2019-11-10 09:04:20
Navjeet Kaur
To cite this article: Navjeet Kaur (2019) Nickel catalysis: six membered heterocycle syntheses,
Synthetic Communications, 49:9, 1103-1133, DOI: 10.1080/00397911.2019.1568499
To link to this article: https://doi.org/10.1080/00397911.2019.1568499
Introduction
The heterocyclic compounds are used in chemical, biological, and industrial fields.[1–3] Heterocyclic compounds form the central core of many biologically active natural products and pharmaceutical agents (such as anti-viral, anti-inflammatory, anti-bacterial, anti-tumoral, and anti-oxidant activities agents), and are used in herbicides and corrosion inhibitors. Heterocyclic compounds, due to their presence in natural products and biologically active compounds are the most explored compounds.[4–6] They are significant not only for the synthesis of drugs, pharmaceuticals, and pigments but also for the development of organic functional materials. Many biologically active pharmaceuticals, agrochemicals, modifiers, and additives used in industrial applications like plastics, cosmetics reprography, and information storage are heterocyclic in nature.[7–10]
Many protocols have been investigated for the synthesis of heterocycles. These protocols for the formation of heterocyclic compounds involve carbon–nitrogen bond-forming reactions such as nucleophilic substitution, reductive amination, or dipolar cycloaddition for ring closure.[11–13] Although various protocols have been developed, their functional group tolerance and substrate scope are often limited. In recent years, for the synthesis of heterocycles, many new protocols have been developed which involve metal and nonmetal assisted carbon–nitrogen bond-forming reactions. These reactions tolerated a number of functional groups, occur under mild conditions, and proceed with high stereoselectivity. The utilization of metal and nonmetal allows highly convergent multi-component coupling approaches, which form many bonds and/or stereocenters in the one-pot protocol.[14–16] This review describes the approaches for the preparation of several six-membered heterocyclic compounds through nickel assisted reactions.
Nickel-catalyzed synthesis of six-membered heterocycles Synthesis of six-membered N-heterocycles
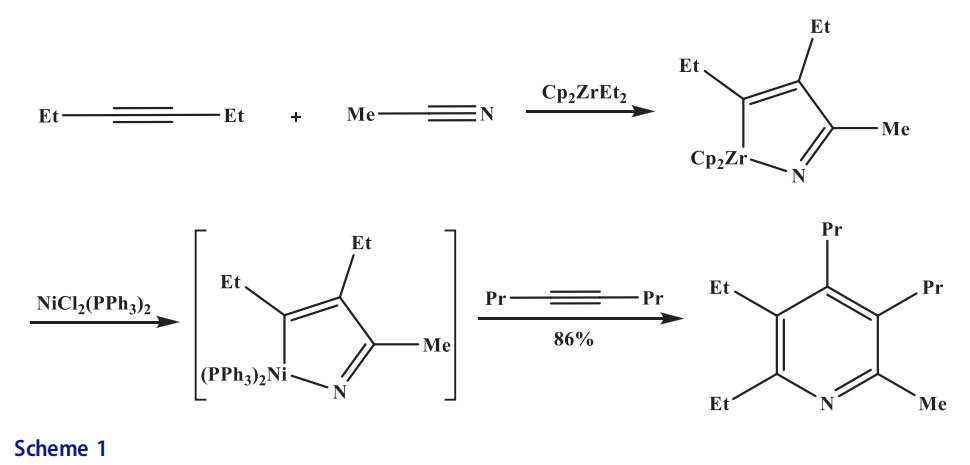
An azazirconacyclopentenone was formed by the coupling of isocyanate and alkyne with Zr. The nickelcyclopentenone was produced when Ni(PPh3)2Cl2 was reacted with azazirconacyclopentenone. The pyridine was formed upon insertion of the second alkyne. This was the first example where nickel was utilized for the preparation of pyridines. Unfortunately, this reaction required both Zr and nickel in stoichiometric amounts (Scheme 1).[17] The nickel and N-heterocyclic carbene, IPr, were utilized in combination with both unactivated nitriles to afford excellent yields of pyridines at room temperature. The
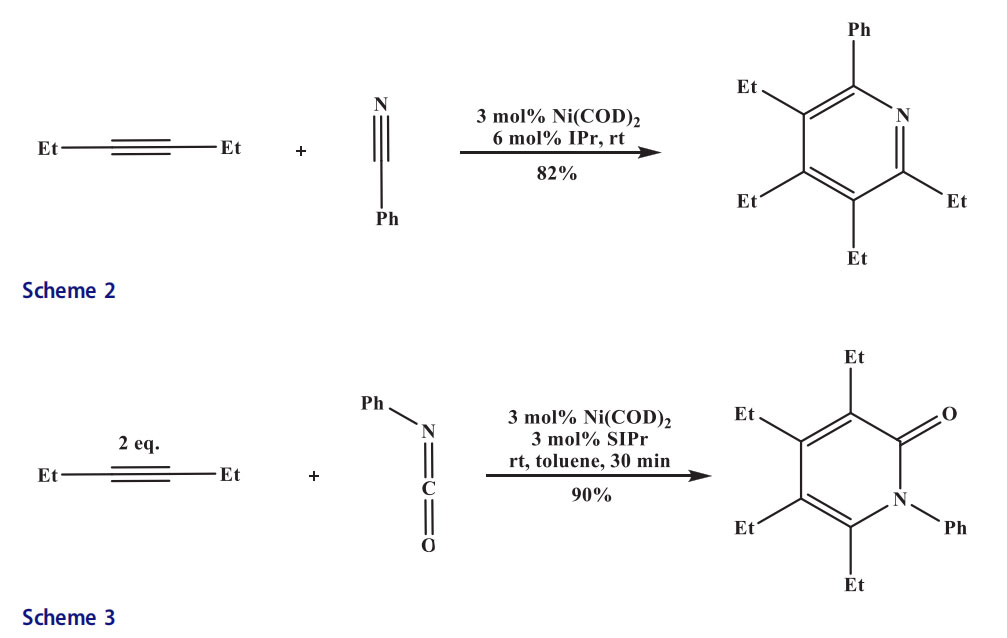
nitriles and alkynes were combined in an intermolecular fashion by this reaction.[18] Pyridines were obtained from nitriles by an effective Ni-catalyzed cycloaddition. The desired pyridines were also formed from untethered alkynes and the unsymmetrical diyne furnished a single regioisomer (Scheme 2).
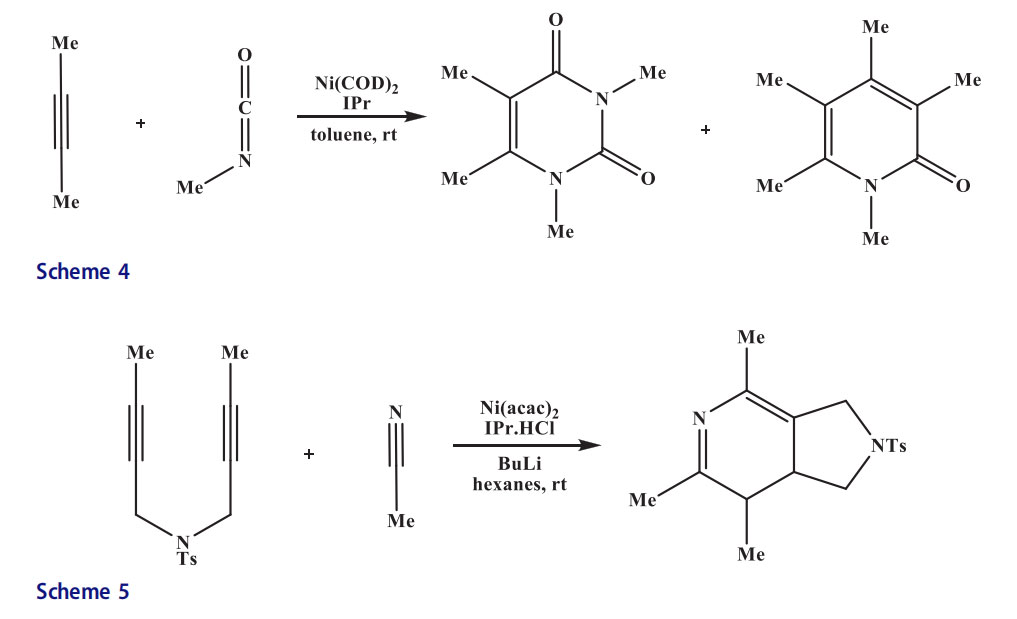
Louie[19] synthesized pyridones using metal complexes as catalysts. High yields of pyridones were obtained when isocyanates and diynes were reacted at ambient temperature employing a catalytic system Ni/SIPr (1,3-bis(2,6-diisopropylphenyl)-imidazolin-2-ylidene). In contrast, high yields of a series of 2-pyridones were reported by a [2 þ 2 þ 2] cycloaddition reaction of isocyanates and diynes using similar catalytic system(Scheme 3).
The catalytic reactions were varied involving carbon–carbon triple and double bonds.
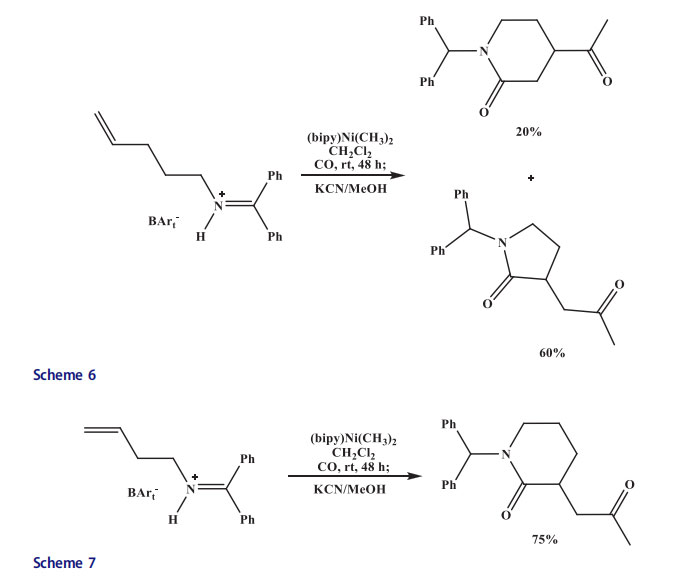
For instance, the 1,3-dienes and aldehydes were coupled with HSiEt3 in the presence of nickel catalyst (Schemes 4 and 5).[20] The BuLi was added to a mixture of imidazolium salt and NiCl2 for the in situ generation of nickel N-heterocyclic carbene complex. Moderate to high yield of (Z)-alkene was obtained. The (E)-alkene was obtained when a phosphane ligand was utilized in place of N-heterocyclic carbene. Jamison et al. performed the coupling of terminal olefins with isocyanates and olefins with aldehydes to afford acrylamides using in situ mixture of IPr (1,3-(2,6-diisopropylphenyl)imidazol-2-ylidene) and Ni(COD)2 as a catalyst. Two isocyanates were coupled with alkynes under similar conditions to provide pyrimidine diones.[23] Variously substituted pyridines were obtained when diynes were reacted with nitriles in the presence of in situ produced nickel N-heterocyclic carbene complex catalyst under basic conditions. Vinyl cyclopropanes were isomerized to cyclopentenes with a mixture of a nickel(0) precursor.
Other reactions with C–C double bonds, for example, the polymerization of styrene[24] and dimerization of ethane[25,26] were carried out with nickel N-heterocyclic carbene complexes. A variety of imines were treated with NaOi-Pr in the presence of IMes and nickel(0) to provide amines by transfer hydrogenation.[27] The dihydrogen gas was produced rapidly by dehydrogenation when a triazol-5-ylidene was reacted with catalytic amounts of Ni(0) and H3NBH3, through hydrogen transfer of ammonia–borane to the carbene carbon, followed by C–H activation by the nickel species.[28]
Longer tethers between the olefin and imine were used to probe the generality of this insertion cascade. Only 20% yield of six-membered lactam was obtained along with five-membered exo-cyclization product in high amounts (60%) when the length of tether was increased by one carbon. No (<5%) seven-membered lactam was obtained and a six-membered lactam was obtained with almost complete selectivity (72% yield) upon extending the tether to three methylene units[29] (Schemes 6 and 7).
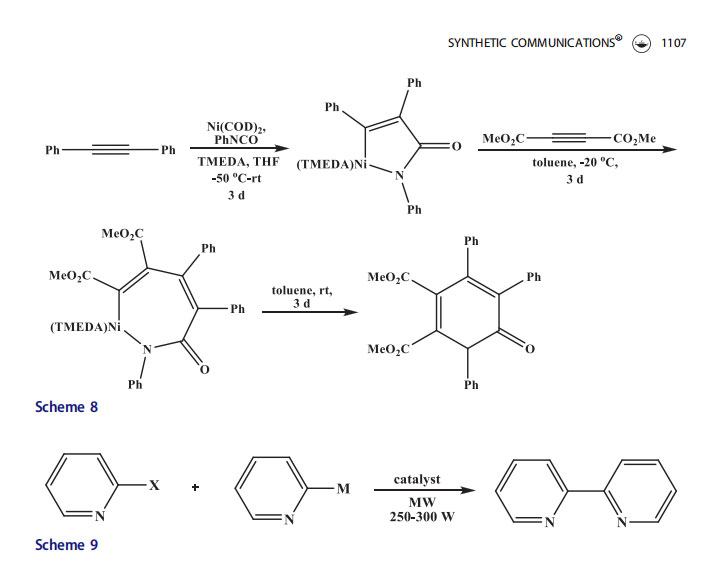
Hoberg[30–32] for first time reported the use of nickel for the preparation of pyridones. Pyridine was formed selectively when an alkyne was reacted with an isocyanate in stoichiometric amounts of Ni (Scheme 8). The oxidative coupling of phenyl isocyanate and alkyne provided nickel acyclopentenone which was coupled with one more equivalent of an alkyne moiety to afford moderate yields of pyridone through nickelacycle. Although this was the first successful method to synthesize pyridones employing nickel, the reaction to construct the nickelacycle needed long reaction time (3 d) and
low temperatures (50 C).
Moore and coworkers[33] synthesized unsymmetrical and symmetrical bipyridines by cross-coupling reactions using heterogeneous palladium and nickel catalysts under MWI(Scheme 9). No phosphine ligand was required when nickel/aluminum oxide-silicon oxide (50 mol% loading) was utilized in the case of nickel catalysis. Under palladium-catalysis moderate yields were obtained; however, only 5 mol% catalyst loading was needed.[6e] Pyridine underwent metalation and functionalization reactions ideally in the presence of metal reagent in catalytic amounts. For the preparation of pyridine derivatives, various synthetic methods were enabled by transition-metal catalysis. For example, cycloaddition reactions in the presence of transition metal catalysts are highly versatile for the synthesis of substituted pyridine from commercially available unsaturated compounds (Scheme 10).
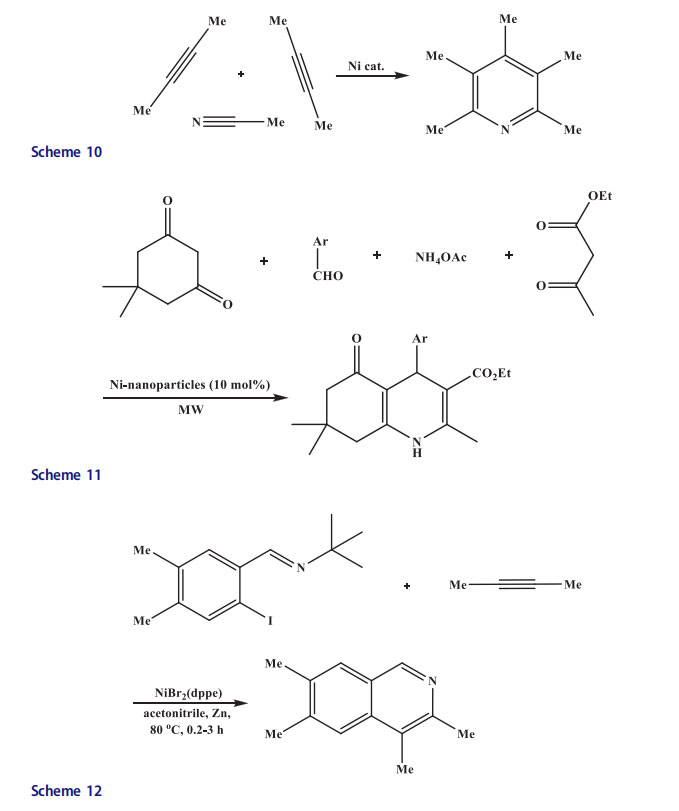
The polyhydroquinoline derivatives were synthesized in high yields by MMS in the absence of solvent using heterogeneous catalyst nanosized Ni particles through Hantzsch protocol (Scheme 11).[36–37]
The tert-butyl imines of 2-iodobenzaldehydes and several alkynes were annulated efficiently in the presence of Ni catalyst to afford a variety of substituted isoquinolines[38](Scheme 12). Two different alkyne insertion routes were there for catalytic reactions as indicated by the regiochemistry of isoquinolines.
