2020-02-14 16:49:20
Yuan Cheng,[a] Xiongyu Ou,[a] Jimei Ma,[a] Linhao Sun,[a] and Zhong-Hua Ma*
Introduction
Using water as a reaction media offers advantages for cost, safety, and environment, and is therefore considered green and ideal for laboratory and industry processes. Hence many efforts have been devoted to the development of aqueous catalytic reactions.[1] In aqueous suspension, some reactions involving organic substrates are accelerated, which has been defined as “on water” reactions by Sharpless.[1b] Several approaches have been aimed to strengthen the interaction of organic substrates under “on water” conditions, including the use of surfactants as amphiphilic additives, the use of phase-transfer catalysts,[2] and the specifically functionalized design of the catalysts structure. Water also brings some benefits for product isolation. Various types of reactions forming C-C and C-heteroatom bond have been carried out in water with enhanced reactivity and selectivity, such as the nucleophilic attack of a carbanion species in conjugate addition reaction, known as Michael-type reactions.[1a,1c] For example, a Michael addition of ß,ß-disubsti- tuted nitroalkenes with malonate derivatives was reported by Song, in which the otherwise unreactive substrate systems were demonstrated as on-water catalytic reaction.[1a]
An effective promotion for nucleophilic addition is via the use of Brønsted or Lewis acids to activate electrophiles.[3] How- ever, the activating ability of Brønsted acids is commonly weak- ened in presence of water. A few studies,[4] initiated by Kobaya- shi's pioneering work,[3e] were developed on Brønsted acid-surfactant-combined catalysts and their applications in aqueous systems. The surfactant segments assemble into micelles as microreactor, whose hydrophobic cores trap the labile sub- strates and exclude water molecules,[3b,5] while the micelle exte- rior remains hydrated, and allows the permeation of protons for substrate activation.[6] This surfactant-combined strategy has also been extended to solid Brønsted acids.
The hydrophobic assembly is probably caused by fluorocar- bon chain except hydrocarbon chain. Perfluoroalkyl chains have a stronger hydrophobic character than hydrocarbon chains due to their greater molecular cross-sectional area.[7] A classic exam- ple is -C8F17, which is applied in fluorous biphasic media for fluorous catalyst recycling.[8] Even relatively short -C4F9 chains exhibit significant hydrophobicity. Our previous research dem- onstrated that -C4F9 hindered access of water molecules to ad- jacent SO2NHSO2 acid sites,[9] and hydrogen was impeded to bind with the negative conjugate base SO2N–SO2.