product.
A range of substituted o-alkynylanilines were shown to un- dergo the one-pot procedure in good yields with groups at the para-position required for a successful reaction. The N-pro- tecting group was also shown to play a crucial role. The reac- tion proceeded smoothly in the presence of an extensive series of silyl enol ethers furnishing the corresponding 4-aceto- nylindole products 53. The authors proposed an iodosylben- zene-mediated oxidative dearomatization of para-substituted o-alkynylanilines 51 (Scheme 17), forming o-alkynylcyclohexa- dienimines 55. AgOTf is then proposed to activate the triple bond to induce heterocyclization, to form intermediate 57, which then undergoes a Mukaiyama-Michael-type addition with the silyl enol ether to generate intermediate 58. Me3SiOTf generated in situ then promotes rearomatization to give indole 60. Subsequent protodemetallation by TfOH produces 4-acetonylindole 53 and regenerates the AgOTf catalyst.
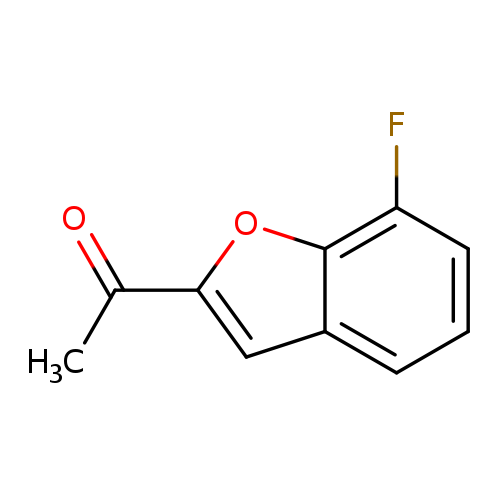
1-(7-fluoro-1-benzofuran-2-yl)ethan-1-oneCatalog No.:AA019XFK CAS No.:1094640-56-8 MDL No.:MFCD11619469 MF:C10H7FO2 MW:178.1598 |
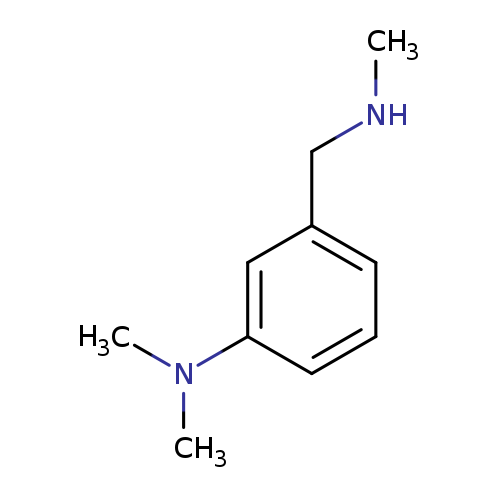
N,N-dimethyl-3-[(methylamino)methyl]anilineCatalog No.:AA01A173 CAS No.:1094641-61-8 MDL No.:MFCD11212378 MF:C10H16N2 MW:164.2474 |
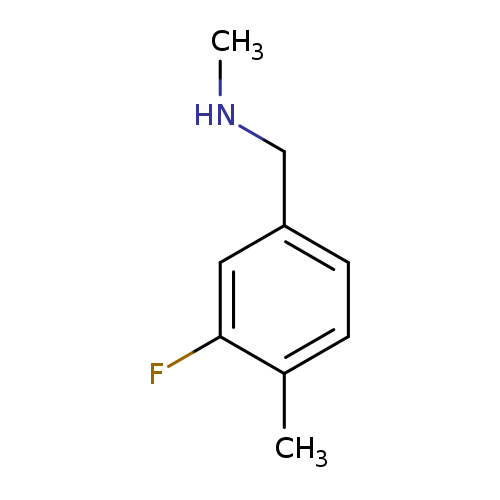
[(3-fluoro-4-methylphenyl)methyl](methyl)amineCatalog No.:AA01BDG0 CAS No.:1094641-63-0 MDL No.:MFCD11212398 MF:C9H12FN MW:153.1967 |
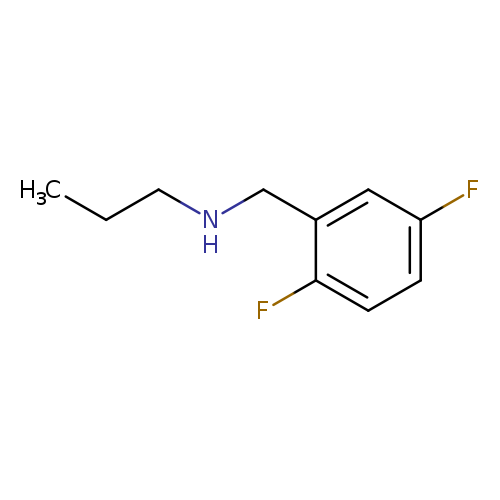
