2020-03-08 12:01:41
Bingmeng Li a, Dan Shu a, Ruifang Wang a, Lanlan Zhai a, Yuye Chai a, Yunjun Lan a, Hongwei Cao b, Chao Zou
1.Introduction
Since the 1970s, the energy and environmental problems have gradually aroused people’s attention. Therefore, people began to exploit renewable energy sources and improve energy efficiency. At present, the use of phase change materials (PCMs) is an effective way to store latent heat in the form of phase change enthalpy [1e4]. As one of the PCMs, PEG has been widely investigated for thermal storage due to its suitable phase change temperature, high latent heat capacity and high thermal and chemical stability after long- term utility period [2,5,6]. However, the leakage in the melting process has limited its applicability in energy storage [6e8].
One of the common solutions is to develop various supporting matrixes, such as polymers [7,9] and inorganic porous materials [6,10e17] to prevent the leakage of melted PEG. PEG/inorganic porous materials composites for energy storage have drawn great interest of researchers, since their thermal properties were better than those of PEG/polymer composites [18]. PEG/inorganic porous materials are usually prepared by physical methods such as ab- sorption blending, and impregnation [6,10e14,16,19]. Karaman et al. [15] prepared a form-stabilized PEG/diatomite composite PCM by vacuum impregnation method, and 50 wt% PEG was incorpo- rated into the pores of diatomite without any seepage. The melting
and freezing latent heats of the composite PCM were 87.09 J/g and 82.22 J/g, respectively. Zhang et al. [16] prepared PEG/silica sheets (GS) PCM by mixing GS with PEG at 70 ○C for 4 h. The highest enthalpy of the composite was 136.3 J/g and the mass fraction of PEG was 80 wt%. Wang et al. [6] prepared PEG/silica fume (SF) composites by physically blending after a series of complex pre- treatment of SF. The highest PEG loading was 47.9% and the highest value of enthalpy was 81.3 J/g. From these reports, the values of phase change enthalpy were rather low compared with that of pure PEG, which means the energy storage capacity of these PCMs was not desirable. Since PEG is absorbed into the pores by capillary force in the above investigations, the amount of PEG absorbed is significantly confined by the absorption capability of porous materials [12]. So the energy storage capability is influenced by the absorption capability of supporting materials.
To improve the absorption capability of supporting materials for higher phase change enthalpy and better energy storage capacity of PEG/porous materials, some measures were took during the syn- theses process, such as vacuum and outgassing. It was reported by Wang [20] that graphite was dried in a vacuum oven at 70 ○C for 20 h, and heated in a furnace at 900 ○C for 50 s. Then PEG/graphite blends were prepared by physically blending for 3 h. The maximum weight percentage of PEG was 90% with the latent heat of 161.2 J/g. Tian et al. [21] mixed PEG and methanol with silica gel (SG) which was outgassed for more than 3 h. The mixture was heated slowly in a water bath at 55 ○C about 9 h to obtain shape-stabilized PEG/SG composites. The pore fillingness of the composites was 80% and the highest enthalpy was 168.84 J/g. Though the enthalpy value of the composites and the mass fraction of PEG were improved after the pretreatment of porous materials, an amount of extra energy and time were consumed to increase the absorption capability of porous materials. Moreover, it was reported that the phase change enthalpy of PEG/porous materials was quantitatively correlated with the average pore diameter and the pore geometrical shape of the supporting materials [21e23] indicating that the absorption
capability essentially depends on the pore structure of porous materials. Pretreatments including vacuum and outgassing are not effective ways to improve the absorption capability of supporting materials, as the pore structure was ignored in the above reports. So the phase change enthalpy or the energy storage capacity of PEG/ porous materials cannot be efficiently increased, no matter how PEG was impregnated into the pore by these conventional syn- theses methods. Then the applications of PEG/porous materials in latent heat storage system are still restrained [24]. To avoid the influence of the absorption capability of porous materials, it has been widely reported that PCMs were encapsulated into SiO2 shell
by sol-gel method through hydrolysis and condensation [18,25e29]. However, most of these reports focused on n-alkane PCMs whose thermal properties are totally different from those of PEG. Moreover, surfactants and catalysts were adopted which have negative effect on the encapsulation ratio and encapsulation effi- ciency [27,28]. The values of the latent heat of the PCM@SiO2 composites decreased significantly in comparison of those of the corresponding pure PCMs.
Recently, the synthesis of mesoporous oxides using PEG sur- factant as structure-directing (templating) agent has been exten- sively studied and substantially reported [30e34], in which PEG is surrounded by hydrolyzable precursors, and then encapsulated into inorganic oxide framework during the process of hydrolysis and condensation. To obtain mesoporous structure, PEG usually needs to be calcined at high temperature or washed with solvents at the end of the synthesis process of mesoporous oxides. In this paper, PEG was packaged into silica framework to prepare PEG@-SiO2 composite in the light of the synthesis method of mesoporous silica without the elimination process of PEG. The mass fraction of the packaged PEG can be increased to the desired contents by the variation of the experimental conditions. The reported preparation by physical blending costs much more time (4 h-9 h) and higher temperature (55e70 ○C), with the pretreatment of vacuum or out-gassing for more than 3 h. In our work, the whole process of the preparation is much simple to operate at low cost without any pretreatment of supporting materials. PEG plays two roles during the preparation of PEG@SiO2, energy storage material and surfac- tant. Additional surfactant and catalyst were avoided, which will decrease the phase change enthalpy as foreign substance for the crystallization of PEG. This preparation method can eliminate the negative effect on both the absorption and the encapsulation of PEG. It benefits higher mass fraction of PEG and thus higher phase change enthalpy. Moreover, To the best of our knowledge, the preparation of PEG@SiO2 inspired by the synthesis of mesoporous materials as shape-stabilized PCM for energy storage has not been reported. The morphology, microstructure and thermal properties of the PEG@SiO2 were investigated by various techniques. Higher phase change enthalpy and energy storage capability of PEG@SiO2 were illustrated. PEG@SiO2 exhibited excellent thermal stability and reliability. Therefore, the facile synthesized composite with comprehensive properties is potential to be applied in energy storage devices such as solar-thermal energy conversion and stor- age systems.
2.Experimental section
2.1.Materials
PEG with an average molecular weight of 6000 (PEG 6000), tetraethoxysilane (TEOS) (>99%, GC grade) and isopropyl alcohol (IPA, 99.9%, GC grade) were purchased from Aladdin Reagent Co (Shanghai, China). N, N-dimethyl formamide (DMF) ( 99.5%, GC grade) was obtained from Guangdong Guanghua Sci-Tech Co, Ltd. Deionized water was used throughout the experiments.
2.2.Preparation of PEG@SiO2
PEG@SiO2 was prepared based on the common synthesis method of mesoporous silica. 5.30 g PEG, 8.64 g deionized water,
3.02 g DMF and 3.02 g IPA were mixed with the molar rate PEG: H2O: DMF: IPA 0.0184:10:0.86:1.05. The solution was then stirred vigorously for 0.5 h at room temperature. Thereafter, 10 g TEOS was added to the mixture under stirring for 1 h at room temperature. Afterwards, the products were washed by absolute ethanol five times and subsequently dried at 50 ○C for a week to completely eliminate the unencapsulated PEG and residual solvent. At last, the composite was obtained. The as-made bulk composite was calcined at 500 ○C for 5 h to obtain the supporting material for comparison.
The schematic illustration of preparation is shown in Fig. 1.
2.3.Sample characterization
Morphological analysis was undertaken using SEM (FEI Nova Nano SEM 200). Energy dispersive X-ray spectroscopy (EDS) coupled with SEM was used to analyze the chemical composition. High-resolution transmission electron microscopy (HRTEM), high- angle annular dark-field (HAADF), scanning transmission electron microscopy (STEM), and EDS were performed on JEOL 2100F mi- croscope. Composition analysis was performed by EDS (oxford INCA). The samples for TEM and STEM-EDS were collected by placing a drop of sample dispersed in absolute ethanol onto carbon- film-supported copper grids. The surface areas, pore volume and pore size distribution of the calcined sample were determined at 77 K using a BELSORP-mini II instrument (Bel Japan Inc.). The calculation of pore size was performed using the Bar- retteJoynereHalenda (BJH) method applied to the adsorption data of the N2 sorption isotherms. An FT-IR spectrometer (Perkin Elmer, USA) was used to characterize the prepared composite. The crystal structures and crystallization properties were studied using wide angle X-ray diffraction (WAXD) (Bruker, D8 advance, Cu Ka radiation using a curved graphite receiving monochromate), with a step of 0.02○ at a speed of 4○/min from 10○ to 80○ at room temperature.
Differential scanning calorimetry (DSC) was carried out to measure the phase change temperature and enthalpy of the samples using Q1000 (TA Instrument, USA) at heating/cooling rate of 10 ○C/min from 10 ○C to 110 ○C in nitrogen atmosphere. The thermal analysis was performed by a thermogravimetric analysis Diamond TG-DTA/Spectrum GX (Perkin Elmer, USA) in nitrogen atmosphere and ox- ygen atmosphere, respectively. The samples were scanned over the temperature range from room temperature to 800 ○C at a heating rate of 10 ○C/min.
