2020-02-11 20:35:40
David Ruel, Esther Yakir and Jonathan D. Bohbot
INTRODUCTION
Aromatic and heterocyclic compounds play an important role in the ecology of adult mosquitoes as indicated by the odor space of the Anopheles gambiae odorant receptor (OR) repertoire (Carey et al., 2010; Wang et al., 2010). Specifically, indole (IUPAC name, 1H-indole) and skatole (IUPAC name, 3-methylindole) are respectively detected by the narrowly tuned Or2 and Or10 paralogous genes found in Culex quinquefasciatus (Hughes et al., 2010; Pelletier et al., 2010), Aedes aegypti and An. gambiae (Carey et al., 2010; Wang et al., 2010) reflecting their ancestral origin (Bohbot et al., 2011). Due to the sensitive and selective nature of the OR2-indole and OR10-skatole interactions, they have been referred to as the "indolergic” receptors (Bohbot and Pitts, 2015).
Indole and skatole are released by a wide variety of organisms but are mainly synthesized by bacteria (Elgaali et al., 2002; Schulz and Dickschat, 2007; Lindh et al., 2008; Hubbard et al., 2015), fungi (Chen et al., 2014; Tomberlin et al., 2017) and plants (Turlings et al., 1991; Frey et al., 2000; Ober, 2005). In adult mosquitoes, both compounds have been proposed to mediate oviposition site (Blackwell and Johnson, 2000) and host-locating behaviors (Cork, 1996). However, their exact ecological role(s) remain complex since indoles are major constituents of floral (Knudsen et al., 2006) and animal scents (Meijerink et al., 2001; Lee et al., 2015). Interestingly, indolic compounds play additional ecological roles in mosquito larvae (Xia et al., 2008; Scialo et al., 2012).
Or2 is expressed in the adult and larval stages of Ae. aegypti (Bohbot et al., 2007) and An. gambiae (Hill et al., 2002; Xia et al., 2008). Or10 expression is more complex: in An. gambiae, Or10 is expressed both in larvae and adults. In Ae. Aegypti, Or10 is only expressed in adults, while a third paralog named Or9, is expressed in the larval antenna (Bohbot et al., 2007). Based on pharmacological studies, we have suggested that receptor sensitivity towards odorants in the nanomolar concentration range is a predictor of OR-semiochemical relationships (Bohbot and Pitts, 2015). The activation of AaegOR9 by indole in the low micromolar concentration range (Bohbot et al., 2011) indicated that a more potent indolic cognate ligand selectively activates this receptor.
Using a reverse chemical ecology approach, we set out to identify a potential cognate ligand for this larval-expressed Or9 gene (Supplementary Table 1). First, we used a panel of 31 indole derivatives from plants and microbes to identify a potent activator of AaegOR9, then we showed that AaegOR9 is narrowly tuned to skatole in the low nanomolar concentration range. Our findings suggest that Culicinae have developed a supersensitive skatole receptor that operates in water where this compound exhibits low solubility. The occurrence of two skatole receptors, each assigned to a different developmental stage indicates the central role of this odorant in the Ae. aegypti life cycle. The deorphanization of AaegOR9: (i) provides a molecular target for future larval behavioral disruption studies;
(ii)improves our understanding of insect OR coding; and
(iii)raises questions on the possible ecological roles of mosquito indolergic receptors.
MATERIALS AND METHODS
Chemical Reagents
The chemicals (Supplementary Table 1) used for the deorphanization of AaegOR9 were obtained from Sigma- Aldrich (Milwaukee, WI, USA), ChemCruz (Dallas, TX, USA), Glentham Life Sciences (Corsham, UK), FluoroChem (Hadfield, UK), SL Moran (Jerusalem, Israel), Holland Moran (Yehun, Israel), Alfa Aesar (Ward Hill, MA, USA) and from the generous contribution of the Dr. Kolodkin-Gal Lab (Weizmann Institute of Science, Israel).
Two-Electrode Voltage Clamp of Xenopus Oocytes Expressing ORs
The methodologies and protocols have been described in details elsewhere (Bohbot and Dickens, 2009). AaegOr9 and Aaeg- ORco cRNAs (Bohbot et al., 2011) were synthesized from linearized pSP64DV expression vectors using the mMESSAGE mMACHINE⑥ SP6 kit (Life Technologies). Stage V-VII oocytes were harvested from Xenopus laevis females, mechanically separated, treated with collagenase (8 mg/mL, 30 min, 18°C) and rinsed in washing solution (96 mM NaCl, 2 mM KCl, 5 mM MgCl2 and 5 mM HEPES, pH 7.6). Oocytes were microinjected with 27.6 ng AaegOr9 and AaegORco cRNAs, incubated at 18° C for 3-4 days in ND96 solution (96 mM NaCl, 2 mM KCl, 5 mM MgCl?, 0.8 mM CaCl? and 5 mM HEPES, pH 7.6), supplemented with 5% dialyzed horse serum, 50 Mg/mL tetracycline, 100 jig/mL streptomycin and 550 Mg/mL sodium pyruvate. Whole-cell currents were recorded using the two-microelectrode voltage-clamp technique. During recording sessions, the holding potential was maintained at -80 mV using an OC-725C oocyte clamp (Warner Instruments, LLC, Hamden, CT, USA). Oocytes placed in a RC-3Z oocyte recording chamber (Warner Instruments, LLC, Hamden, CT, USA) were exposed to odorants for 8 s. Current was allowed to return to baseline between odorant applications. Data acquisition and concentration-response analyses were carried out with a Digidata 1550A and the pCLAMP10 software (Molecular Devices, Sunnyvale, CA, USA), and analyzed using GraphPad Prism 7 (GraphPad Software Inc., La Jolla, CA, USA). Stock concentration of odorants (10-2 M) were dissolved in ringer solution containing 2% dimethyl sulfoxide (DMSO) in order to solubilize the hydrophobic indolic compounds.
Pharmacological Characterization
The response profile was established using multiple sessions, each including six compounds at a time and indole as an internal reference. The order in which these compounds were administered was reversed within a session to mitigate against any potential sequence effects between compounds (none were observed). All the response values were normalized to the indole reference in each recording session (Supplementary Figure 1).
For the establishment of the concentration-response curves, oocytes were exposed to increasing concentrations of indole, skatole and indole-3-carboxaldehyde (I3C; Supplementary Figure 2). Quantitative characterization of OR sensitivity was estimated using the averaged effective concentration at 50% of the maximal response (EC50) over the sample population. The data to establish the concentration response curve and EC50 of AaegOR10-skatole was extracted from a previous study (Bohbot and Dickens, 2012).
Phylogeny OR Intron-Exon Structure and Phylogeny
All the sequences used in our phylogenic analysis were obtained from the VectorBase and NCBI databases using AaegOr2/9/10 as query (for accession numbers, see Supplementary Table 3). DNA sequences for Toxorhynchites Or2 and Or10 can be accessed here: http://dx.doi.org/10.6084/m9.figshare.1092617. MAFFT version 7 (Nakamura et al., 2018) was used for multiple amino-acid sequence alignment. The phylogenic software IQ-TREE (Nguyen et al., 2015; Kalyaanamoorthy et al., 2017; Hoang et al., 2018) and the FigTree phylogenic tree based on the maximum likelihood method (Model: JC, UFbootstrap: 5,000). Using MAFFT (default parameters) and the Vectorbase database, we located the intron positions on the indolergic receptor genes.