[(2,5-Difluorophenyl)methyl](propyl)amineCatalog No.:AA01AYH2 CAS No.:1094641-70-9 MDL No.:MFCD11212459 MF:C10H13F2N MW:185.2137 |
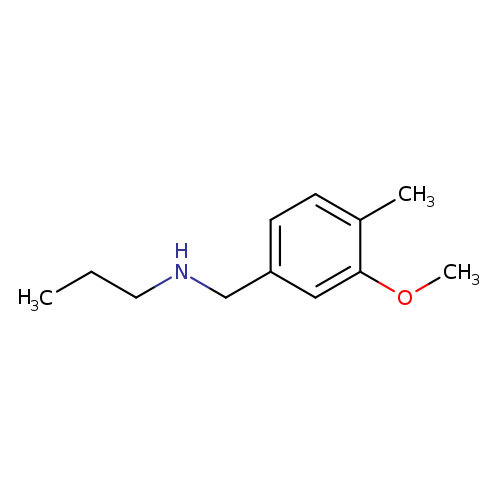
[(3-methoxy-4-methylphenyl)methyl](propyl)amineCatalog No.:AA00IW4T CAS No.:1094641-71-0 MDL No.:MFCD11212463 MF:C12H19NO MW:193.2854 |
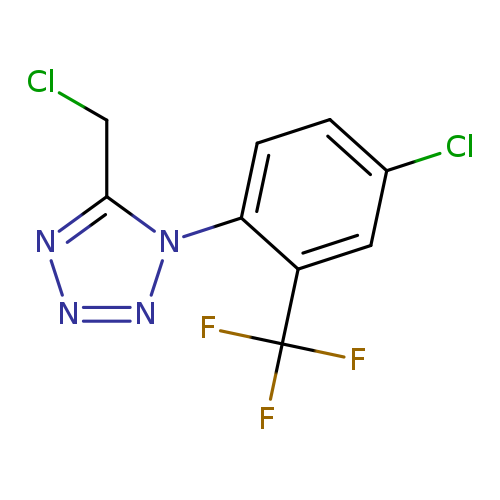
1-[4-chloro-2-(trifluoromethyl)phenyl]-5-(chloromethyl)-1H-1,2,3,4-tetrazoleCatalog No.:AA019WGM CAS No.:1094642-43-9 MDL No.:MFCD11523870 MF:C9H5Cl2F3N4 MW:297.0640 |
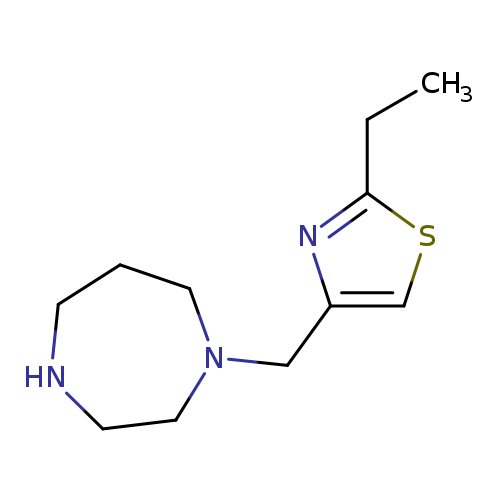
1-[(2-ethyl-1,3-thiazol-4-yl)methyl]-1,4-diazepaneCatalog No.:AA01AB8H CAS No.:1094643-90-9 MDL No.:MFCD11211707 MF:C11H19N3S MW:225.3537 |
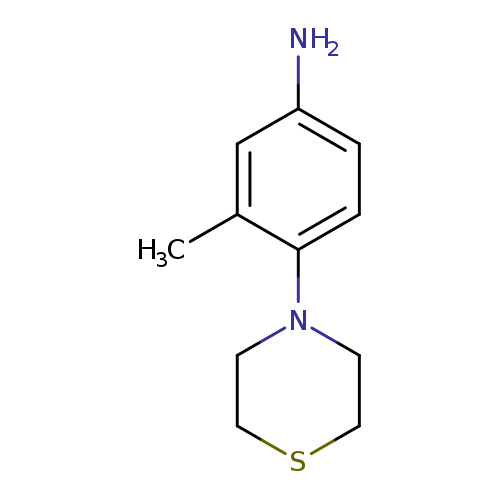
3-methyl-4-(thiomorpholin-4-yl)anilineCatalog No.:AA01A8AS CAS No.:1094646-31-7 MDL No.:MFCD11214021 MF:C11H16N2S MW:208.3231 |
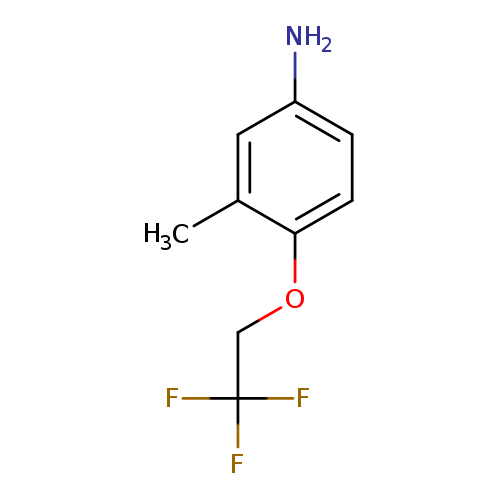
3-methyl-4-(2,2,2-trifluoroethoxy)anilineCatalog No.:AA01AJPC CAS No.:1094646-56-6 MDL No.:MFCD11214142 MF:C9H10F3NO MW:205.1770 |
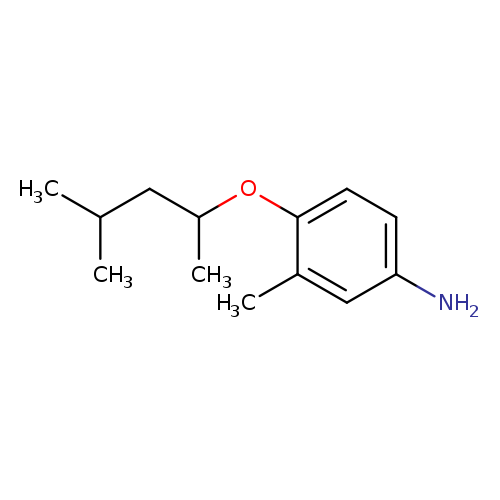
3-methyl-4-[(4-methylpentan-2-yl)oxy]anilineCatalog No.:AA01A7YU CAS No.:1094646-62-4 MDL No.:MFCD11214162 MF:C13H21NO MW:207.3119 |
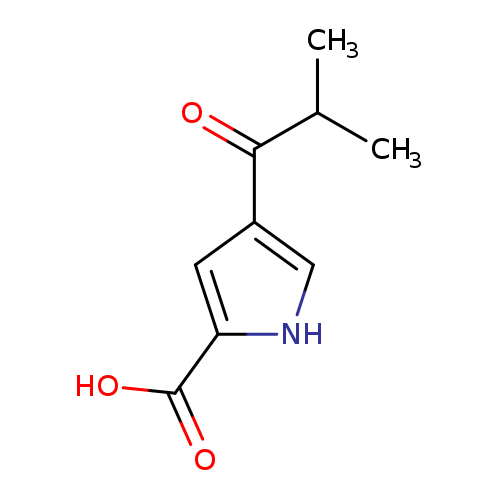
4-(2-methylpropanoyl)-1H-pyrrole-2-carboxylic acidCatalog No.:AA01C2GX CAS No.:1094648-86-8 MDL No.:MFCD11211486 MF:C9H11NO3 MW:181.1885 |
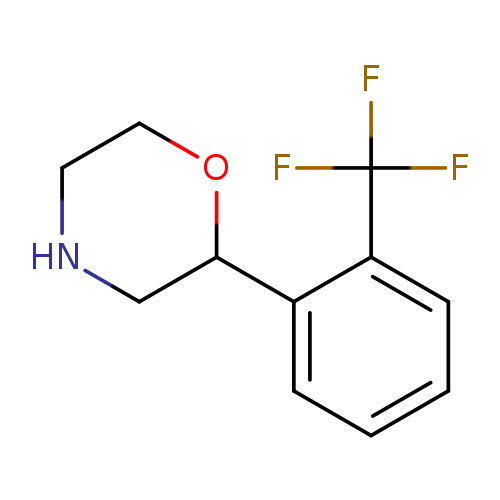
2-[2-(trifluoromethyl)phenyl]morpholineCatalog No.:AA01A5TS CAS No.:1094649-72-5 MDL No.:MFCD11646231 MF:C11H12F3NO MW:231.2143 |
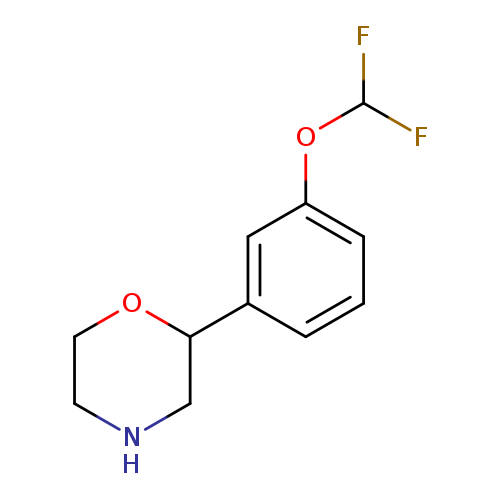