[10] Similarly, in Wang's reports, pyrrolidine perfluorobutyl sulfonamide was used in aqueous catalysis.[11] On the other hand, perfluoroalkyl sulfonylimides, (RFSO2)2NH, known as nitrogen acids, is widely applied in catalytic field instead of SO3H analogues to practice challenging tasks because of their strong acidity.[3c,3d,9] Gener- ally, compounds with high fluorine content (> 60 %) strongly prefer to go into fluorous phase in fluorocarbon–hydrocarbon systems.[12] These distinct properties could provide water- shielded microenvironments for aqueous reactions. However, too long fluorocarbon chain will inevitably lead to low miscibility of organic and fluorous substrates, weaken the interaction of organic substrates, and increas the cost.
Herein, in the structure of designed sulfonimide catalyst 1a/ 1b, fluorine content and acid density are well balanced by split- ting long perfluoroalkyl chain into two relatively short perfluoroalkyl chains, attached to respective imide Brønsted acid sites (Scheme 1). This method adjusts fluorine content at relative low level with elevated acid density per molecule. The acidity is examined by using the Hammett acidity function, the 31P NMR shift of Et3P=O probe, and conductance titration. The binary acid catalyst has been applied in water to promote the nucleo- philic addition (the Friedel–Crafts reactions). The substrates used herein were indoles, a widely distributed core structure in nature, and there is a large number of applications of synthetic indoles as pharmaceuticals and agrochemicals.[3a,13] With the use of 1a, Friedel–Crafts alkylation of indoles in water was easily performed, via 1,4-addition with ß-monosubstituted vinyl ket- ones and condensation with aldehydes, respectively. The prod- ucts are isolated without column chromatography or recrystalli- zation. The aqueous solution containing 1a is recyclable at least three runs. Furthermore, typically poor-reactive ß,ß-disubsti- tuted vinyl ketones were also activated by the strong acidity of the catalyst, giving medium to good product yields.
Results and Discussion
Catalyst Design