[1-(2-Methylphenyl)-1h-1,2,3-triazol-4-yl]methanolCatalog No.:AA01B98U CAS No.:1096130-75-4 MDL No.:MFCD11844961 MF:C10H11N3O MW:189.2138 |
(1S,3R)-3-aminocyclopentane-1-carboxylic acid HClCatalog No.:AA008YZ3 CAS No.:1096155-68-8 MDL No.:MFCD00492872 MF:C6H12ClNO2 MW:165.6180 |
3-(1-Bromoethyl)benzonitrileCatalog No.:AA01B161 CAS No.:1096159-01-1 MDL No.:MFCD19625763 MF:C9H8BrN MW:210.0705 |
2-Propenoic acid, 3-(3-chlorophenyl)-2-methyl-, (2E)-Catalog No.:AA00822N CAS No.:109619-78-5 MDL No.:MFCD00666054 MF:C10H9ClO2 MW:196.6303 |
(+)-TRANS-(1R,2R)-U-50488 HYDROCHLORIDECatalog No.:AA008XAX CAS No.:109620-49-7 MDL No.:MFCD00211360 MF:C19H27Cl3N2O MW:405.7895 |
N-(2-aminoethyl)-1-methyl-1H-pyrrole-3-sulfonamideCatalog No.:AA01A1VC CAS No.:1096270-51-7 MDL No.:MFCD11633199 MF:C7H13N3O2S MW:203.2620 |
(cyclobutylmethyl)[(2,4-dimethoxyphenyl)methyl]amineCatalog No.:AA01B8SG CAS No.:1096278-17-9 MDL No.:MFCD11634814 MF:C14H21NO2 MW:235.3220 |
1H-BenziMidazole, 2-(ethylthio)-, MonohydrobroMideCatalog No.:AA0095VT CAS No.:109628-14-0 MDL No.:MFCD01596149 MF:C9H11BrN2S MW:259.1660 |
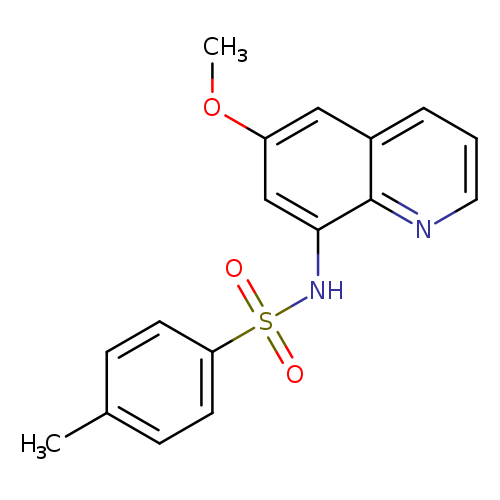
TSQCatalog No.:AA008RKJ CAS No.:109628-27-5 MDL No.:MFCD00467847 MF:C17H16N2O3S MW:328.3855 |
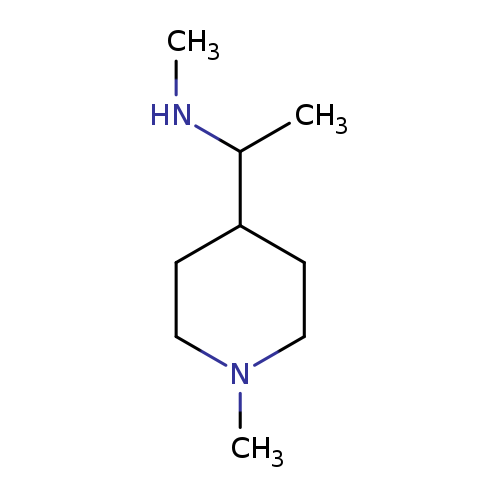
methyl[1-(1-methylpiperidin-4-yl)ethyl]amineCatalog No.:AA019XIE CAS No.:1096282-35-7 MDL No.:MFCD11637866 MF:C9H20N2 MW:156.2685 |
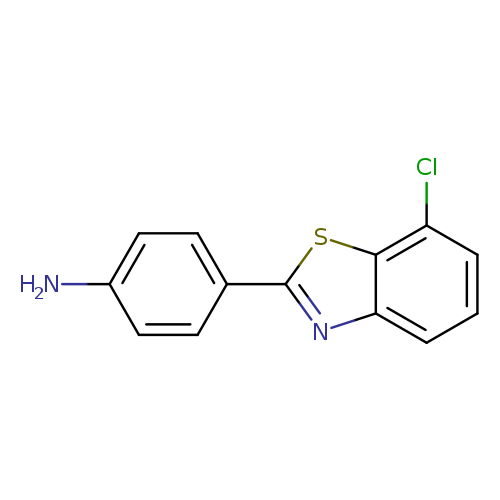
4-(7-chloro-1,3-benzothiazol-2-yl)anilineCatalog No.:AA019YG1 CAS No.:1096285-07-2 MDL No.:MFCD11637006 MF:C13H9ClN2S MW:260.7420 |