2-[3-(difluoromethoxy)phenyl]morpholineCatalog No.:AA01A2Y0 CAS No.:1094649-73-6 MDL No.:MFCD11646246 MF:C11H13F2NO2 MW:229.2232 |
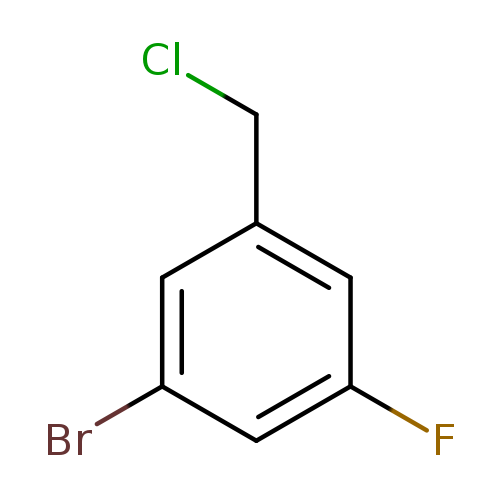
1-Bromo-3-(chloromethyl)-5-fluorobenzeneCatalog No.:AA008STD CAS No.:1094651-50-9 MDL No.:MFCD11619429 MF:C7H5BrClF MW:223.4700 |
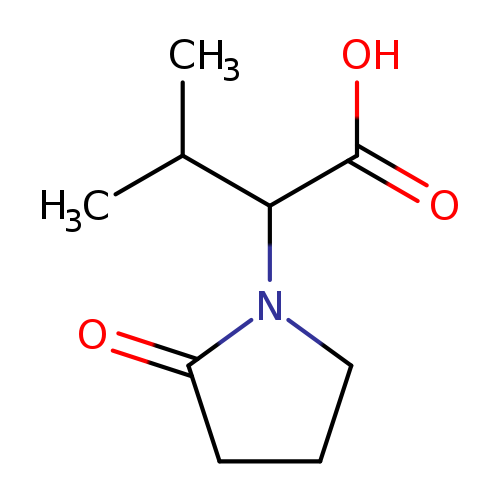
3-methyl-2-(2-oxopyrrolidin-1-yl)butanoic acidCatalog No.:AA01A7YX CAS No.:1094652-93-3 MDL No.:MFCD11214951 MF:C9H15NO3 MW:185.2203 |
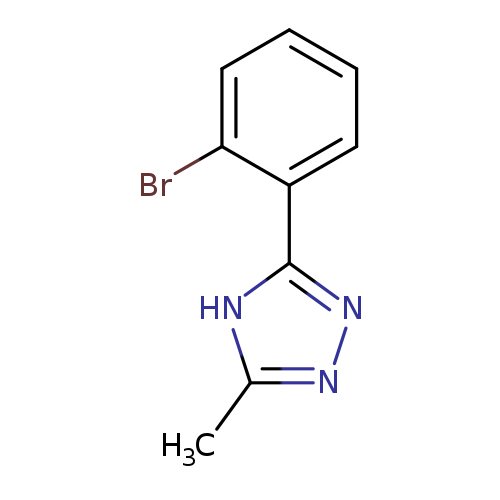
3-(2-bromophenyl)-5-methyl-4H-1,2,4-triazoleCatalog No.:AA01DX7T CAS No.:1094653-15-2 MDL No.:MFCD19381956 MF:C9H8BrN3 MW:238.0839 |
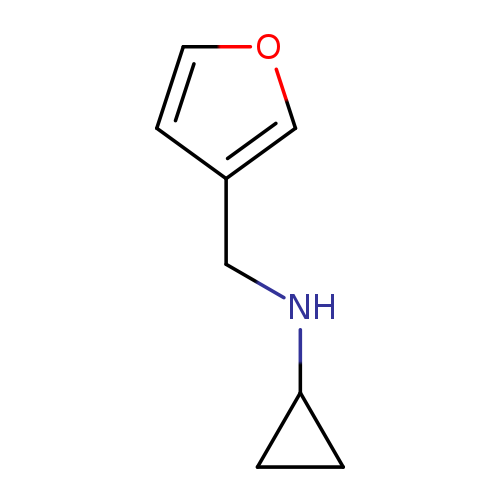
N-[(furan-3-yl)methyl]cyclopropanamineCatalog No.:AA00JWHU CAS No.:1094655-04-5 MDL No.:MFCD11212572 MF:C8H11NO MW:137.1790 |
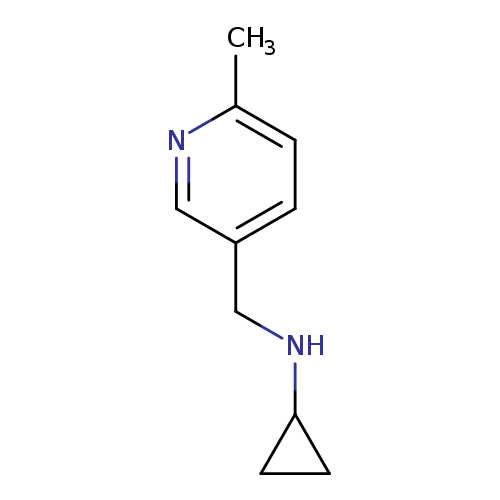
N-[(6-Methylpyridin-3-yl)methyl]cyclopropanamineCatalog No.:AA01BTEM CAS No.:1094655-05-6 MDL No.:MFCD11212576 MF:C10H14N2 MW:162.2316 |
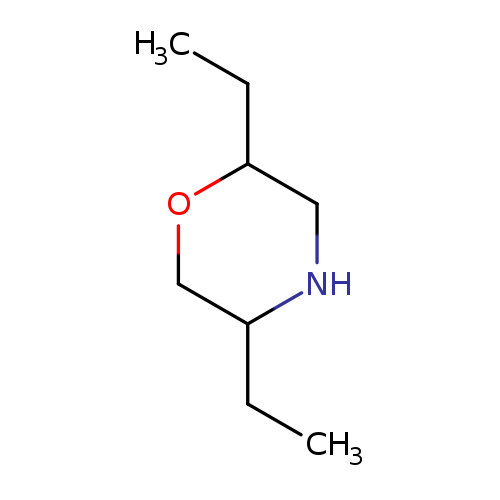
2,5-diethylmorpholineCatalog No.:AA01C5FD CAS No.:1094657-75-6 MDL No.:MFCD11652784 MF:C8H17NO MW:143.2267 |
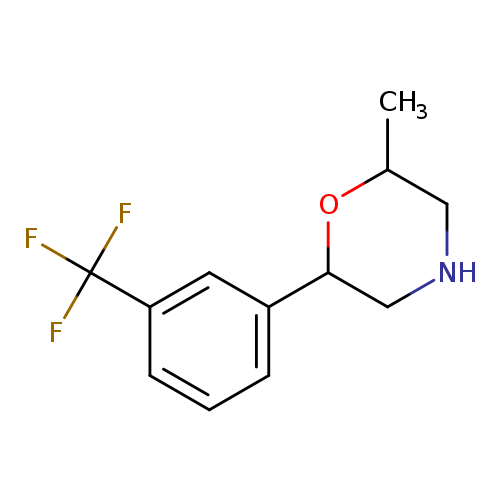
2-methyl-6-[3-(trifluoromethyl)phenyl]morpholineCatalog No.:AA01ALD6 CAS No.:1094657-85-8 MDL No.:MFCD11652907 MF:C12H14F3NO MW:245.2409 |
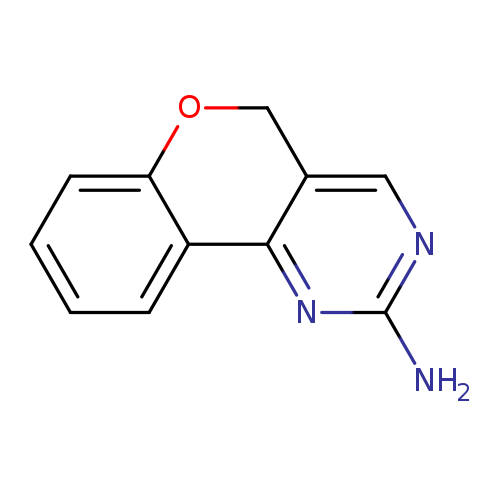
5H-Chromeno[4,3-d]pyrimidin-2-amineCatalog No.:AA00IYEQ CAS No.:109466-23-1 MDL No.:MFCD04125744 MF:C11H9N3O MW:199.2087 |
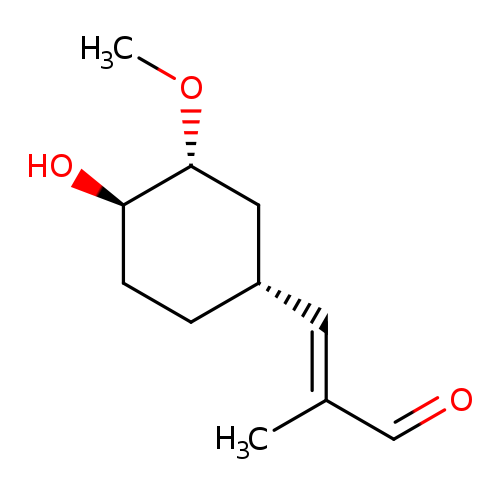
TacroliMus Methyl Acryl AldehydeCatalog No.:AA008WAI CAS No.:109466-74-2 MDL No.: MF:C11H18O3 MW:198.2588 |