Compounds 1a/1b comprise two N-H groups, respectively at- tached to perfluoroalkyl groups (Scheme 1). Perfluoroalkyl groups bring some advantages of stabilizing the anionic charge of the conjugate bases due to its electron-withdrawing effect. Furthermore, there is an extensive delocalization system over N, O and phenyl ring, which conspire to increase the propensity of dissociation of N–H bonds, largely improving the intrinsic acidity and decreases the dependence on external solvation.[14] An example is the analogue (CF3SO2)2NH, whose pKa values were 2.8, 2.7 and 2.1, respectively in H2O, MeOH, and DMSO. The values demonstrate the effect of the two factors on high acidity.[15] Compounds 1a/1b is thus reasoned to serve as strong acid.
1b was given by acidification of 1b·2Na, but decomposed when handled at ca. 55–65 °C to remove the remained solvent; whereas 1a was obtained. The result suggests that a certain linkage in 1b is more susceptible to the intrinsic acidity at higher temperature than that in 1a. The most likely linkage is -CONH-, which is possibly decomposed by the strong acidity, but stabilized by -C4F9 in 1a. It is inferred that the nonpolar segments, including -C4F9 groups and phenyl ring, aggregate to protect -CONH- group. Especially in the presence of water, due to the hydrophobic hydration effect, the aggregation splits each other's H-bond association of the water molecules near hydrophobic segments, and the bulk water thus hardly en- croaches -CONH- group.[6a,16] The similar aggregation much less efficiently happens to the shorter -CF3 of 1b. 1a is thus a stable amphiphile.
A simple Tyndall effect trial was conducted to confirm the formation of the hydrophobic aggregation (Figure 1a). The Tyn- dall effect is often used as a measurement of a colloid, and the intensity of the effect is proportional to the mean volume of the particles. In our trial, obvious Tyndall phenomenon hap- pened to 1a solution, showing the nano-level aggregation ex- isted. As a comparison, no similar phenomenon was observed for CF3SO3H solution. Furthermore, TEM images of 1a aqueous solution is shown in Figure 1(b). A large amount of nano-size aggregates was observed, with a size of 15–20 nm. Their rela- tively small diameters implied a stacked hydrophobic segment among molecules.
Ethyl 2-(2,6-dichlorophenyl)sulfanylacetateCatalog No.:AA01FAQX CAS No.:1097071-89-0 MDL No.:MFCD11641651 MF:C10H10Cl2O2S MW:265.1562 |
(4-chlorophenyl)[3-(difluoromethoxy)phenyl]methanoneCatalog No.:AA01EJVO CAS No.:1097076-61-3 MDL No.:MFCD11934980 MF:C14H9ClF2O2 MW:282.6699 |
6-(2,6-difluorobenzoyl)-2,3-dihydro-1,4-benzodioxineCatalog No.:AA01BHKO CAS No.:1097078-33-5 MDL No.:MFCD13660710 MF:C15H10F2O3 MW:276.2349 |
Dipotassium ({3-[(sulfanidylmethanethioyl)-amino]propyl}carbamothioyl)sulfanideCatalog No.:AA01F3UE CAS No.:109708-90-9 MDL No.:MFCD00955699 MF:C5H8K2N2S4 MW:302.5870 |