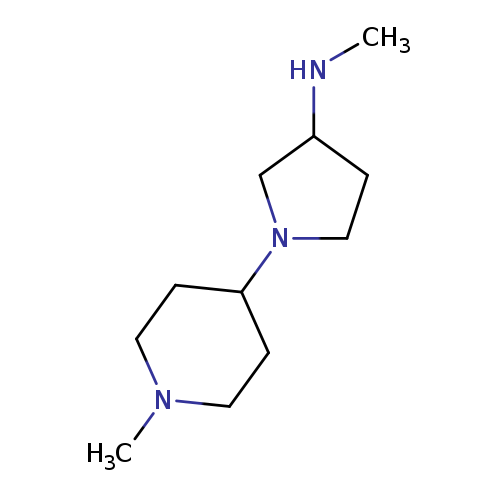
N-methyl-1-(1-methylpiperidin-4-yl)pyrrolidin-3-amineCatalog No.:AA01AH9O CAS No.:1096289-19-8 MDL No.:MFCD12167334 MF:C11H23N3 MW:197.3204 |
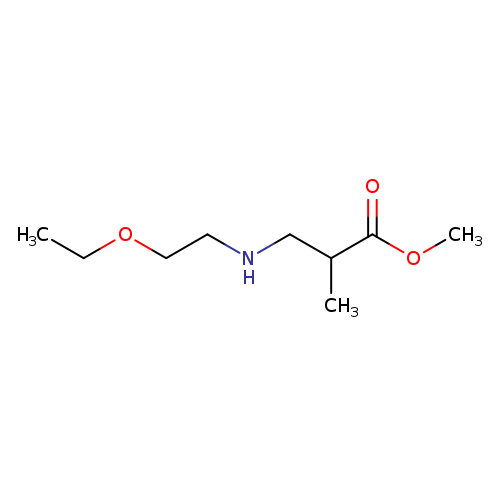
methyl 3-[(2-ethoxyethyl)amino]-2-methylpropanoateCatalog No.:AA00JT5B CAS No.:109629-29-0 MDL No.:MFCD12079827 MF:C9H19NO3 MW:189.2521 |
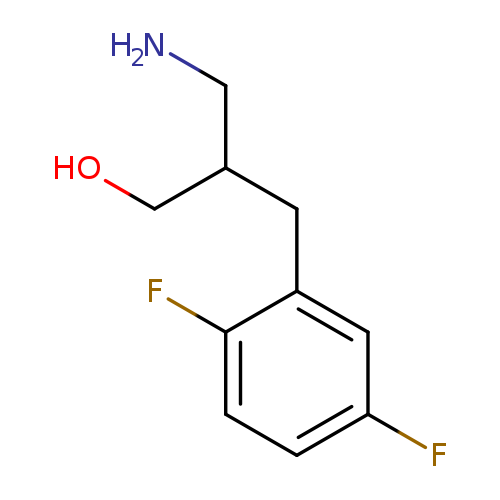
3-amino-2-[(2,5-difluorophenyl)methyl]propan-1-olCatalog No.:AA019SBR CAS No.:1096296-04-6 MDL No.:MFCD11639648 MF:C10H13F2NO MW:201.2131 |
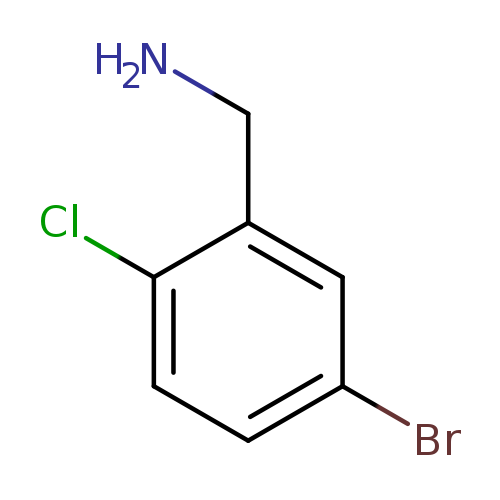
5-Bromo-2-chlorobenzylamineCatalog No.:AA007SRQ CAS No.:1096296-85-3 MDL No.:MFCD12026286 MF:C7H7BrClN MW:220.4942 |
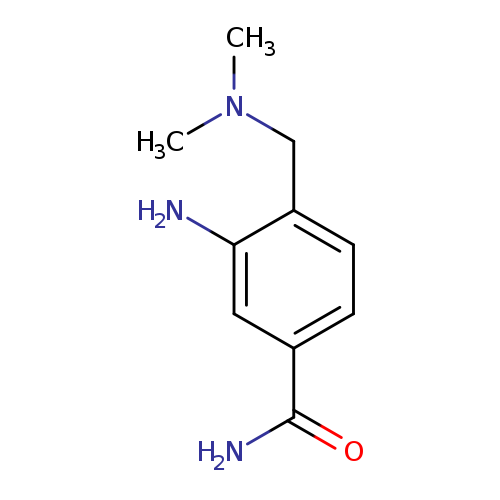
3-amino-4-[(dimethylamino)methyl]benzamideCatalog No.:AA01AJTQ CAS No.:1096299-39-6 MDL No.:MFCD12167950 MF:C10H15N3O MW:193.2456 |
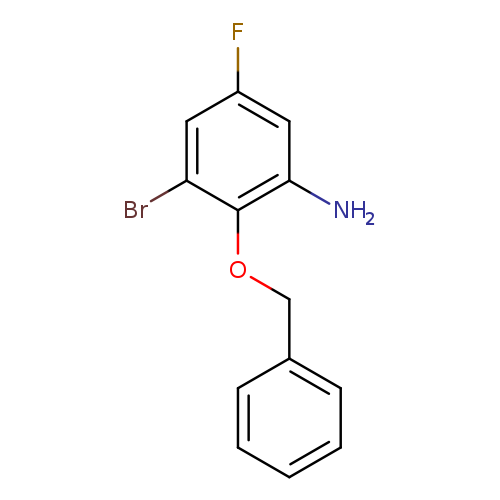
2-(Benzyloxy)-3-bromo-5-fluoroanilineCatalog No.:AA009472 CAS No.:1096303-75-1 MDL No.:MFCD12408888 MF:C13H11BrFNO MW:296.1349 |
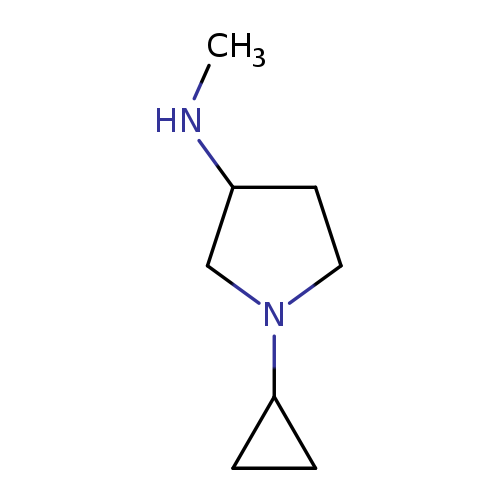
1-cyclopropyl-N-methylpyrrolidin-3-amineCatalog No.:AA01AHF6 CAS No.:1096304-44-7 MDL No.:MFCD11640858 MF:C8H16N2 MW:140.2260 |
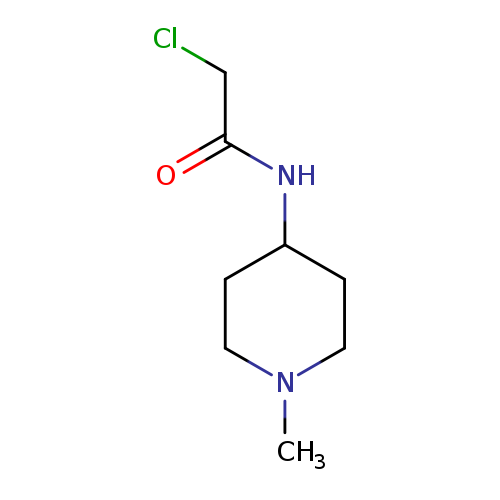
2-Chloro-N-(1-methylpiperidin-4-yl)acetamideCatalog No.:AA01BR1L CAS No.:1096305-49-5 MDL No.:MFCD12166284 MF:C8H15ClN2O MW:190.6705 |
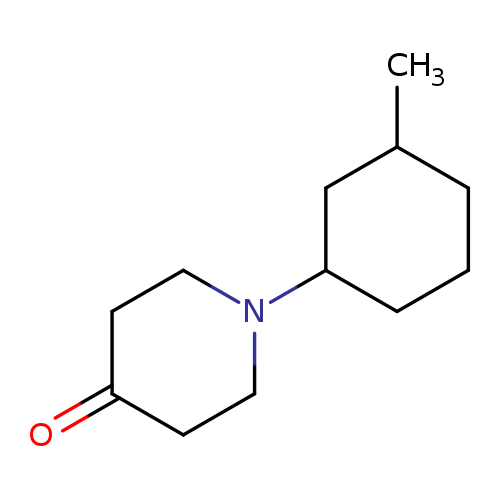
1-(3-methylcyclohexyl)piperidin-4-oneCatalog No.:AA01AH3I CAS No.:1096306-10-3 MDL No.:MFCD12166449 MF:C12H21NO MW:195.3012 |