2-BROMO-4-(TRIFLUOROMETHOXY)BENZALDEHYDECatalog No.:AA003855 CAS No.:1114808-87-5 MDL No.:MFCD09835093 MF:C8H4BrF3O2 MW:269.0154 |
1-(2-Cyanophenyl)piperazineCatalog No.:AA003CRF CAS No.:111373-03-6 MDL No.:MFCD00040793 MF:C11H13N3 MW:187.2410 |
3-(Ethylamino)pyrrolidineCatalog No.:AA003I5D CAS No.:111390-22-8 MDL No.:MFCD00191357 MF:C6H14N2 MW:114.1888 |
3-Ethyl 5-methyl 2-((2-aminoethoxy)methyl)-4-(2-chlorophenyl)-6-methyl-1,4-dihydropyridine-3,5-dicarboxylate benzenesulfonateCatalog No.:AA003NRB CAS No.:111470-99-6 MDL No.:MFCD00887594 MF:C26H31ClN2O8S MW:567.0509 |
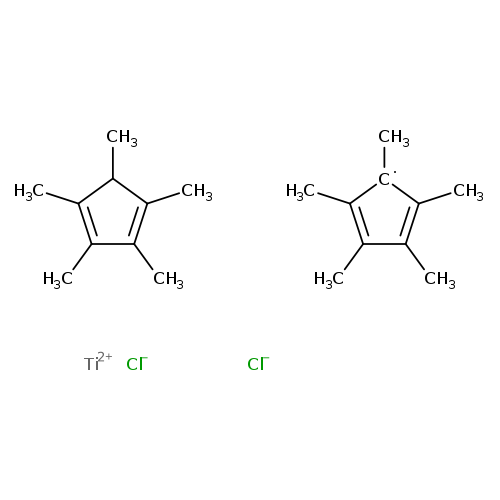
Dichlorobis(pentamethylcyclopentadienyl)titaniumCatalog No.:AA003O91 CAS No.:11136-36-0 MDL No.:MFCD00049153 MF:C20H30Cl2Ti MW:389.2252 |
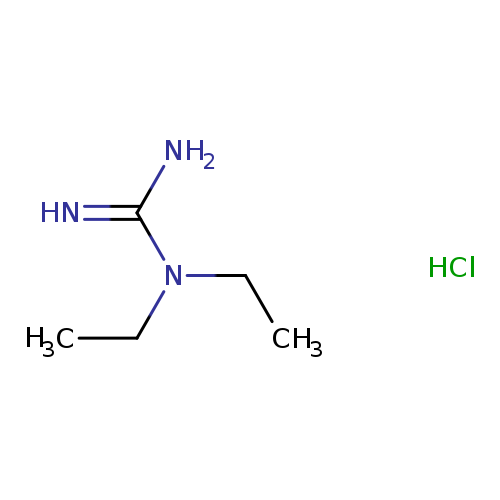
1,1-Diethylguanidine hydrochlorideCatalog No.:AA003QQV CAS No.:1114-39-2 MDL No.:MFCD00035042 MF:C5H14ClN3 MW:151.6378 |
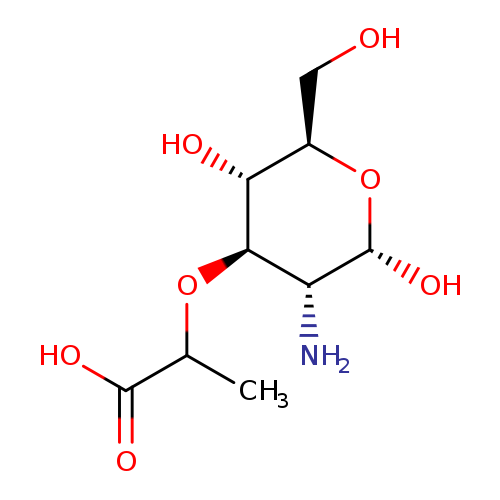
(R)-2-(((2R,3R,4R,5R)-2-Amino-4,5,6-trihydroxy-1-oxohexan-3-yl)oxy)propanoic acidCatalog No.:AA003S3X CAS No.:1114-41-6 MDL No.:MFCD00213429 MF:C9H17NO7 MW:251.2338 |
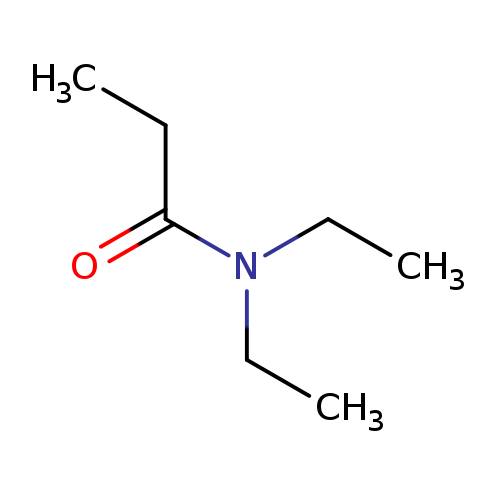
N,N-DiethylpropionamideCatalog No.:AA003SH8 CAS No.:1114-51-8 MDL No.:MFCD00009302 MF:C7H15NO MW:129.2001 |
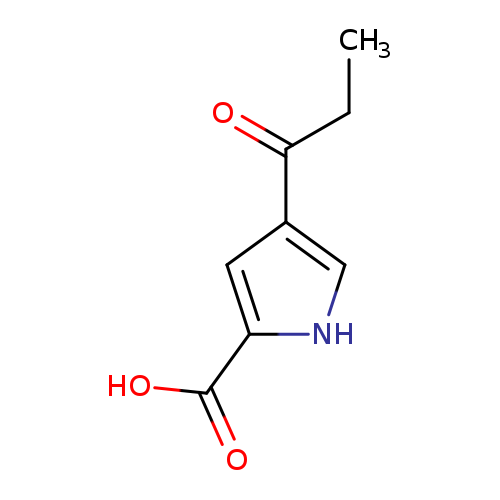
4-Propionyl-1H-pyrrole-2-carboxylic acidCatalog No.:AA007AH8 CAS No.:111468-94-1 MDL No.:MFCD06200986 MF:C8H9NO3 MW:167.1620 |
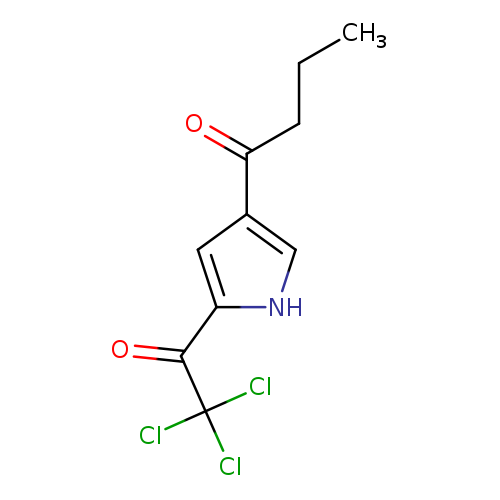
1-(5-(2,2,2-Trichloroacetyl)-1H-pyrrol-3-yl)butan-1-oneCatalog No.:AA007AHA CAS No.:111468-91-8 MDL No.:MFCD03001293 MF:C10H10Cl3NO2 MW:282.5509 |
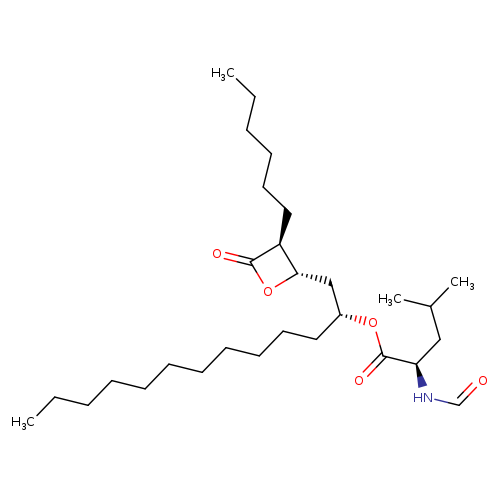
(S,R,S,S)-OrlistatCatalog No.:AA007AHC CAS No.:111466-63-8 MDL No.:MFCD23160335 MF:C29H53NO5 MW:495.7348 |
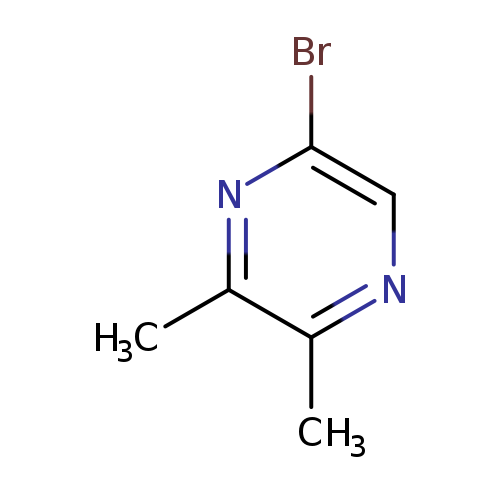
5-Bromo-2,3-dimethylpyrazineCatalog No.:AA007AHL CAS No.:111454-68-3 MDL No.:MFCD12404946 MF:C6H7BrN2 MW:187.0372 |
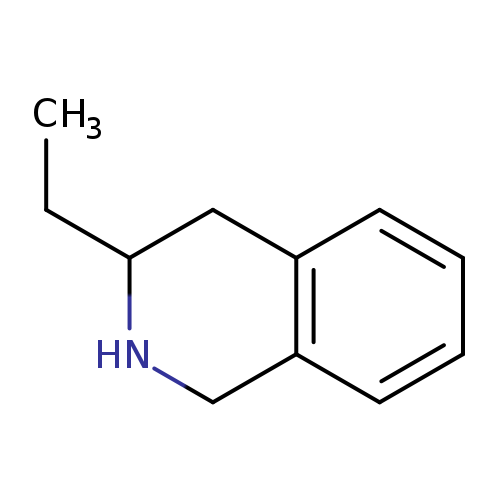