4-[(3-methylphenyl)methoxy]benzene-1-sulfonamideCatalog No.:AA01AGJT CAS No.:1097083-50-5 MDL No.:MFCD12569426 MF:C14H15NO3S MW:277.3388 |
1-(3-Chlorophenyl)-6-methyl-1h,4h,5h-pyrazolo[3,4-d]pyrimidin-4-oneCatalog No.:AA019XDD CAS No.:1097095-35-6 MDL No.:MFCD11544235 MF:C12H9ClN4O MW:260.6791 |
4-(benzyloxy)-3-bromobenzene-1-sulfonyl chlorideCatalog No.:AA01B6JH CAS No.:1097095-65-2 MDL No.:MFCD12568335 MF:C13H10BrClO3S MW:361.6387 |
1-acetamidocycloheptane-1-carboxylic acidCatalog No.:AA019KUC CAS No.:1097102-48-1 MDL No.:MFCD13633884 MF:C10H17NO3 MW:199.2469 |
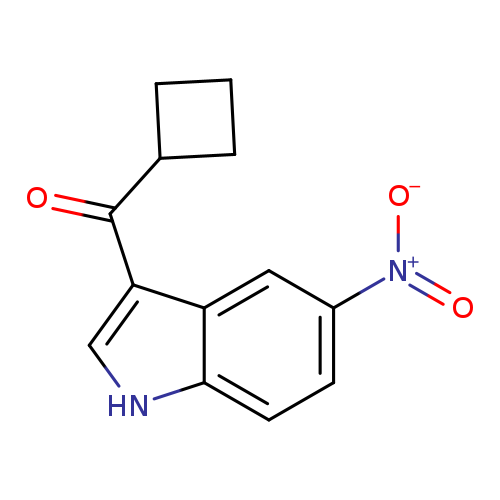
3-cyclobutanecarbonyl-5-nitro-1H-indoleCatalog No.:AA01AC52 CAS No.:1097117-03-7 MDL No.:MFCD11545710 MF:C13H12N2O3 MW:244.2460 |

2-[(2,4-difluorophenyl)sulfanyl]-2-phenylacetic acidCatalog No.:AA01AB3D CAS No.:1097125-21-7 MDL No.:MFCD12569683 MF:C14H10F2O2S MW:280.2898 |
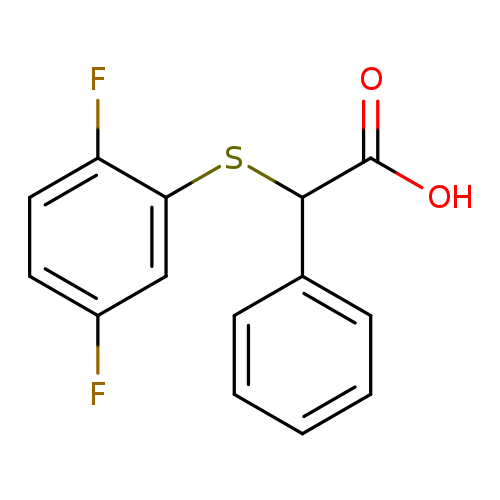
2-[(2,5-difluorophenyl)sulfanyl]-2-phenylacetic acidCatalog No.:AA01AB60 CAS No.:1097125-22-8 MDL No.:MFCD12569684 MF:C14H10F2O2S MW:280.2898 |
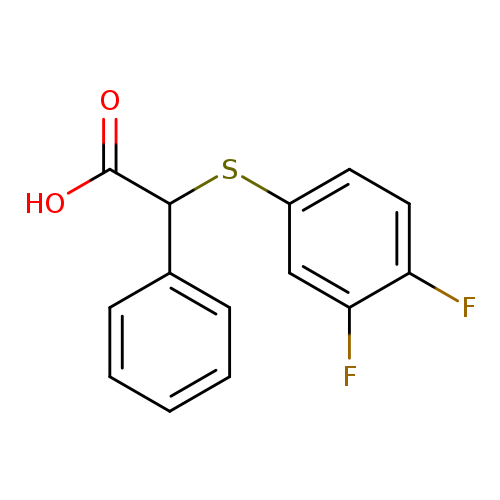
2-[(3,4-difluorophenyl)sulfanyl]-2-phenylacetic acidCatalog No.:AA01AB4C CAS No.:1097125-23-9 MDL No.:MFCD12654648 MF:C14H10F2O2S MW:280.2898 |
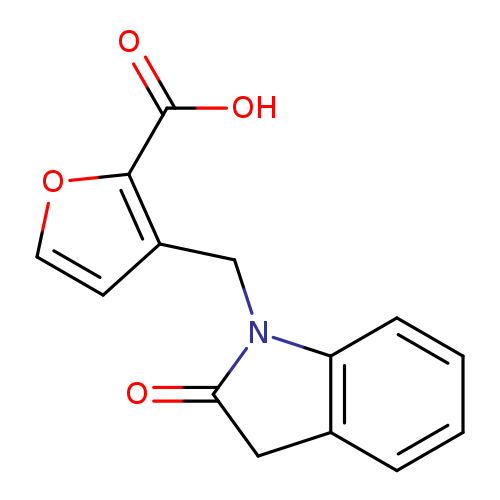
3-[(2-Oxo-2,3-dihydro-1h-indol-1-yl)methyl]furan-2-carboxylic acidCatalog No.:AA01AAKW CAS No.:1097140-95-8 MDL No.:MFCD12652091 MF:C14H11NO4 MW:257.2414 |
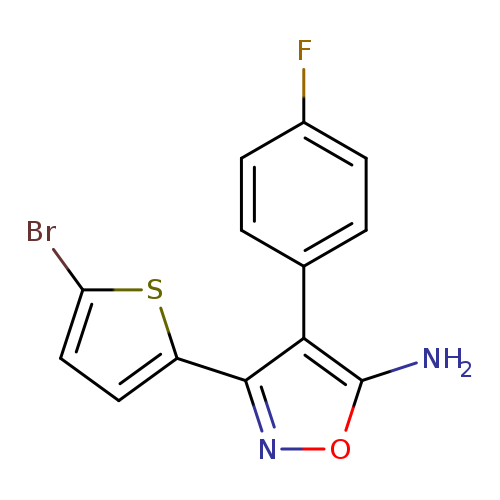
3-(5-bromothiophen-2-yl)-4-(4-fluorophenyl)-1,2-oxazol-5-amineCatalog No.:AA01AHYI CAS No.:1097153-77-9 MDL No.:MFCD12653048 MF:C13H8BrFN2OS MW:339.1828 |
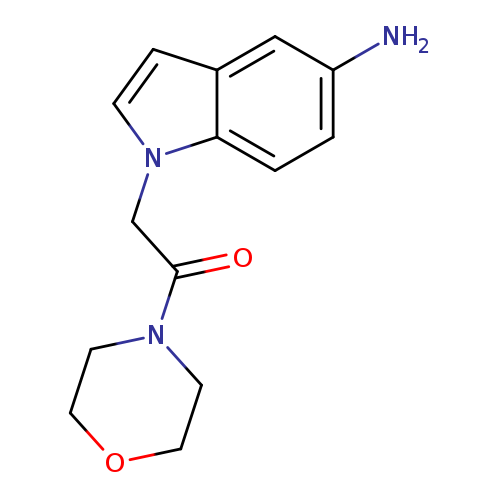
2-(5-amino-1H-indol-1-yl)-1-(morpholin-4-yl)ethan-1-oneCatalog No.:AA01A7W3 CAS No.:1097161-86-8 MDL No.:MFCD12654343 MF:C14H17N3O2 MW:259.3037 |
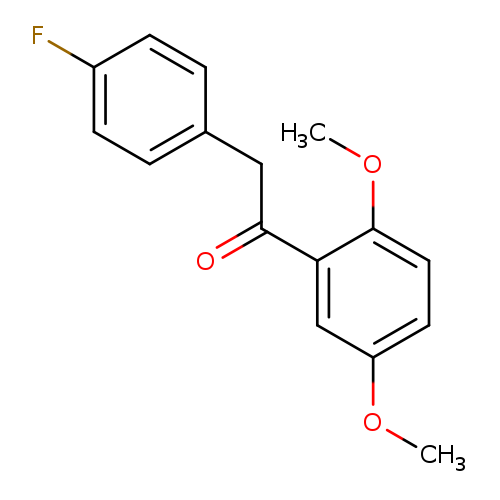
1-(2,5-Dimethoxyphenyl)-2-(4-fluorophenyl)ethanoneCatalog No.:AA019EJP CAS No.:1097164-22-1 MDL No.:MFCD13660606 MF:C16H15FO3 MW:274.2869 |
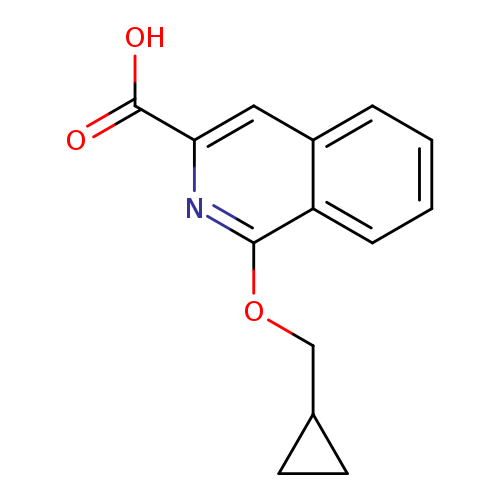
1-(cyclopropylmethoxy)isoquinoline-3-carboxylic acidCatalog No.:AA01AIAX CAS No.:1097166-34-1 MDL No.:MFCD11546080 MF:C14H13NO3 MW:243.2579 |
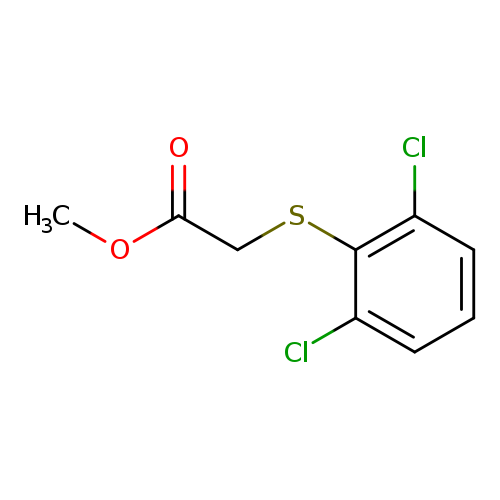
Methyl 2-(2,6-dichlorophenyl)sulfanylacetateCatalog No.:AA01FAGV CAS No.:1097167-77-5 MDL No.:MFCD11641638 MF:C9H8Cl2O2S MW:251.1296 |
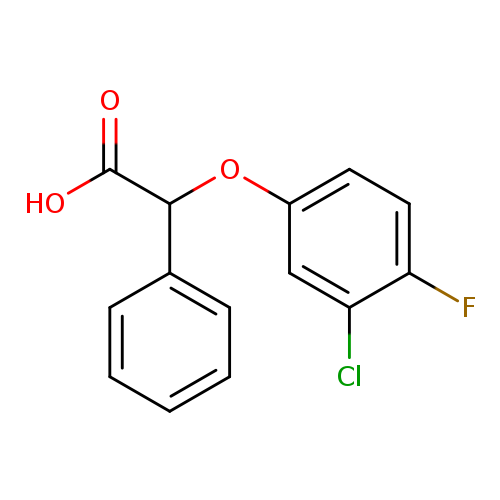
2-(3-Chloro-4-fluorophenoxy)-2-phenylacetic acidCatalog No.:AA01AGIN CAS No.:1097168-10-9 MDL No.:MFCD12542597 MF:C14H10ClFO3 MW:280.6788 |
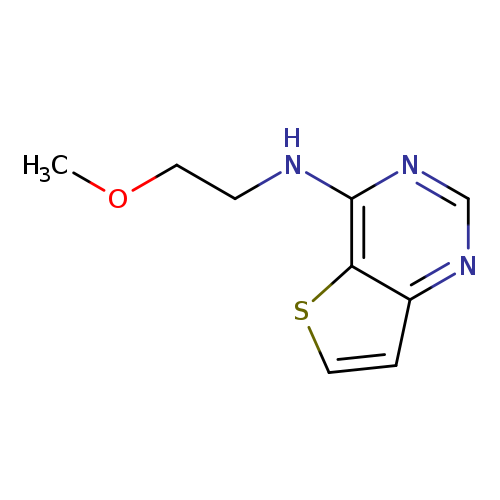