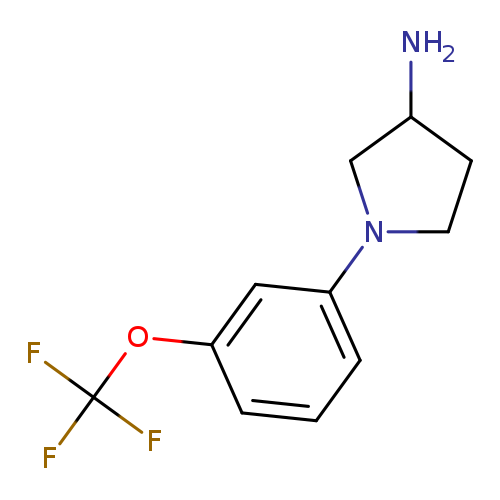
1-[3-(Trifluoromethoxy)phenyl]pyrrolidin-3-amineCatalog No.:AA01AI9D CAS No.:1096306-23-8 MDL No.:MFCD11641008 MF:C11H13F3N2O MW:246.2289 |
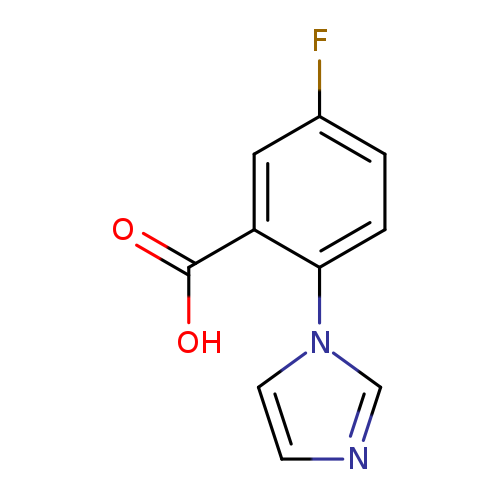
5-fluoro-2-(1H-imidazol-1-yl)benzoic acidCatalog No.:AA01AK7M CAS No.:1096306-42-1 MDL No.:MFCD11639849 MF:C10H7FN2O2 MW:206.1732 |
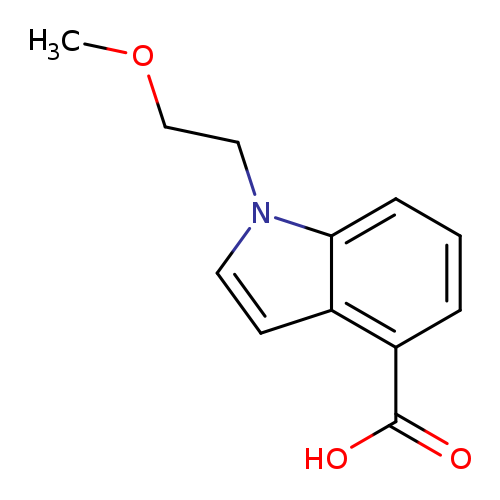
1-(2-Methoxyethyl)-1h-indole-4-carboxylic acidCatalog No.:AA00HBH7 CAS No.:1096306-76-1 MDL No.:MFCD11641383 MF:C12H13NO3 MW:219.2365 |
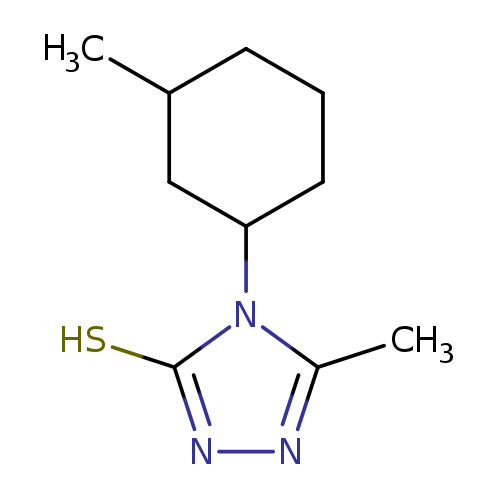
5-Methyl-4-(3-methylcyclohexyl)-4h-1,2,4-triazole-3-thiolCatalog No.:AA01A2Z6 CAS No.:1096306-89-6 MDL No.:MFCD12166493 MF:C10H17N3S MW:211.3271 |
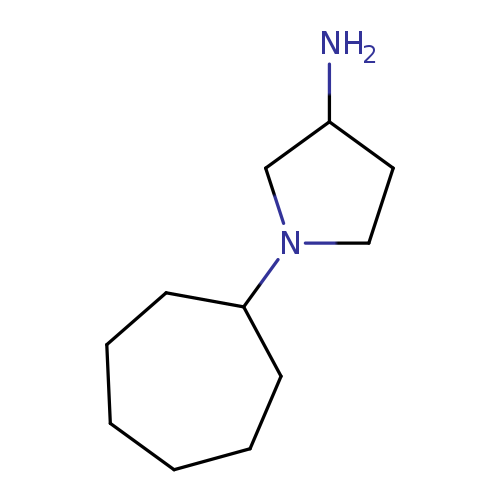
1-cycloheptylpyrrolidin-3-amineCatalog No.:AA01A937 CAS No.:1096307-23-1 MDL No.:MFCD11641073 MF:C11H22N2 MW:182.3058 |
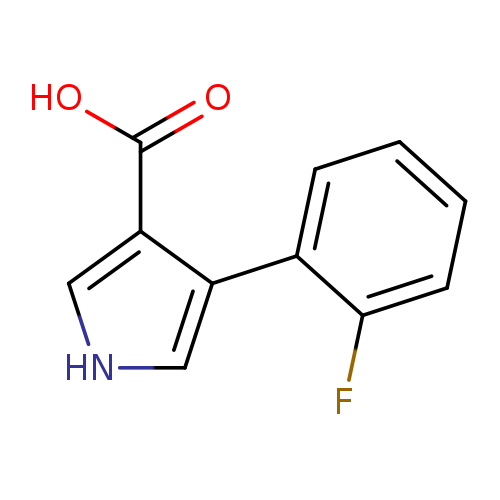
4-(2-fluorophenyl)-1H-pyrrole-3-carboxylic acidCatalog No.:AA01BF76 CAS No.:1096308-11-0 MDL No.:MFCD09746414 MF:C11H8FNO2 MW:205.1851 |
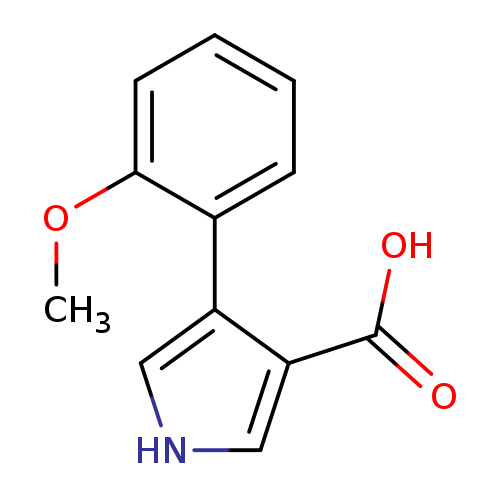
4-(2-Methoxyphenyl)-1h-pyrrole-3-carboxylic acidCatalog No.:AA01A94T CAS No.:1096308-15-4 MDL No.:MFCD11217946 MF:C12H11NO3 MW:217.2206 |
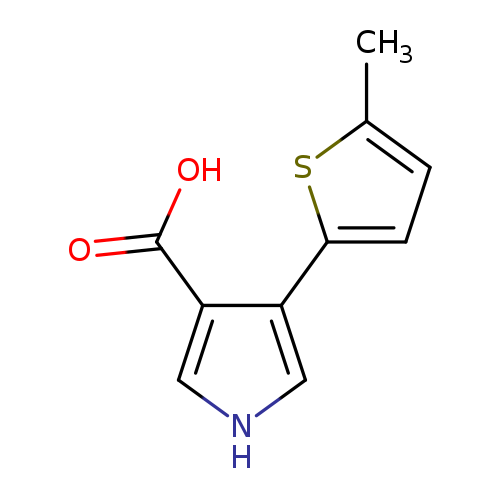
4-(5-methylthiophen-2-yl)-1H-pyrrole-3-carboxylic acidCatalog No.:AA01AFAB CAS No.:1096308-28-9 MDL No.:MFCD11641424 MF:C10H9NO2S MW:207.2490 |