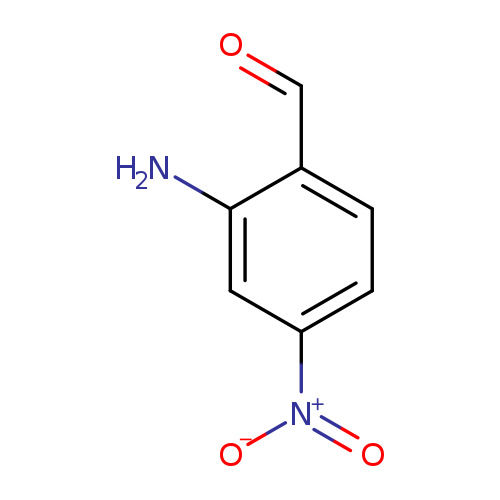
2-Amino-4-nitrobenzaldehydeCatalog No.:AA0096KQ CAS No.:109466-84-4 MDL No.:MFCD01319035 MF:C7H6N2O3 MW:166.1341 |
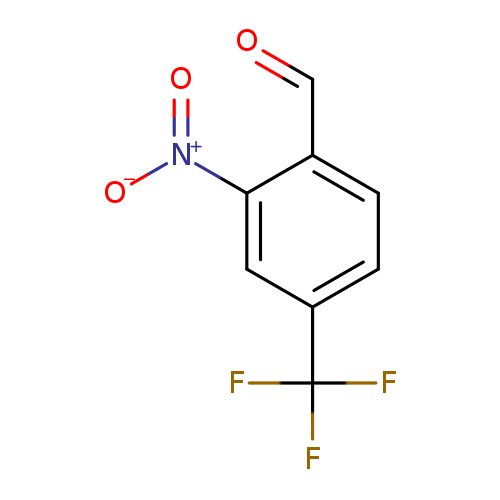
2-Nitro-4-(trifluoromethyl)benzaldehydeCatalog No.:AA0033BY CAS No.:109466-87-7 MDL No.:MFCD06797985 MF:C8H4F3NO3 MW:219.1175 |
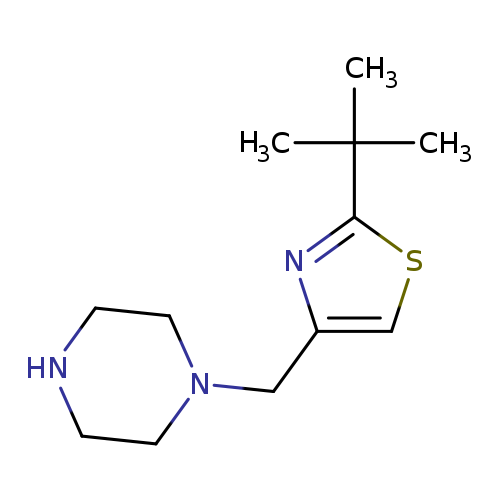
1-[(2-tert-butyl-1,3-thiazol-4-yl)methyl]piperazineCatalog No.:AA019MWB CAS No.:1094661-15-0 MDL No.:MFCD12068349 MF:C12H21N3S MW:239.3802 |
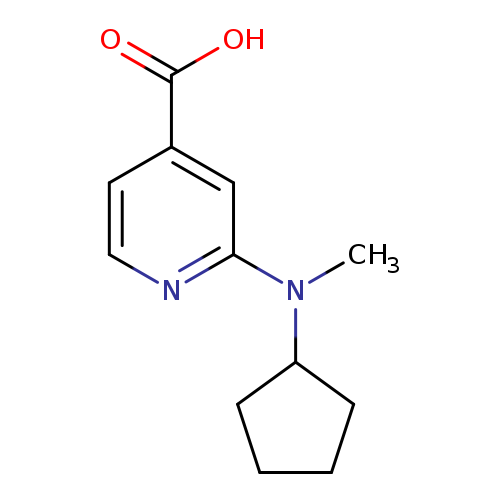
2-[Cyclopentyl(methyl)amino]pyridine-4-carboxylic acidCatalog No.:AA019T1G CAS No.:1094661-79-6 MDL No.:MFCD11618576 MF:C12H16N2O2 MW:220.2676 |
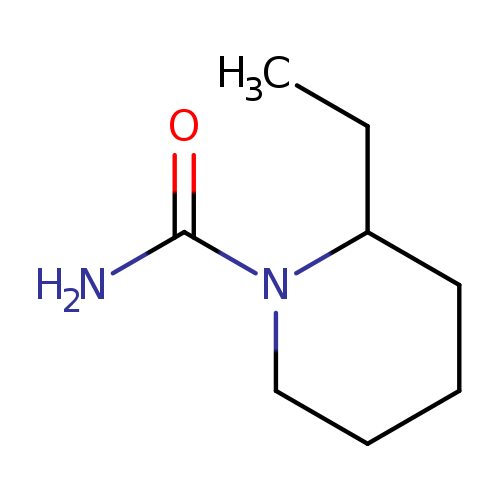
2-Ethylpiperidine-1-carboxamideCatalog No.:AA01A8SD CAS No.:1094661-84-3 MDL No.:MFCD11618582 MF:C8H16N2O MW:156.2254 |
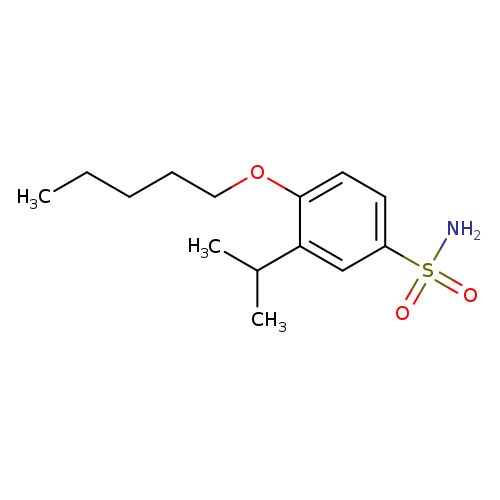
4-(Pentyloxy)-3-(propan-2-yl)benzene-1-sulfonamideCatalog No.:AA01DUW6 CAS No.:1094662-80-2 MDL No.:MFCD12542273 MF:C14H23NO3S MW:285.4023 |
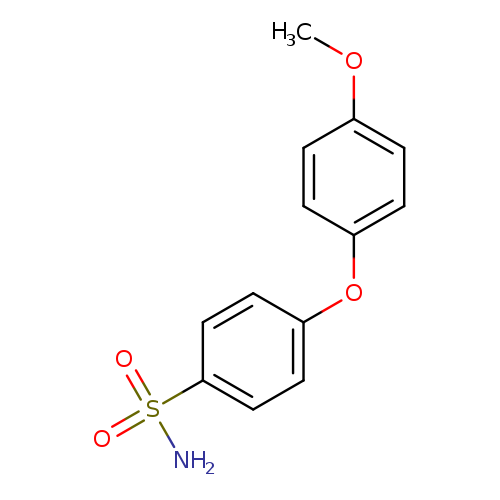
4-(4-methoxyphenoxy)benzene-1-sulfonamideCatalog No.:AA01AKEQ CAS No.:1094663-05-4 MDL No.:MFCD12569451 MF:C13H13NO4S MW:279.3116 |
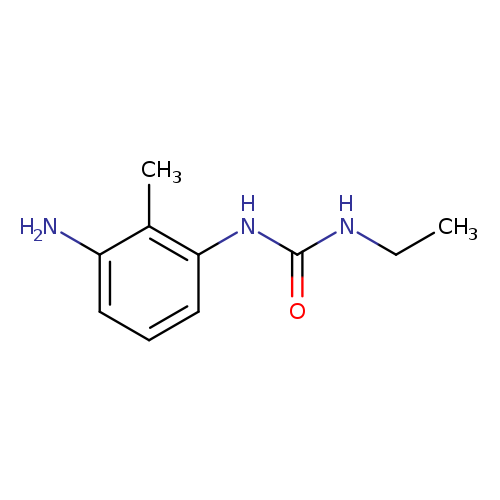
1-(3-amino-2-methylphenyl)-3-ethylureaCatalog No.:AA01BFU3 CAS No.:1094663-60-1 MDL No.:MFCD11213431 MF:C10H15N3O MW:193.2456 |
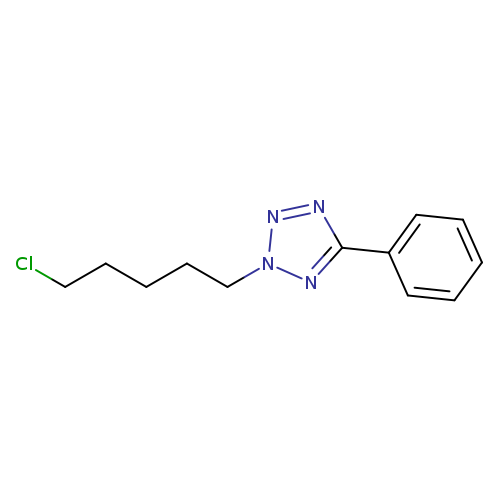
2-(5-chloropentyl)-5-phenyl-2H-1,2,3,4-tetrazoleCatalog No.:AA01A9WK CAS No.:1094667-82-9 MDL No.:MFCD11645006 MF:C12H15ClN4 MW:250.7273 |
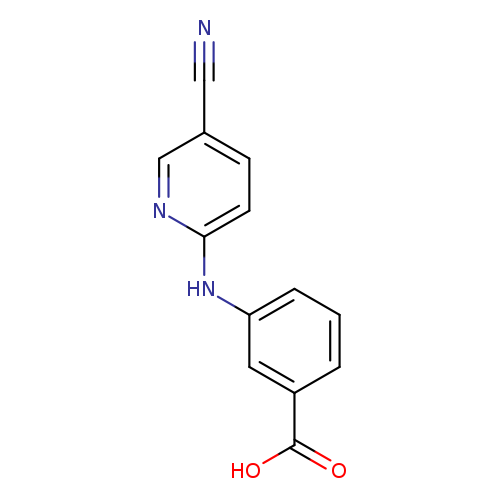