Terminal alkynes, as well as various unsymmetrical and symmetrical internal alkynes, provided isoquinolones in very high yields. However, varied regioselectivity was shown by unsymmetrical alkynes. Murakami et al.[39] reported that 1,2,3-benzotriazinones also served as good substrates in denitrogenative transannulation reactions. Thus, 1,2,3-benzotriazinones were reacted with alkynes in the presence of Ni catalyst to synthesize isoquinolones. Initially, nickel(0) was inserted into the N,N linkage of 1,2,3-benzotriazinones, which lost dinitrogen to afford azanickelacycle.[40] The alkyne was inserted into the nickel–carbon bond to produce a seven-membered nickelacycle intermediate, which provided the final product isoquinolones after the reductive elimination and the regeneration of the nickel(0) catalyst (Scheme 13). This reaction did not occur with N-unsubstituted benzotriazinone.
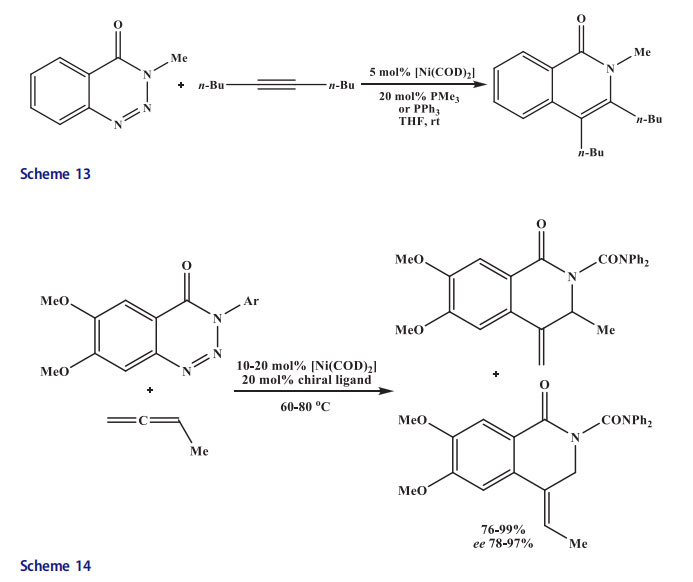
The 1,2,3-benzotriazinones were transannulated with a wide range of monosubstituted allenes by a catalytic version of this reaction (20 mol% PMe3, 5 mol% [Ni(cod)2], 60 C, THF). Both the electron-donating and electron-withdrawing substituents present at the aromatic ring of the benzotriazinone and at the N atom of the triazole moiety worked well and a major regioisomer of differently substituted isoquinolones were provided.[41] The regiochemistry was reversed completely in the reaction with trialkylsilyland tert-butyl-substituted allenes due to sterics. The use of phosphinooxazoline ligand(S,S)-iPr-foxap[42] afforded very high enantio- and regioselectivities (Scheme 14).
Murakami and coworkers[43] reported that 1,2,3-benzotriazinones underwent denitrogenative transannulation with allenes in the presence of Ni catalyst. A five-membered azanickelacycle intermediate was produced when triazene was reacted with [Ni(cod)2] and dppbenz in stoichiometric amounts. Treatment of azanickelacycle intermediate with an allene at 60 C in tetrahydrofuran provided 3,4 dihydroisoquinolin-1(2H)-ones(54:46) in 99% yield as an isomeric mixture (Scheme 15).
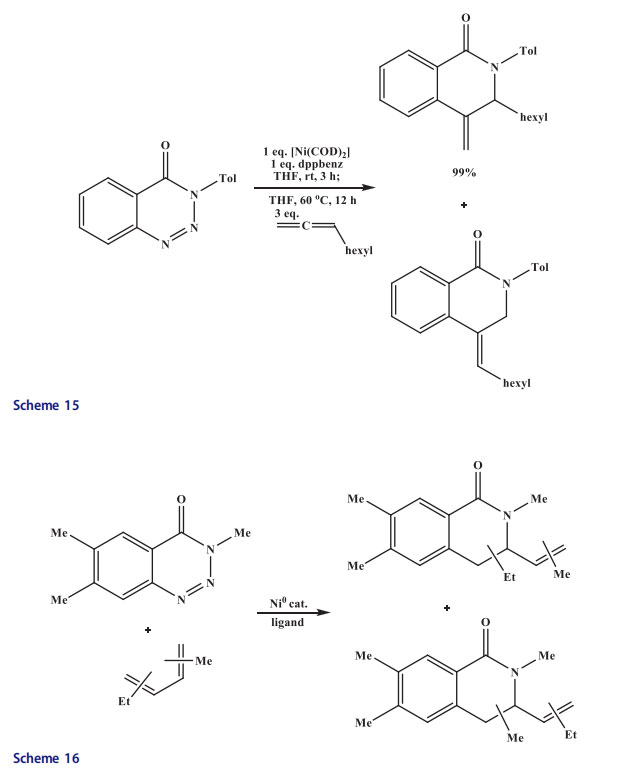
Various N-protected isoquinolones were synthesized when differently substituted benzotriazinones were reacted with symmetrical 1,3-dienes using 1,1’-bis(diphenylphosphino)ferrocene (10 mol%) and [Ni(cod)2] (10 mol%) at 60 C in tetrahydrofuran (Scheme 16). The yields were high with all other substrates with the exception of N-benzyl-substituted benzotriazinone (24%). Benzotriazinones with both electron-withdrawing and electron-donating groups at the benzene ring were equally competent in this reaction. Isoquinolones were obtained as major regioisomers using unsymmetrical dienes.[43]
The benzotriazinones were transannulated with activated alkenes and 1,3-dienes in the presence of Ni catalyst.[44] The quinoline was obtained in only trace amounts when Ni complex was mixed with 1,3-diene without a phosphine ligand. However, quinoline was formed in 40% yield upon addition of 1,1’-bis(diphenylphosphino)ferrocene ligand(Scheme 17).
The benzotriazinones underwent transannulation with activated alkenes in the presence of P(n-Bu)3 and [Ni(cod)2]. Thus, excellent yields of dehydroisoquinolinones were obtained when electron-deficient alkenes, like acrylonotrile, acrylamide, and methyl acrylate underwent transannulation smoothly with benzotriazinones. Pyridyl-possessing alkenes were efficient, whereas with styrene product was formed in a low yield. Electron-rich and electron-neutral alkenes did not participate in the reaction at all (Scheme 18).
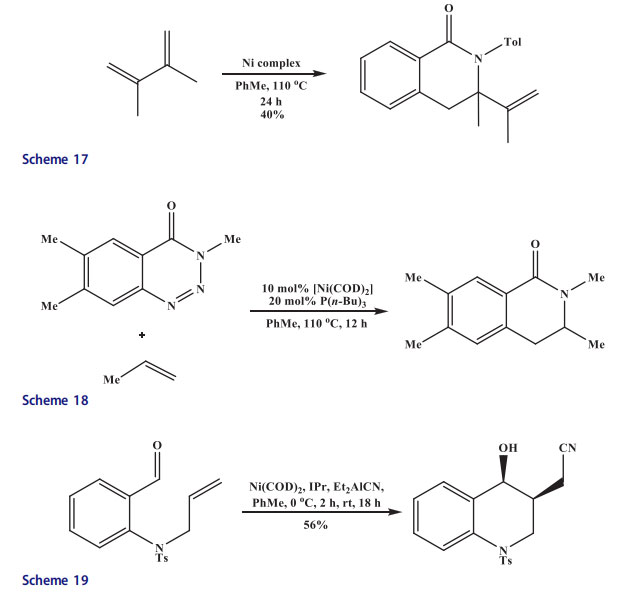
Ho[45] reported that N-allyl aniline through cyanative alkene aldehyde coupling afforded quinoline derivative with the help of nickel(0) catalyst-N-heterocyclic carbene/Et2AlCN (Scheme 19). The isoquinoline derivatives were produced in good yields by a [2 þ 2 þ 2] co-cyclization in the presence of Ni(0) catalyst at room temperature[46] (Scheme 20). But steric hindrance influenced the cyclization. No cyclized isoquinoline products were obtained when disubstituted alkynes were used in place of acetylene. The diphenylacetylene and azazirconacyclopentadiene were coupled in the presence of NiCl2(PPh3)2 catalyst to afford a substituted isoquinoline derivative[47] (Scheme 21).
The coupling reaction occurred in high regioselectivity and furnished only single products when unsymmetrical alkynes like 1-phenyl-1-butyne and 1-phenyl-1-propyne were utilized as the second alkyne.
Louie[48] reported successfully that there was no need of stoichiometric amounts of nickel to prepare pyridines. Alkyne trimerization with nickel catalysts delivered mixtures of highly functionalized benzene derivatives. One alkyne was replaced with a nitrile to synthesize pyridines and various other nitrogen-containing heterocycles through similar intermediates. A metallocyclopentadiene was prepared when diyne was stitched together, the resulting compound was reacted with a third alkyne in a Diels–Alder reaction. The formed metal-bridged norbornadiene was aromatized with the loss of nickel catalyst
and the cycle begins again (Scheme 22).
The 2-aminopyridine is important, therefore authors developed an efficient protocol for the synthesis of this motif from an easily available and simple starting material in a single step. Authors reported a [2 þ 2 þ 2] cycloaddition of readily available cyanamides and diynes. The cycloaddition reaction of various cyanamides and diynes was also reported employing a nickel-N-heterocyclic carbene catalyst system.[49] A Fe-catalyzed protocol to obtain 2-aminopyridines has not yet been reported (Scheme 23).
The ynals by reductive cyclization reactions provided fused azabicycle in the presence of nickel catalyst.[50] The reaction occurred with high stereoselectivity due to the formation of highly organized oxametallacycles, which underwent reduction with triethylsilane to synthesize diverse indolizidine and pyrrolizidine products. This protocol was applicable in Montgomery’s total preparation of indolizidine alkaloid allopumiliotoxin through the stereoselective reductive cyclization followed by two subsequent protecting group manipulations (Scheme 24).

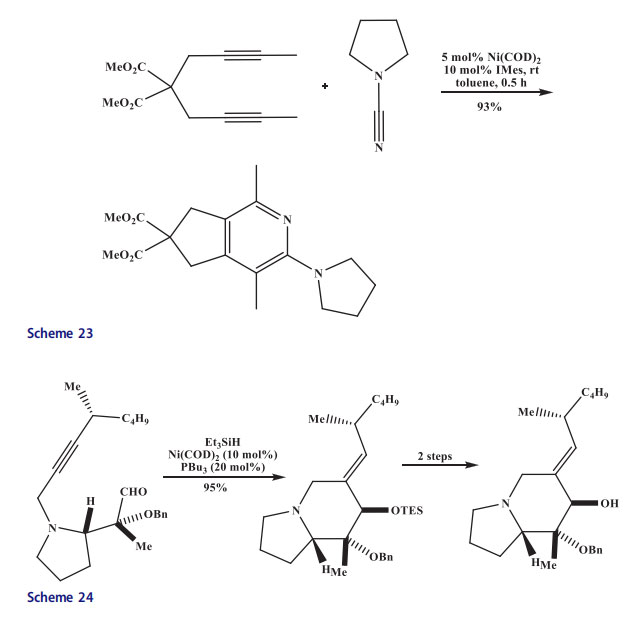
The gem-dialkyl groups bearing dipropargyl amines [52,53] were transformed in the presence of cobalt(II) chloride/manganese catalytic system. The reaction temperature was decreased to 80 C and yield of dihydropyrrolopyridines were increased up to 90–98% due to the presence of gem-dialkyl groups in diacetylene (Scheme 25).[54]
This method was extended for the construction of DE-ring skeleton of the yohimbine(Scheme 26) by initiating the synthesis of tryptophylsubstituted amide in three steps by sequential alkylation,[55] acylation, and Boc protection [56] of tryptamine. The cycloaddition of tryptophylsubstituted amide in the presence of Ni catalyst afforded quinoline in 88% yield at room temperature, while the alternative synthetic thermal method needed 150 C temperature to cleave the Boc protecting group and to give 1,4-cyclohexadiene product in 45% yield only. The resulting product was elaborated when the sterically less encumbered olefin was hydrogenated selectively, the vinylsilane underwent protiodesilylation[57] with concomitant removal of the Boc protecting group, and the C-ring underwent Bischler-Napieralski closure to afford the pentacyclic 19,20-didehydroyohimban in 86% yield. The 19,20-didehydroyohimban was hydrogenated catalytically in platinum oxide to afford alloyohimban and yohimban in a 1:1 ratio.[58–60]
Cook and coworkers[61,62] synthesized opioid agonistic alkaloid mitragynine through the formation of allylamine intermediate. An asymmetric Pictet–Spengler reaction and a cyclization in the presence of Ni(cod)2 provided allylamine intermediate (Scheme 27).