N-(2-methoxyethyl)thieno[3,2-d]pyrimidin-4-amineCatalog No.:AA01C45C CAS No.:1097168-21-2 MDL No.:MFCD11641715 MF:C9H11N3OS MW:209.2681 |
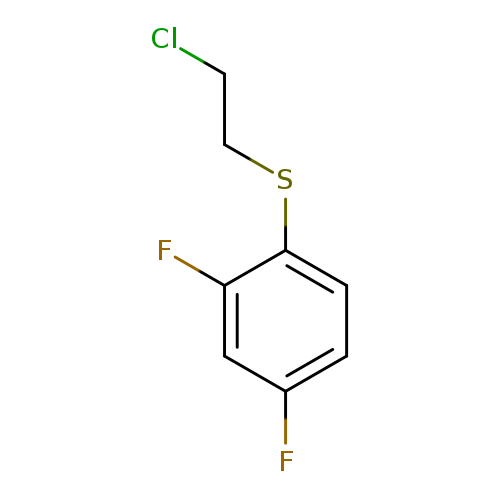
1-[(2-chloroethyl)sulfanyl]-2,4-difluorobenzeneCatalog No.:AA01DQ8Y CAS No.:1097168-39-2 MDL No.:MFCD11641763 MF:C8H7ClF2S MW:208.6560 |
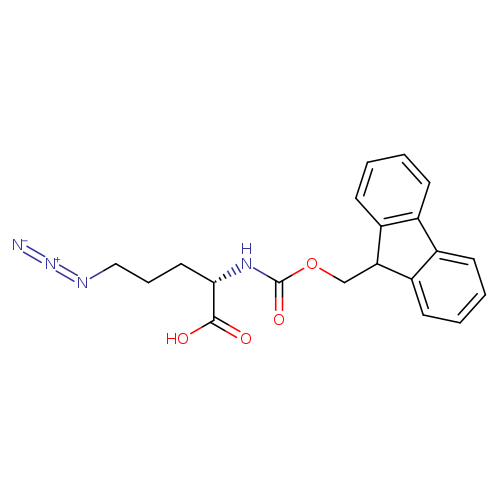
Fmoc-l-delta-azidoornithineCatalog No.:AA003CK1 CAS No.:1097192-04-5 MDL No.:MFCD11052921 MF:C20H20N4O4 MW:380.3972 |
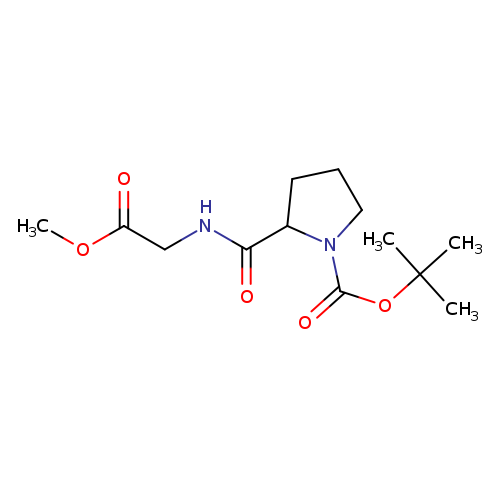
1-Boc-2-(2-methoxy-2-oxoethylcarbamoyl)pyrrolidineCatalog No.:AA009LNQ CAS No.:1097194-13-2 MDL No.:MFCD01046139 MF:C13H22N2O5 MW:286.3242 |
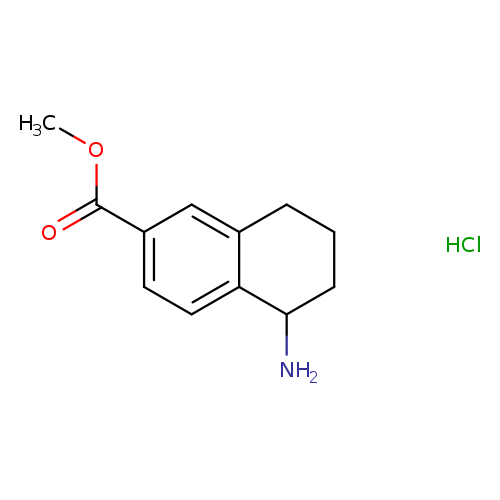
Methyl 5-amino-5,6,7,8-tetrahydronaphthalene-2-carboxylate hydrochlorideCatalog No.:AA00HBI4 CAS No.:1097196-62-7 MDL No.:MFCD22571285 MF:C12H16ClNO2 MW:241.7139 |
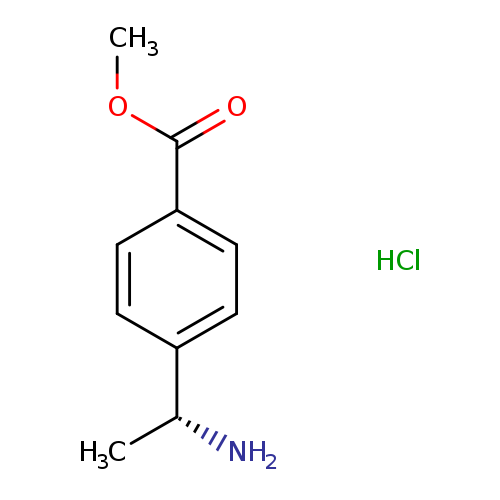
(R)-4-(1-Amino-ethyl)-benzoic acid methyl ester hydrochlorideCatalog No.:AA00928U CAS No.:1097196-96-7 MDL No.:MFCD12910780 MF:C10H14ClNO2 MW:215.6767 |
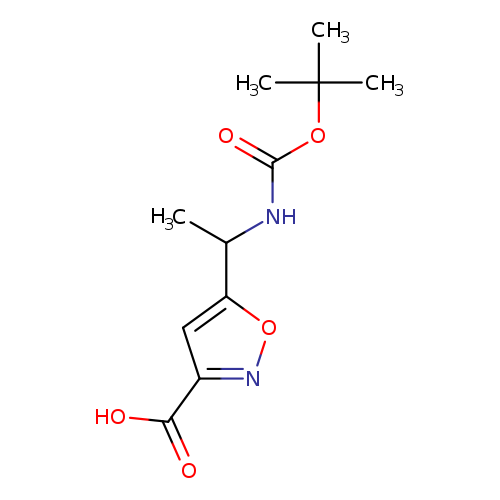
5-(1-((tert-Butoxycarbonyl)amino)ethyl)isoxazole-3-carboxylic acidCatalog No.:AA0099AC CAS No.:1097257-19-6 MDL No.:MFCD24532820 MF:C11H16N2O5 MW:256.2551 |
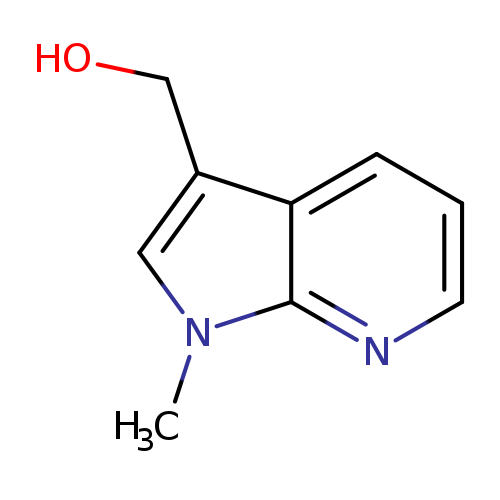
(1-Methyl-1h-pyrrolo[2,3-b]pyridin-3-yl)methanolCatalog No.:AA00HBID CAS No.:1097323-08-4 MDL No.:MFCD21769727 MF:C9H10N2O MW:162.1885 |
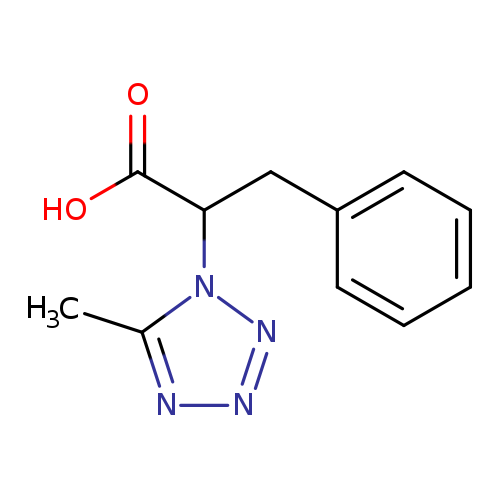
2-(5-methyl-1H-1,2,3,4-tetrazol-1-yl)-3-phenylpropanoic acidCatalog No.:AA01AJA4 CAS No.:109734-83-0 MDL No.:MFCD19282299 MF:C11H12N4O2 MW:232.2386 |
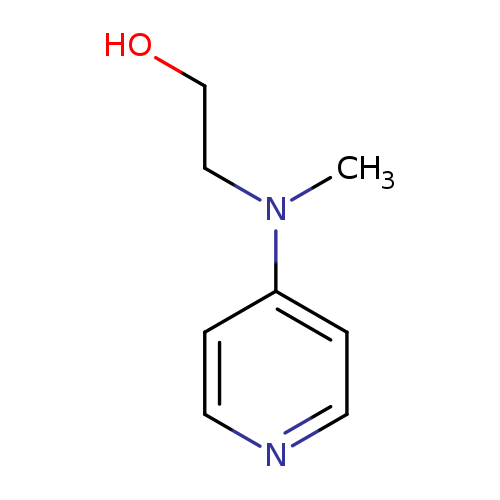
2-(Methyl(pyridin-4-yl)amino)ethanolCatalog No.:AA007SEJ CAS No.:109736-44-9 MDL No.:MFCD14588720 MF:C8H12N2O MW:152.1937 |
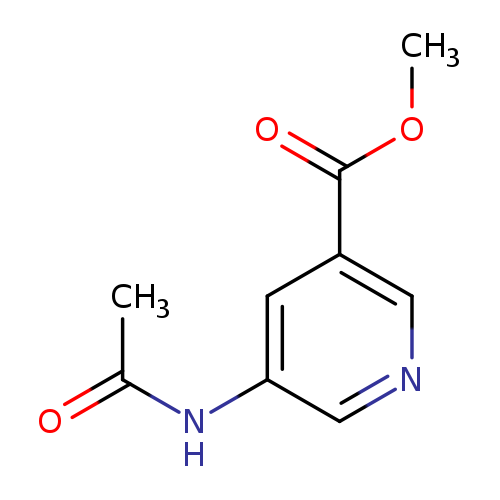
Methyl 5-acetamidopyridine-3-carboxylateCatalog No.:AA009TR6 CAS No.:109737-31-7 MDL No.:MFCD22392129 MF:C9H10N2O3 MW:194.1873 |
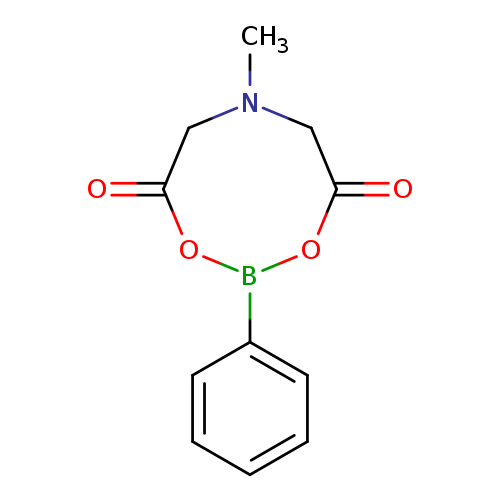
6-Methyl-2-phenyl-1,3,6,2-dioxazaborocane-4,8-dioneCatalog No.:AA003TMU CAS No.:109737-57-7 MDL No.:MFCD11215250 MF:C11H12BNO4 MW:233.0283 |
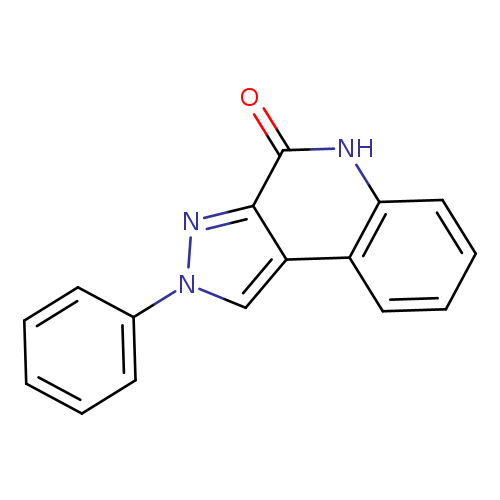
2-PHENYL-2,5-DIHYDRO-4H-PYRAZOLO[3,4-C]QUINOLIN-4-ONECatalog No.:AA009TR7 CAS No.:109740-09-2 MDL No.:MFCD07339082 MF:C16H11N3O MW:261.2780 |
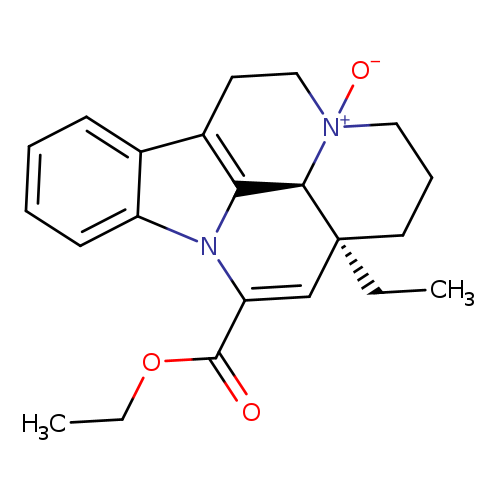
ApovincaMinic Acid Ethyl Ester N-OxideCatalog No.:AA00961N CAS No.:109741-24-4 MDL No.:MFCD30530035 MF:C22H26N2O3 MW:366.4534 |
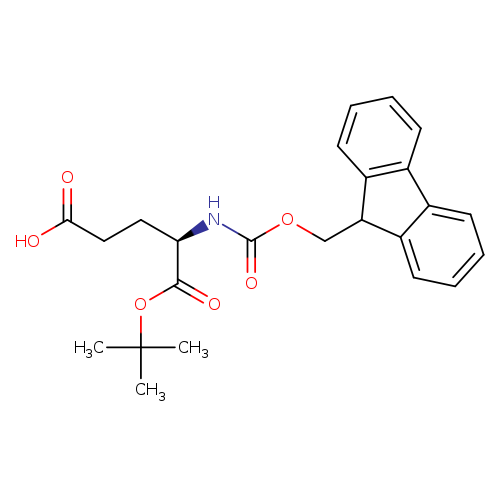