3-(5-cyanopyridin-2-ylamino)benzoic acidCatalog No.:AA0095YZ CAS No.:1094668-69-5 MDL No.:MFCD11548992 MF:C13H9N3O2 MW:239.2295 |
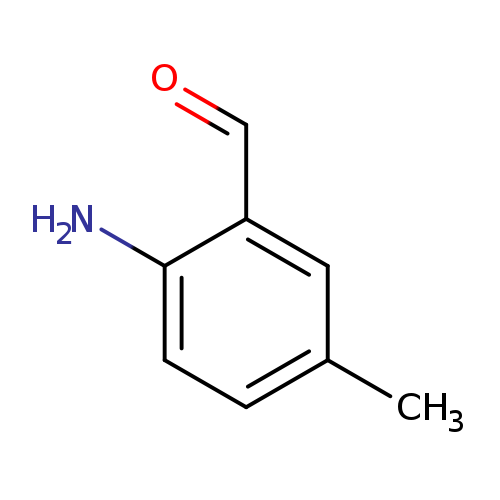
2-Amino-5-methylbenzaldehydeCatalog No.:AA0096WS CAS No.:109467-00-7 MDL No.:MFCD10696883 MF:C8H9NO MW:135.1632 |
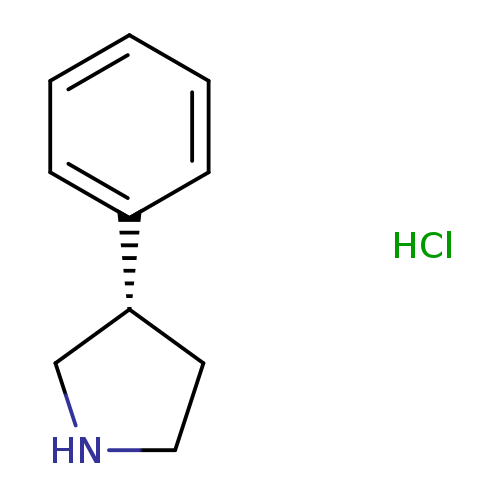
(S)-3-Phenyl-pyrrolidine, HClCatalog No.:AA008S4F CAS No.:1094670-20-8 MDL No.:MFCD06796635 MF:C10H14ClN MW:183.6779 |
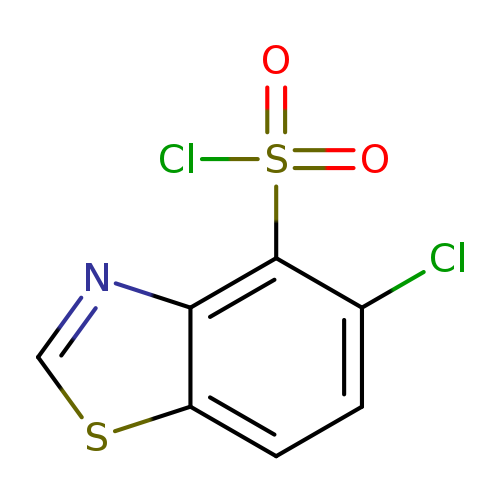
5-Chloro-1,3-benzothiazole-4-sulfonyl chlorideCatalog No.:AA01ABJS CAS No.:1094671-88-1 MDL No.:MFCD11650767 MF:C7H3Cl2NO2S2 MW:268.1402 |
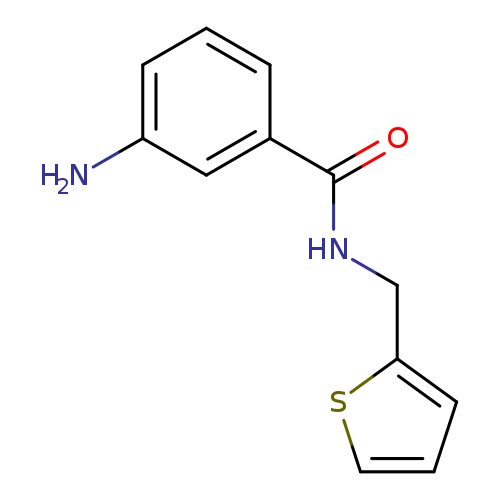
3-amino-N-(thiophen-2-ylmethyl)benzamideCatalog No.:AA01A1SD CAS No.:1094671-91-6 MDL No.:MFCD11650816 MF:C12H12N2OS MW:232.3015 |
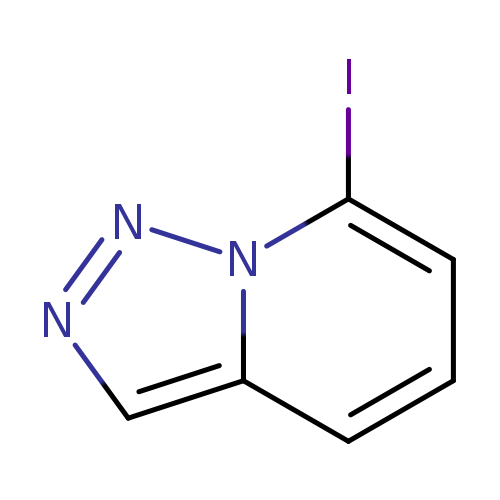
7-iodo-[1,2,3]triazolo[1,5-a]pyridineCatalog No.:AA01ELB9 CAS No.:1094674-57-3 MDL No.:MFCD31666749 MF:C6H4IN3 MW:245.0205 |
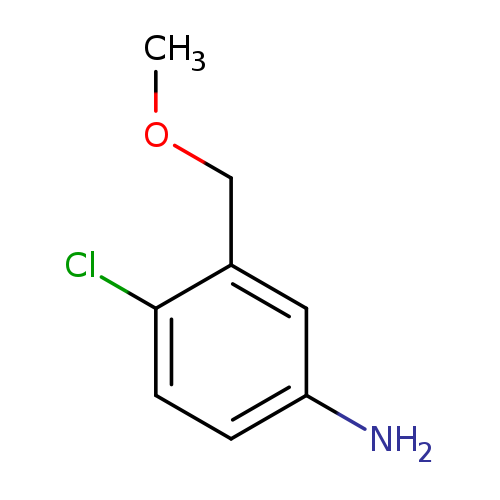
4-chloro-3-(methoxymethyl)anilineCatalog No.:AA01DUW7 CAS No.:1094675-66-7 MDL No.:MFCD11645905 MF:C8H10ClNO MW:171.6241 |
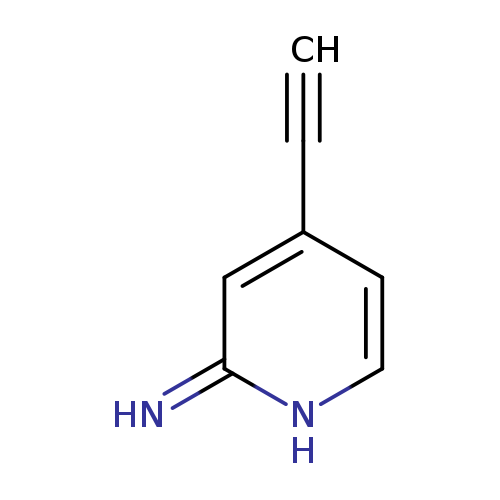
4-Ethynylpyridin-2-amineCatalog No.:AA019ENI CAS No.:1094679-27-2 MDL No.:MFCD13189750 MF:C7H6N2 MW:118.1359 |
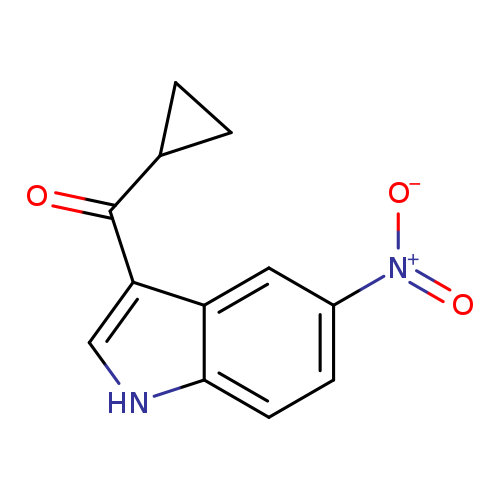
3-cyclopropanecarbonyl-5-nitro-1H-indoleCatalog No.:AA01ACB9 CAS No.:1094681-83-0 MDL No.:MFCD11211546 MF:C12H10N2O3 MW:230.2194 |
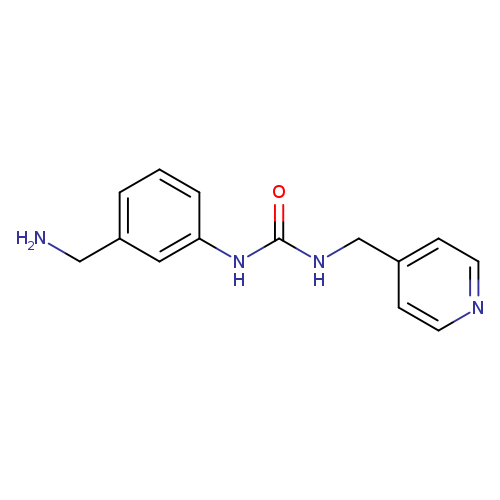
3-[3-(aminomethyl)phenyl]-1-(pyridin-4-ylmethyl)ureaCatalog No.:AA01A9MU CAS No.:1094683-17-6 MDL No.:MFCD12550861 MF:C14H16N4O MW:256.3030 |