4-(6-methoxynaphthalen-2-yl)-1H-pyrrole-3-carboxylic acidCatalog No.:AA019WVI CAS No.:1096308-30-3 MDL No.:MFCD11217988 MF:C16H13NO3 MW:267.2793 |
methyl 4-amino-3-[(methylcarbamoyl)methoxy]benzoateCatalog No.:AA01BV6G CAS No.:1096309-23-7 MDL No.:MFCD12410241 MF:C11H14N2O4 MW:238.2399 |
1-chloro-4-[2-(chloromethyl)-4-fluorophenoxy]-2-ethylbenzeneCatalog No.:AA01E8MU CAS No.:1096309-52-2 MDL No.:MFCD12456612 MF:C15H13Cl2FO MW:299.1675 |
1-(4-TERT-BUTYLPHENYL)CYCLOPROPANE-1-CARBONITRILECatalog No.:AA01EJOW CAS No.:1096317-25-7 MDL No.:MFCD12410112 MF:C14H17N MW:199.2915 |
[1-(4-tert-butylphenyl)cyclopentyl]methanamineCatalog No.:AA01A25U CAS No.:1096317-29-1 MDL No.:MFCD11898956 MF:C16H25N MW:231.3764 |
{[2-(4-chloro-2-methylphenoxy)-5-fluorophenyl]methyl}(methyl)amineCatalog No.:AA01E8JZ CAS No.:1096319-05-9 MDL No.:MFCD12456707 MF:C15H15ClFNO MW:279.7371 |
1-(Ethylamino)-3-(4-(2-methoxyethyl)phenoxy)propan-2-olCatalog No.:AA007AC9 CAS No.:109632-08-8 MDL No.:MFCD11549256 MF:C14H23NO3 MW:253.3373 |
2-[(4-methylpiperazin-1-yl)methyl]-1H-1,3-benzodiazol-6-amineCatalog No.:AA019XI9 CAS No.:1096321-61-7 MDL No.:MFCD11978899 MF:C13H19N5 MW:245.3235 |
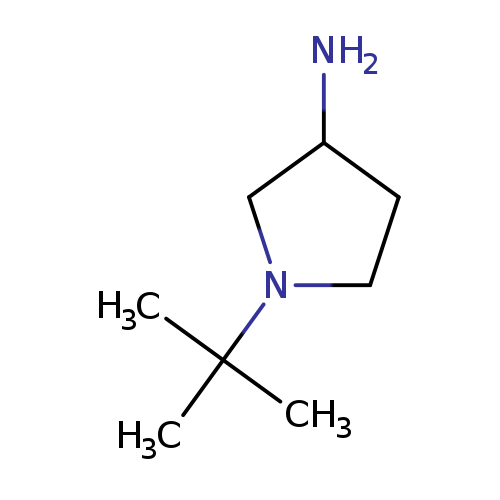
1-tert-butyl-3-pyrrolidinamineCatalog No.:AA00J2IQ CAS No.:1096325-48-2 MDL No.:MFCD11640606 MF:C8H18N2 MW:142.2419 |
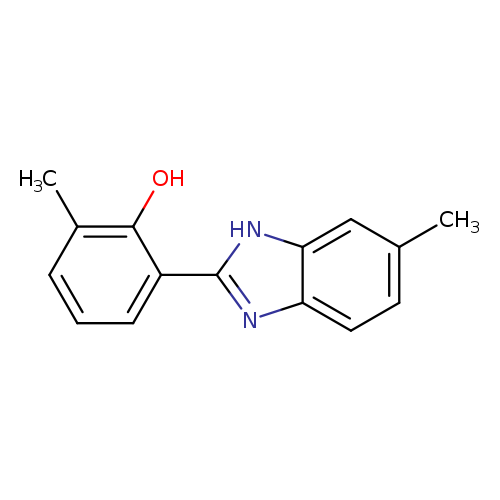
2-methyl-6-(6-methyl-1H-1,3-benzodiazol-2-yl)phenolCatalog No.:AA01A3EM CAS No.:1096325-53-9 MDL No.:MFCD12069244 MF:C15H14N2O MW:238.2845 |
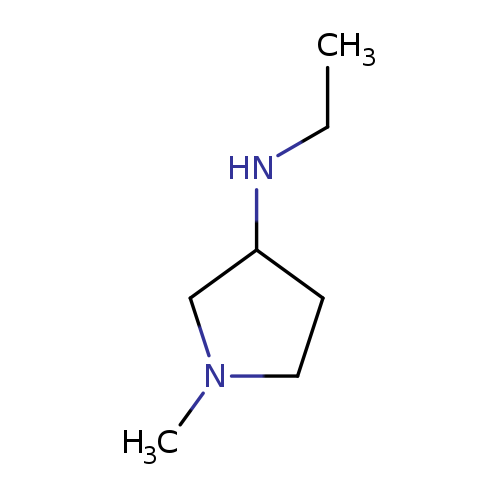