3-Ethyl-1,2,3,4-tetrahydroisoquinolineCatalog No.:AA007AHZ CAS No.:111422-13-0 MDL No.:MFCD16743958 MF:C11H15N MW:161.2435 |
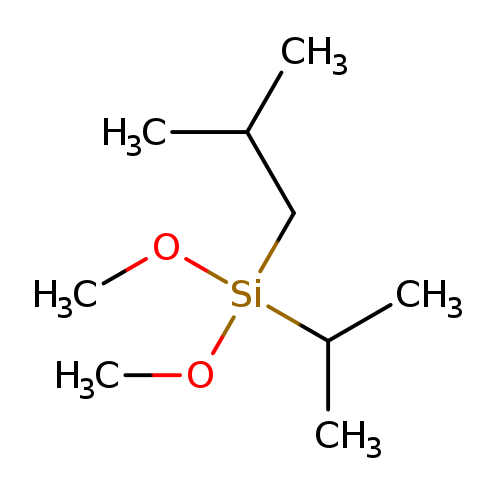
IsobutylisopropyldimethoxysilaneCatalog No.:AA007AHV CAS No.:111439-76-0 MDL No.:MFCD00239345 MF:C9H22O2Si MW:190.3553 |
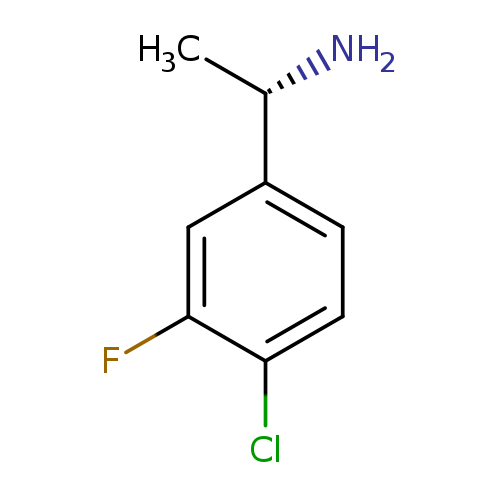
(S)-1-(4-Chloro-3-fluorophenyl)ethanamineCatalog No.:AA007AHK CAS No.:1114559-11-3 MDL No.:MFCD07772560 MF:C8H9ClFN MW:173.6152 |
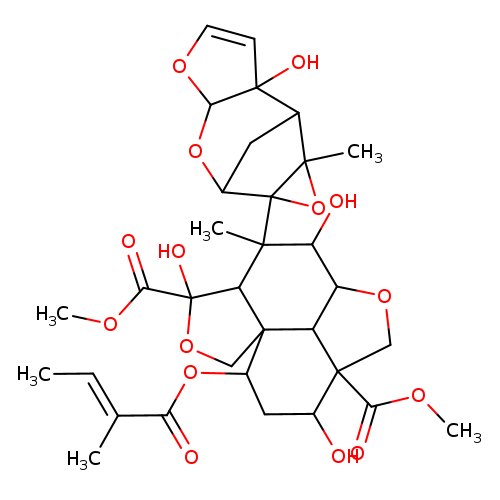
azadirachtinCatalog No.:AA007AI2 CAS No.:11141-17-6 MDL No.:MFCD00083241 MF:C33H42O15 MW:678.6776 |
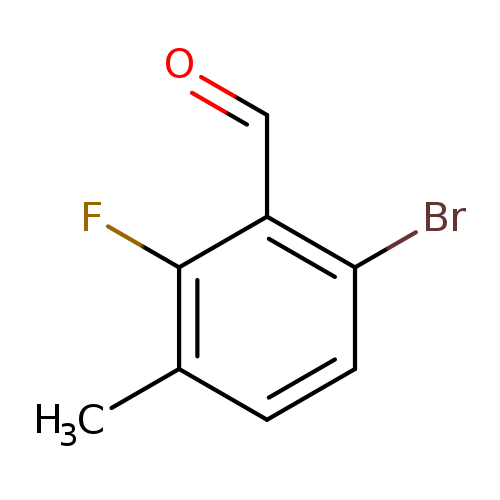
6-Bromo-2-fluoro-3-methylbenzaldehydeCatalog No.:AA007SST CAS No.:1114809-22-1 MDL No.:MFCD11520668 MF:C8H6BrFO MW:217.0350 |
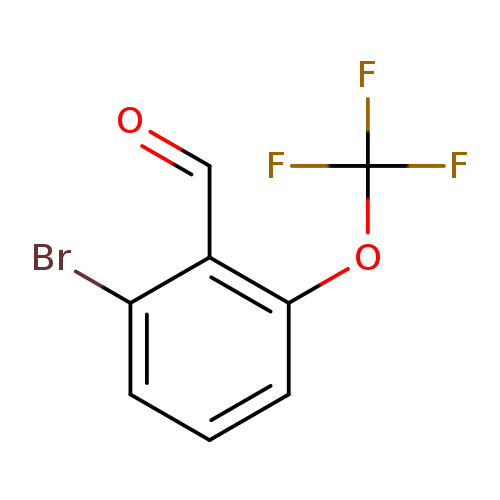
2-Bromo-6-(trifluoromethoxy)benzaldehydeCatalog No.:AA007SSU CAS No.:1114809-17-4 MDL No.:MFCD11856022 MF:C8H4BrF3O2 MW:269.0154 |
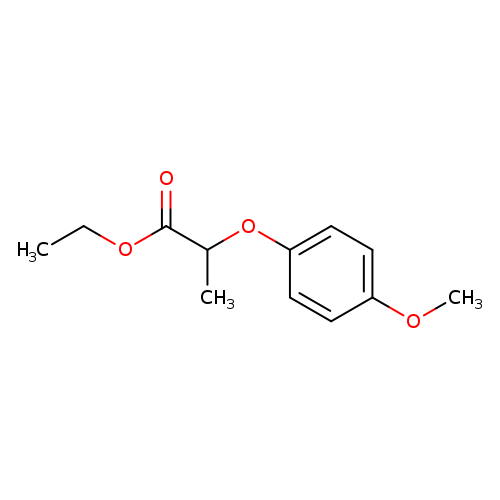
Ethyl 2-(4-methoxyphenoxy)propanoateCatalog No.:AA007SSY CAS No.:111479-08-4 MDL No.:MFCD16041984 MF:C12H16O4 MW:224.2530 |
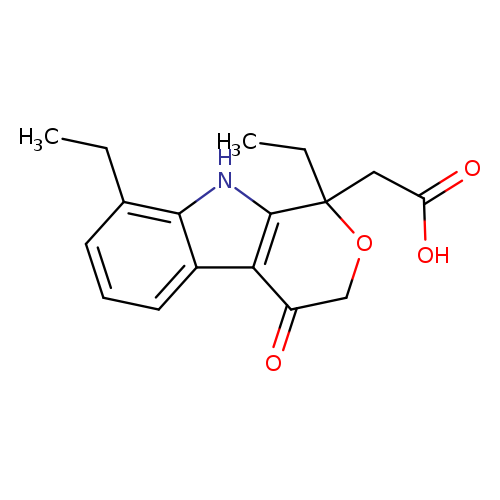
Pyrano[3,4-b]indole-1-aceticacid, 1,8-diethyl-1,3,4,9-tetrahydro-4-oxo-Catalog No.:AA007SSZ CAS No.:111478-86-5 MDL No.:MFCD09841133 MF:C17H19NO4 MW:301.3371 |
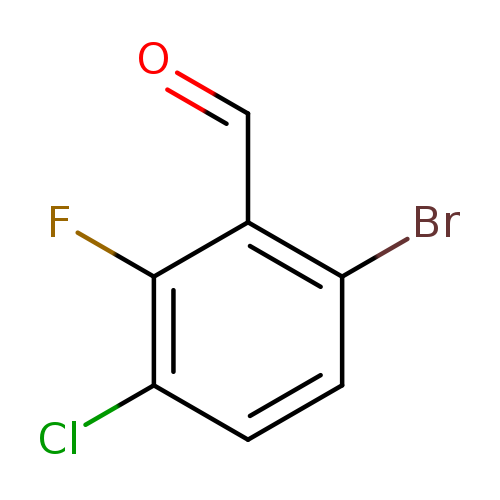
6-Bromo-3-chloro-2-fluorobenzaldehydeCatalog No.:AA007SSV CAS No.:1114809-02-7 MDL No.:MFCD11520661 MF:C7H3BrClFO MW:237.4535 |
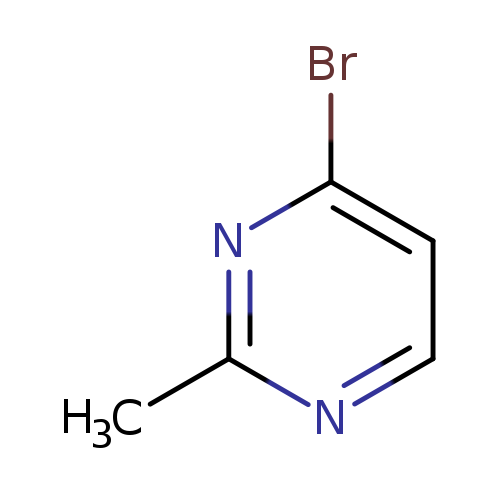
4-Bromo-2-methylpyrimidineCatalog No.:AA007STO CAS No.:1114560-76-7 MDL No.:MFCD11223245 MF:C5H5BrN2 MW:173.0106 |
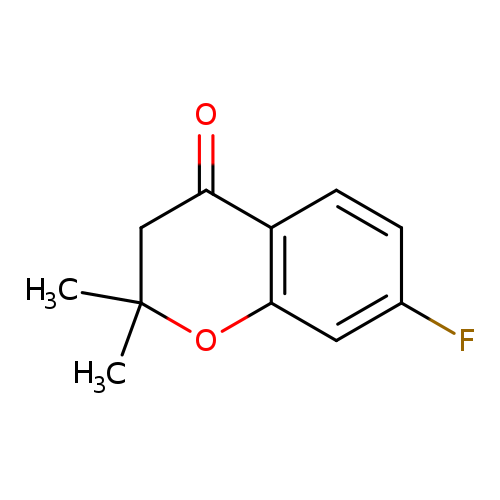
7-Fluoro-2,2-dimethylchroman-4-oneCatalog No.:AA007ST0 CAS No.:111477-98-6 MDL No.:MFCD15527098 MF:C11H11FO2 MW:194.2022 |
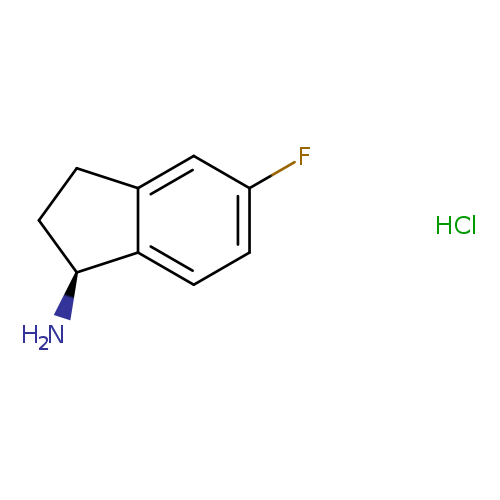
(S)-5-Fluoro-2,3-dihydro-1H-inden-1-amineCatalog No.:AA007STY CAS No.:1114333-11-7 MDL No.:MFCD06761876 MF:C9H10FN MW:151.1808 |