3-(4-Bromo-2-fluorophenyl)-1h-1,2,4-triazol-5-amineCatalog No.:AA01EL5T CAS No.:1016771-38-2 MDL No.:MFCD18380681 MF:C8H6BrFN4 MW:257.0624 |

4-(3-aminophenyl)-4,5-dihydro-1H-1,2,4-triazol-5-oneCatalog No.:AA01A970 CAS No.:1016771-84-8 MDL No.:MFCD09933328 MF:C8H8N4O MW:176.1753 |
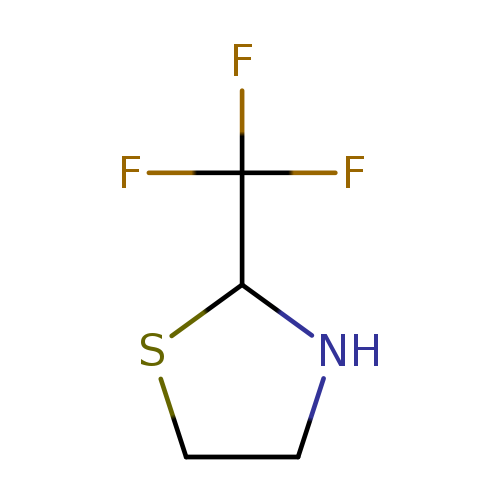
2-(trifluoromethyl)-1,3-thiazolidineCatalog No.:AA01B6VJ CAS No.:1016771-85-9 MDL No.:MFCD09934579 MF:C4H6F3NS MW:157.1573 |
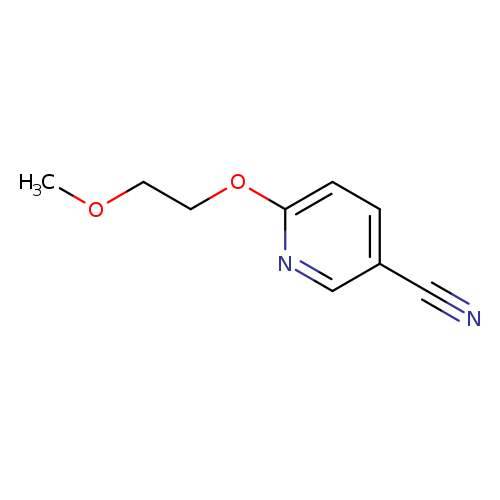
6-(2-Methoxyethoxy)pyridine-3-carbonitrileCatalog No.:AA01A3N9 CAS No.:1016772-36-3 MDL No.:MFCD09933599 MF:C9H10N2O2 MW:178.1879 |
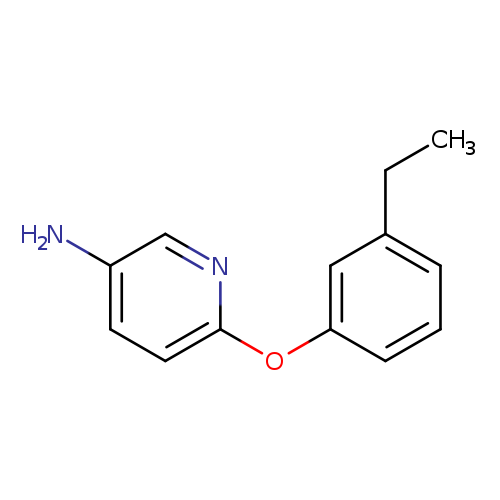
6-(3-Ethylphenoxy)pyridin-3-amineCatalog No.:AA019UNA CAS No.:1016772-75-0 MDL No.:MFCD09934445 MF:C13H14N2O MW:214.2631 |
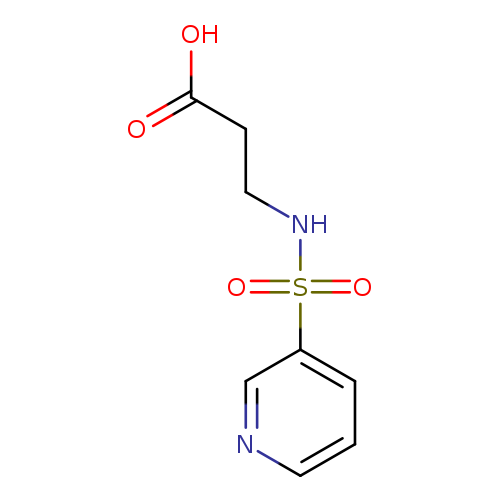
3-(Pyridine-3-sulfonamido)propanoic acidCatalog No.:AA019LJ1 CAS No.:1016775-40-8 MDL No.:MFCD09933429 MF:C8H10N2O4S MW:230.2410 |
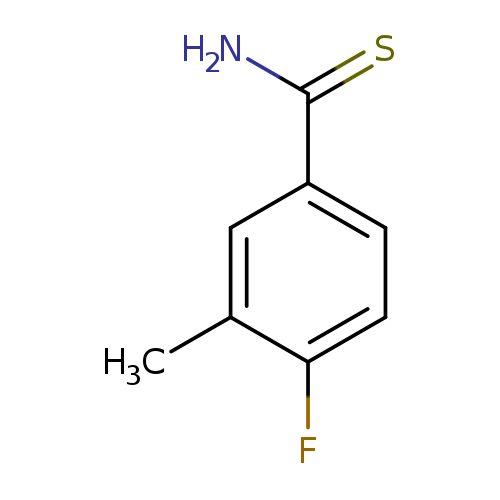
4-Fluoro-3-methylbenzenecarbothioamideCatalog No.:AA00H9GM CAS No.:1016775-44-2 MDL No.:MFCD09757567 MF:C8H8FNS MW:169.2192 |
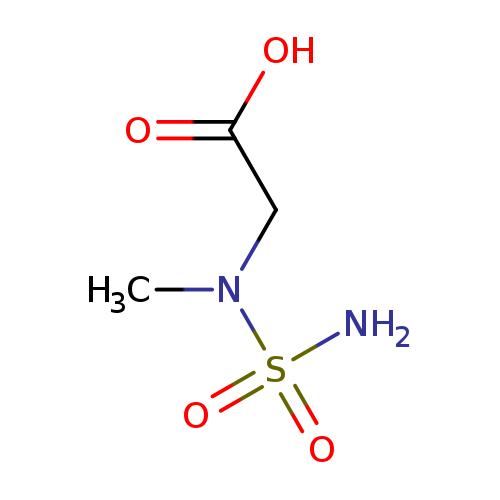
[(Aminosulfonyl)(methyl)amino]acetic acidCatalog No.:AA019N51 CAS No.:1016775-64-6 MDL No.:MFCD09935151 MF:C3H8N2O4S MW:168.1716 |
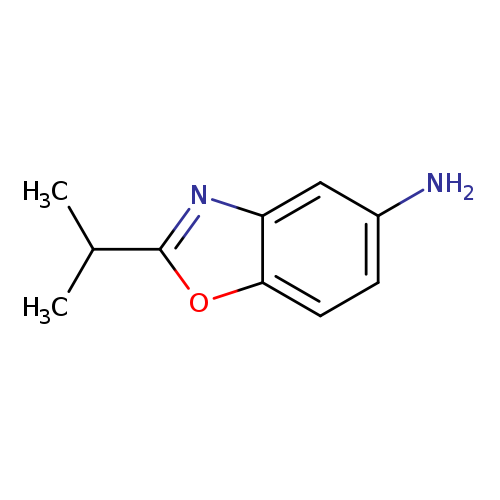
5-Benzoxazolamine, 2-(1-methylethyl)-Catalog No.:AA000549 CAS No.:1016776-39-8 MDL No.:MFCD09934235 MF:C10H12N2O MW:176.2151 |
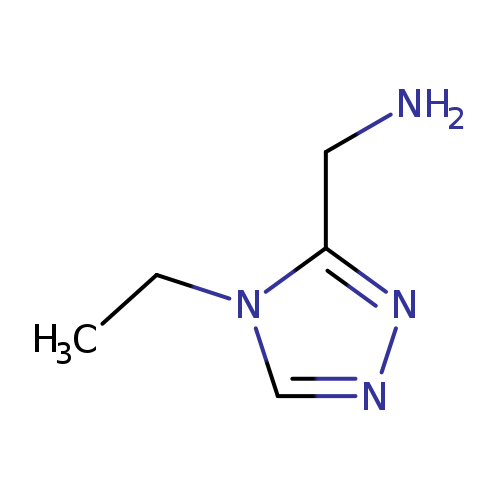
(4-ethyl-4H-1,2,4-triazol-3-yl)methanamineCatalog No.:AA019UA8 CAS No.:1016776-40-1 MDL No.:MFCD08456980 MF:C5H10N4 MW:126.1597 |
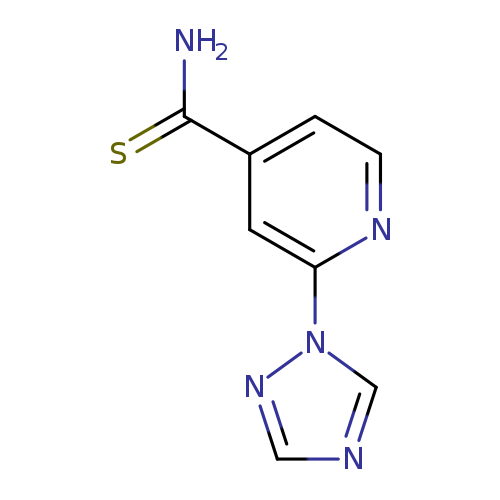
2-(1H-1,2,4-triazol-1-yl)pyridine-4-carbothioamideCatalog No.:AA019MLL CAS No.:1016776-89-8 MDL No.:MFCD09933631 MF:C8H7N5S MW:205.2397 |
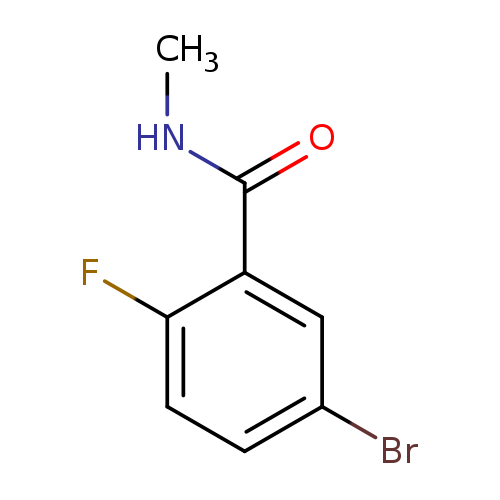
5-Bromo-2-fluoro-n-methylbenzamideCatalog No.:AA00H9GO CAS No.:1016777-86-8 MDL No.:MFCD09934823 MF:C8H7BrFNO MW:232.0497 |
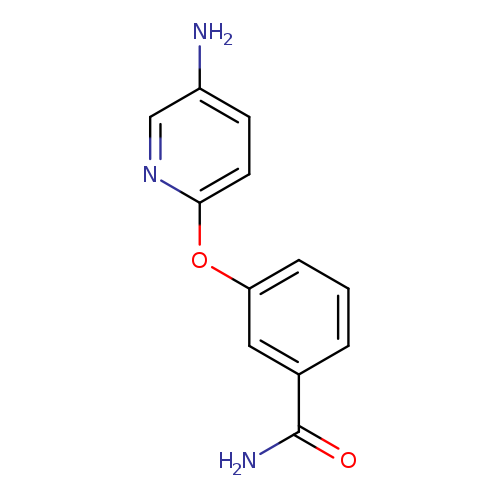
3-[(5-aminopyridin-2-yl)oxy]benzamideCatalog No.:AA01A8OX CAS No.:1016778-72-5 MDL No.:MFCD09933308 MF:C12H11N3O2 MW:229.2346 |
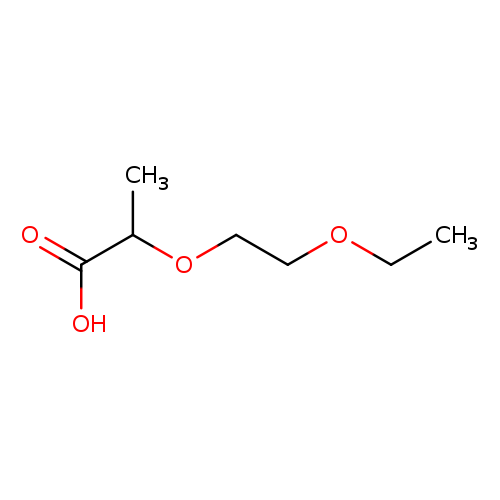
2-(2-Ethoxyethoxy)propanoic acidCatalog No.:AA019WF9 CAS No.:1016779-46-6 MDL No.:MFCD09933427 MF:C7H14O4 MW:162.1837 |
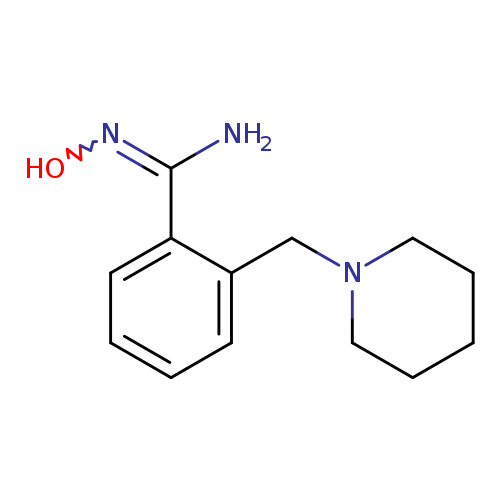
N'-hydroxy-2-(piperidin-1-ylmethyl)benzene-1-carboximidamideCatalog No.:AA01A7G0 CAS No.:1016780-05-4 MDL No.:MFCD16572287 MF:C13H19N3O MW:233.3095 |
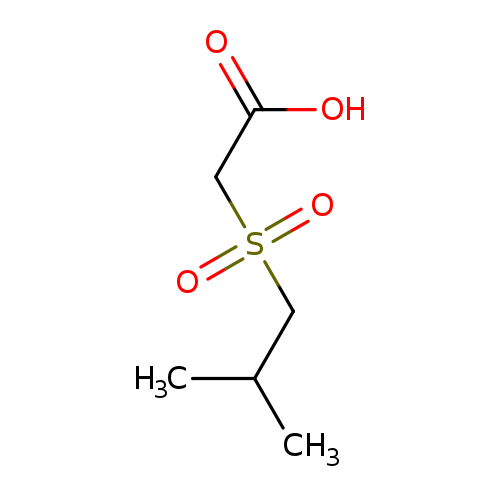
2-(2-methylpropanesulfonyl)acetic acidCatalog No.:AA019UL2 CAS No.:1016780-08-7 MDL No.:MFCD09935247 MF:C6H12O4S MW:180.2221 |
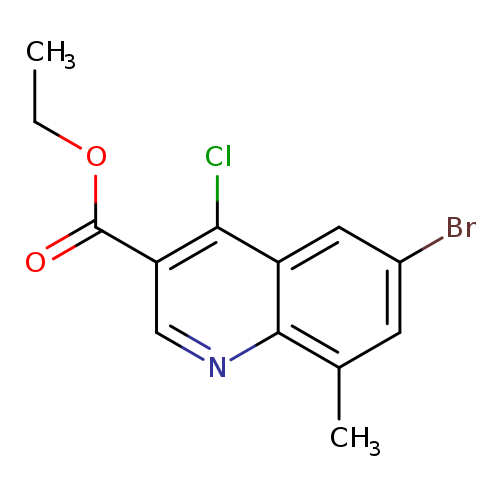
Ethyl 6-bromo-4-chloro-8-methylquinoline-3-carboxylateCatalog No.:AA0093E6 CAS No.:1016780-82-7 MDL No.:MFCD09933645 MF:C13H11BrClNO2 MW:328.5889 |
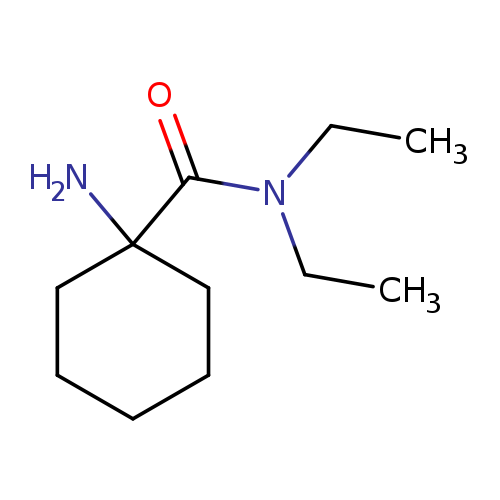