Ethyl-(1-methyl-pyrrolidin-3-yl)-amineCatalog No.:AA00HBHA CAS No.:1096325-56-2 MDL No.:MFCD11640615 MF:C7H16N2 MW:128.2153 |
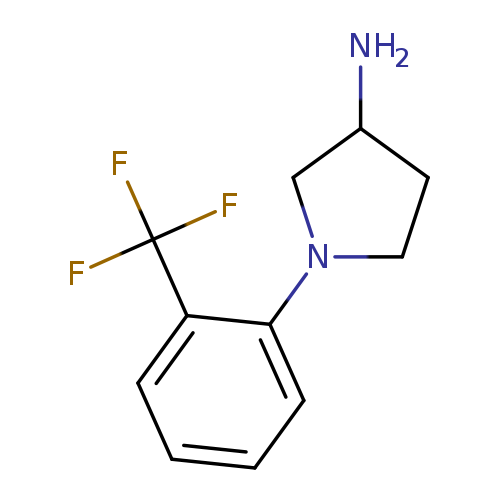
1-[2-(trifluoromethyl)phenyl]pyrrolidin-3-amineCatalog No.:AA01AF50 CAS No.:1096326-52-1 MDL No.:MFCD11640657 MF:C11H13F3N2 MW:230.2295 |
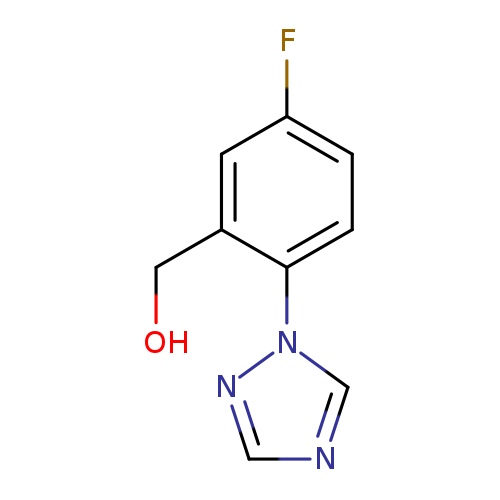
[5-fluoro-2-(1H-1,2,4-triazol-1-yl)phenyl]methanolCatalog No.:AA01BFF0 CAS No.:1096329-29-1 MDL No.:MFCD11639940 MF:C9H8FN3O MW:193.1777 |
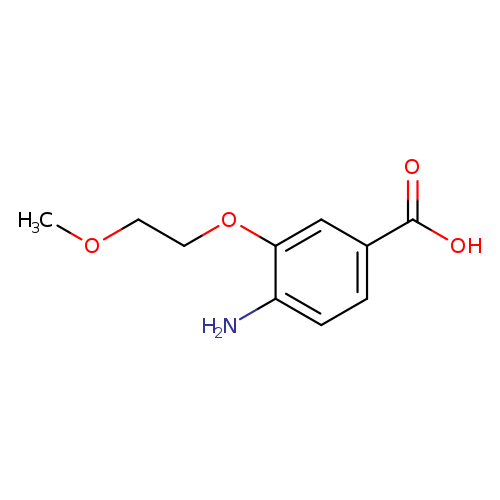
4-Amino-3-(2-methoxyethoxy)benzoic acidCatalog No.:AA01BVDZ CAS No.:1096330-19-6 MDL No.:MFCD12168716 MF:C10H13NO4 MW:211.2145 |
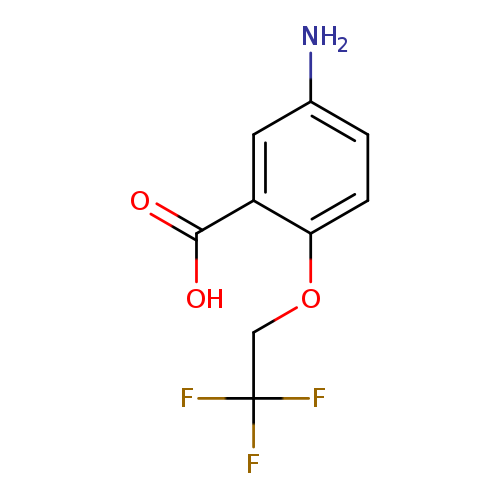
5-amino-2-(2,2,2-trifluoroethoxy)benzoic acidCatalog No.:AA01AGS8 CAS No.:1096332-51-2 MDL No.:MFCD12168743 MF:C9H8F3NO3 MW:235.1599 |
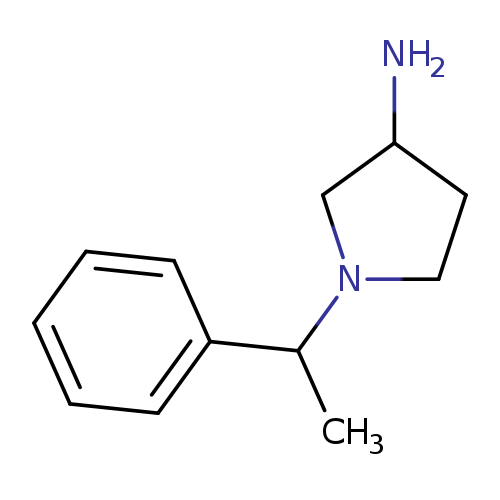
1-(1-Phenylethyl)pyrrolidin-3-amineCatalog No.:AA019WTJ CAS No.:1096332-61-4 MDL No.:MFCD11640767 MF:C12H18N2 MW:190.2847 |
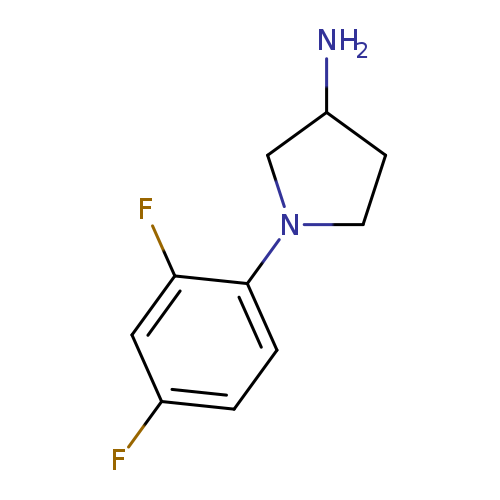
1-(2,4-Difluorophenyl)pyrrolidin-3-amineCatalog No.:AA01ABBW CAS No.:1096333-56-0 MDL No.:MFCD11640823 MF:C10H12F2N2 MW:198.2125 |
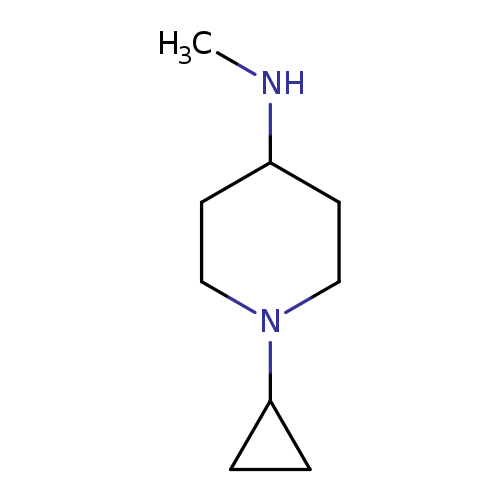
(1-CYCLOPROPYL-PIPERIDIN-4-YL)-METHYL-AMINECatalog No.:AA01DX82 CAS No.:1096334-18-7 MDL No.:MFCD11640861 MF:C9H18N2 MW:154.2526 |
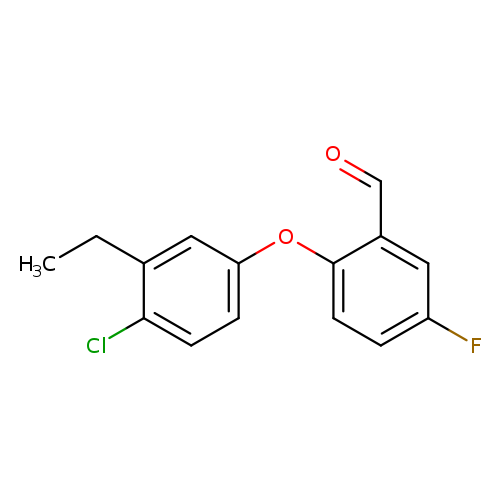
2-(4-chloro-3-ethylphenoxy)-5-fluorobenzaldehydeCatalog No.:AA01EA5T CAS No.:1096334-56-3 MDL No.:MFCD12456598 MF:C15H12ClFO2 MW:278.7060 |
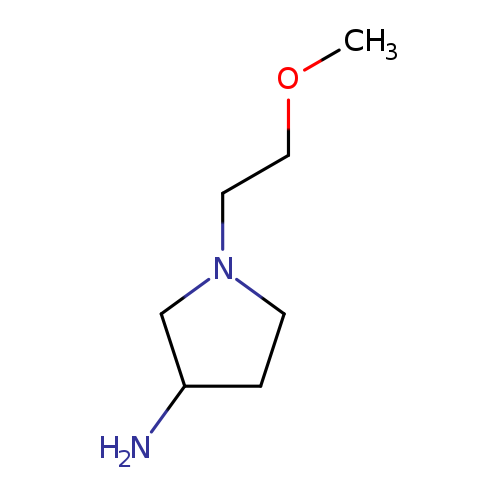