Ristomycin sulfateCatalog No.:AA007SU4 CAS No.:11140-99-1 MDL No.:MFCD00076121 MF:CH6O4S MW:114.1209 |
Imidazo[1,2-a]pyridin-8-ylmethanolCatalog No.:AA007ST1 CAS No.:111477-17-9 MDL No.:MFCD13183234 MF:C8H8N2O MW:148.1619 |
2-EthylbutanamideCatalog No.:AA007STW CAS No.:1114-38-1 MDL No.:MFCD00025512 MF:C6H13NO MW:115.1735 |
5-Amino-2-chlorobenzamideCatalog No.:AA007T2N CAS No.:111362-50-6 MDL No.:MFCD07787477 MF:C7H7ClN2O MW:170.5963 |
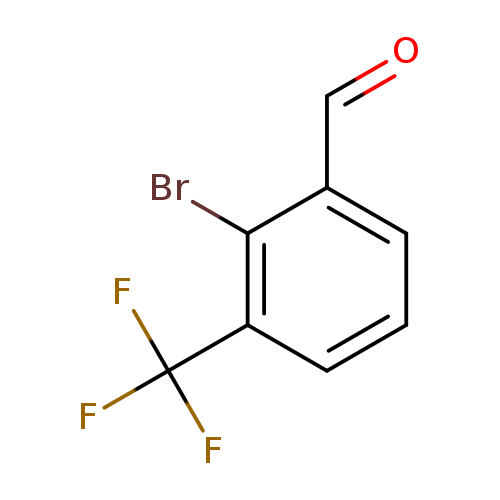
2-Bromo-3-(trifluoromethyl)benzaldehydeCatalog No.:AA00825S CAS No.:1114808-95-5 MDL No.:MFCD11520657 MF:C8H4BrF3O MW:253.0160 |
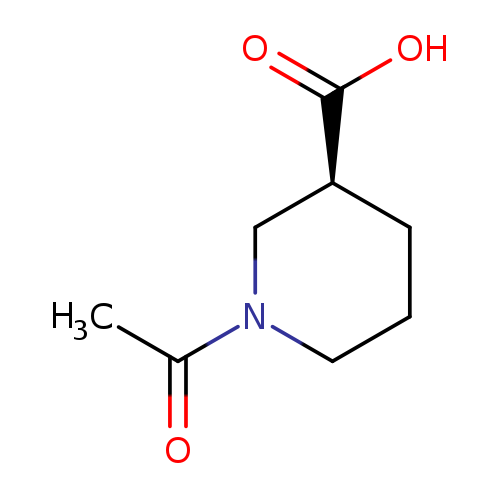
(S)-1-Acetylpiperidine-3-carboxylic acidCatalog No.:AA008278 CAS No.:111479-21-1 MDL No.:MFCD11617910 MF:C8H13NO3 MW:171.1937 |
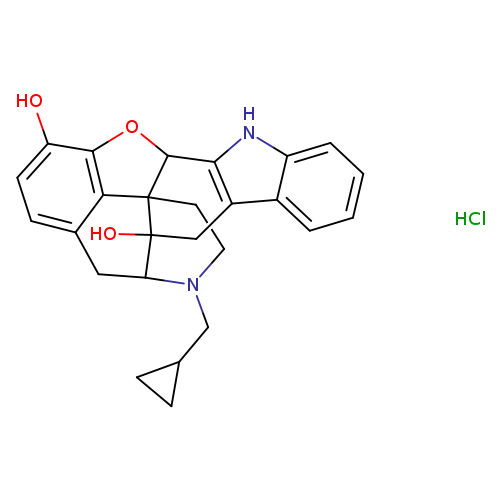
NALTRINDOLE HYDROCHLORIDECatalog No.:AA00827F CAS No.:111469-81-9 MDL No.:MFCD00069313 MF:C26H27ClN2O3 MW:450.9572 |
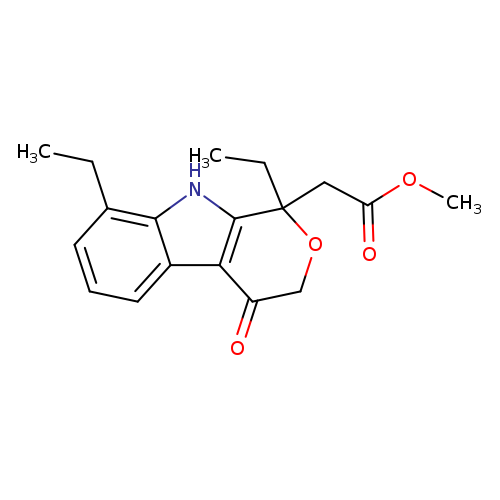
1,8-Diethyl-1,3,4,9-tetrahydro-4-oxo-pyrano[3,4-b]indole-1-acetic Acid Methyl EsterCatalog No.:AA00827A CAS No.:111478-84-3 MDL No.:MFCD09840461 MF:C18H21NO4 MW:315.3636 |
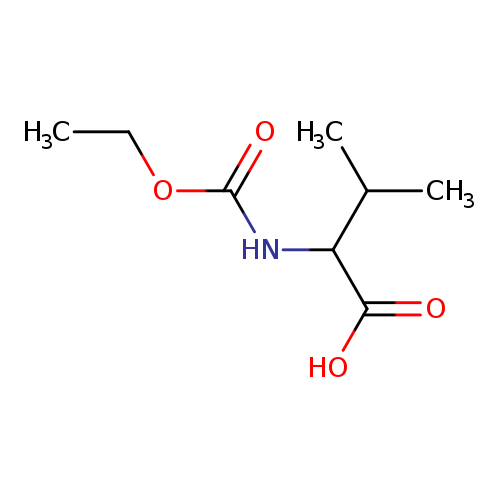
2-[(ethoxycarbonyl)amino]-3-methylbutanoic acidCatalog No.:AA008280 CAS No.:111421-95-5 MDL No.:MFCD09807004 MF:C8H15NO4 MW:189.2090 |
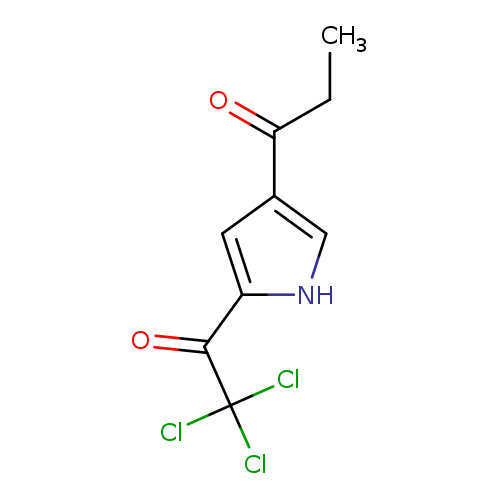
1-[5-(2,2,2-trichloroacetyl)-1H-pyrrol-3-yl] -1-propanoneCatalog No.:AA00827I CAS No.:111468-90-7 MDL No.:MFCD00141837 MF:C9H8Cl3NO2 MW:268.5243 |
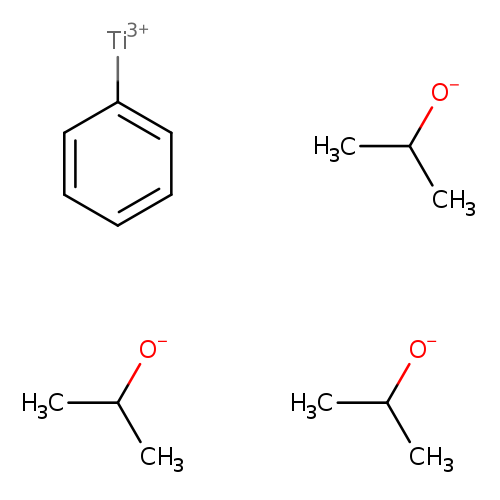
Titanium,phenyltripropoxy-, (T-4)-Catalog No.:AA00827N CAS No.:111452-11-0 MDL No.:MFCD08275495 MF:C15H26O3Ti MW:302.2321 |
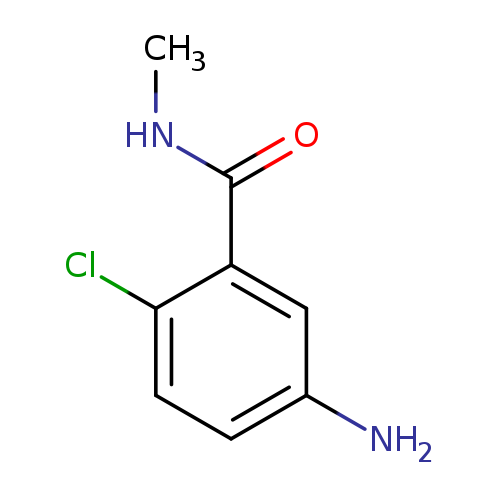
5-Amino-2-chloro-N-methylbenzamideCatalog No.:AA0082FF CAS No.:111362-49-3 MDL No.:MFCD09044637 MF:C8H9ClN2O MW:184.6229 |
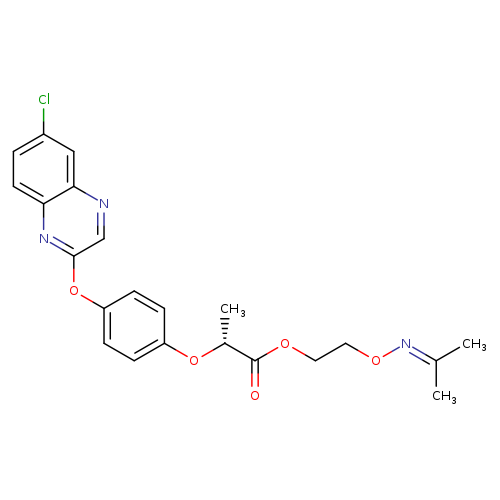
PropaquizafopCatalog No.:AA008S2P CAS No.:111479-05-1 MDL No.:MFCD01697482 MF:C22H22ClN3O5 MW:443.8802 |