1-amino-N,N-diethylcyclohexane-1-carboxamideCatalog No.:AA019WG5 CAS No.:1016782-22-1 MDL No.:MFCD09935128 MF:C11H22N2O MW:198.3052 |
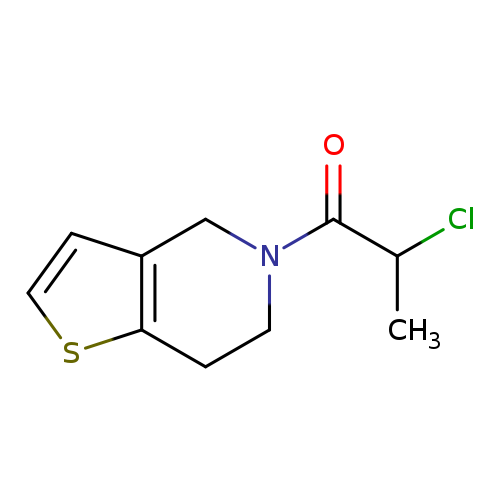
2-chloro-1-{4H,5H,6H,7H-thieno[3,2-c]pyridin-5-yl}propan-1-oneCatalog No.:AA019KPL CAS No.:1016783-06-4 MDL No.:MFCD09935285 MF:C10H12ClNOS MW:229.7264 |
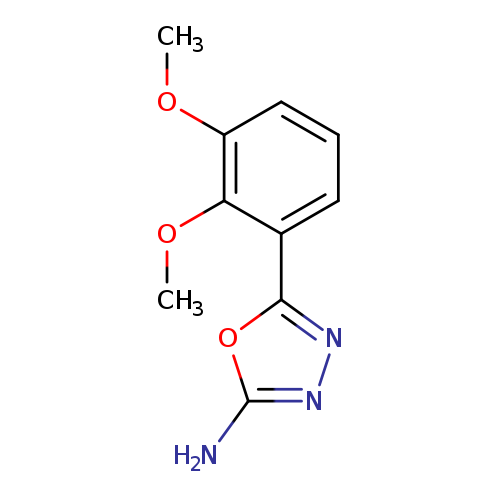
5-(2,3-dimethoxyphenyl)-1,3,4-oxadiazol-2-amineCatalog No.:AA01AAD6 CAS No.:1016783-13-3 MDL No.:MFCD09933173 MF:C10H11N3O3 MW:221.2126 |
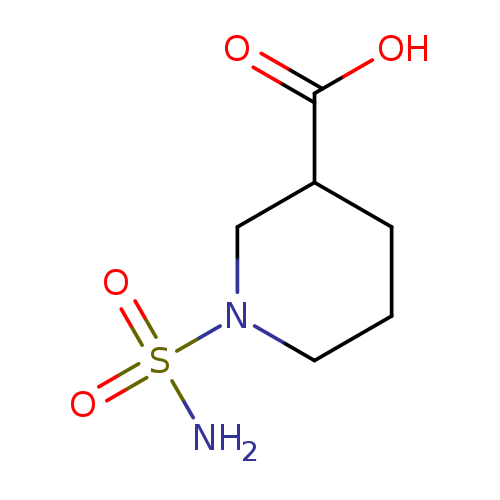
1-Sulfamoylpiperidine-3-carboxylic acidCatalog No.:AA01AL38 CAS No.:1016784-46-5 MDL No.:MFCD09933431 MF:C6H12N2O4S MW:208.2355 |
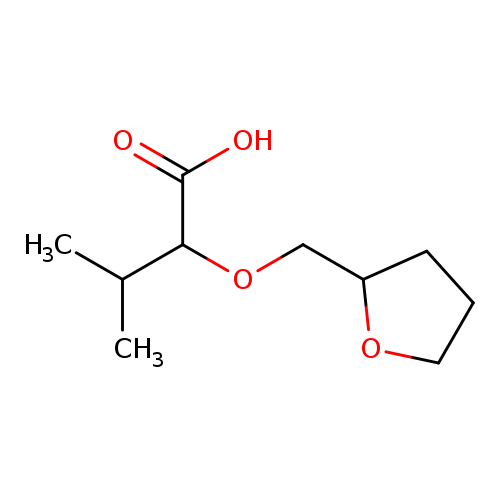
3-methyl-2-(oxolan-2-ylmethoxy)butanoic acidCatalog No.:AA01A7HP CAS No.:1016785-00-4 MDL No.:MFCD09941758 MF:C10H18O4 MW:202.2475 |
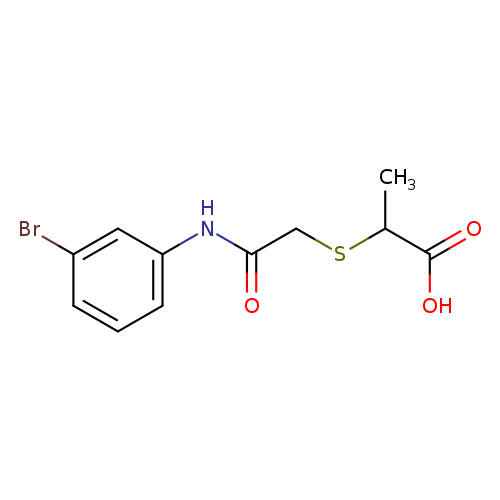
2-((2-[(3-Bromophenyl)amino]-2-oxoethyl)thio)propanoic acidCatalog No.:AA019N75 CAS No.:1016786-75-6 MDL No.:MFCD09946366 MF:C11H12BrNO3S MW:318.1869 |
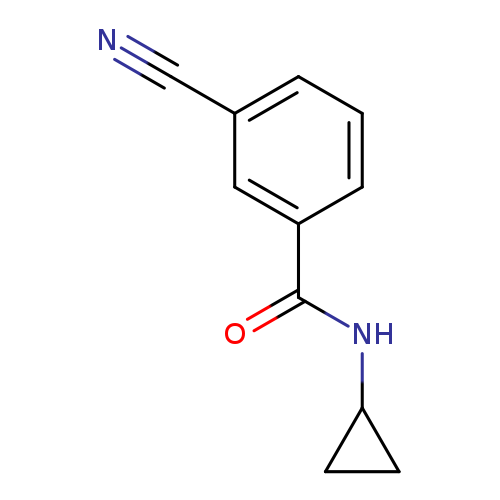
3-Cyano-N-cyclopropylbenzamideCatalog No.:AA00H9GP CAS No.:1016788-06-9 MDL No.:MFCD09946575 MF:C11H10N2O MW:186.2099 |
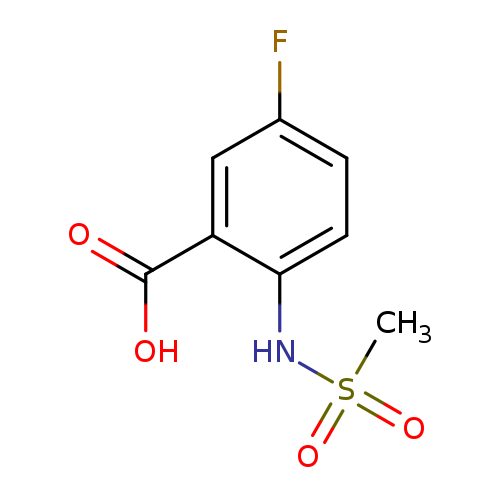
5-Fluoro-2-(methylsulfonamido)benzoic acidCatalog No.:AA000548 CAS No.:1016788-23-0 MDL No.:MFCD09949279 MF:C8H8FNO4S MW:233.2168 |
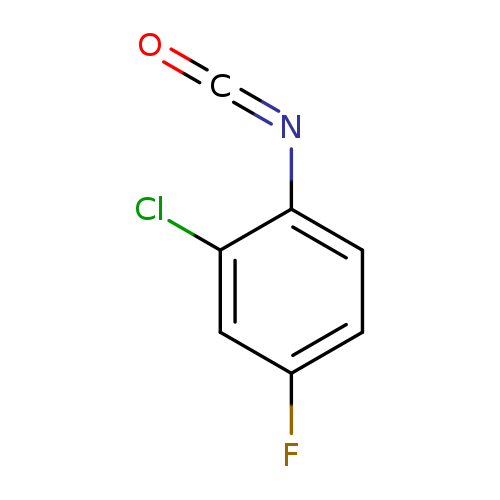
2-chloro-4-fluoro-1-isocyanatobenzeneCatalog No.:AA019UH9 CAS No.:1016788-39-8 MDL No.:MFCD09946620 MF:C7H3ClFNO MW:171.5562 |
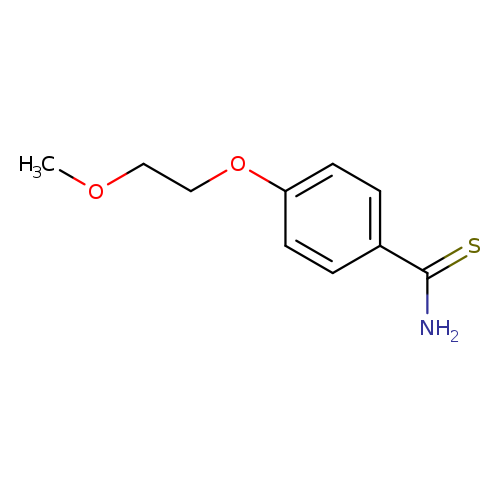
4-(2-methoxyethoxy)benzene-1-carbothioamideCatalog No.:AA01A201 CAS No.:1016788-46-7 MDL No.:MFCD09942523 MF:C10H13NO2S MW:211.2807 |
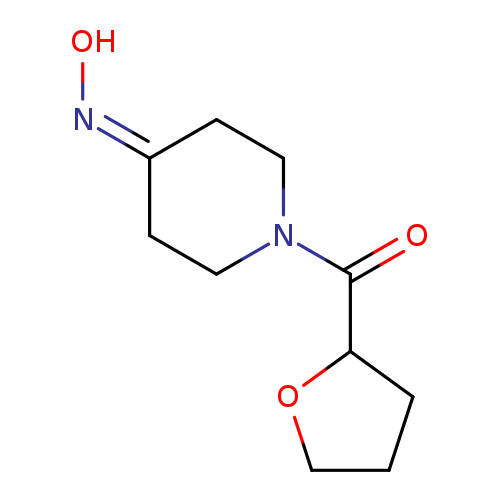
N-[1-(oxolane-2-carbonyl)piperidin-4-ylidene]hydroxylamineCatalog No.:AA019LX8 CAS No.:1016789-76-6 MDL No.:MFCD09935502 MF:C10H16N2O3 MW:212.2456 |
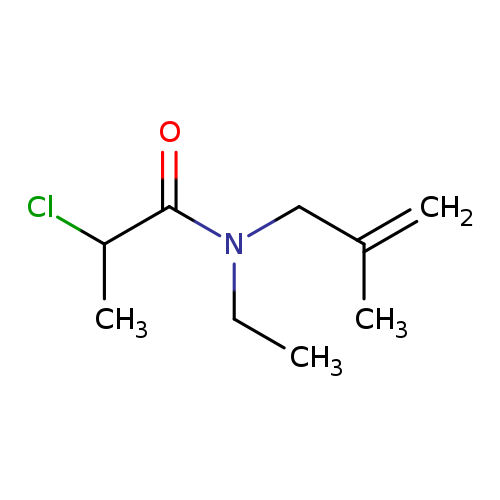
2-Chloro-n-ethyl-n-(2-methylprop-2-en-1-yl)propanamideCatalog No.:AA019KOW CAS No.:1016790-17-2 MDL No.:MFCD09942842 MF:C9H16ClNO MW:189.6824 |
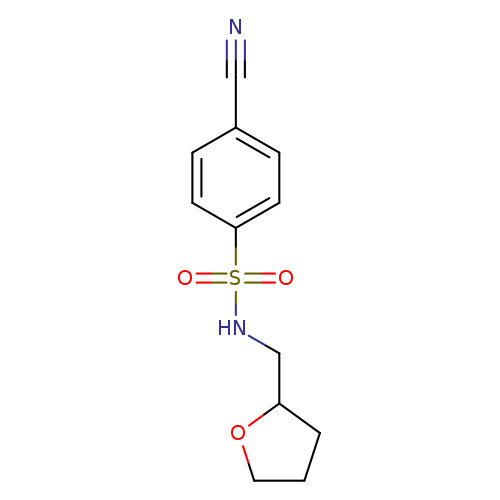
4-Cyano-n-(oxolan-2-ylmethyl)benzene-1-sulfonamideCatalog No.:AA019UE0 CAS No.:1016790-21-8 MDL No.:MFCD09942846 MF:C12H14N2O3S MW:266.3162 |
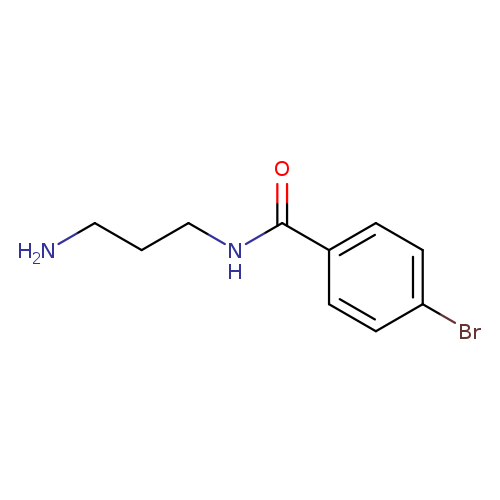
N-(3-aminopropyl)-4-bromobenzamideCatalog No.:AA01A9BE CAS No.:1016790-51-4 MDL No.:MFCD01069850 MF:C10H13BrN2O MW:257.1270 |
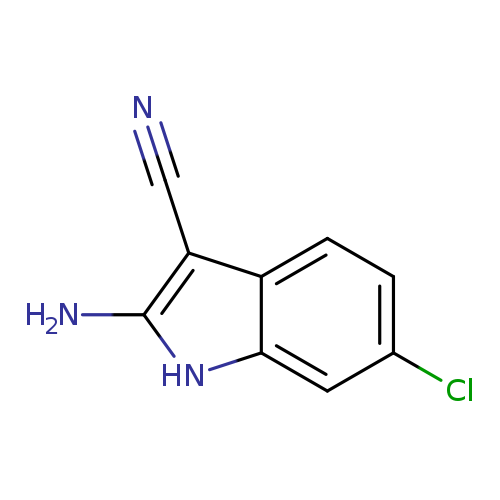
2-amino-6-chloro-1H-indole-3-carbonitrileCatalog No.:AA01A97H CAS No.:1016791-11-9 MDL No.:MFCD09935690 MF:C9H6ClN3 MW:191.6170 |
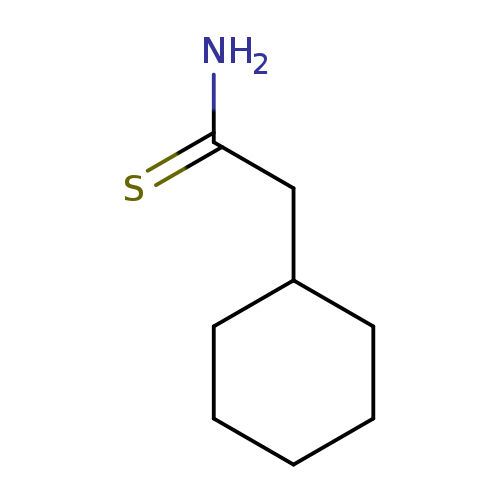
2-cyclohexylethanethioamideCatalog No.:AA019QZZ CAS No.:1016791-35-7 MDL No.:MFCD09935702 MF:C8H15NS MW:157.2764 |
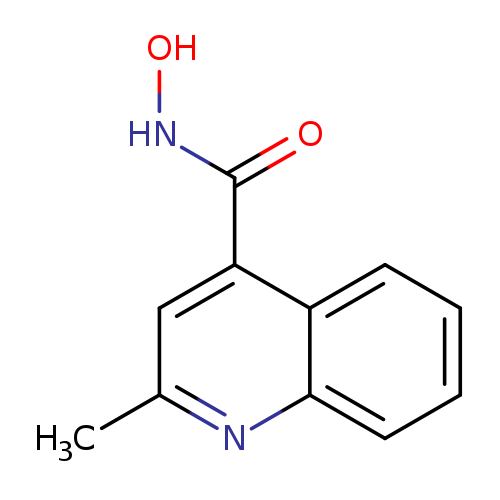
N-hydroxy-2-methylquinoline-4-carboxamideCatalog No.:AA01BAAZ CAS No.:1016791-55-1 MDL No.:MFCD09935718 MF:C11H10N2O2 MW:202.2093 |
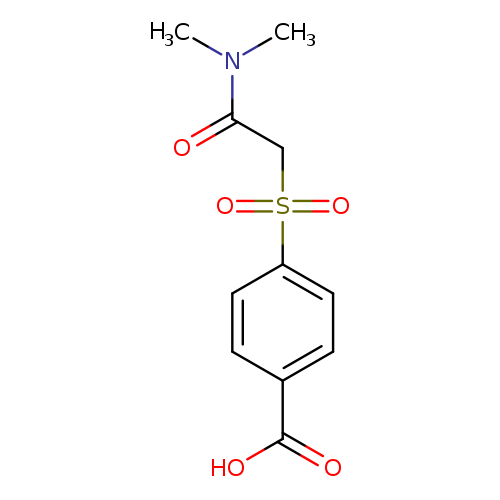