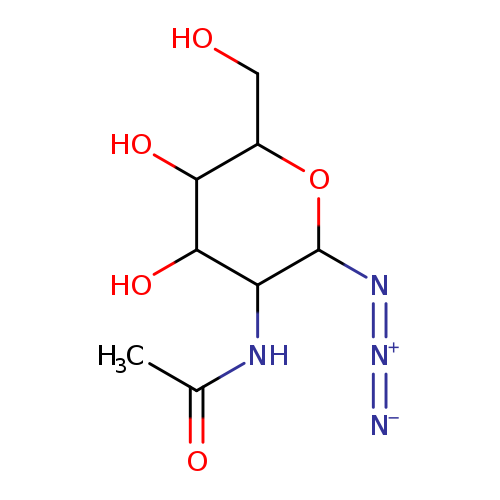
N-(2-AZIDO-4,5-DIHYDROXY-6-HYDROXYMETHYL-TETRAHYDRO-PYRAN-3-YL)-ACETAMIDECatalog No.:AA01AQUF CAS No.:1094684-86-2 MDL No.:MFCD04336512 MF:C8H14N4O5 MW:246.2206 |
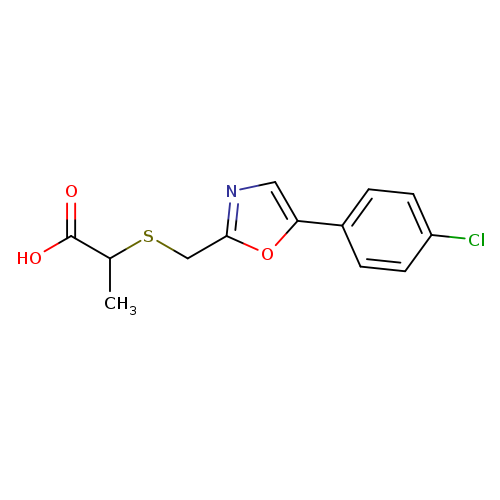
2-({[5-(4-chlorophenyl)-1,3-oxazol-2-yl]methyl}sulfanyl)propanoic acidCatalog No.:AA019XBK CAS No.:1094685-51-4 MDL No.:MFCD12655852 MF:C13H12ClNO3S MW:297.7573 |
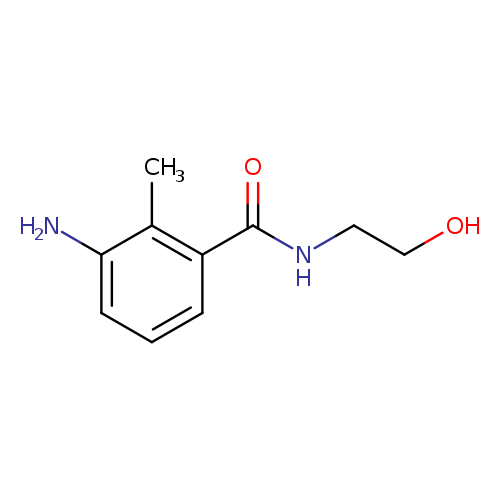
3-amino-N-(2-hydroxyethyl)-2-methylbenzamideCatalog No.:AA019X4J CAS No.:1094686-54-0 MDL No.:MFCD11642866 MF:C10H14N2O2 MW:194.2304 |
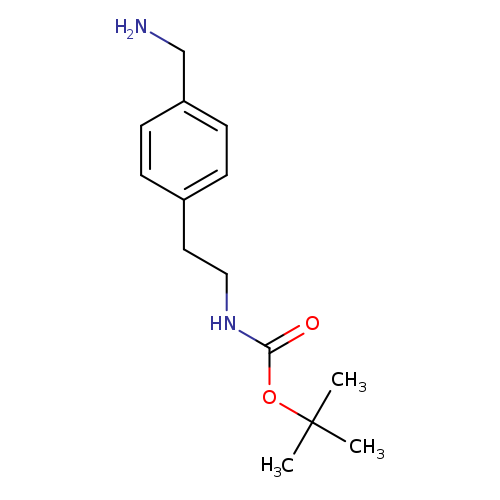
tert-butyl 4-(aminomethyl)phenethylcarbamateCatalog No.:AA01A90E CAS No.:1094689-12-9 MDL No.:MFCD11525087 MF:C14H22N2O2 MW:250.3367 |
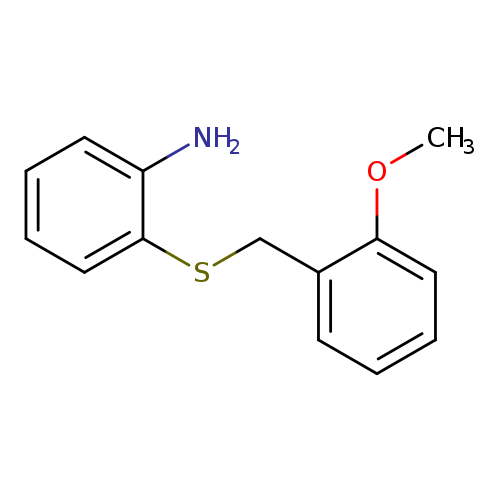
2-{[(2-methoxyphenyl)methyl]sulfanyl}anilineCatalog No.:AA01A18N CAS No.:1094690-40-0 MDL No.:MFCD11619585 MF:C14H15NOS MW:245.3400 |
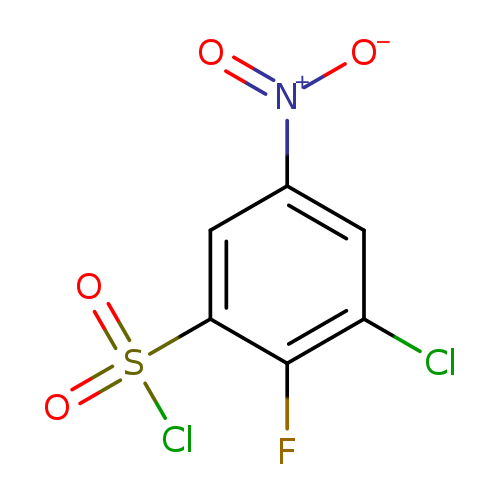
3-chloro-2-fluoro-5-nitrobenzene-1-sulfonyl chlorideCatalog No.:AA019NB5 CAS No.:1094690-77-3 MDL No.:MFCD11212857 MF:C6H2Cl2FNO4S MW:274.0538 |
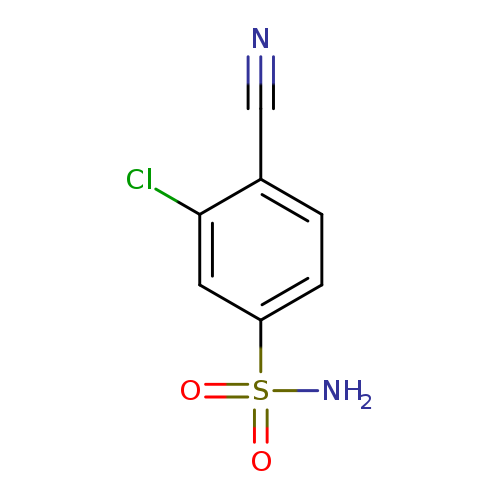
3-chloro-4-cyanobenzene-1-sulfonamideCatalog No.:AA01AB51 CAS No.:1094691-00-5 MDL No.:MFCD11212967 MF:C7H5ClN2O2S MW:216.6448 |
quinolin-8-ylmethanesulfonamideCatalog No.:AA01A333 CAS No.:1094691-01-6 MDL No.:MFCD11212982 MF:C10H10N2O2S MW:222.2636 |
4-[4-(2-methoxyethyl)phenoxy]butanoic acidCatalog No.:AA01AB2A CAS No.:1094693-27-2 MDL No.:MFCD11647674 MF:C13H18O4 MW:238.2796 |