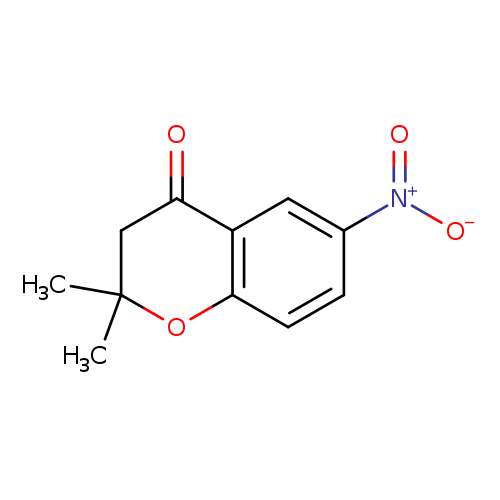
2,2-DIMETHYL-6-NITRO-CHROMAN-4-ONECatalog No.:AA008S52 CAS No.:111478-49-0 MDL No.:MFCD07369941 MF:C11H11NO4 MW:221.2093 |
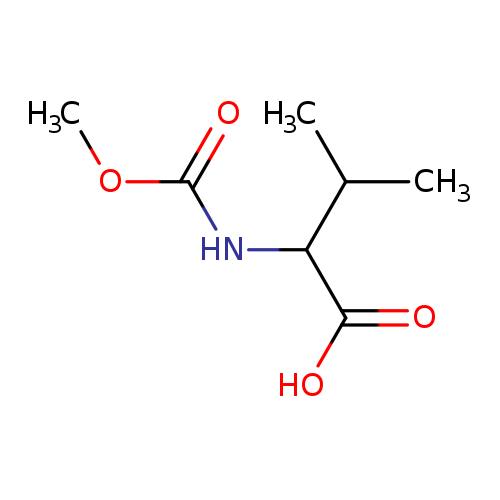
(Methoxycarbonyl)valineCatalog No.:AA008SC3 CAS No.:111398-44-8 MDL No.:MFCD11128988 MF:C7H13NO4 MW:175.1824 |
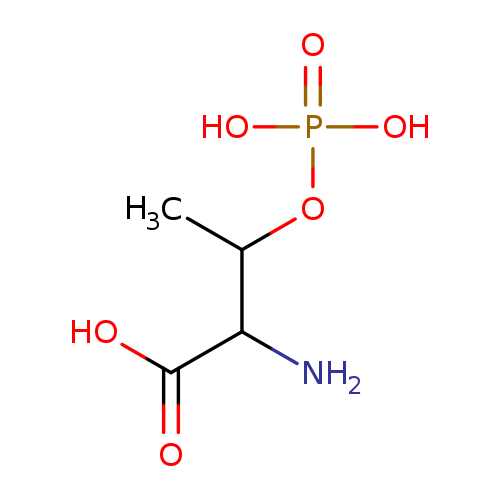
Phospho-L-threonineCatalog No.:AA008SHV CAS No.:1114-81-4 MDL No.:MFCD00069578 MF:C4H10NO6P MW:199.0991 |
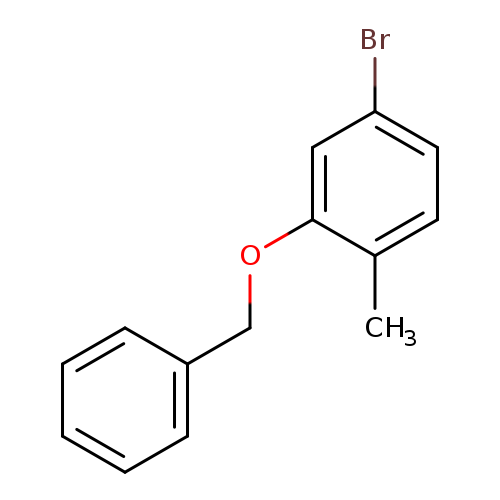
2-Benzyloxy-4-bromotolueneCatalog No.:AA008SSC CAS No.:1114808-93-3 MDL No.:MFCD11520656 MF:C14H13BrO MW:277.1564 |
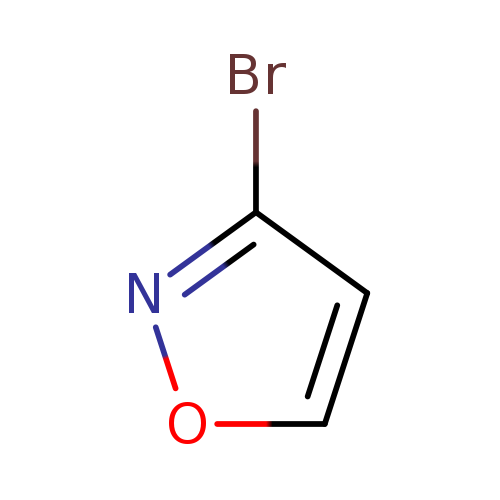
3-BromoisoxazoleCatalog No.:AA008T5X CAS No.:111454-71-8 MDL No.:MFCD07778379 MF:C3H2BrNO MW:147.9581 |
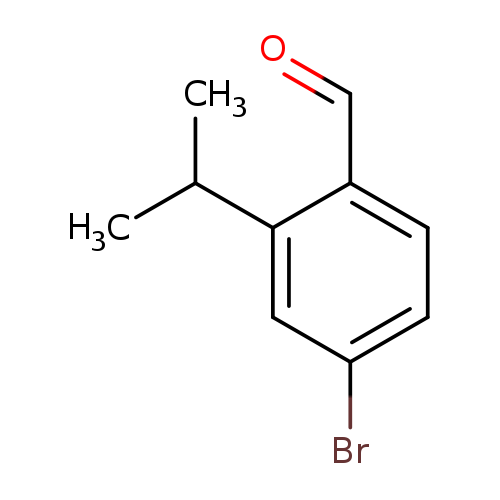
4-Bromo-2-isopropylbenzaldehydeCatalog No.:AA008TSA CAS No.:1114808-82-0 MDL No.:MFCD09835100 MF:C10H11BrO MW:227.0977 |
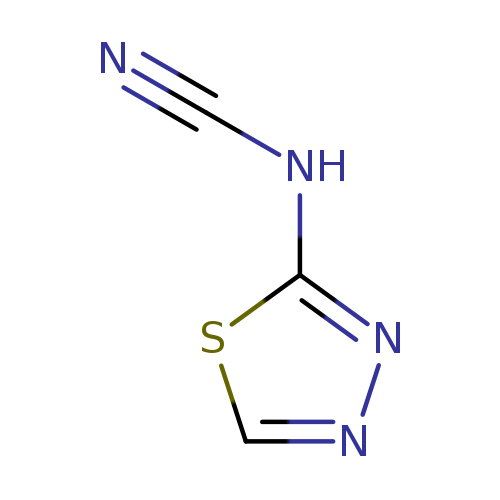
AmitivirCatalog No.:AA008UUV CAS No.:111393-84-1 MDL No.:MFCD00866935 MF:C3H2N4S MW:126.1398 |
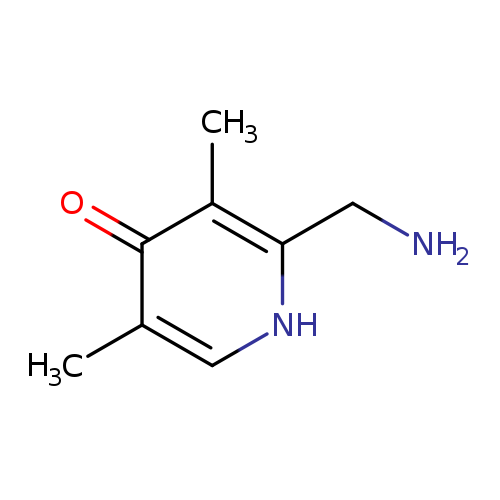
2-(Aminomethyl)-3,5-dimethylpyridin-4(1H)-one hydrochlorideCatalog No.:AA008V61 CAS No.:1114596-81-4 MDL No.:MFCD12026994 MF:C8H12N2O MW:152.1937 |
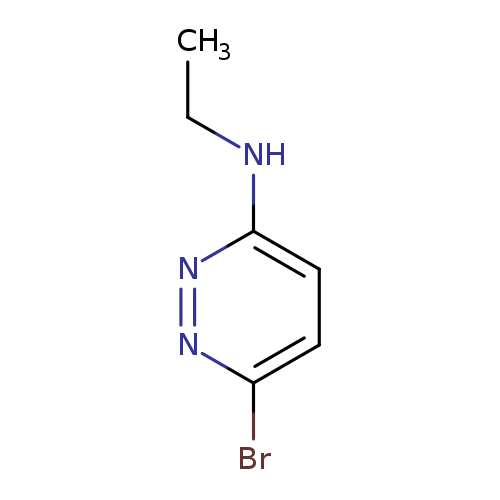
6-bromo-N-ethyl-3-pyridazinamineCatalog No.:AA008VDW CAS No.:111454-69-4 MDL No.:MFCD16556124 MF:C6H8BrN3 MW:202.0518 |
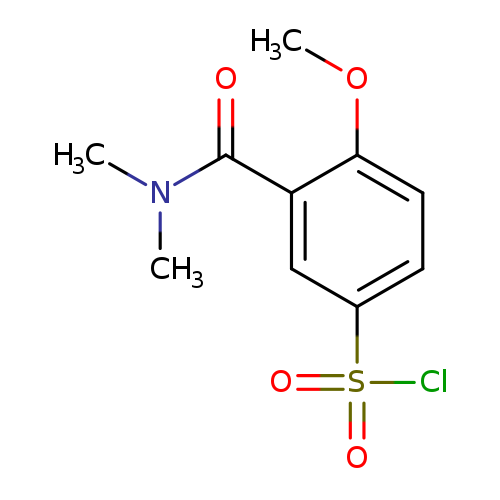
3-(Dimethylcarbamoyl)-4-methoxybenzene-1-sulfonyl chlorideCatalog No.:AA008VHV CAS No.:1114594-34-1 MDL No.:MFCD12827497 MF:C10H12ClNO4S MW:277.7246 |
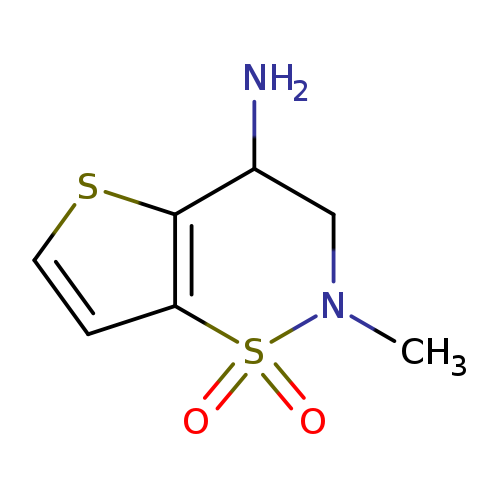
2-Methyl-3,4-dihydro-2H-thieno[2,3-e][1,2]thiazin-4-amine 1,1-dioxideCatalog No.:AA008VJN CAS No.:1114597-89-5 MDL No.:MFCD12027043 MF:C7H10N2O2S2 MW:218.2965 |
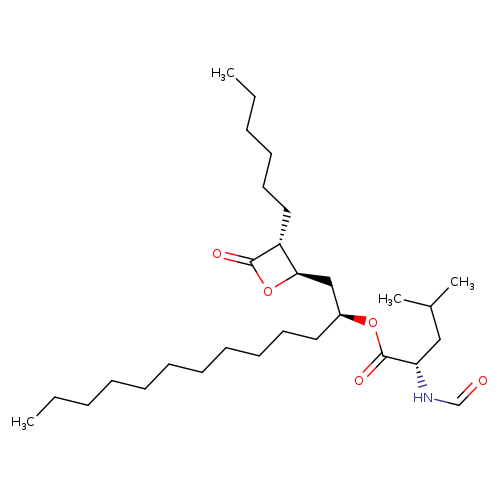
(S,S,R,R)-OrlistatCatalog No.:AA008W2T CAS No.:111466-62-7 MDL No.:MFCD23160336 MF:C29H53NO5 MW:495.7348 |