4-[(dimethylcarbamoyl)methanesulfonyl]benzoic acidCatalog No.:AA019S2G CAS No.:1016792-92-9 MDL No.:MFCD09951225 MF:C11H13NO5S MW:271.2896 |
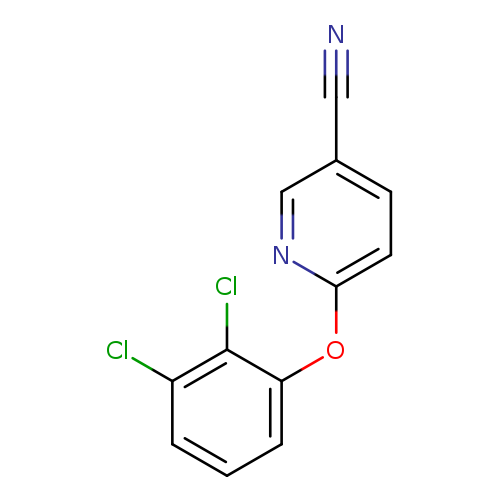
6-(2,3-Dichlorophenoxy)pyridine-3-carbonitrileCatalog No.:AA019T8X CAS No.:1016793-12-6 MDL No.:MFCD09951261 MF:C12H6Cl2N2O MW:265.0948 |
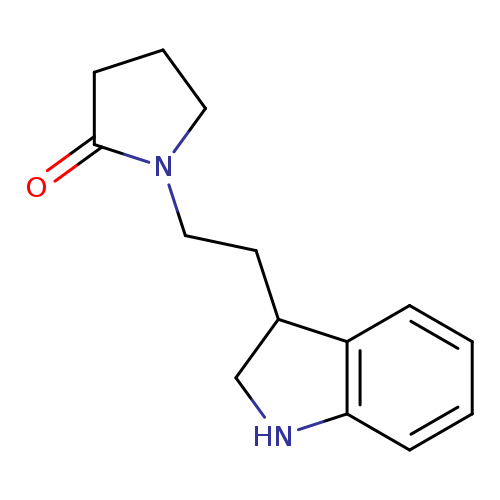
1-[2-(2,3-dihydro-1H-indol-3-yl)ethyl]pyrrolidin-2-oneCatalog No.:AA019YNE CAS No.:1016794-28-7 MDL No.:MFCD09939360 MF:C14H18N2O MW:230.3055 |
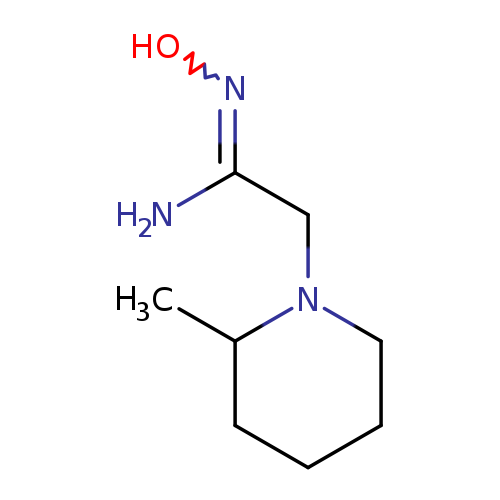
N'-hydroxy-2-(2-methylpiperidin-1-yl)ethanimidamideCatalog No.:AA019UZN CAS No.:1016794-51-6 MDL No.:MFCD11858097 MF:C8H17N3O MW:171.2401 |
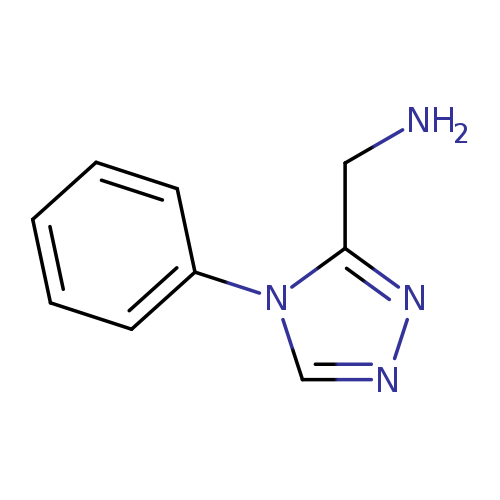
(4-Phenyl-4H-1,2,4-triazol-3-yl)methanamine dihydrochlorideCatalog No.:AA00VRYB CAS No.:1016794-97-0 MDL No.:MFCD09940257 MF:C9H10N4 MW:174.2025 |
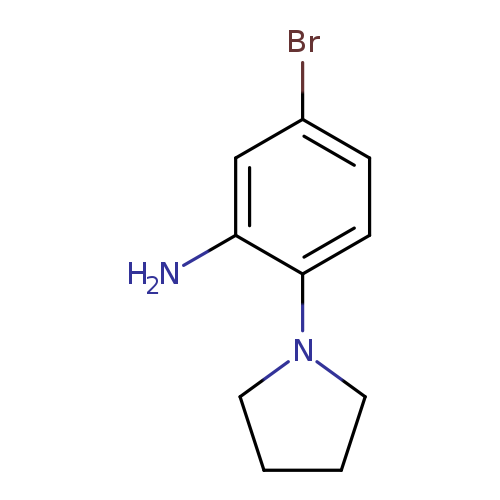
5-bromo-2-(pyrrolidin-1-yl)anilineCatalog No.:AA01A0E4 CAS No.:1016795-07-5 MDL No.:MFCD09939470 MF:C10H13BrN2 MW:241.1276 |
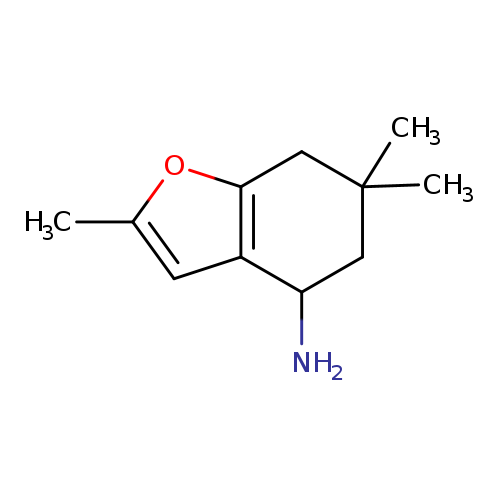
2,6,6-trimethyl-4,5,6,7-tetrahydro-1-benzofuran-4-amineCatalog No.:AA019UIV CAS No.:1016796-75-0 MDL No.:MFCD09936557 MF:C11H17NO MW:179.2588 |
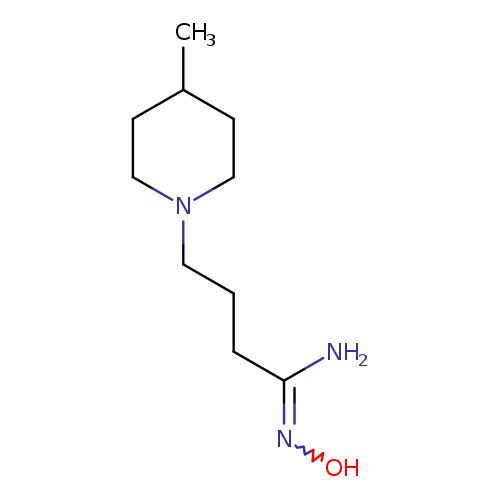
N'-hydroxy-4-(4-methylpiperidin-1-yl)butanimidamideCatalog No.:AA01A7WU CAS No.:1016797-22-0 MDL No.:MFCD09936637 MF:C10H21N3O MW:199.2932 |
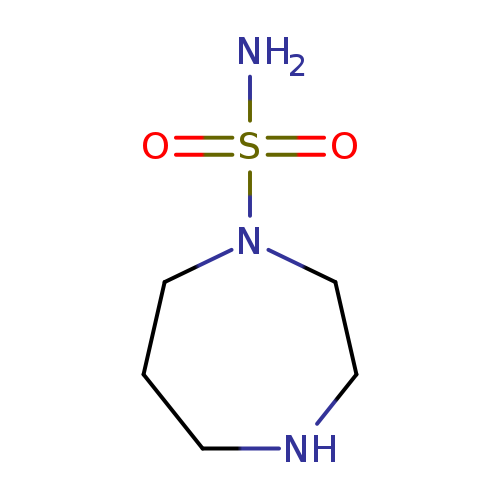
1,4-diazepane-1-sulfonamideCatalog No.:AA01AH4N CAS No.:1016797-88-8 MDL No.:MFCD09944819 MF:C5H13N3O2S MW:179.2406 |
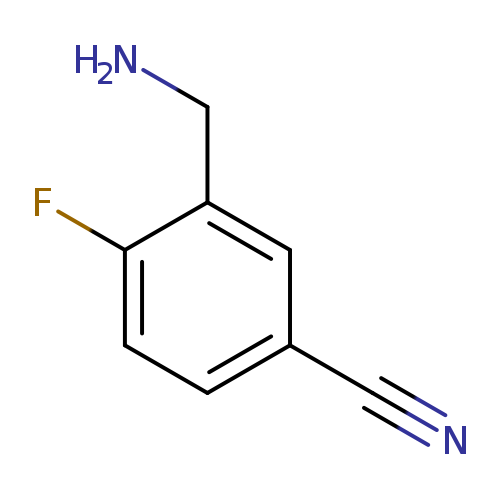
3-(aminomethyl)-4-fluorobenzonitrileCatalog No.:AA019R42 CAS No.:1016798-28-9 MDL No.:MFCD09947814 MF:C8H7FN2 MW:150.1530 |
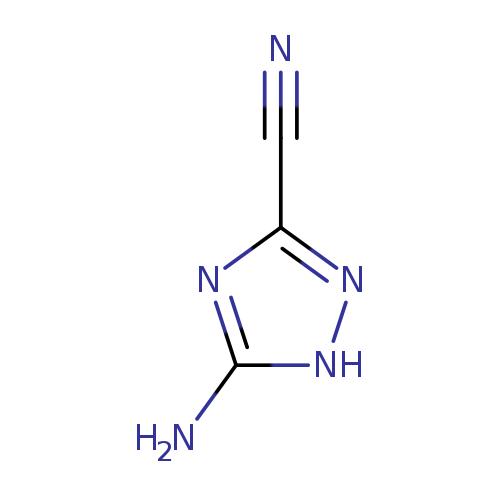
5-amino-1H-1,2,4-triazole-3-carbonitrileCatalog No.:AA01ABZY CAS No.:1016798-54-1 MDL No.:MFCD30090052 MF:C3H3N5 MW:109.0894 |
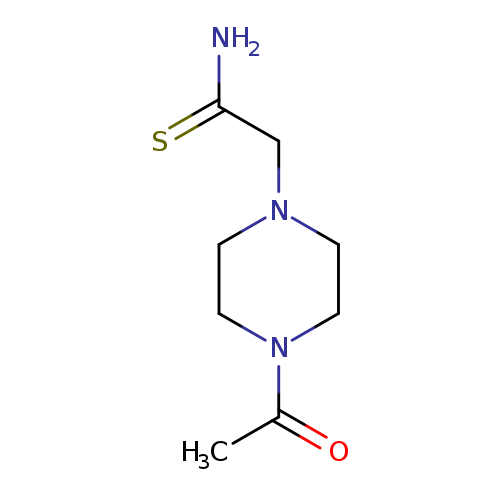
2-(4-Acetyl-piperazin-1-yl)-thioacetamideCatalog No.:AA018MJX CAS No.:1016799-62-4 MDL No.:MFCD09945165 MF:C8H15N3OS MW:201.2892 |
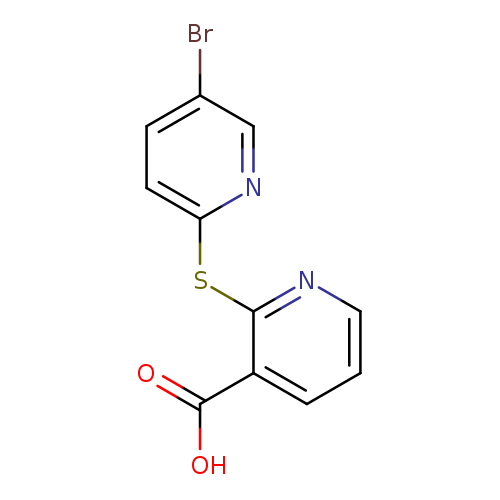
2-[(5-bromopyridin-2-yl)sulfanyl]pyridine-3-carboxylic acidCatalog No.:AA019WU5 CAS No.:1016799-66-8 MDL No.:MFCD09940986 MF:C11H7BrN2O2S MW:311.1545 |

Pyridine, 4-bromo-3-methyl-Catalog No.:AA00054H CAS No.:10168-00-0 MDL No.:MFCD01646140 MF:C6H6BrN MW:172.0225 |
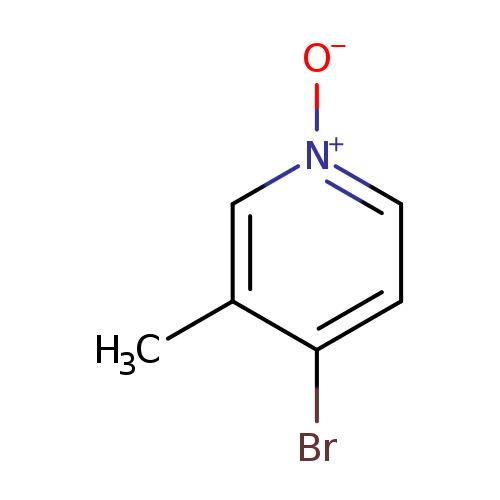
4-Bromo-3-methylpyridine 1-oxideCatalog No.:AA00054G CAS No.:10168-58-8 MDL No.:MFCD11870743 MF:C6H6BrNO MW:188.0219 |
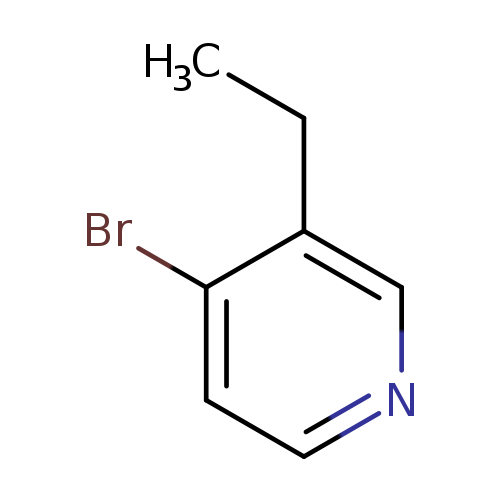
4-Bromo-3-ethylpyridine hydrobromideCatalog No.:AA00054F CAS No.:10168-60-2 MDL No.:MFCD19543895 MF:C7H8BrN MW:186.0491 |
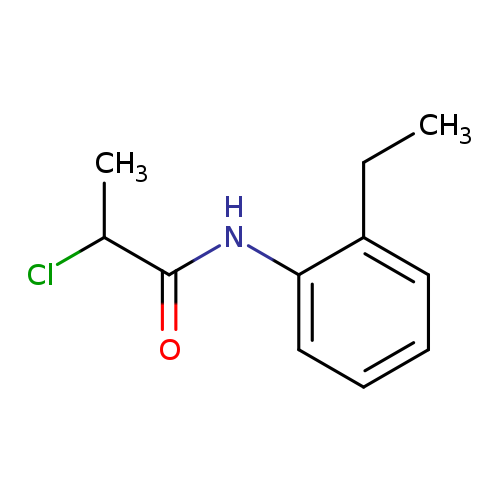
2-chloro-N-(2-ethylphenyl)propanamideCatalog No.:AA019KNA CAS No.:1016802-55-3 MDL No.:MFCD09949608 MF:C11H14ClNO MW:211.6880 |
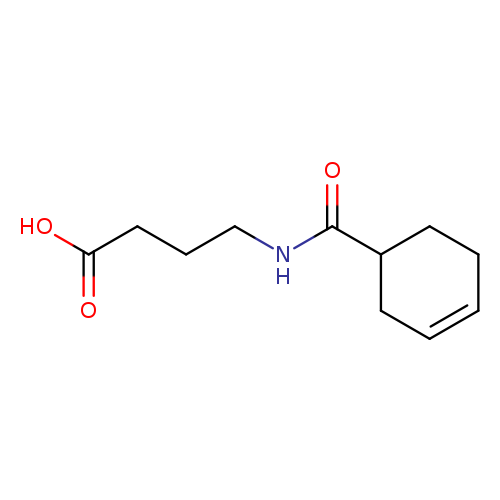