1-(2-amino-6-methylphenyl)-3-ethylureaCatalog No.:AA01C2AL CAS No.:1094697-45-6 MDL No.:MFCD11213447 MF:C10H15N3O MW:193.2456 |
2-(Chloromethyl)-5-[(2-methylphenyl)methyl]-1,3,4-oxadiazoleCatalog No.:AA019ZXD CAS No.:1094697-61-6 MDL No.:MFCD11213536 MF:C11H11ClN2O MW:222.6708 |
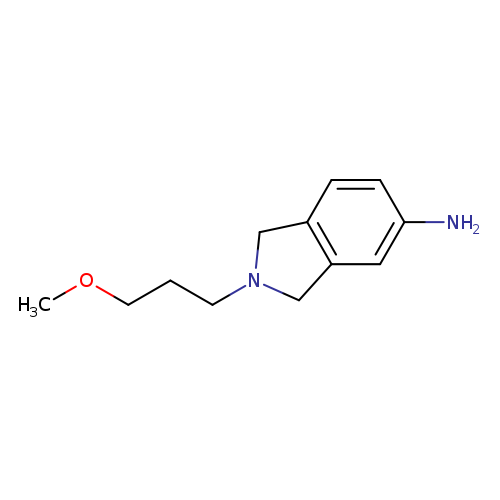
2-(3-methoxypropyl)-2,3-dihydro-1H-isoindol-5-amineCatalog No.:AA01A8YY CAS No.:1094699-77-0 MDL No.:MFCD11655098 MF:C12H18N2O MW:206.2841 |
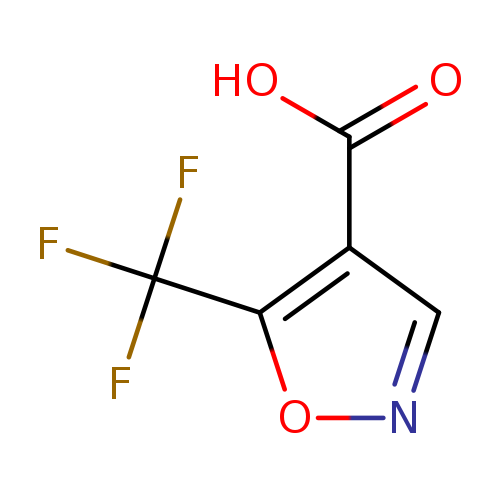
5-(Trifluoromethyl)isoxazole-4-carboxylic acidCatalog No.:AA0092HN CAS No.:1094702-34-7 MDL No.:MFCD11620299 MF:C5H2F3NO3 MW:181.0695 |
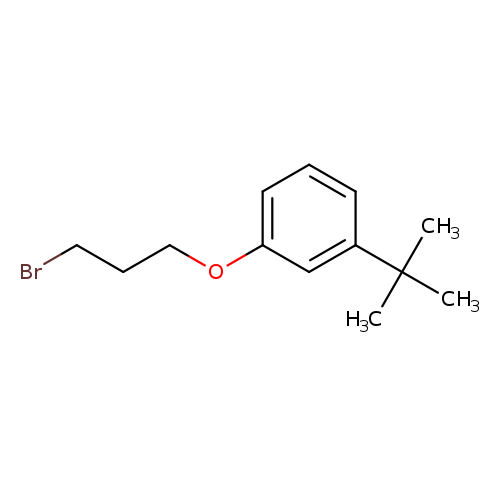
1-(3-Bromopropoxy)-3-tert-butylbenzeneCatalog No.:AA01AAKQ CAS No.:1094702-92-7 MDL No.:MFCD11647763 MF:C13H19BrO MW:271.1934 |
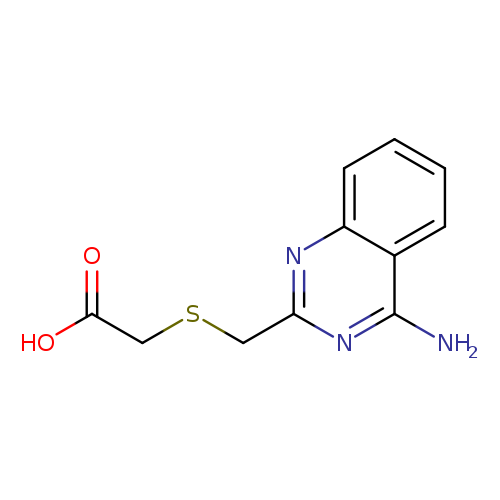
2-{[(4-aminoquinazolin-2-yl)methyl]sulfanyl}acetic acidCatalog No.:AA01A32D CAS No.:1094703-32-8 MDL No.:MFCD11647904 MF:C11H11N3O2S MW:249.2889 |
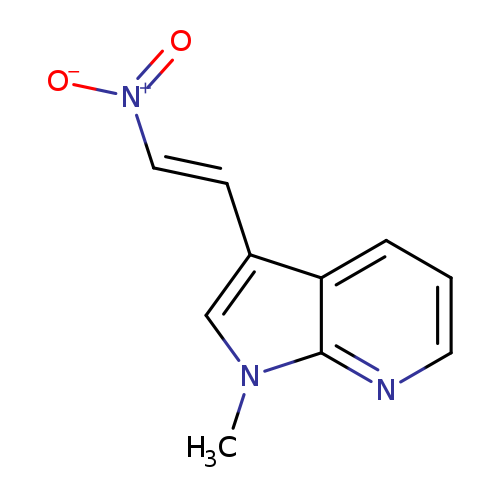
(E)-1-Methyl-3-(2-nitrovinyl)-1H-pyrrolo[2,3-b]pyridineCatalog No.:AA00HBFL CAS No.:1094709-34-8 MDL No.:MFCD22121412 MF:C10H9N3O2 MW:203.1974 |
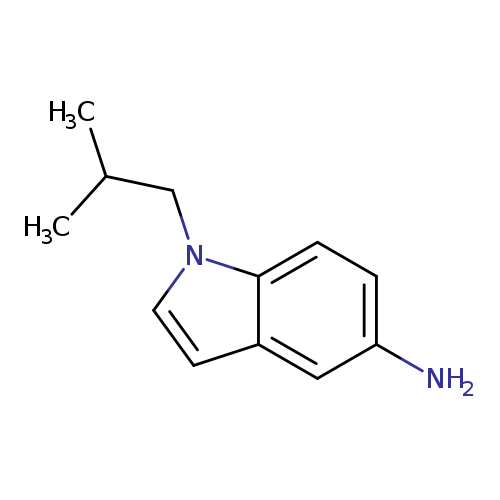
1-isobutyl-1H-indol-5-amineCatalog No.:AA00JK4K CAS No.:1094710-32-3 MDL No.:MFCD11213924 MF:C12H16N2 MW:188.2688 |
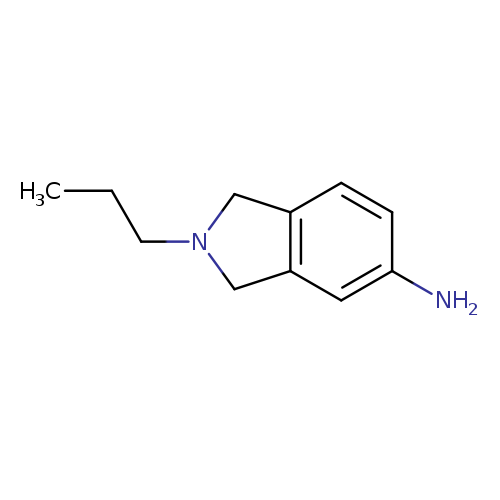
2-propyl-2,3-dihydro-1H-isoindol-5-amineCatalog No.:AA01A8JJ CAS No.:1094711-70-2 MDL No.:MFCD11655080 MF:C11H16N2 MW:176.2581 |
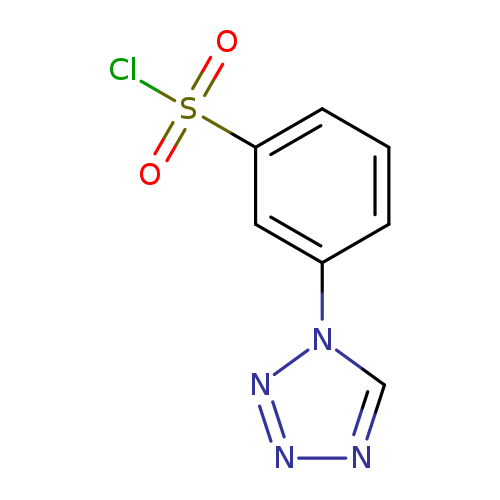