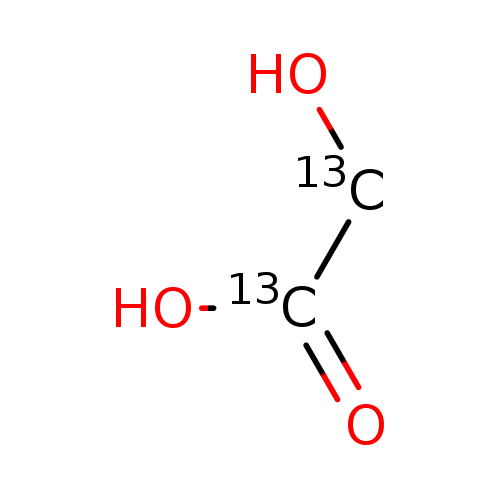
Glycolic Acid-13C2Catalog No.:AA008WH9 CAS No.:111389-68-5 MDL No.:MFCD04118149 MF:C2H4O3 MW:78.0367 |
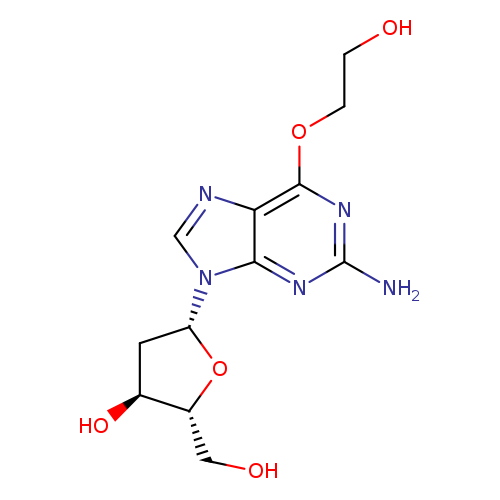
O6-(2-Hydroxyethyl)-2'-deoxyguanosineCatalog No.:AA008WNC CAS No.:111447-35-9 MDL No.: MF:C12H17N5O5 MW:311.2939 |
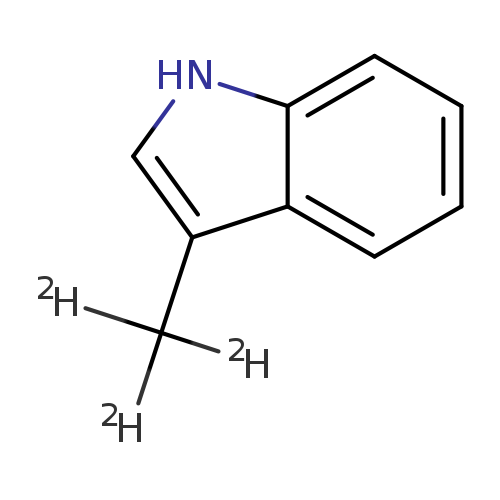
Skatole-d3Catalog No.:AA008WSU CAS No.:111399-60-1 MDL No.:MFCD22565753 MF:C9H6D3N MW:134.1929 |
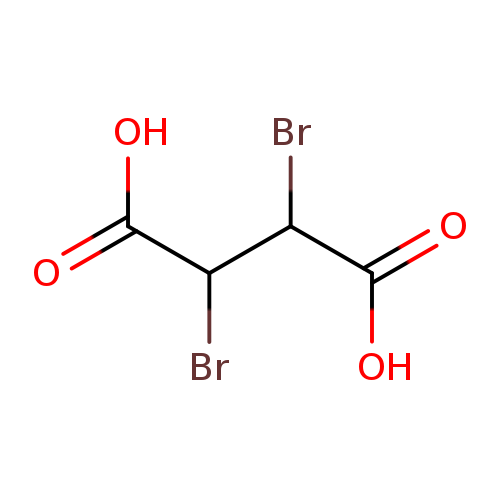
(2R,3S)-2,3-Dibromosuccinic acidCatalog No.:AA008YT0 CAS No.:1114-00-7 MDL No.:MFCD00066439 MF:C4H4Br2O4 MW:275.8802 |
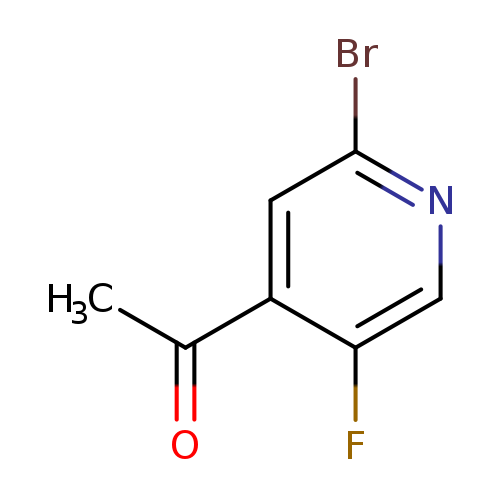
1-(2-Bromo-5-fluoropyridin-4-yl)ethanoneCatalog No.:AA008Z08 CAS No.:1114523-56-6 MDL No.:MFCD15526922 MF:C7H5BrFNO MW:218.0231 |
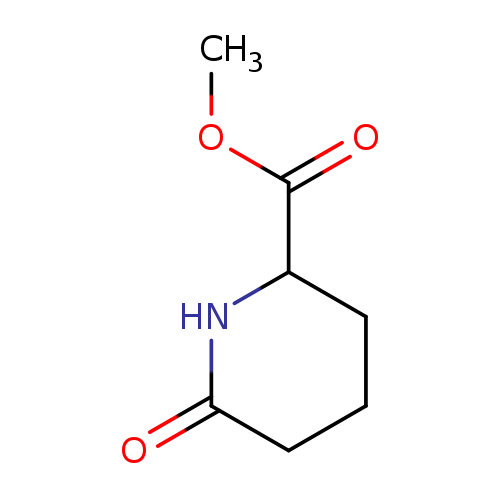
Methyl 6-oxopiperidine-2-carboxylateCatalog No.:AA0092IH CAS No.:111479-60-8 MDL No.:MFCD11111644 MF:C7H11NO3 MW:157.1671 |
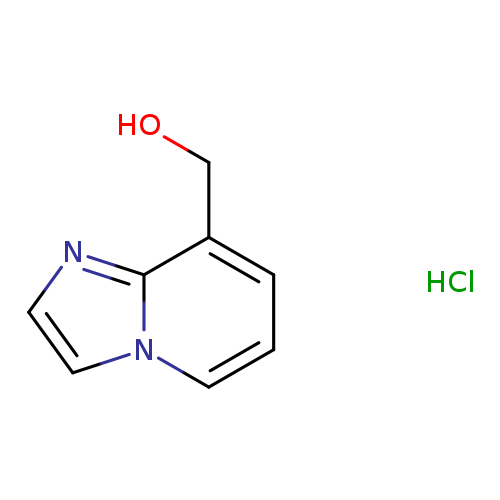
Imidazo[1,2-a]pyridin-8-yl-methanol hydrochlorideCatalog No.:AA0093I6 CAS No.:111477-18-0 MDL No.:MFCD28134240 MF:C8H9ClN2O MW:184.6229 |
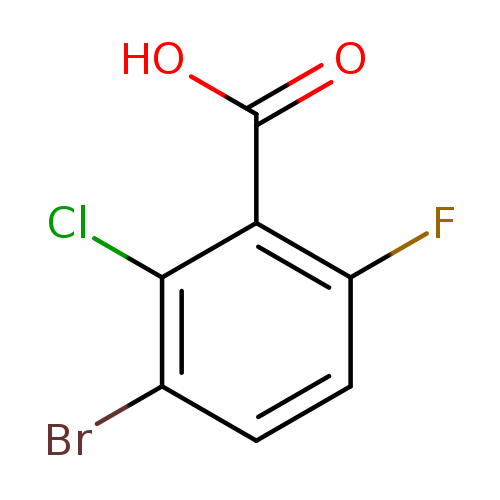
3-Bromo-2-chloro-6-fluorobenzoic acidCatalog No.:AA0094CY CAS No.:1114809-13-0 MDL No.:MFCD11856020 MF:C7H3BrClFO2 MW:253.4529 |
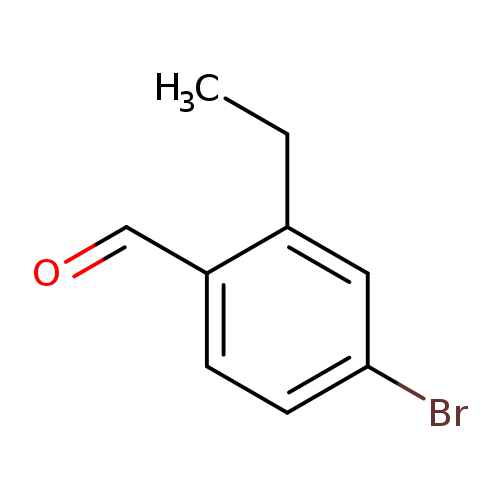
4-Bromo-2-ethylbenzaldehydeCatalog No.:AA0094DU CAS No.:1114808-89-7 MDL No.:MFCD09835094 MF:C9H9BrO MW:213.0712 |
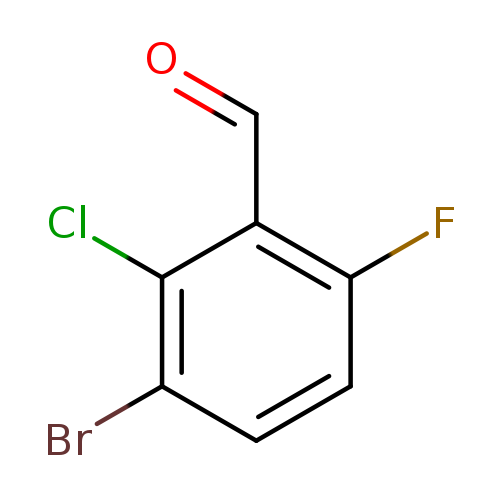
3-Bromo-2-chloro-6-fluorobenzaldehydeCatalog No.:AA0094K2 CAS No.:1114809-11-8 MDL No.:MFCD11110260 MF:C7H3BrClFO MW:237.4535 |
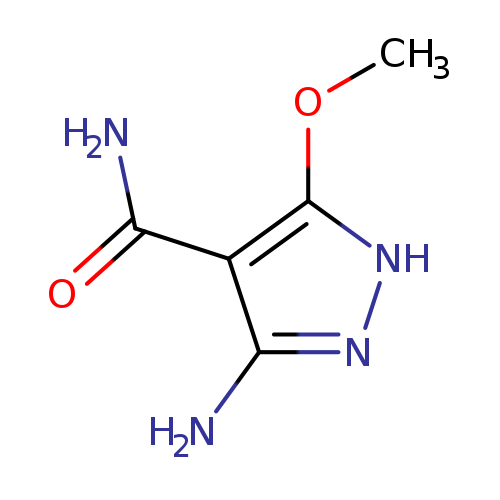
3-Amino-5-methoxy-1H-pyrazole-4-carboxamideCatalog No.:AA0094N4 CAS No.:111375-25-8 MDL No.:MFCD16619822 MF:C5H8N4O2 MW:156.1426 |
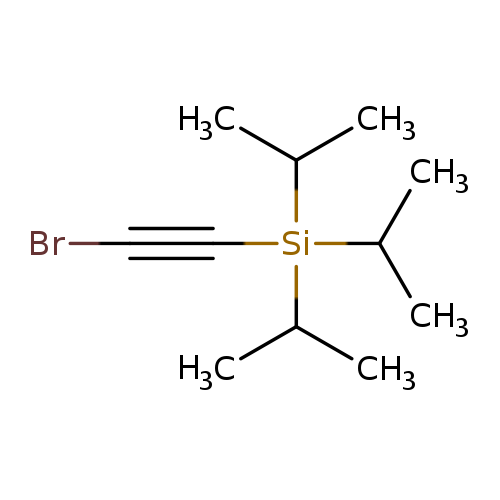
(Bromoethynyl)triisopropylsilaneCatalog No.:AA0095E0 CAS No.:111409-79-1 MDL No.:MFCD11036302 MF:C11H21BrSi MW:261.2739 |