1-(2-Methoxyethyl)pyrrolidin-3-amineCatalog No.:AA01AGIJ CAS No.:1096335-57-7 MDL No.:MFCD11640980 MF:C7H16N2O MW:144.2147 |
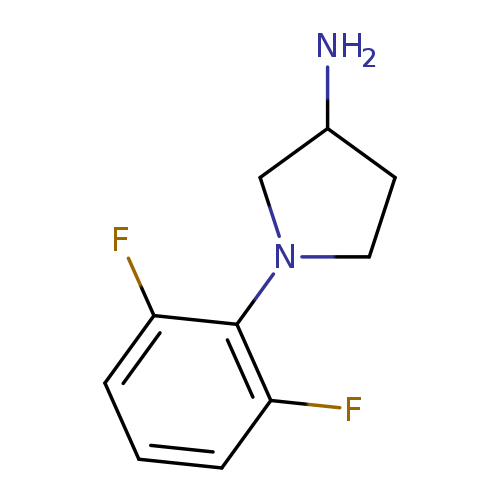
1-(2,6-Difluorophenyl)pyrrolidin-3-amineCatalog No.:AA019Z55 CAS No.:1096336-44-5 MDL No.:MFCD11641048 MF:C10H12F2N2 MW:198.2125 |
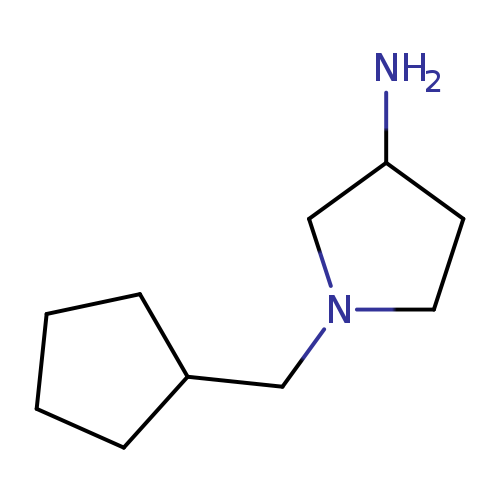
1-(cyclopentylmethyl)pyrrolidin-3-amineCatalog No.:AA019SCX CAS No.:1096341-29-5 MDL No.:MFCD12166677 MF:C10H20N2 MW:168.2792 |
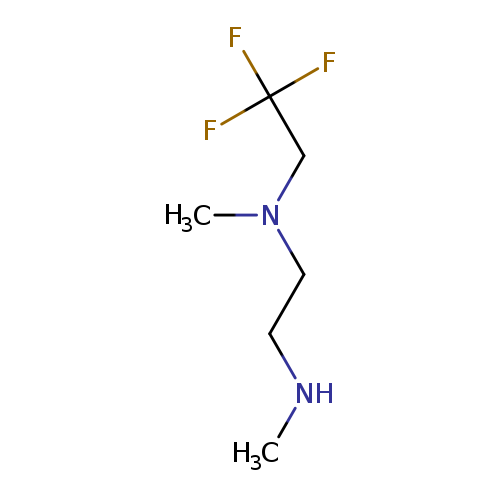
methyl({2-[methyl(2,2,2-trifluoroethyl)amino]ethyl})amineCatalog No.:AA01AHYY CAS No.:1096348-60-5 MDL No.:MFCD12167703 MF:C6H13F3N2 MW:170.1760 |
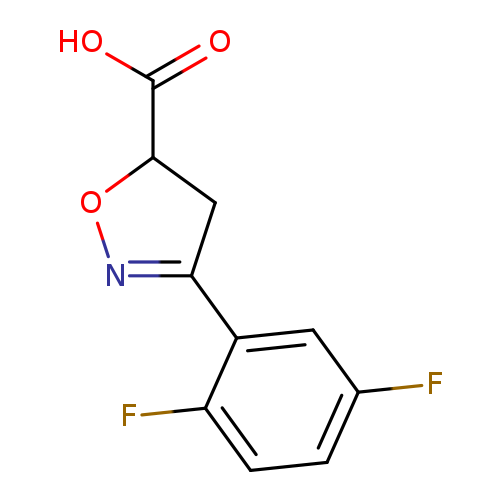
3-(2,5-Difluorophenyl)-4,5-dihydro-1,2-oxazole-5-carboxylic acidCatalog No.:AA01AHKE CAS No.:1096348-81-0 MDL No.:MFCD11639661 MF:C10H7F2NO3 MW:227.1643 |
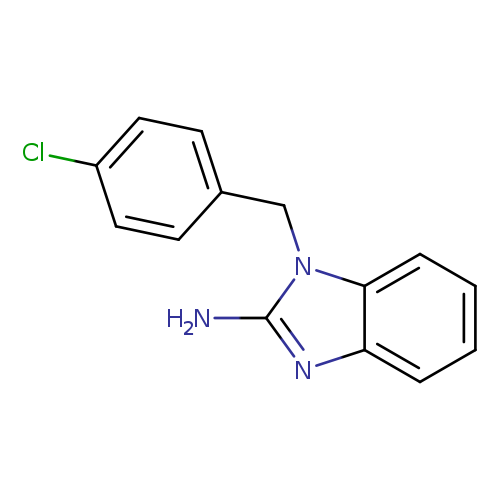
1-(4-Chlorobenzyl)-1H-benzo[d]imidazol-2-amineCatalog No.:AA009LVZ CAS No.:109635-38-3 MDL No.:MFCD00667028 MF:C14H12ClN3 MW:257.7182 |
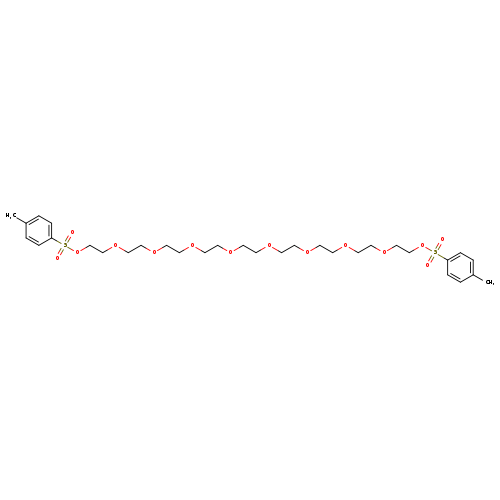
Nonaethylenel di(p-toluenesulfonate)Catalog No.:AA00HBHE CAS No.:109635-64-5 MDL No.:MFCD25372003 MF:C32H50O14S2 MW:722.8610 |
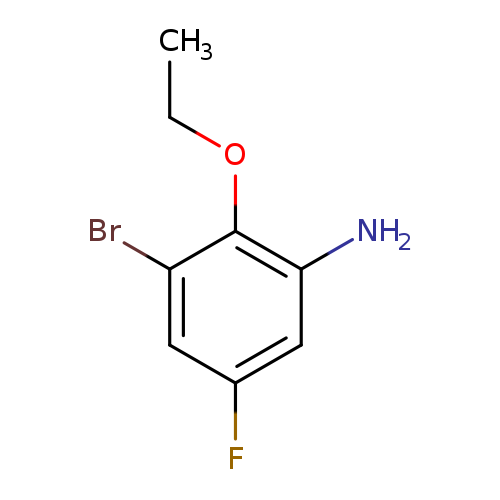
3-Bromo-2-ethoxy-5-fluoroanilineCatalog No.:AA007SRM CAS No.:1096354-40-3 MDL No.:MFCD13623652 MF:C8H9BrFNO MW:234.0656 |
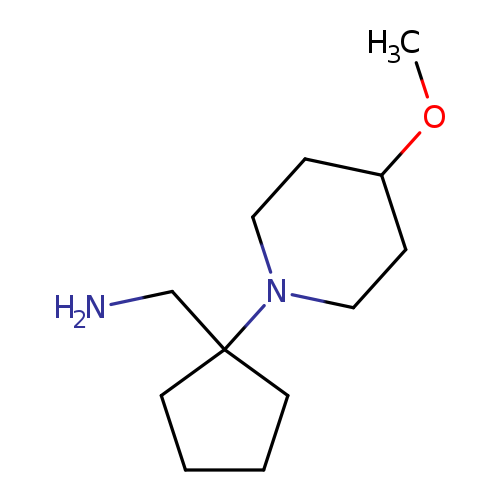
[1-(4-methoxypiperidin-1-yl)cyclopentyl]methanamineCatalog No.:AA01AGL8 CAS No.:1096355-79-1 MDL No.:MFCD12167165 MF:C12H24N2O MW:212.3318 |