Benzenesulfonylchloride, 3-(1H-tetrazol-1-yl)-Catalog No.:AA0082CL CAS No.:1094713-89-9 MDL No.:MFCD11650652 MF:C7H5ClN4O2S MW:244.6582 |
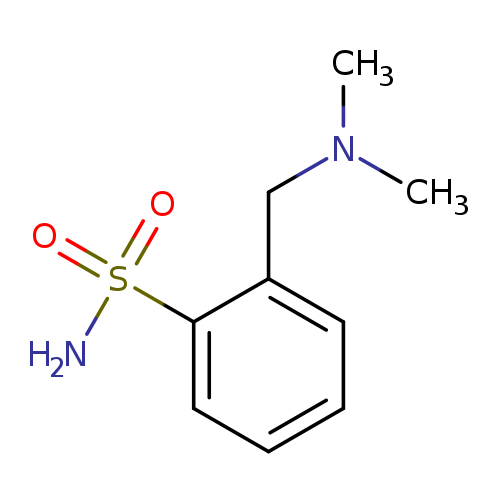
2-[(dimethylamino)methyl]benzene-1-sulfonamideCatalog No.:AA01B2SS CAS No.:1094713-95-7 MDL No.:MFCD11650781 MF:C9H14N2O2S MW:214.2847 |
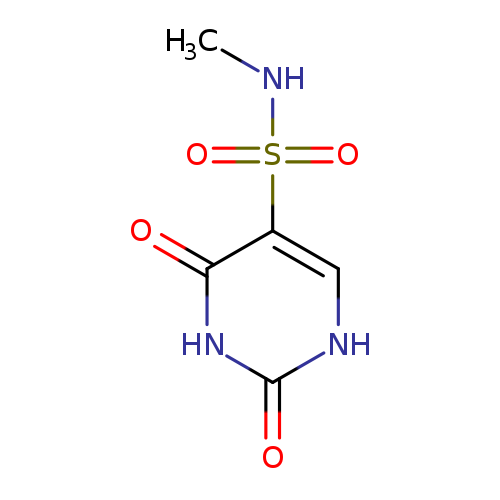
N-Methyl-2,4-dioxo-1,2,3,4-tetrahydropyrimidine-5-sulfonamideCatalog No.:AA01E877 CAS No.:1094715-34-0 MDL No.:MFCD00791451 MF:C5H7N3O4S MW:205.1918 |
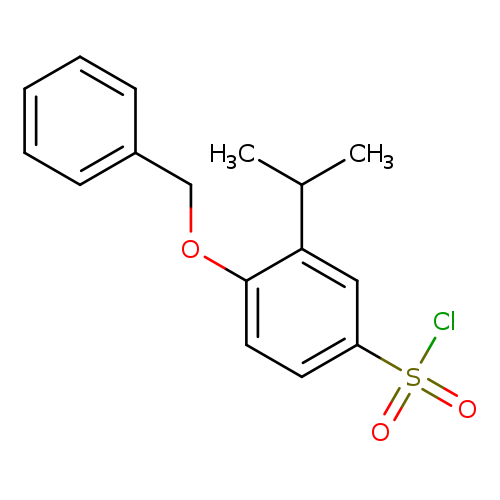
4-(benzyloxy)-3-(propan-2-yl)benzene-1-sulfonyl chlorideCatalog No.:AA01EIVC CAS No.:1094721-09-1 MDL No.:MFCD13635109 MF:C16H17ClO3S MW:324.8224 |
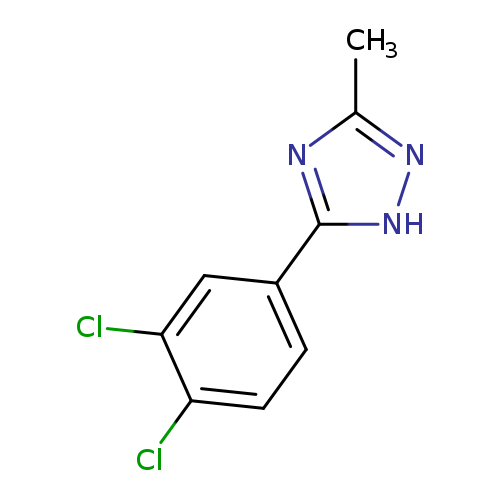
5-(3,4-dichlorophenyl)-3-methyl-1H-1,2,4-triazoleCatalog No.:AA019XMA CAS No.:1094730-06-9 MDL No.:MFCD11215127 MF:C9H7Cl2N3 MW:228.0780 |
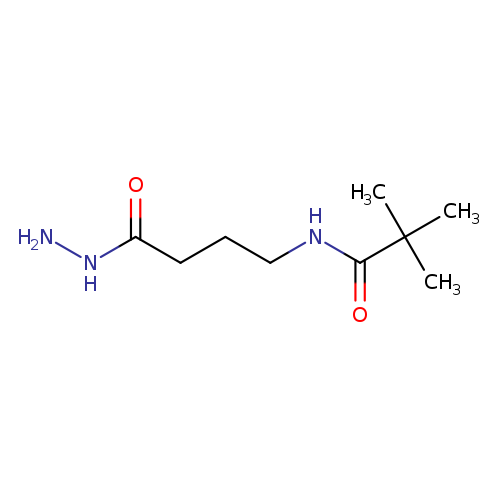
N-[3-(hydrazinecarbonyl)propyl]-2,2-dimethylpropanamideCatalog No.:AA01B5LC CAS No.:1094734-08-3 MDL No.:MFCD11643882 MF:C9H19N3O2 MW:201.2661 |
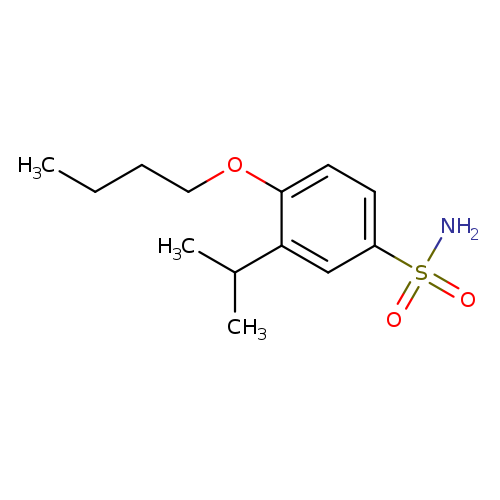
4-Butoxy-3-(propan-2-yl)benzene-1-sulfonamideCatalog No.:AA01BTJ2 CAS No.:1094735-99-5 MDL No.:MFCD11213165 MF:C13H21NO3S MW:271.3757 |
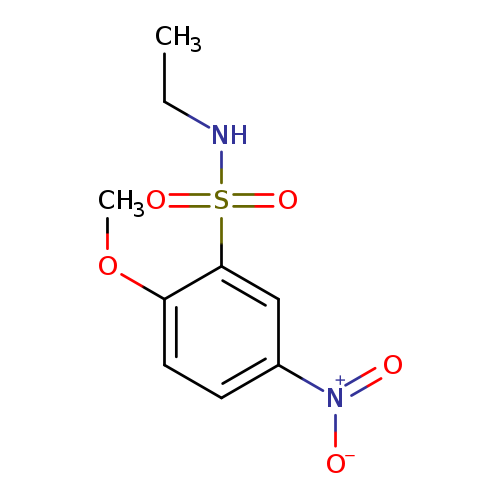
N-Ethyl-2-methoxy-5-nitrobenzenesulfonamideCatalog No.:AA01EH3C CAS No.:1094737-95-7 MDL No.:MFCD11618487 MF:C9H12N2O5S MW:260.2670 |
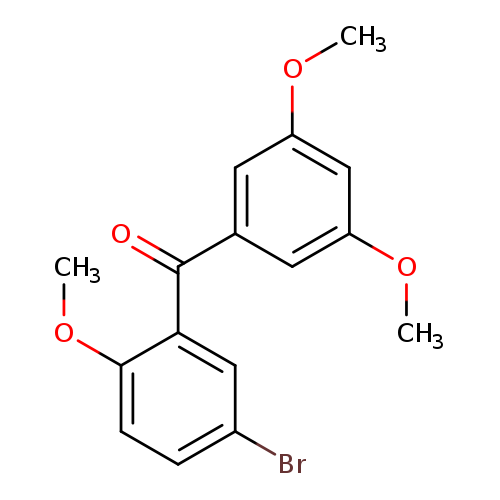
(5-Bromo-2-methoxyphenyl)(3,5-dimethoxyphenyl)methanoneCatalog No.:AA00HBFN CAS No.:1094749-23-1 MDL No.:MFCD13468856 MF:C16H15BrO4 MW:351.1919 |