Allyl Octadecyl MethylsilaneCatalog No.:AA0095DV CAS No.:1114235-08-3 MDL No.: MF:C22H46Si MW:338.6861 |
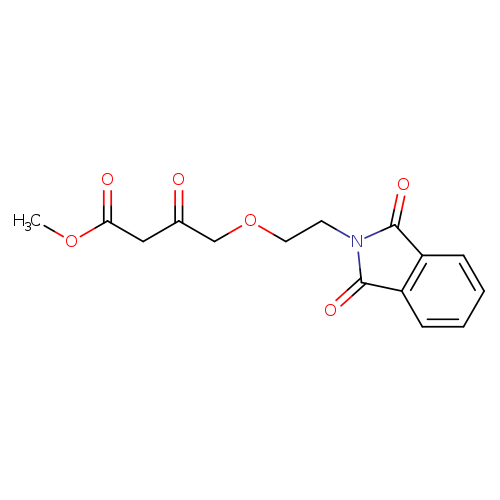
Methyl 4-(2-(1,3-dioxoisoindolin-2-yl)ethoxy)-3-oxobutanoateCatalog No.:AA00962O CAS No.:111429-90-4 MDL No.:MFCD26385799 MF:C15H15NO6 MW:305.2827 |
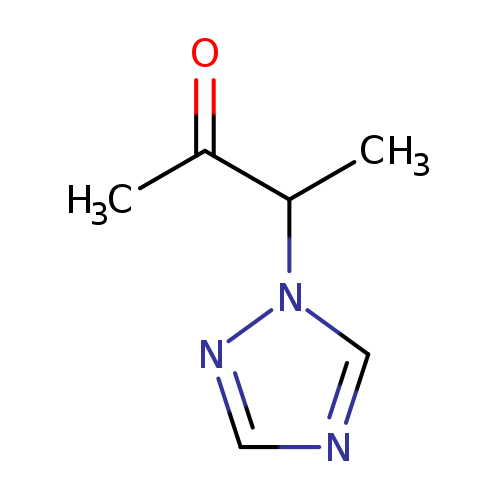
3-(1H-1,2,4-Triazol-1-yl)-2-butanoneCatalog No.:AA009MBA CAS No.:111451-34-4 MDL No.:MFCD00142027 MF:C6H9N3O MW:139.1552 |
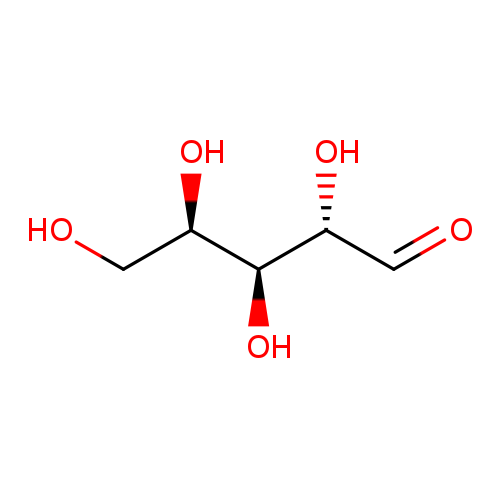
(2S,3S,4R)-2,3,4,5-TetrahydroxypentanalCatalog No.:AA00HC2D CAS No.:1114-34-7 MDL No.:MFCD00064362 MF:C5H10O5 MW:150.1299 |
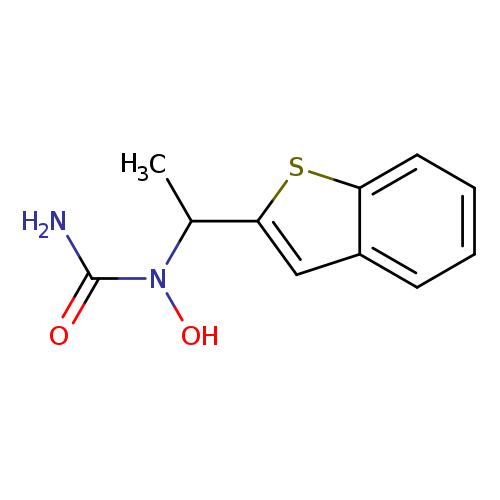
ZileutonCatalog No.:AA00HC2E CAS No.:111406-87-2 MDL No.:MFCD00866097 MF:C11H12N2O2S MW:236.2902 |
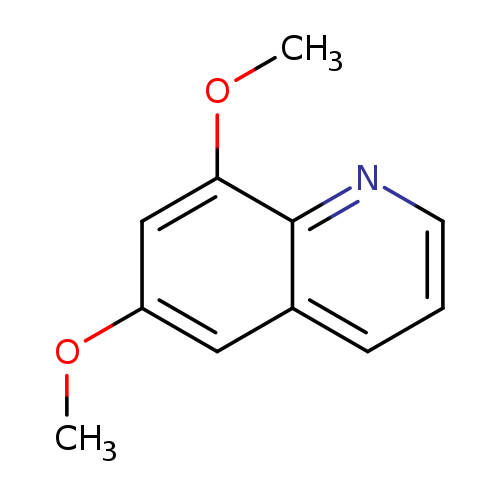
6,8-DimethoxyquinolineCatalog No.:AA00HC2H CAS No.:111454-91-2 MDL No.:MFCD14705144 MF:C11H11NO2 MW:189.2105 |
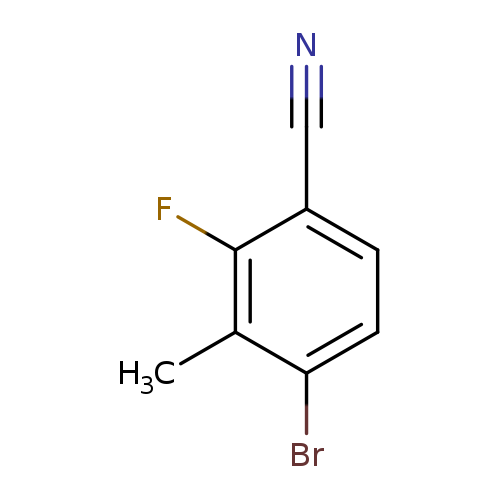
4-Bromo-2-fluoro-3-methylbenzonitrileCatalog No.:AA00HC2I CAS No.:1114546-30-3 MDL No.:MFCD23707391 MF:C8H5BrFN MW:214.0344 |
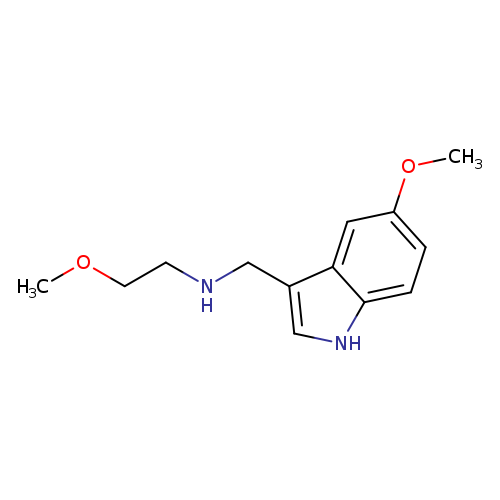
N-(2-Methoxyethyl)-n-[(5-methoxy-1h-indol-3-yl)methyl]amineCatalog No.:AA00HC2Z CAS No.:1114597-69-1 MDL No.:MFCD11982917 MF:C13H18N2O2 MW:234.2942 |
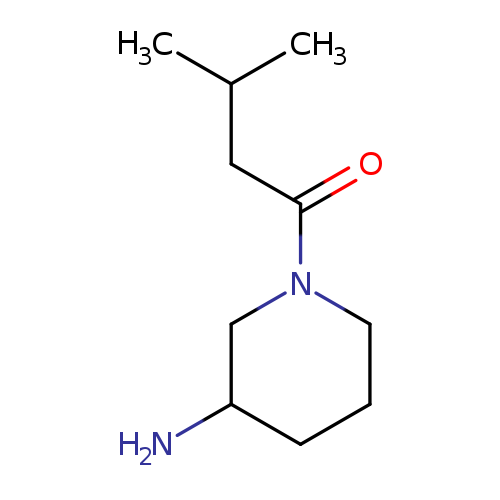
1-(3-Methylbutanoyl)piperidin-3-amineCatalog No.:AA00HC2S CAS No.:1114596-49-4 MDL No.:MFCD12026978 MF:C10H20N2O MW:184.2786 |
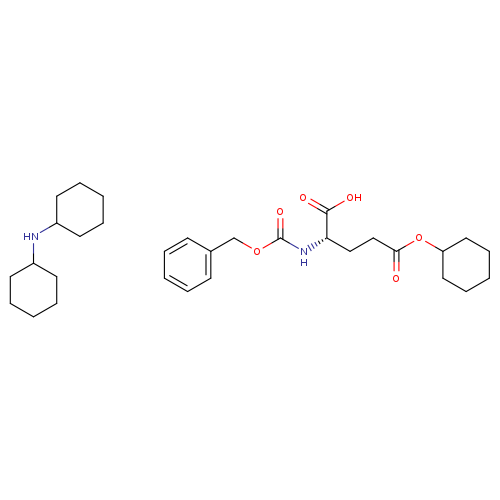
Z-L-Glu(ochex)-oh dchaCatalog No.:AA00HC32 CAS No.:111479-43-7 MDL No.:MFCD19707054 MF:C31H48N2O6 MW:544.7226 |
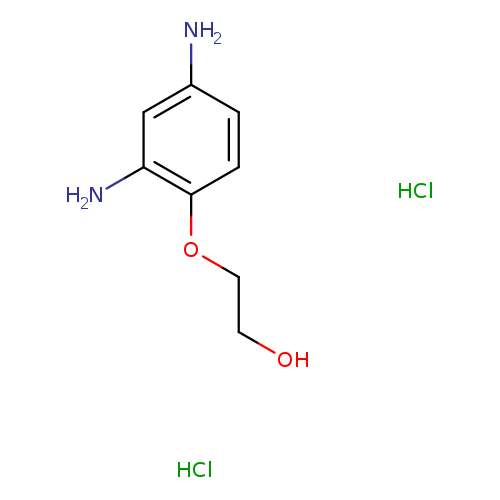
Xanthan GumCatalog No.:AA00IKC8 CAS No.:11138-66-2 MDL No.:MFCD00131256 MF:C48H82O29 MW:1123.1473 |
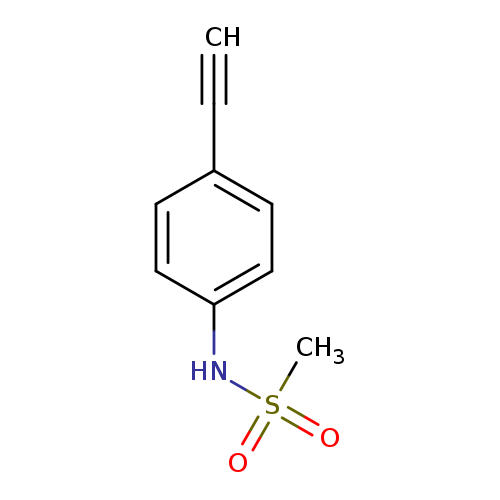
N-(4-Ethynylphenyl)methanesulfonamideCatalog No.:AA00IO17 CAS No.:111448-81-8 MDL No.:MFCD03012027 MF:C9H9NO2S MW:195.2383 |
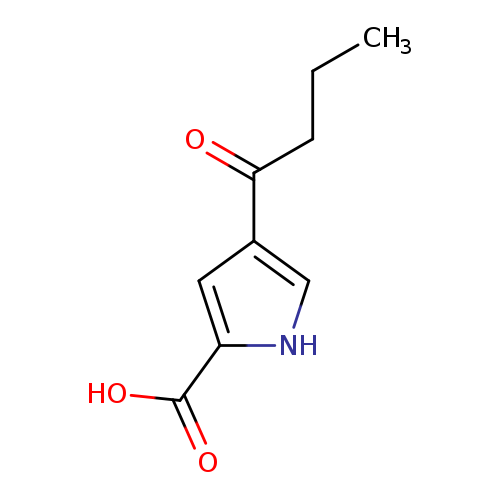