4-(cyclohex-3-en-1-ylformamido)butanoic acidCatalog No.:AA01B7QG CAS No.:1016804-29-7 MDL No.:MFCD09949909 MF:C11H17NO3 MW:211.2576 |
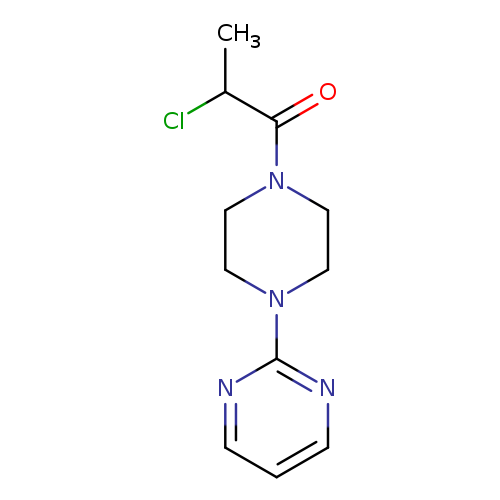
2-chloro-1-[4-(pyrimidin-2-yl)piperazin-1-yl]propan-1-oneCatalog No.:AA019KOU CAS No.:1016805-23-4 MDL No.:MFCD09946025 MF:C11H15ClN4O MW:254.7160 |
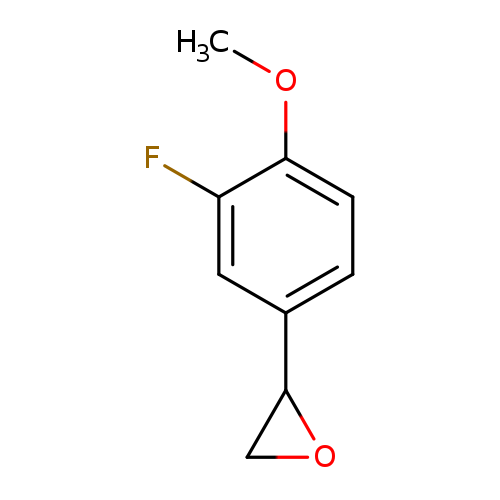
2-(3-fluoro-4-methoxyphenyl)oxiraneCatalog No.:AA019WIW CAS No.:1016806-32-8 MDL No.:MFCD09937964 MF:C9H9FO2 MW:168.1650 |
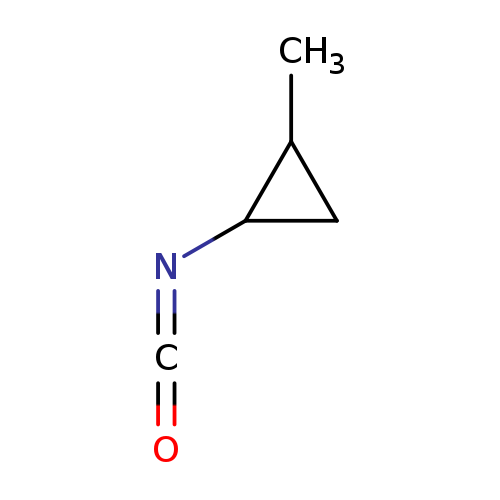
1-Isocyanato-2-methylcyclopropaneCatalog No.:AA01A1EN CAS No.:1016807-38-7 MDL No.:MFCD09950508 MF:C5H7NO MW:97.1152 |
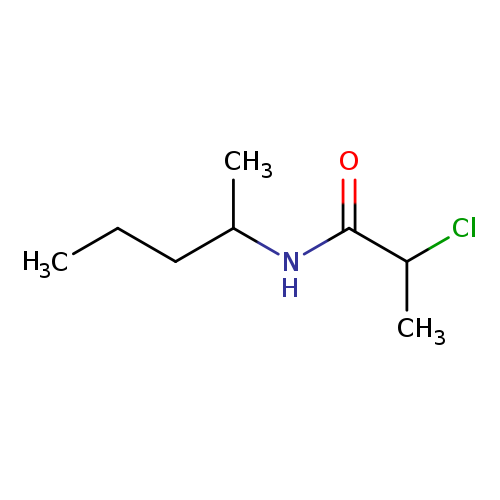
2-Chloro-n-(pentan-2-yl)propanamideCatalog No.:AA019KN2 CAS No.:1016808-29-9 MDL No.:MFCD09950655 MF:C8H16ClNO MW:177.6717 |
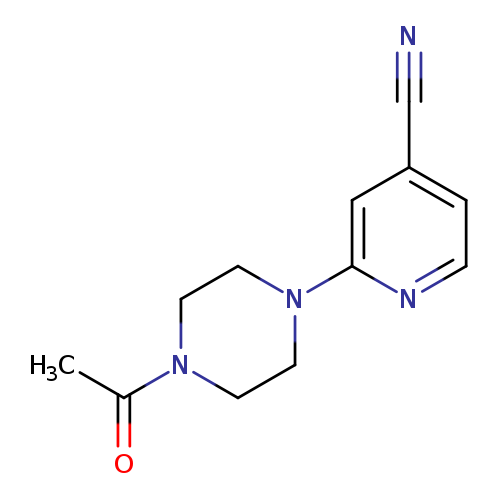
2-(4-acetylpiperazin-1-yl)pyridine-4-carbonitrileCatalog No.:AA019MOV CAS No.:1016809-01-0 MDL No.:MFCD09950750 MF:C12H14N4O MW:230.2658 |
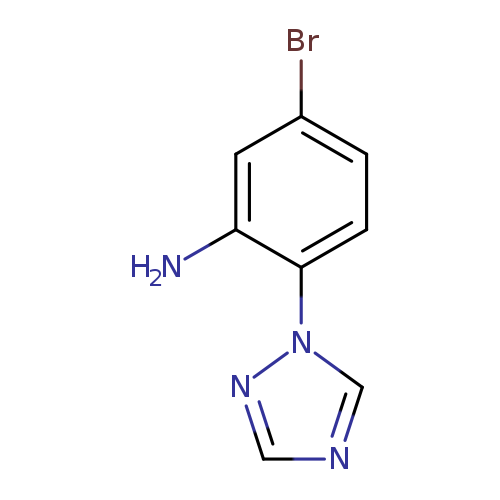
5-bromo-2-(1H-1,2,4-triazol-1-yl)anilineCatalog No.:AA01A854 CAS No.:1016809-06-5 MDL No.:MFCD09946703 MF:C8H7BrN4 MW:239.0720 |
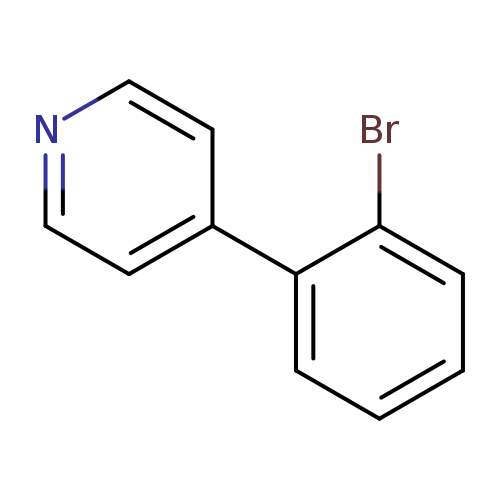
4-(2-Bromophenyl)pyridineCatalog No.:AA0091Z8 CAS No.:101681-34-9 MDL No.:MFCD18415515 MF:C11H8BrN MW:234.0919 |
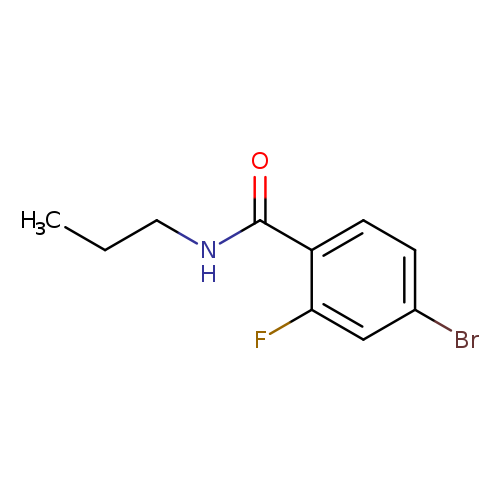
Propyl 4-bromo-2-fluorobenzamideCatalog No.:AA000540 CAS No.:1016811-41-8 MDL No.:MFCD09935765 MF:C10H11BrFNO MW:260.1028 |
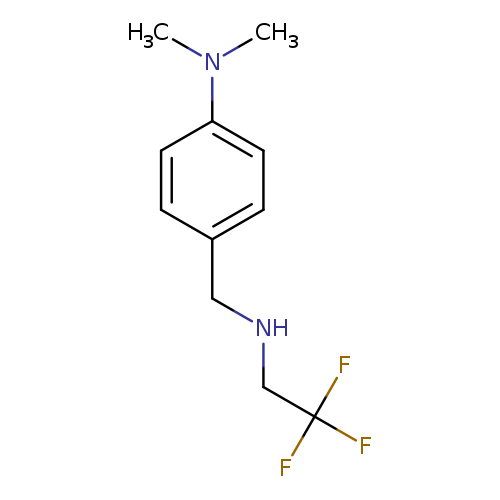
N,N-dimethyl-4-{[(2,2,2-trifluoroethyl)amino]methyl}anilineCatalog No.:AA01AAOP CAS No.:1016811-90-7 MDL No.:MFCD09938951 MF:C11H15F3N2 MW:232.2454 |
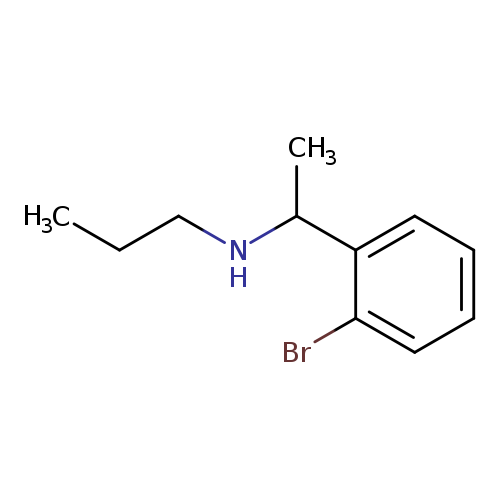
[1-(2-Bromophenyl)ethyl](propyl)amineCatalog No.:AA019UTW CAS No.:1016812-00-2 MDL No.:MFCD09935872 MF:C11H16BrN MW:242.1554 |
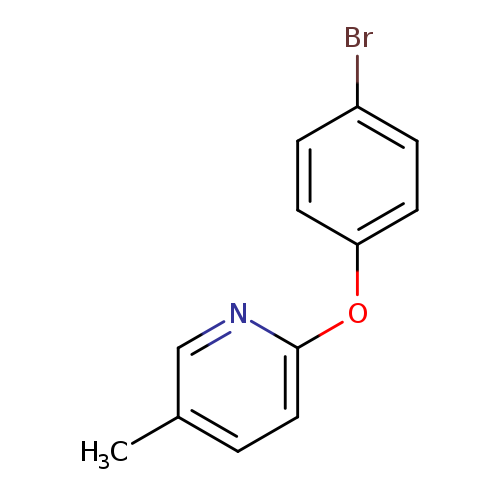
2-(4-bromophenoxy)-5-methylpyridineCatalog No.:AA019UAC CAS No.:1016812-08-0 MDL No.:MFCD09938980 MF:C12H10BrNO MW:264.1179 |
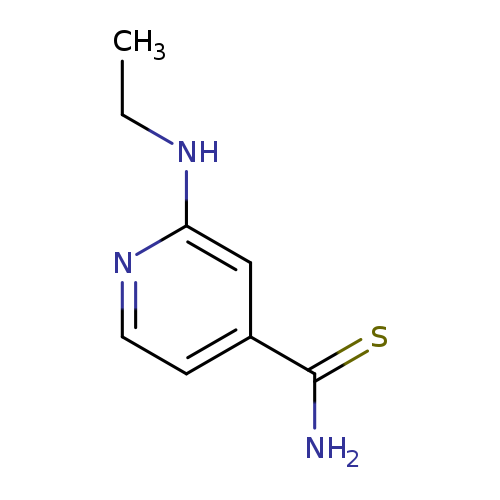
2-(ethylamino)pyridine-4-carbothioamideCatalog No.:AA01B9HG CAS No.:1016812-14-8 MDL No.:MFCD09938989 MF:C8H11N3S MW:181.2580 |
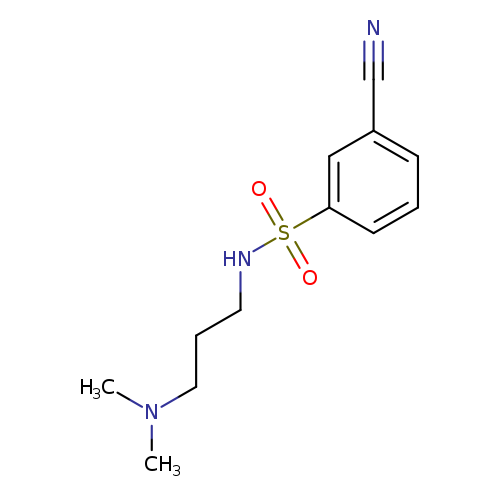
3-cyano-N-[3-(dimethylamino)propyl]benzene-1-sulfonamideCatalog No.:AA019VVQ CAS No.:1016813-13-0 MDL No.:MFCD09943317 MF:C12H17N3O2S MW:267.3473 |
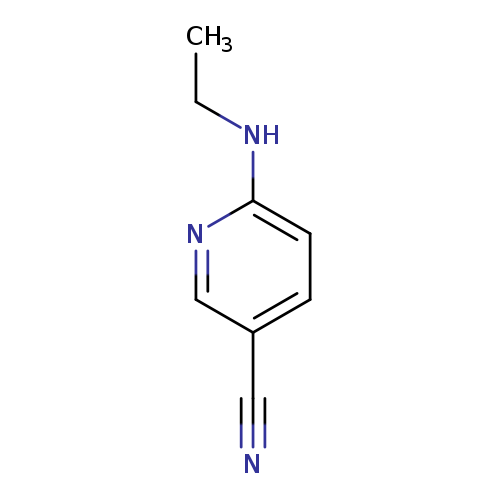
6-(Ethylamino)pyridine-3-carbonitrileCatalog No.:AA00948R CAS No.:1016813-34-5 MDL No.:MFCD09940529 MF:C8H9N3 MW:147.1772 |
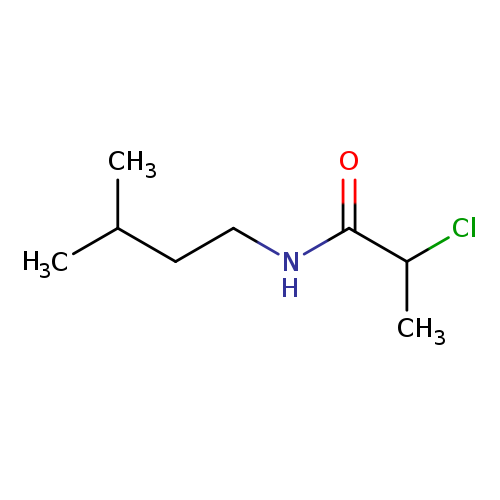
2-chloro-N-(3-methylbutyl)propanamideCatalog No.:AA019KMR CAS No.:1016813-78-7 MDL No.:MFCD09947369 MF:C8H16ClNO MW:177.6717 |
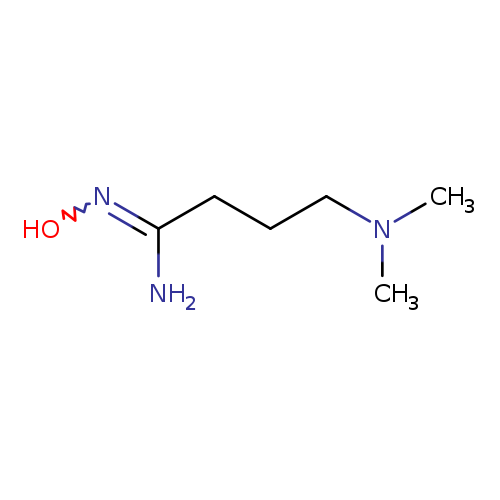
4-(dimethylamino)-N'-hydroxybutanimidamideCatalog No.:AA01A13L CAS No.:1016814-60-0 MDL No.:MFCD09940729 MF:C6H15N3O MW:145.2028 |
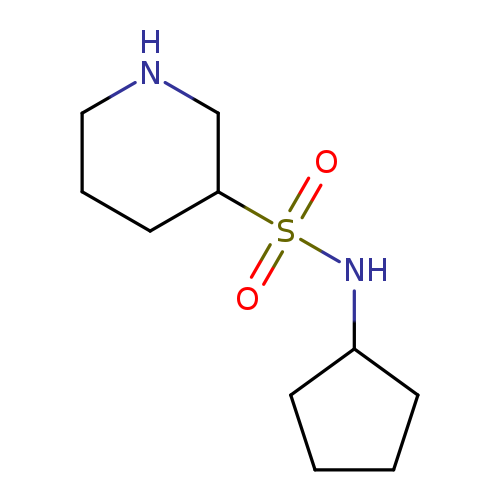
N-cyclopentylpiperidine-3-sulfonamideCatalog No.:AA01A62H CAS No.:1016814-99-5 MDL No.:MFCD09936747 MF:C10H20N2O2S MW:232.3430 |