Fmoc-D-Glu-OtBuCatalog No.:AA00793W CAS No.:109745-15-5 MDL No.:MFCD00077054 MF:C24H27NO6 MW:425.4743 |
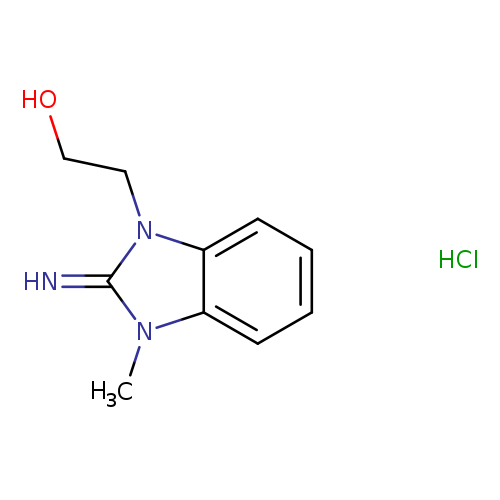
2-(2-imino-3-methyl-2,3-dihydro-1H-benzimidazol-1-yl)ethanol hydrochlorideCatalog No.:AA00J3NL CAS No.:109753-28-8 MDL No.:MFCD01455425 MF:C10H14ClN3O MW:227.6907 |
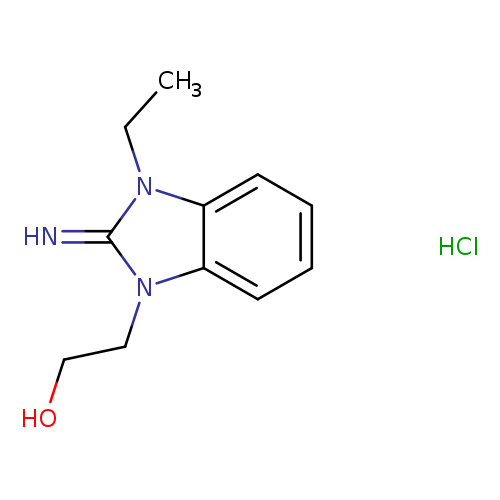
2-(3-ethyl-2-imino-2,3-dihydro-1H-benzimidazol-1-yl)ethanol hydrochlorideCatalog No.:AA00J3NM CAS No.:109753-29-9 MDL No.:MFCD01455261 MF:C11H16ClN3O MW:241.7172 |
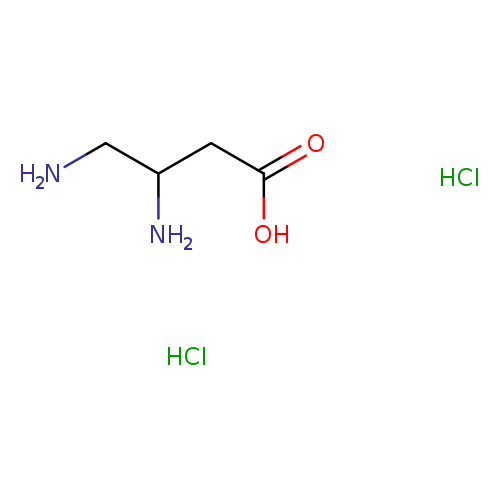
3,4-Diaminobutanoic acid dihydrochlorideCatalog No.:AA01AHYB CAS No.:109754-82-7 MDL No.:MFCD09836079 MF:C4H12Cl2N2O2 MW:191.0563 |
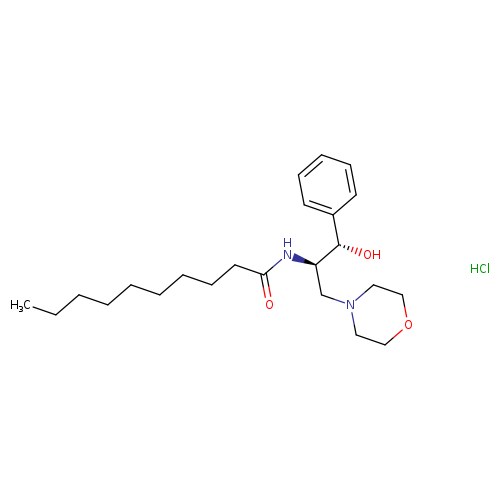
D,L-ERYTHRO-1-PHENYL-2-DECANOYLAMINO-3-MORPHOLINO-1-PROPANOL HCLCatalog No.:AA008RA3 CAS No.:109760-77-2 MDL No.:MFCD08703011 MF:C23H39ClN2O3 MW:427.0204 |
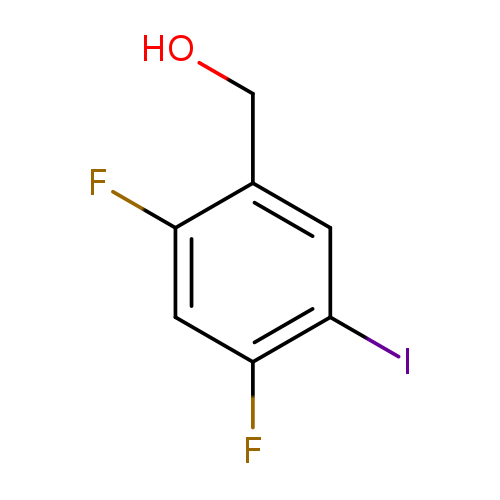
(2,4-Difluoro-5-iodo-phenyl)-methanolCatalog No.:AA0093K4 CAS No.:1097626-21-5 MDL No.:MFCD28348701 MF:C7H5F2IO MW:270.0153 |
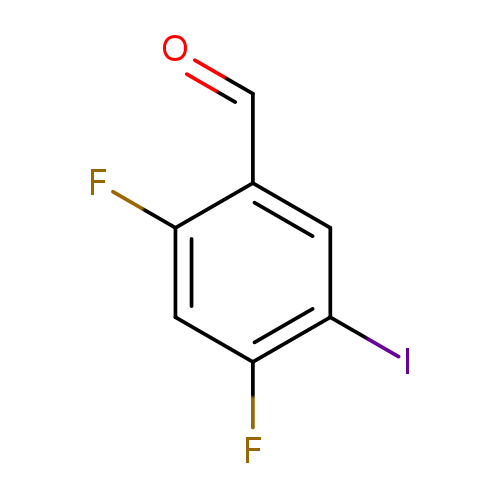
2,4-Difluoro-5-iodo-benzaldehydeCatalog No.:AA0093KR CAS No.:1097626-30-6 MDL No.:MFCD28789181 MF:C7H3F2IO MW:267.9994 |
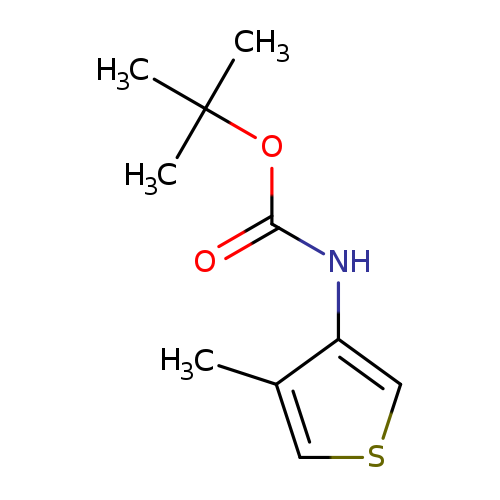
tert-butyl N-(4-methylthiophen-3-yl)carbamateCatalog No.:AA01ACAF CAS No.:1097629-78-1 MDL No.:MFCD24471163 MF:C10H15NO2S MW:213.2966 |
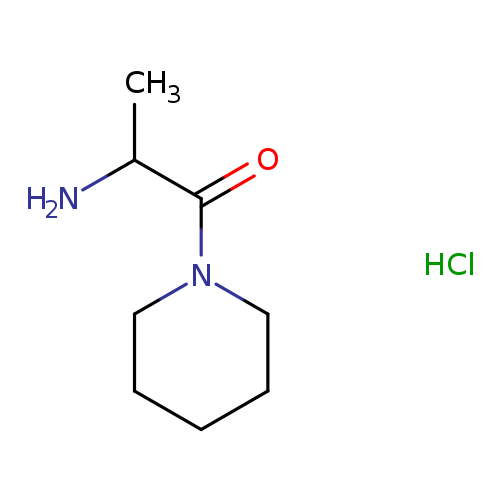
2-Amino-1-(1-piperidinyl)-1-propanonehydrochlorideCatalog No.:AA0097ZV CAS No.:1097653-04-7 MDL No.:MFCD09028974 MF:C8H17ClN2O MW:192.6864 |
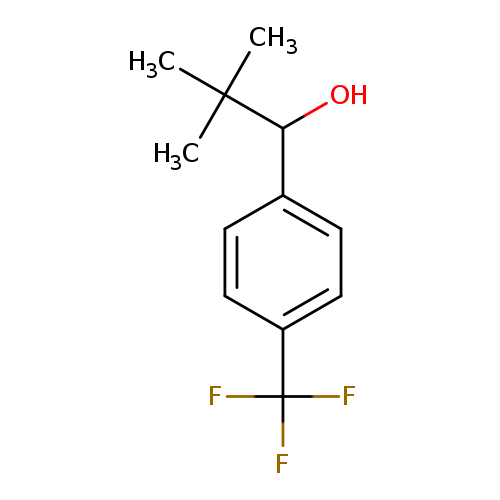
2,2-dimethyl-1-[4-(trifluoromethyl)phenyl]propan-1-olCatalog No.:AA01B1F2 CAS No.:109769-57-5 MDL No.:MFCD16316341 MF:C12H15F3O MW:232.2421 |

H-ASP-ALA-GLU-PHE-ARG-HIS-ASP-SER-GLY-TYR-GLU-VAL-HIS-HIS-GLN-LYS-LEU-VAL-PHE-PHE-ALA-GLU-ASP-VAL-GLY-SER-ASN-LYS-OHCatalog No.:AA008RK8 CAS No.:109770-29-8 MDL No.:MFCD00133074 MF: MW: |
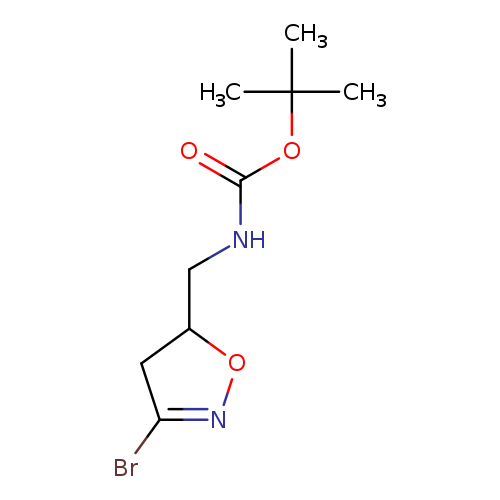
tert-Butyl ((3-bromo-4,5-dihydroisoxazol-5-yl)methyl)carbamateCatalog No.:AA003UFZ CAS No.:109770-82-3 MDL No.:MFCD08741480 MF:C9H15BrN2O3 MW:279.1310 |
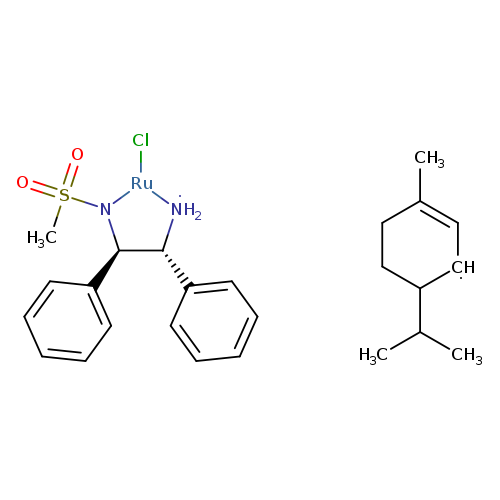
Chloro(p-cymene)[(1R,2R)-(-)-2-amino-1,2-diphenylethyl(methylsulfonylamido)]ruthenium(II)Catalog No.:AA008WJR CAS No.:1097730-63-6 MDL No.:MFCD16294973 MF:C25H36ClN2O2RuS MW:565.1535 |
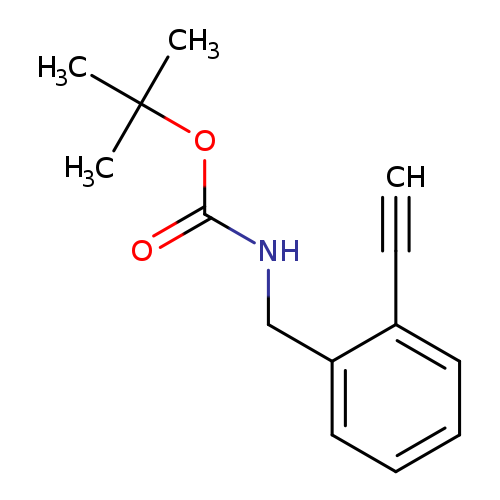
tert-Butyl 2-ethynylbenzylcarbamateCatalog No.:AA0093XX CAS No.:1097731-47-9 MDL No.:MFCD24471043 MF:C14H17NO2 MW:231.2903 |
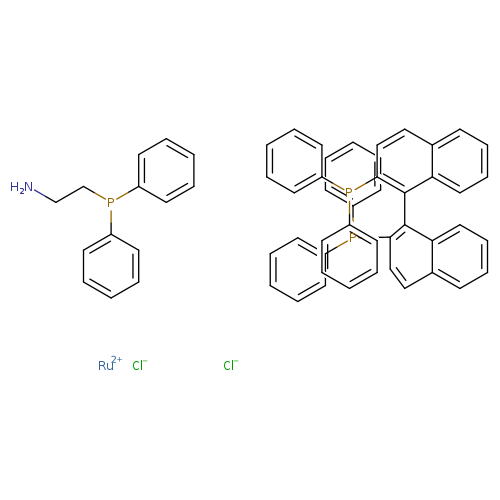