4-butyryl-1H-pyrrole-2-carboxylic acidCatalog No.:AA00ISG4 CAS No.:111468-95-2 MDL No.:MFCD05663820 MF:C9H11NO3 MW:181.1885 |
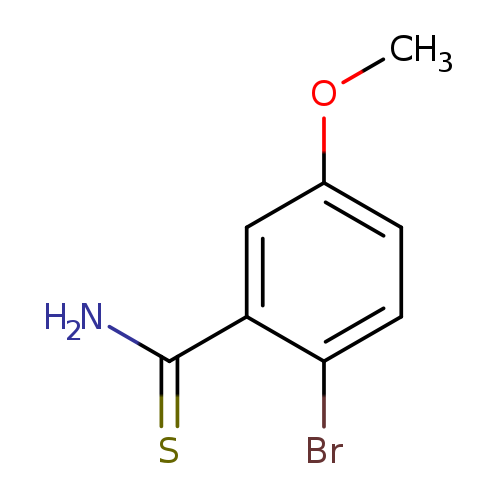
2-bromo-5-methoxybenzene-1-carbothioamideCatalog No.:AA019N1T CAS No.:1114822-69-3 MDL No.:MFCD11857769 MF:C8H8BrNOS MW:246.1242 |
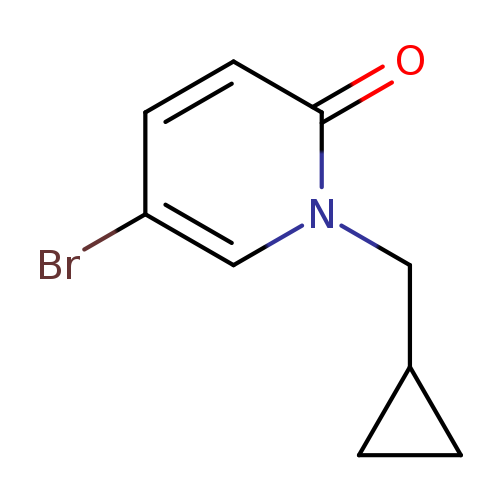
5-Bromo-1-(cyclopropylmethyl)-1,2-dihydropyridin-2-oneCatalog No.:AA01ACA0 CAS No.:1114573-41-9 MDL No.:MFCD21746683 MF:C9H10BrNO MW:228.0858 |
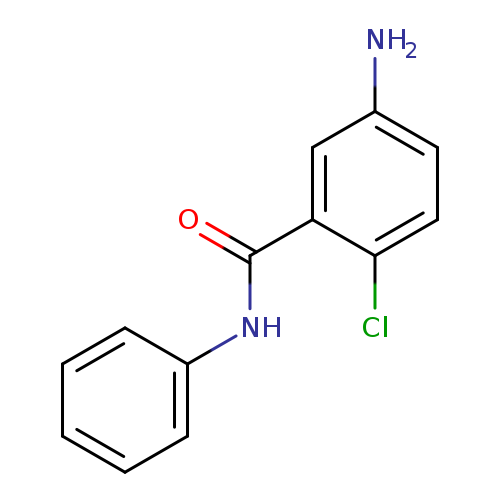
5-amino-2-chloro-N-phenylbenzamideCatalog No.:AA01AILA CAS No.:111362-57-3 MDL No.:MFCD00447349 MF:C13H11ClN2O MW:246.6922 |
(Hept-6-en-1-yl)(methyl)amineCatalog No.:AA01AK5I CAS No.:111475-66-2 MDL No.:MFCD09048143 MF:C8H17N MW:127.2273 |
6-Bromo-N-cyclopropylpyridine-3-carboxamideCatalog No.:AA01BRM8 CAS No.:1114563-25-5 MDL No.:MFCD14702800 MF:C9H9BrN2O MW:241.0846 |
rac-(1R,2S)-1-methyl-2-phenylcyclopropan-1-amine hydrochlorideCatalog No.:AA01DUWK CAS No.:111466-77-4 MDL No.:MFCD31617780 MF:C10H14ClN MW:183.6779 |
methyl 2-(3-methoxycyclohexyl)acetateCatalog No.:AA01EHQB CAS No.:1114554-12-9 MDL No.:MFCD28402811 MF:C10H18O3 MW:186.2481 |
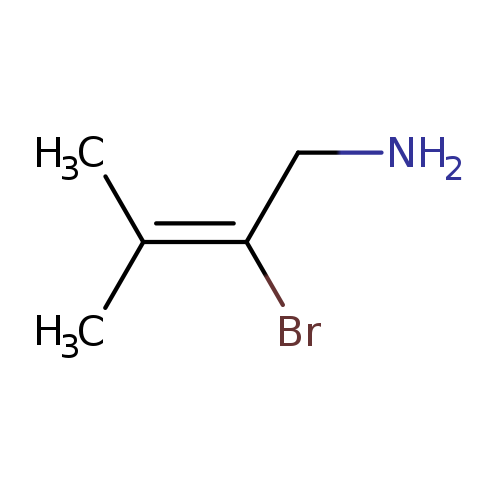
2-bromo-3-methylbut-2-en-1-amineCatalog No.:AA01ELWS CAS No.:111463-45-7 MDL No.:MFCD20696246 MF:C5H10BrN MW:164.0436 |
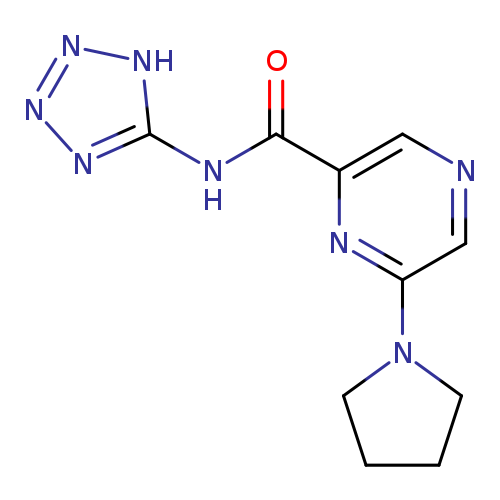
HSR6071Catalog No.:AA01ENUU CAS No.:111374-21-1 MDL No.:MFCD00879807 MF:C10H12N8O MW:260.2553 |
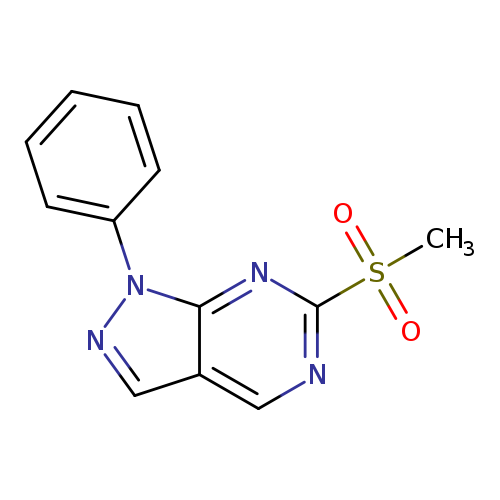
6-(methylsulfonyl)-1-phenyl-1H-pyrazolo[3,4-d]pyrimidineCatalog No.:AA01FFPQ CAS No.:1114555-03-1 MDL No.:MFCD19105137 MF:C12H10N4O2S MW:274.2984 |
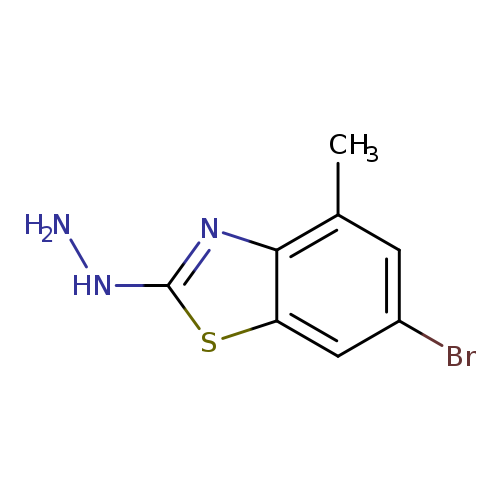
6-bromo-2-hydrazinyl-4-methyl-1,3-benzothiazoleCatalog No.:AA01FMWF CAS No.:1114594-94-3 MDL No.:MFCD11867941 MF:C8H8BrN3S MW:258.1382 |
ethyl 2-(7-ethyl-1H-indol-3-yl)-2-oxoacetateCatalog No.:AA008RXT CAS No.:111478-90-1 MDL No.:MFCD00189390 MF:C14H15NO3 MW:245.2738 |