5-thia-8-azaspiro[3.4]octaneCatalog No.:AA01BKCG CAS No.:109640-45-1 MDL No.:MFCD23920108 MF:C6H11NS MW:129.2232 |
methyl 2-[(5-chloro-1,3-dimethyl-1H-pyrazol-4-yl)formamido]-2-phenylacetateCatalog No.:AA00ISEW CAS No.:1096440-16-2 MDL No.:MFCD03305825 MF:C15H16ClN3O3 MW:321.7588 |
3-Amino-3-cyclohexylpropan-1-olCatalog No.:AA008SJD CAS No.:109647-95-2 MDL No.:MFCD03412684 MF:C9H19NO MW:157.2533 |

Neuronostatin-13 trifluoroacetate saltCatalog No.:AA009791 CAS No.:1096485-24-3 MDL No.:MFCD11975025 MF: MW: |
3-Phenyl-6,7,8,9-tetrahydro-5h-imidazo[1,5-a]azepine-1-carboxylic acidCatalog No.:AA019LW3 CAS No.:109650-67-1 MDL No.:MFCD09971640 MF:C15H16N2O2 MW:256.2997 |
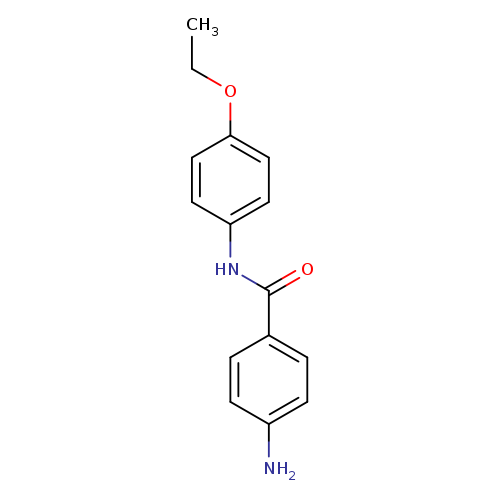
4-AMINO-N-(4-ETHOXYPHENYL)BENZAMIDE HYDROCHLORIDECatalog No.:AA008ZCF CAS No.:109651-02-7 MDL No.:MFCD08447182 MF:C15H16N2O2 MW:256.2997 |
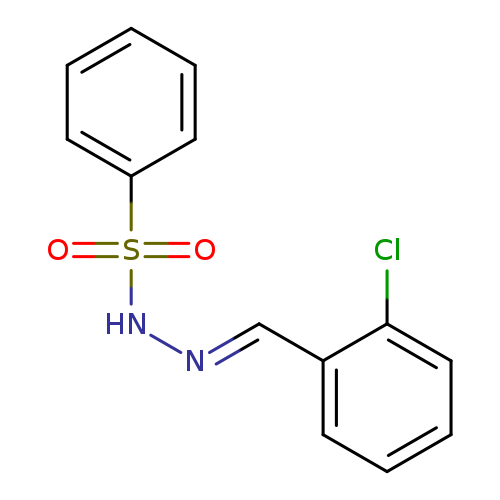
N'-[(1E)-(2-chlorophenyl)methylidene]benzenesulfonohydrazideCatalog No.:AA00ITNF CAS No.:1096535-32-8 MDL No.:MFCD01225032 MF:C13H11ClN2O2S MW:294.7566 |
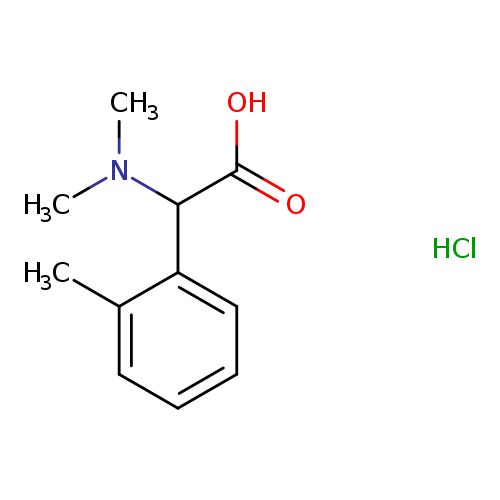
(Dimethylamino)(2-methylphenyl)acetic acid hydrochlorideCatalog No.:AA00J1BC CAS No.:1096586-07-0 MDL No.:MFCD18483397 MF:C11H16ClNO2 MW:229.7032 |
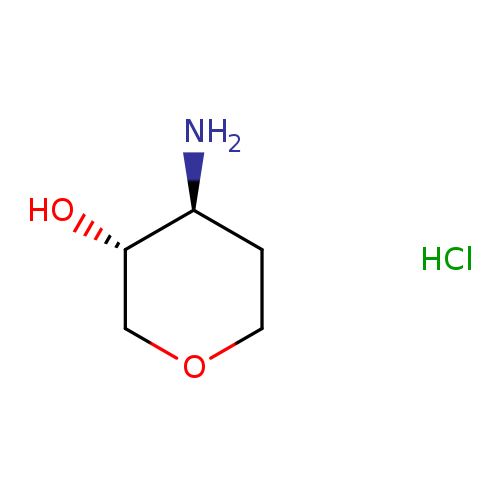
(3R,4S)-4-Aminooxan-3-ol hydrochlorideCatalog No.:AA00HBHK CAS No.:1096594-11-4 MDL No.:MFCD23159220 MF:C5H12ClNO2 MW:153.6073 |
2-(4,4-Dimethyl-4,5-dihydro-2-oxazolyl)quinolineCatalog No.:AA003ERQ CAS No.:109660-13-1 MDL No.:MFCD19443638 MF:C14H14N2O MW:226.2738 |