Dichloro[(R)-bis(diphenylphosphino)-1,1-binaphthyl][2-(diphenylphosphino)ethylamine]ruthenium(II)Catalog No.:AA008VJT CAS No.:1097731-98-0 MDL No.:MFCD17018812 MF:C58H48Cl2NP3Ru MW:1023.9057 |
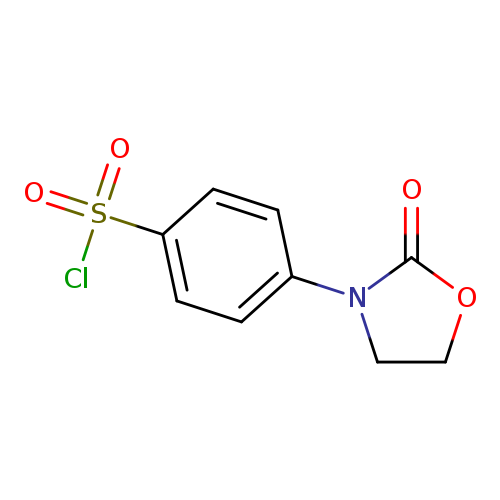
4-(2-Oxooxazolidin-3-yl)benzenesulfonyl chlorideCatalog No.:AA00VTUM CAS No.:1097732-13-2 MDL No.:MFCD14529489 MF:C9H8ClNO4S MW:261.6821 |
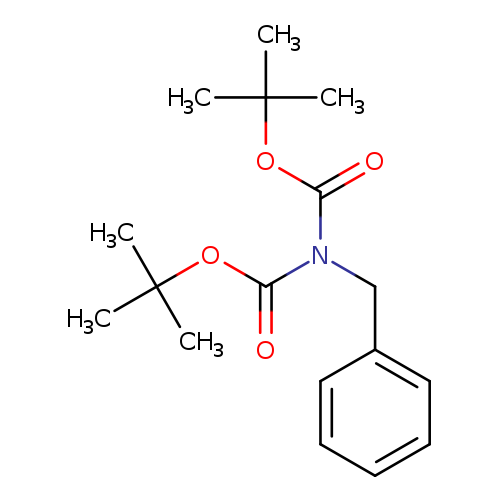
Di-tert-butyl benzylimidodicarbonateCatalog No.:AA00HBIH CAS No.:109774-57-4 MDL No.:MFCD24465674 MF:C17H25NO4 MW:307.3847 |
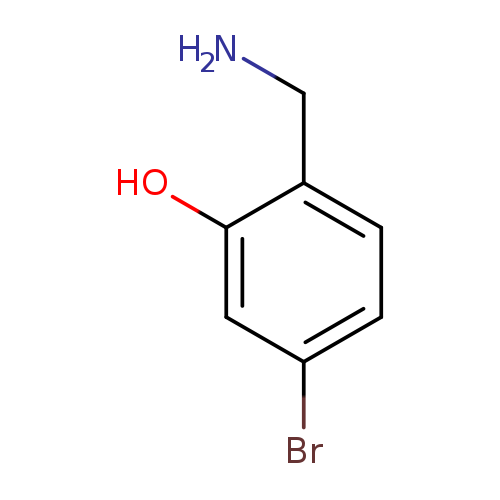
2-(Aminomethyl)-5-bromophenolCatalog No.:AA0092CG CAS No.:1097778-99-8 MDL No.:MFCD09999926 MF:C7H8BrNO MW:202.0485 |
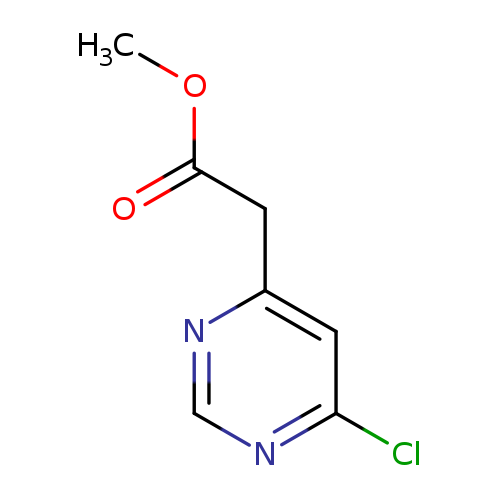
Methyl 2-(6-chloropyrimidin-4-yl)acetateCatalog No.:AA003RI5 CAS No.:1097779-00-4 MDL No.:MFCD11520447 MF:C7H7ClN2O2 MW:186.5957 |
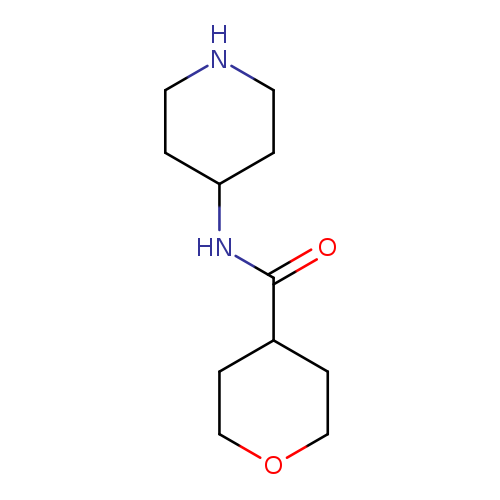
N-(piperidin-4-yl)oxane-4-carboxamideCatalog No.:AA01AGKX CAS No.:1097781-67-3 MDL No.:MFCD11642775 MF:C11H20N2O2 MW:212.2887 |
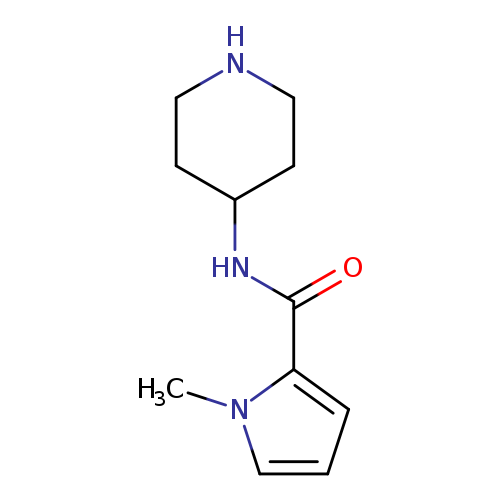
1-methyl-N-(piperidin-4-yl)-1H-pyrrole-2-carboxamideCatalog No.:AA01A8PW CAS No.:1097782-07-4 MDL No.:MFCD11642823 MF:C11H17N3O MW:207.2722 |
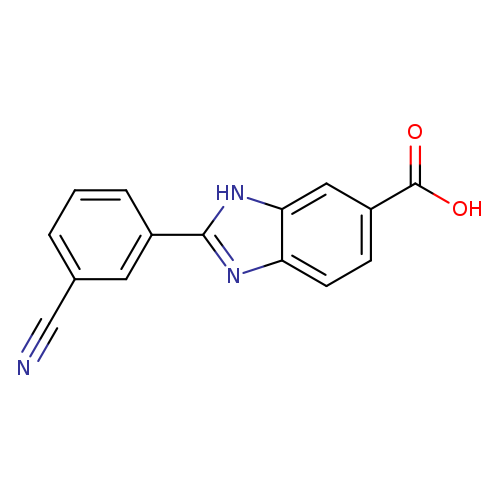
2-(3-Cyanophenyl)-1H-benzo[d]imidazole-6-carboxylic acidCatalog No.:AA01F9CL CAS No.:1097783-82-8 MDL No.:MFCD20265179 MF:C15H9N3O2 MW:263.2509 |
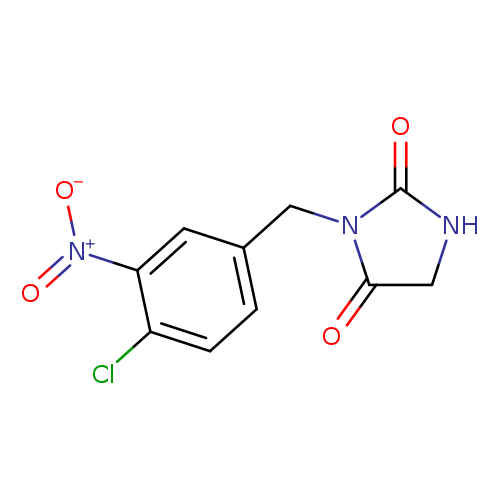
3-[(4-chloro-3-nitrophenyl)methyl]imidazolidine-2,4-dioneCatalog No.:AA01A0A2 CAS No.:1097788-31-2 MDL No.:MFCD26407868 MF:C10H8ClN3O4 MW:269.6412 |
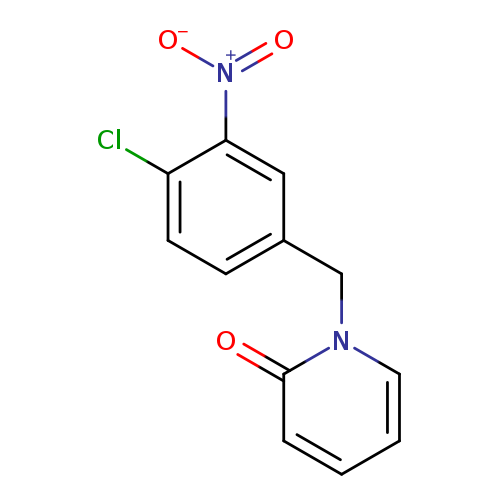
1-[(4-chloro-3-nitrophenyl)methyl]-1,2-dihydropyridin-2-oneCatalog No.:AA01A0A4 CAS No.:1097788-36-7 MDL No.:MFCD11547889 MF:C12H9ClN2O3 MW:264.6645 |
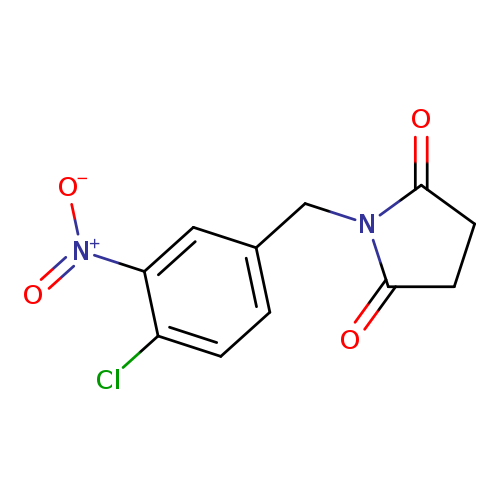
1-[(4-chloro-3-nitrophenyl)methyl]pyrrolidine-2,5-dioneCatalog No.:AA01A0A5 CAS No.:1097788-45-8 MDL No.:MFCD11547891 MF:C11H9ClN2O4 MW:268.6532 |
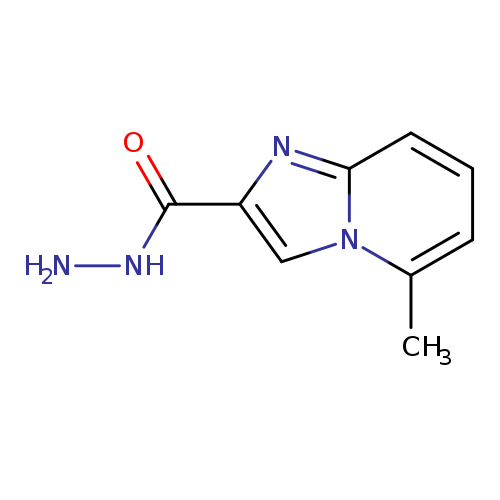
5-Methylimidazo[1,2-a]pyridine-2-carbohydrazideCatalog No.:AA009481 CAS No.:1097788-54-9 MDL No.:MFCD03419473 MF:C9H10N4O MW:190.2019 |
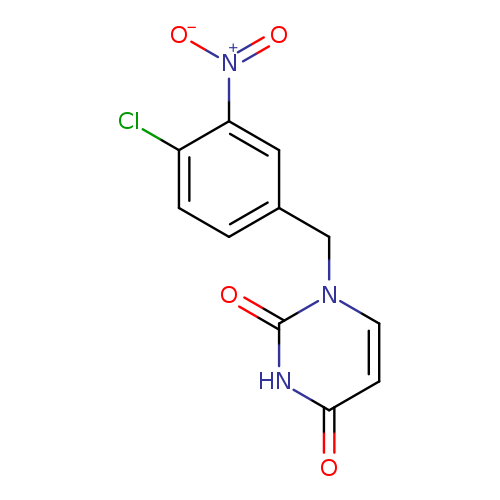
1-[(4-Chloro-3-nitrophenyl)methyl]-1,2,3,4-tetrahydropyrimidine-2,4-dioneCatalog No.:AA01A2C9 CAS No.:1097789-53-1 MDL No.:MFCD12543547 MF:C11H8ClN3O4 MW:281.6519 |
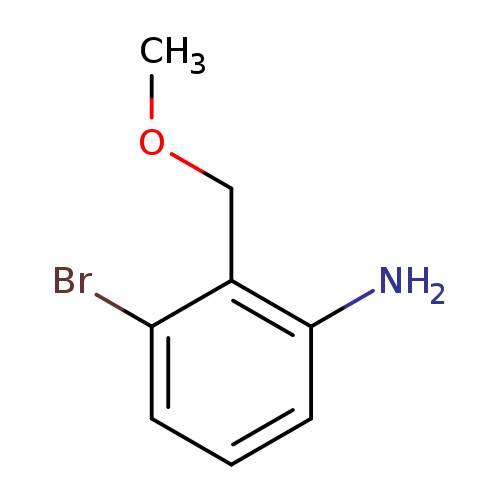
3-bromo-2-(methoxymethyl)anilineCatalog No.:AA019WLU CAS No.:1097790-44-7 MDL No.:MFCD11645709 MF:C8H10BrNO MW:216.0751 |
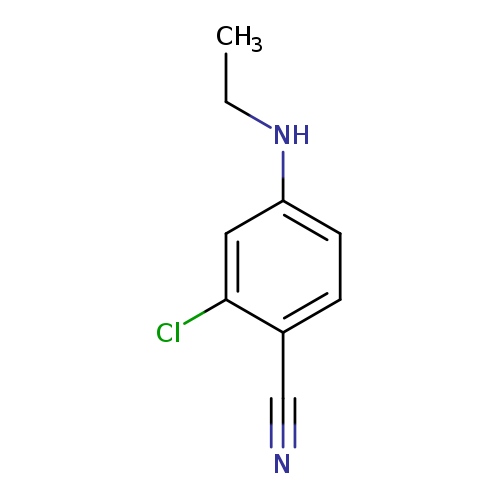