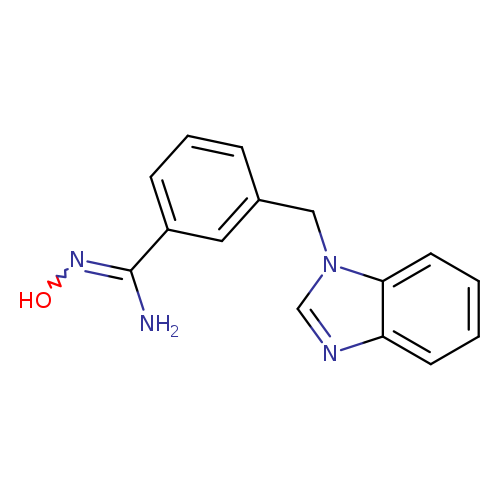
3-(1H-1,3-benzodiazol-1-ylmethyl)-N'-hydroxybenzene-1-carboximidamideCatalog No.:AA01A7FZ CAS No.:1016815-21-6 MDL No.:MFCD14705695 MF:C15H14N4O MW:266.2979 |
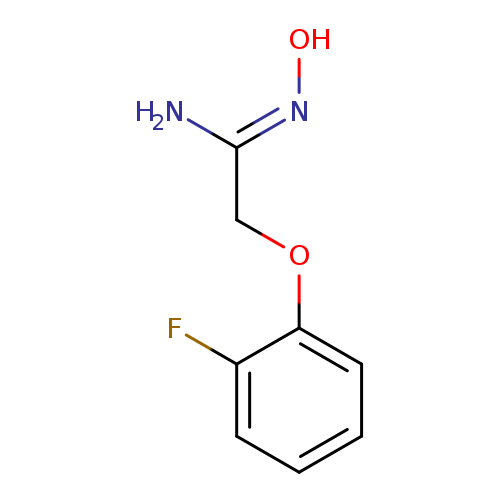
(Z)-2-(2-fluorophenoxy)-N'-hydroxyethenimidamideCatalog No.:AA00VVO8 CAS No.:1016817-25-6 MDL No.:MFCD09939824 MF:C8H9FN2O2 MW:184.1677 |
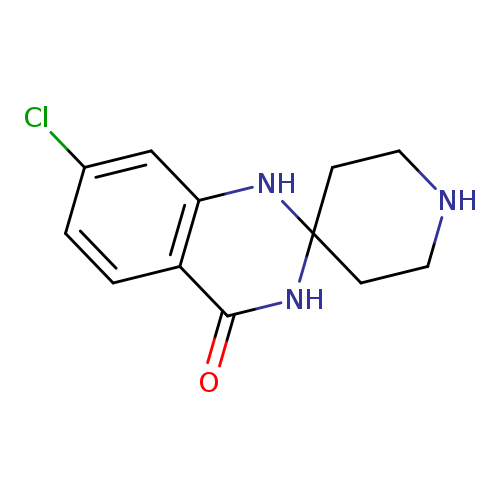
7'-Chloro-1'h-spiro[piperidine-4,2'-quinazolin]-4'(3'h)-oneCatalog No.:AA00H9GT CAS No.:1016818-09-9 MDL No.:MFCD10455857 MF:C12H14ClN3O MW:251.7121 |
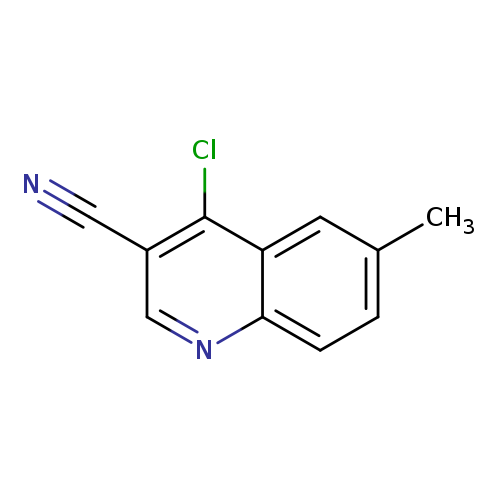
4-Chloro-6-methylquinoline-3-carbonitrileCatalog No.:AA00053V CAS No.:1016818-79-3 MDL No.:MFCD09948299 MF:C11H7ClN2 MW:202.6397 |
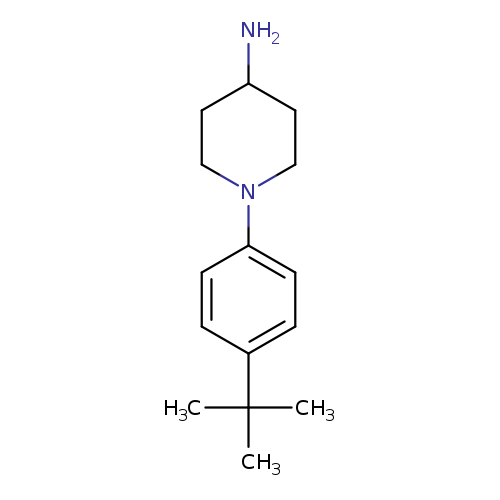
1-(4-tert-Butylphenyl)piperidin-4-amineCatalog No.:AA019WY3 CAS No.:1016818-89-5 MDL No.:MFCD09948321 MF:C15H24N2 MW:232.3645 |
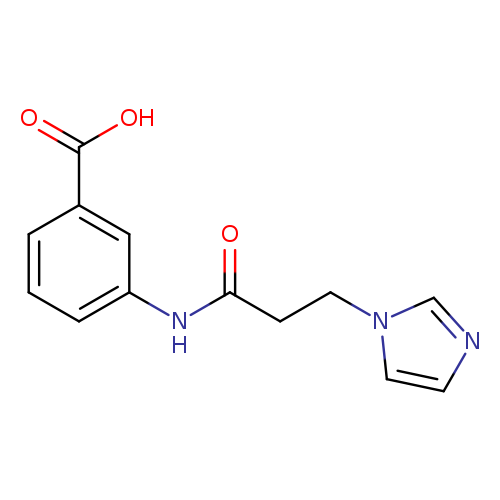
3-[3-(1H-Imidazol-1-yl)propanamido]benzoic acidCatalog No.:AA019VCS CAS No.:1016819-96-7 MDL No.:MFCD09945541 MF:C13H13N3O3 MW:259.2606 |
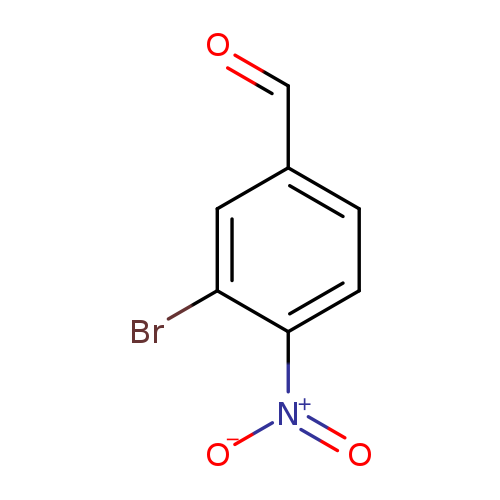
3-Bromo-4-nitrobenzaldehydeCatalog No.:AA00054A CAS No.:101682-68-2 MDL No.:MFCD00968939 MF:C7H4BrNO3 MW:230.0156 |
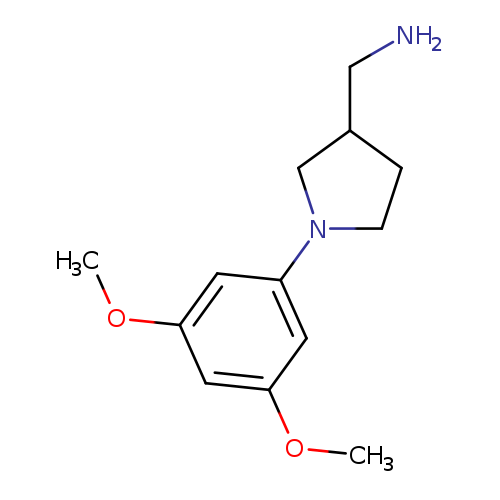
[1-(3,5-dimethoxyphenyl)pyrrolidin-3-yl]methanamineCatalog No.:AA019TXE CAS No.:1016820-27-1 MDL No.:MFCD09944506 MF:C13H20N2O2 MW:236.3101 |
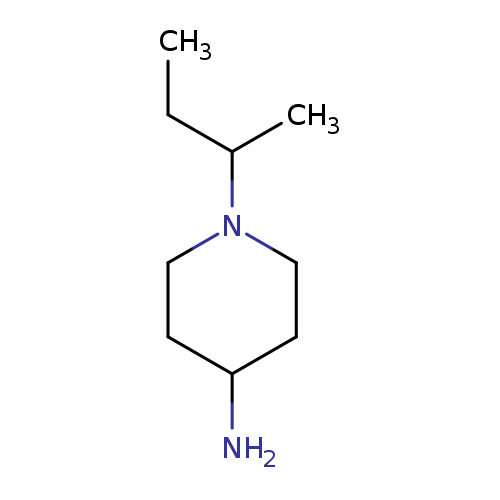
1-(butan-2-yl)piperidin-4-amineCatalog No.:AA01DX2J CAS No.:1016820-77-1 MDL No.:MFCD09945700 MF:C9H20N2 MW:156.2685 |
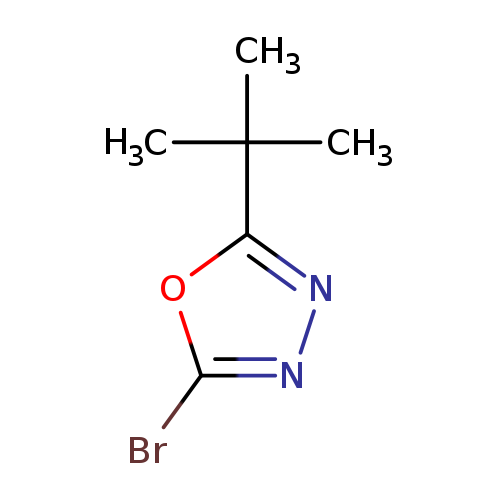
2-Bromo-5-(tert-butyl)-1,3,4-oxadiazoleCatalog No.:AA00VUIZ CAS No.:1016820-98-6 MDL No.:MFCD09947546 MF:C6H9BrN2O MW:205.0525 |
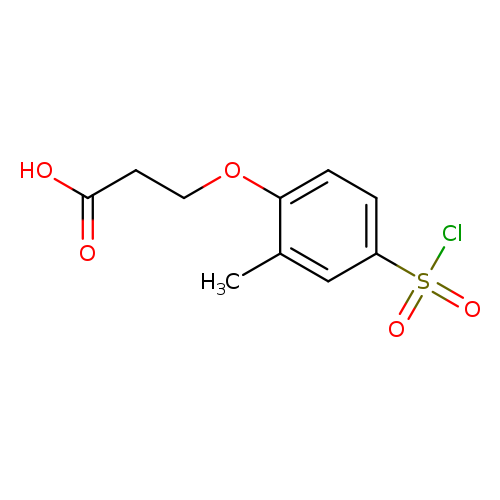
3-[4-(Chlorosulfonyl)-2-methylphenoxy]propanoic acidCatalog No.:AA01A7GA CAS No.:1016822-12-0 MDL No.:MFCD09944876 MF:C10H11ClO5S MW:278.7093 |
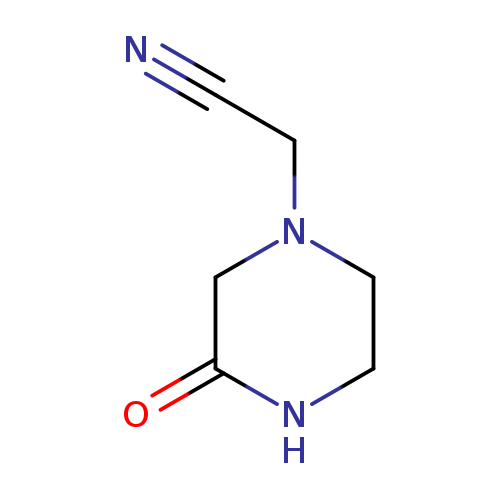
2-(3-oxopiperazin-1-yl)acetonitrileCatalog No.:AA01AJCD CAS No.:1016823-35-0 MDL No.:MFCD09944049 MF:C6H9N3O MW:139.1552 |
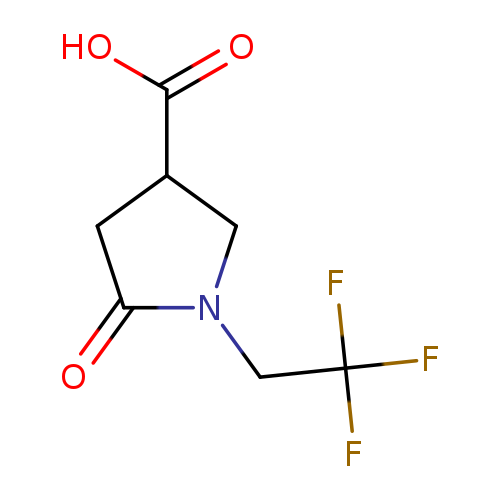
5-oxo-1-(2,2,2-trifluoroethyl)pyrrolidine-3-carboxylic acidCatalog No.:AA019M7W CAS No.:1016823-41-8 MDL No.:MFCD09944063 MF:C7H8F3NO3 MW:211.1385 |
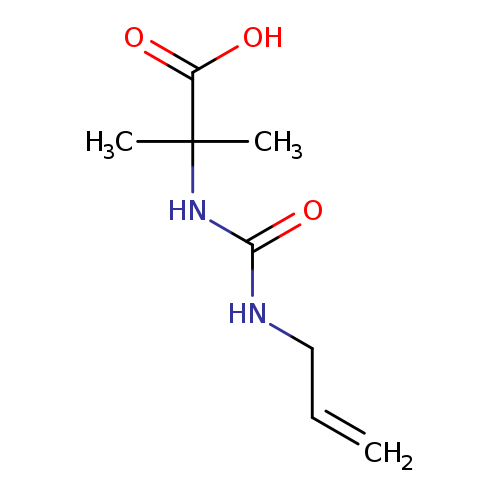
2-methyl-2-{[(prop-2-en-1-yl)carbamoyl]amino}propanoic acidCatalog No.:AA019WKL CAS No.:1016823-58-7 MDL No.:MFCD09941258 MF:C8H14N2O3 MW:186.2084 |
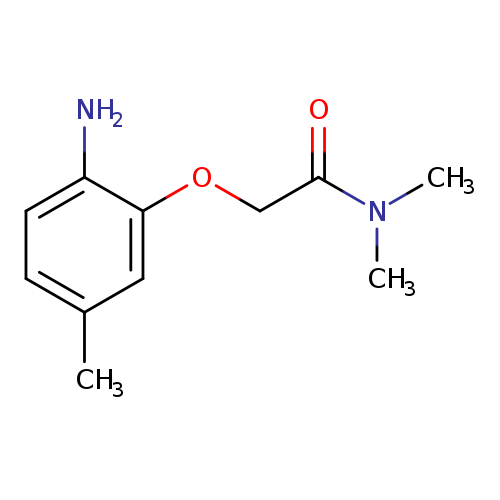
2-(2-amino-5-methylphenoxy)-N,N-dimethylacetamideCatalog No.:AA019WO2 CAS No.:1016824-15-9 MDL No.:MFCD09945239 MF:C11H16N2O2 MW:208.2569 |
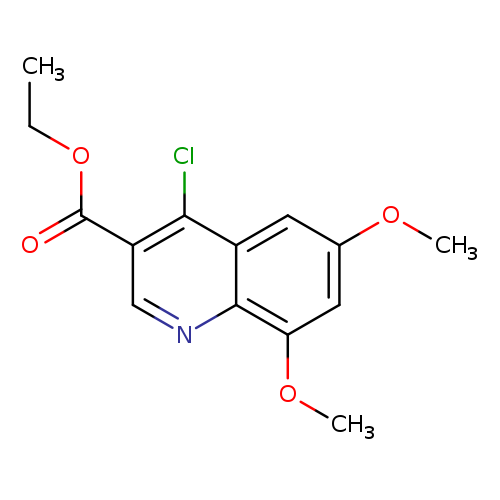
Ethyl 4-chloro-6,8-dimethoxyquinoline-3-carboxylateCatalog No.:AA019X70 CAS No.:1016825-43-6 MDL No.:MFCD09937855 MF:C14H14ClNO4 MW:295.7183 |
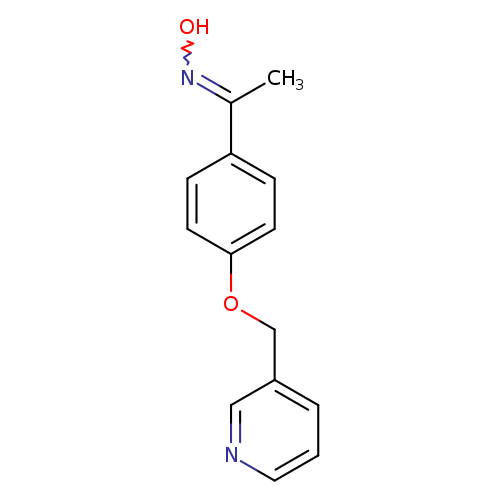