2-chloro-4-(ethylamino)benzonitrileCatalog No.:AA01A2C0 CAS No.:1097793-74-2 MDL No.:MFCD11644849 MF:C9H9ClN2 MW:180.6342 |
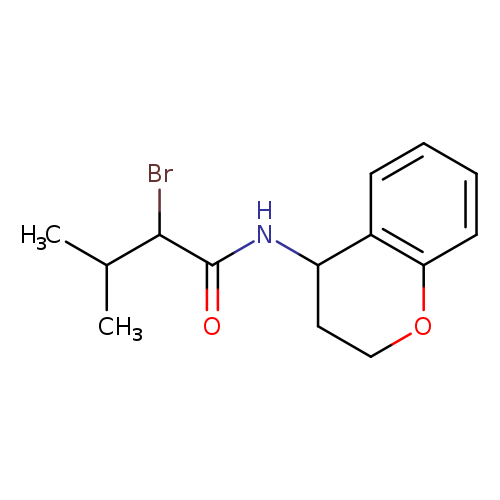
2-Bromo-n-(3,4-dihydro-2h-1-benzopyran-4-yl)-3-methylbutanamideCatalog No.:AA019KWE CAS No.:1097794-77-8 MDL No.:MFCD11547037 MF:C14H18BrNO2 MW:312.2022 |
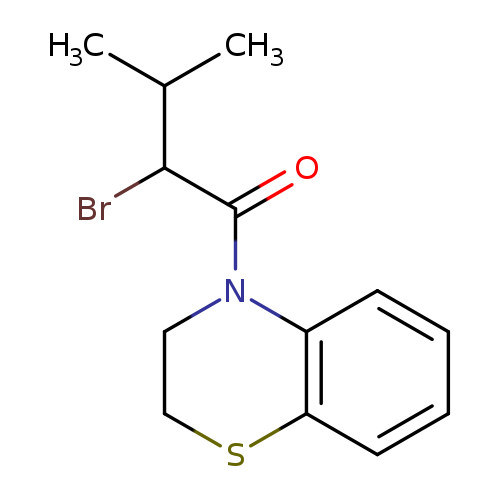
2-bromo-1-(3,4-dihydro-2H-1,4-benzothiazin-4-yl)-3-methylbutan-1-oneCatalog No.:AA019KWV CAS No.:1097794-82-5 MDL No.:MFCD11643992 MF:C13H16BrNOS MW:314.2412 |
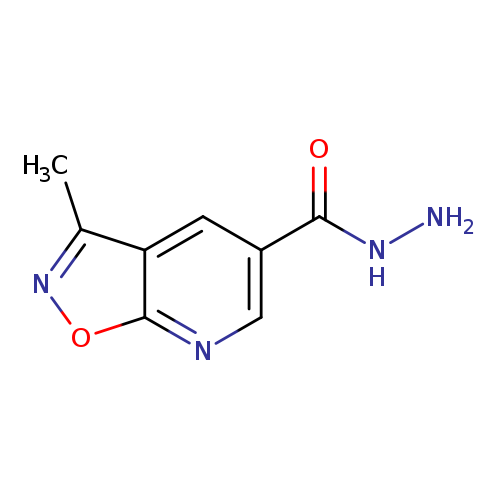
3-methyl-[1,2]oxazolo[5,4-b]pyridine-5-carbohydrazideCatalog No.:AA01AH6O CAS No.:1097794-89-2 MDL No.:MFCD11644000 MF:C8H8N4O2 MW:192.1747 |
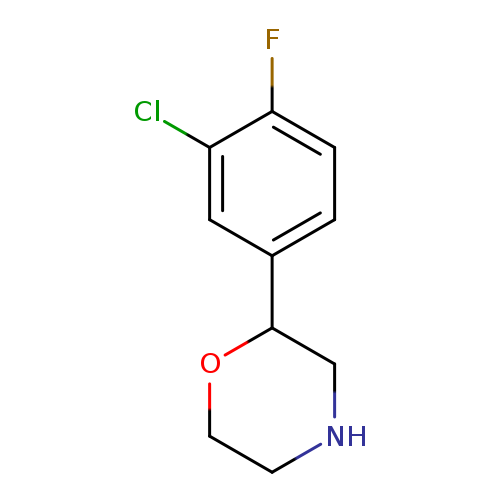
2-(3-chloro-4-fluorophenyl)morpholineCatalog No.:AA01A9RV CAS No.:1097796-68-3 MDL No.:MFCD11646191 MF:C10H11ClFNO MW:215.6518 |
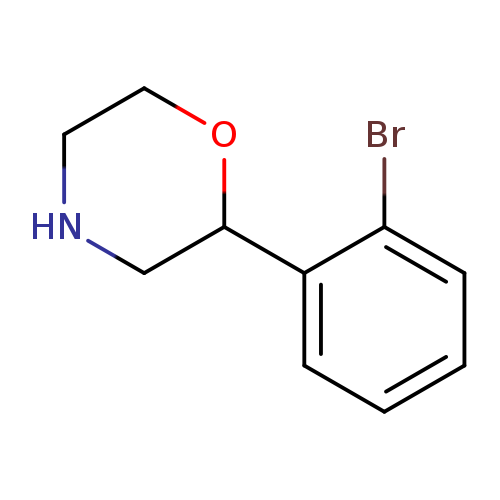
2-(2-Bromophenyl)morpholineCatalog No.:AA00HBIM CAS No.:1097796-83-2 MDL No.:MFCD11646196 MF:C10H12BrNO MW:242.1124 |
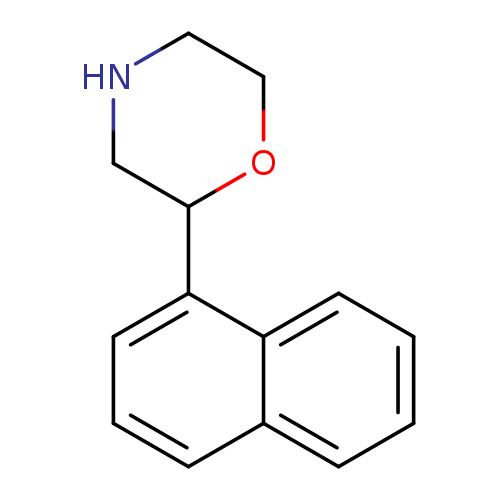
2-(NAPHTHALEN-1-YL)MORPHOLINECatalog No.:AA01EHRM CAS No.:1097797-03-9 MDL No.:MFCD11646204 MF:C14H15NO MW:213.2750 |
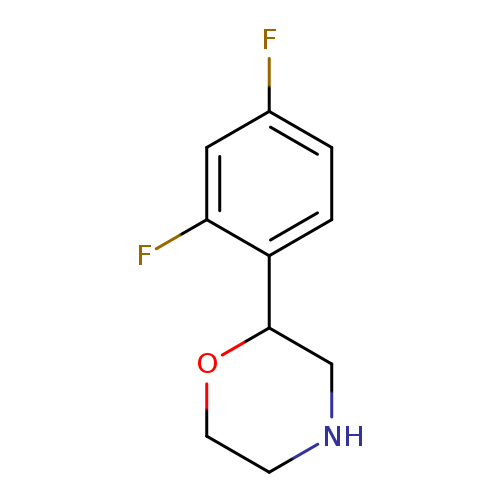
2-(2,4-difluorophenyl)morpholineCatalog No.:AA01AFQH CAS No.:1097797-34-6 MDL No.:MFCD11646218 MF:C10H11F2NO MW:199.1972 |
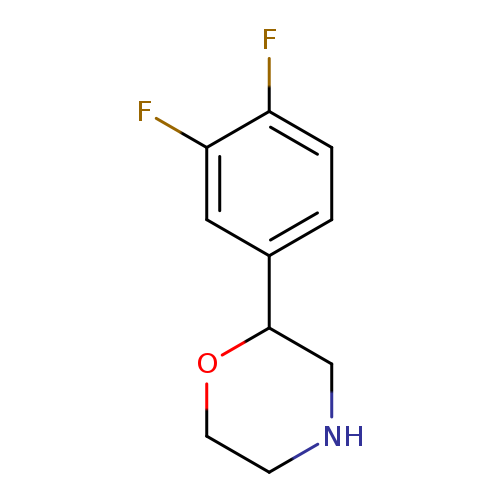
2-(3,4-difluorophenyl)morpholineCatalog No.:AA0095D2 CAS No.:1097797-38-0 MDL No.:MFCD11646219 MF:C10H11F2NO MW:199.1972 |
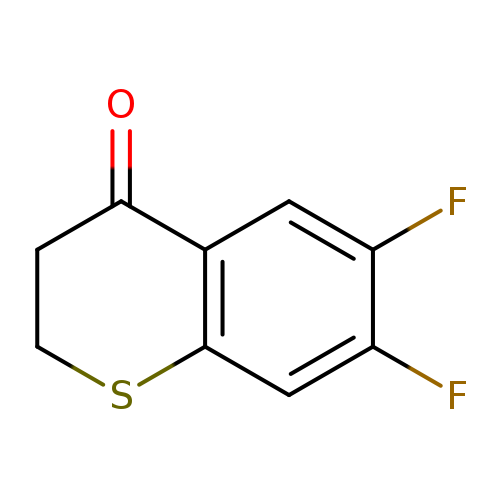
6,7-Difluoro-2,3-dihydro-4H-1-benzothiopyran-4-oneCatalog No.:AA008SGG CAS No.:1097802-78-2 MDL No.:MFCD11642456 MF:C9H6F2OS MW:200.2051 |
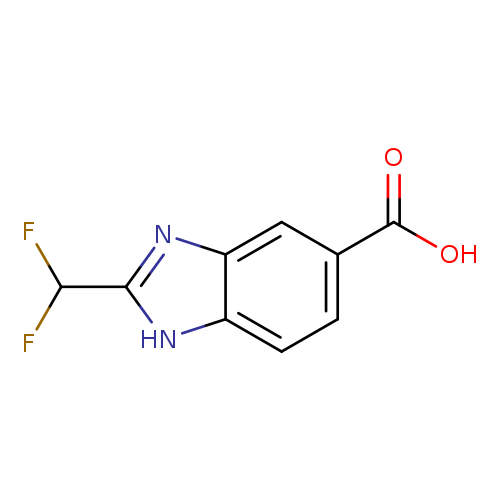
2-(Difluoromethyl)-1H-benzo[d]imidazole-5-carboxylic acidCatalog No.:AA01A9VX CAS No.:1097804-95-9 MDL No.:MFCD11645380 MF:C9H6F2N2O2 MW:212.1529 |
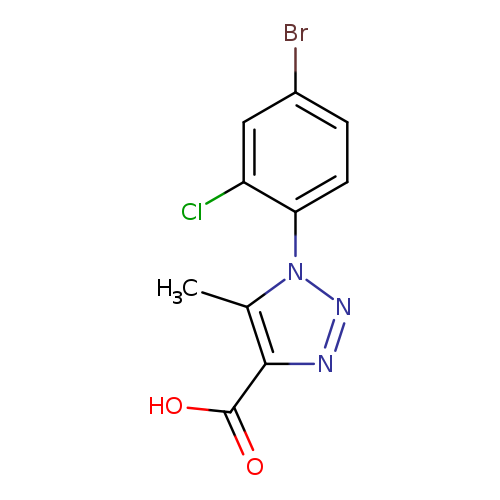
1-(4-Bromo-2-chlorophenyl)-5-methyl-1H-1,2,3-triazole-4-carboxylic acidCatalog No.:AA01AH6T CAS No.:1097809-53-4 MDL No.:MFCD11644681 MF:C10H7BrClN3O2 MW:316.5385 |
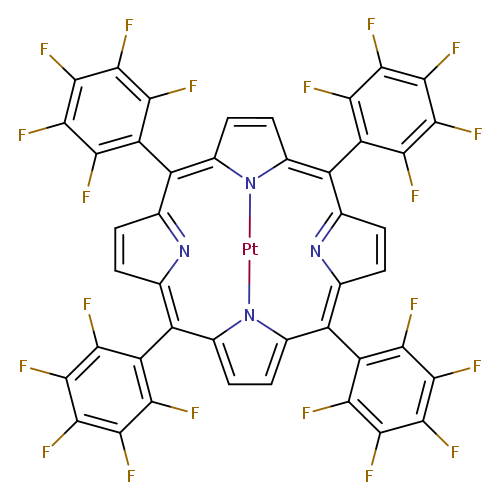
5,10,15,20-Tetrakis(pentafluorophenyl)-21h,23h-porphine, platinum(ii)Catalog No.:AA00HBIN CAS No.:109781-47-7 MDL No.:MFCD00672302 MF:C44H8F20N4Pt MW:1167.6072 |
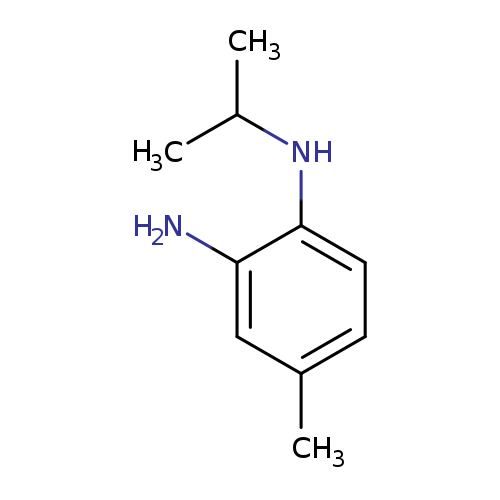
4-methyl-1-N-(propan-2-yl)benzene-1,2-diamineCatalog No.:AA019VC3 CAS No.:1097810-17-7 MDL No.:MFCD11645461 MF:C10H16N2 MW:164.2474 |
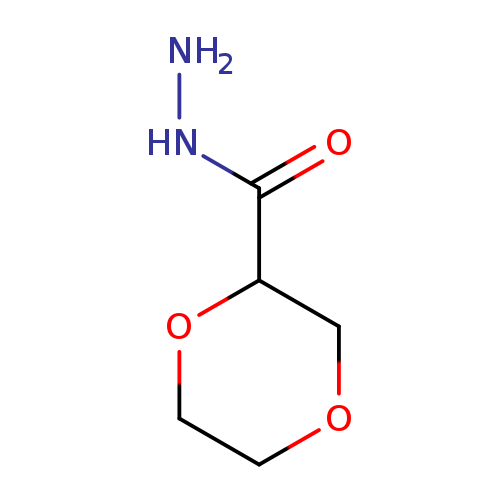
1,4-Dioxane-2-carbohydrazideCatalog No.:AA019T5H CAS No.:1097812-58-2 MDL No.:MFCD11645181 MF:C5H10N2O3 MW:146.1445 |