N-(1-[4-(Pyridin-3-ylmethoxy)phenyl]ethylidene)hydroxylamineCatalog No.:AA01A7Q1 CAS No.:1016826-30-4 MDL No.:MFCD09949618 MF:C14H14N2O2 MW:242.2732 |
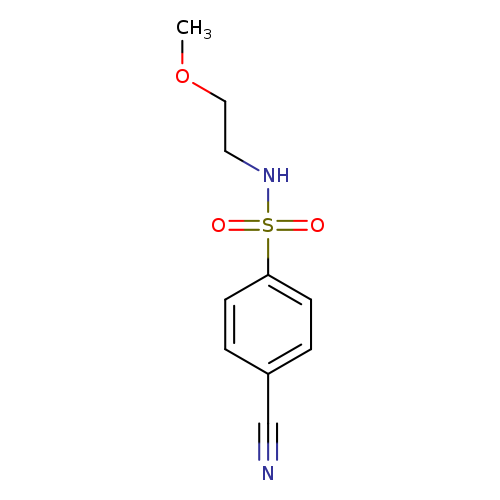
4-cyano-N-(2-methoxyethyl)benzene-1-sulfonamideCatalog No.:AA019UDZ CAS No.:1016826-53-1 MDL No.:MFCD09949671 MF:C10H12N2O3S MW:240.2789 |
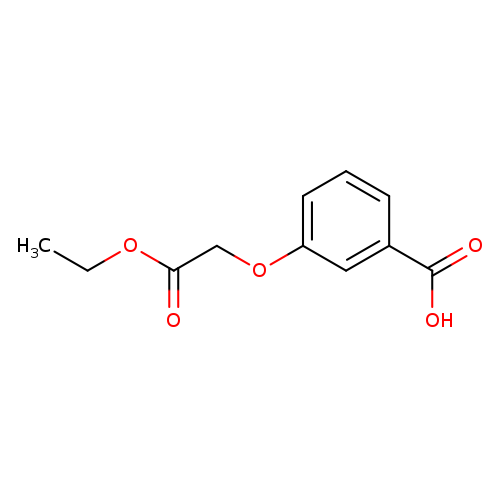
3-(2-Ethoxy-2-oxoethoxy)benzoic acidCatalog No.:AA019WJL CAS No.:1016826-94-0 MDL No.:MFCD09945858 MF:C11H12O5 MW:224.2100 |
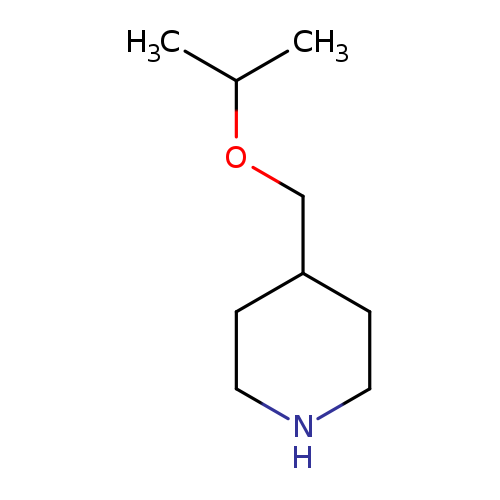
4-(Isopropoxymethyl)piperidineCatalog No.:AA008VEJ CAS No.:1016827-31-8 MDL No.:MFCD09948547 MF:C9H19NO MW:157.2533 |
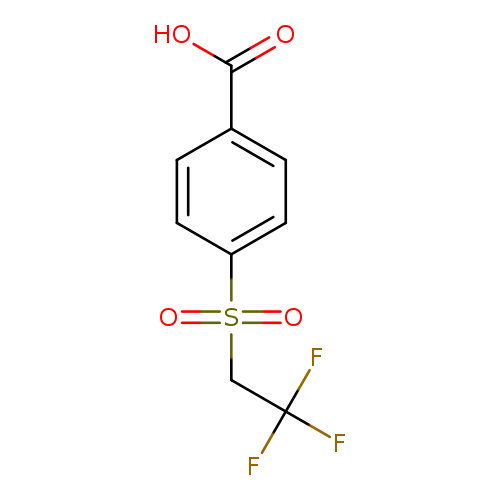
4-[(2,2,2-Trifluoroethane)sulfonyl]benzoic acidCatalog No.:AA019T2Y CAS No.:1016827-86-3 MDL No.:MFCD09942164 MF:C9H7F3O4S MW:268.2097 |
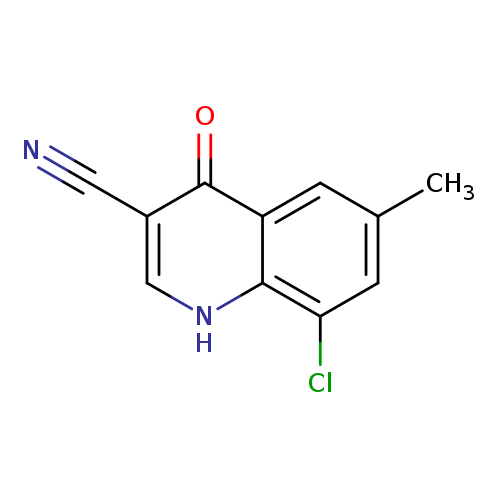
8-chloro-6-methyl-4-oxo-1,4-dihydroquinoline-3-carbonitrileCatalog No.:AA01EB6M CAS No.:1016827-93-2 MDL No.:MFCD09946007 MF:C11H7ClN2O MW:218.6391 |
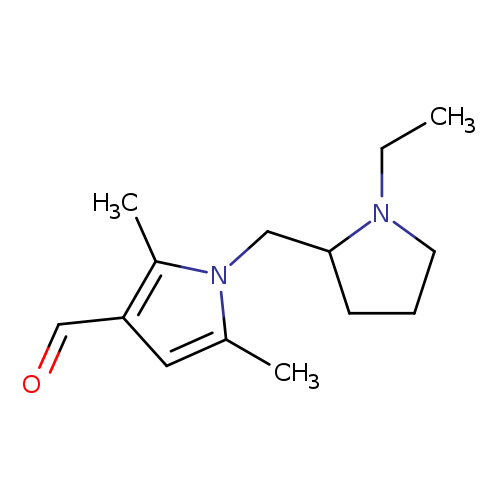
1-[(1-ethylpyrrolidin-2-yl)methyl]-2,5-dimethyl-1H-pyrrole-3-carbaldehydeCatalog No.:AA019MD3 CAS No.:1016828-00-4 MDL No.:MFCD02669932 MF:C14H22N2O MW:234.3373 |
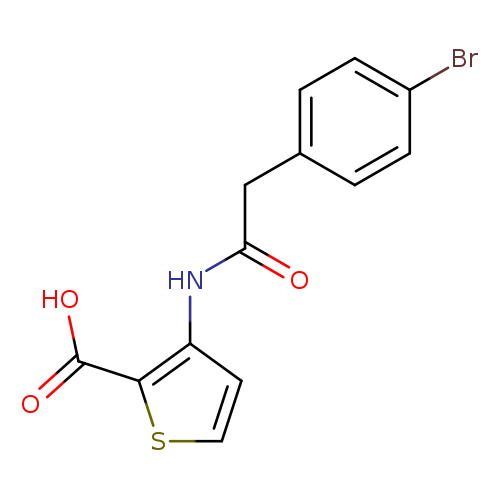
3-([(4-Bromophenyl)acetyl]amino)thiophene-2-carboxylic acidCatalog No.:AA019UB1 CAS No.:1016828-19-5 MDL No.:MFCD09948668 MF:C13H10BrNO3S MW:340.1924 |
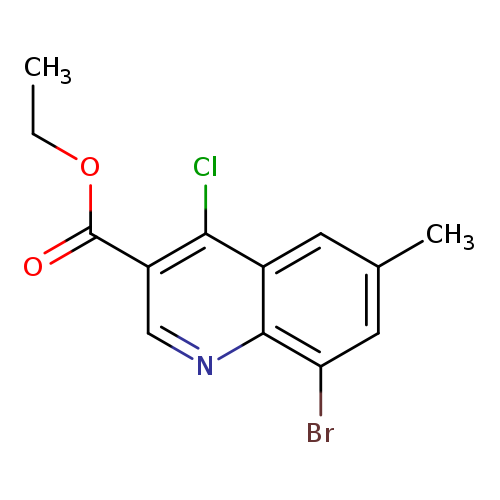
8-Bromo-4-chloro-6-methylquinoline-3-carboxylic acid ethyl esterCatalog No.:AA0093FU CAS No.:1016828-40-2 MDL No.:MFCD09946074 MF:C13H11BrClNO2 MW:328.5889 |
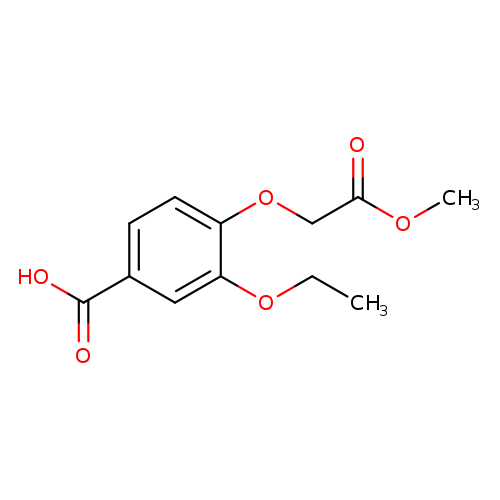
3-Ethoxy-4-(2-methoxy-2-oxoethoxy)benzoic acidCatalog No.:AA01AAGI CAS No.:1016829-67-6 MDL No.:MFCD09938653 MF:C12H14O6 MW:254.2360 |
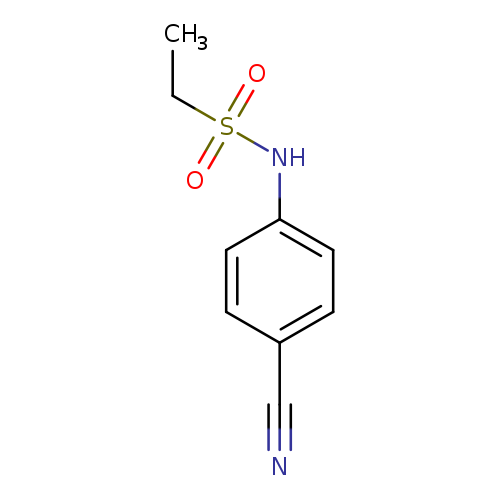
N-(4-cyanophenyl)ethane-1-sulfonamideCatalog No.:AA01AGSM CAS No.:1016830-93-5 MDL No.:MFCD09939863 MF:C9H10N2O2S MW:210.2529 |
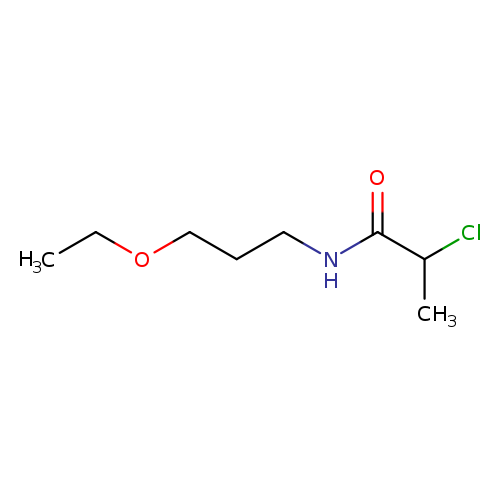
2-chloro-N-(3-ethoxypropyl)propanamideCatalog No.:AA019KN0 CAS No.:1016831-56-3 MDL No.:MFCD09939950 MF:C8H16ClNO2 MW:193.6711 |
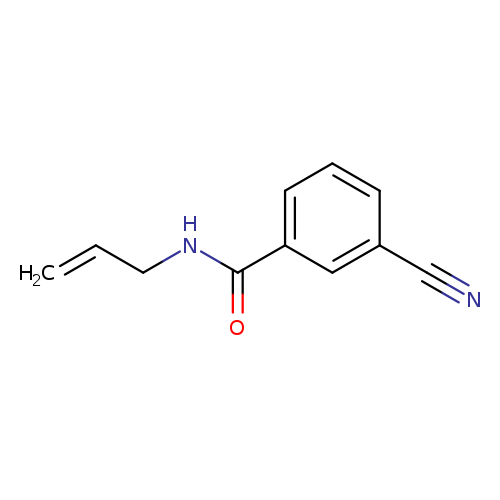
3-cyano-N-(prop-2-en-1-yl)benzamideCatalog No.:AA019XIO CAS No.:1016832-46-4 MDL No.:MFCD09940078 MF:C11H10N2O MW:186.2099 |
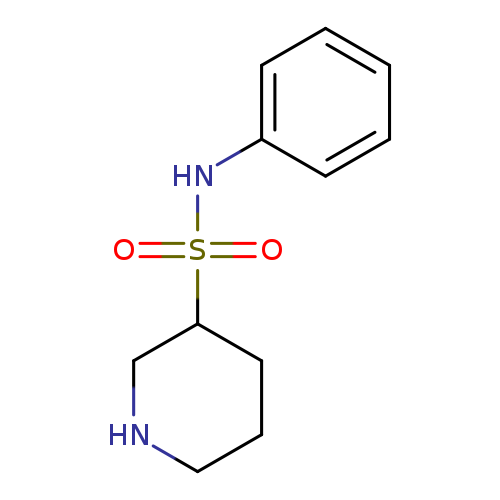
N-phenylpiperidine-3-sulfonamideCatalog No.:AA01A62G CAS No.:1016833-04-7 MDL No.:MFCD09936263 MF:C11H16N2O2S MW:240.3219 |
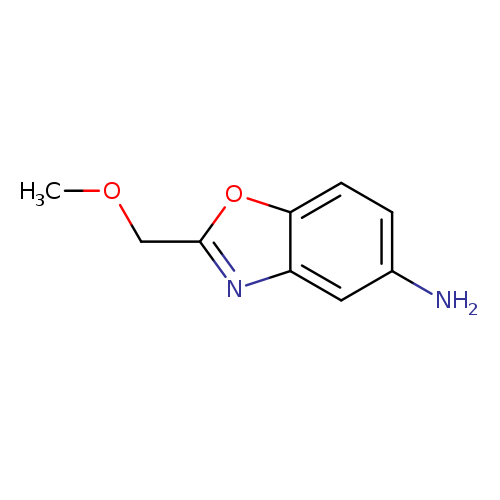
2-(methoxymethyl)-1,3-benzoxazol-5-amineCatalog No.:AA019YIC CAS No.:1016833-15-0 MDL No.:MFCD09942764 MF:C9H10N2O2 MW:178.1879 |
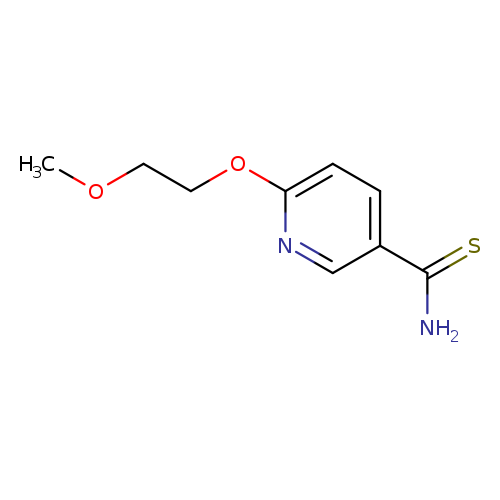
6-(2-Methoxyethoxy)pyridine-3-carbothioamideCatalog No.:AA01BENU CAS No.:1016833-69-4 MDL No.:MFCD09940236 MF:C9H12N2O2S MW:212.2688 |
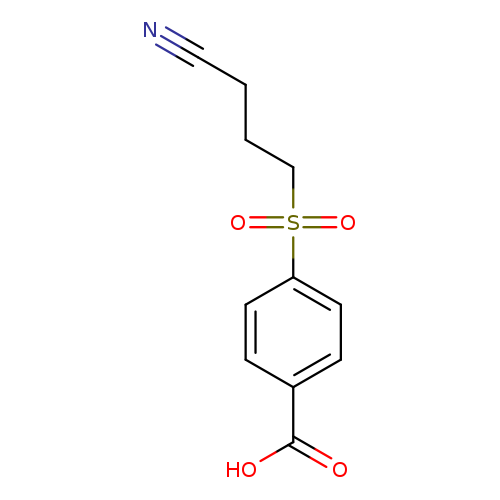
4-(3-cyanopropanesulfonyl)benzoic acidCatalog No.:AA01A869 CAS No.:1016834-20-0 MDL No.:MFCD09942888 MF:C11H11NO4S MW:253.2743 |
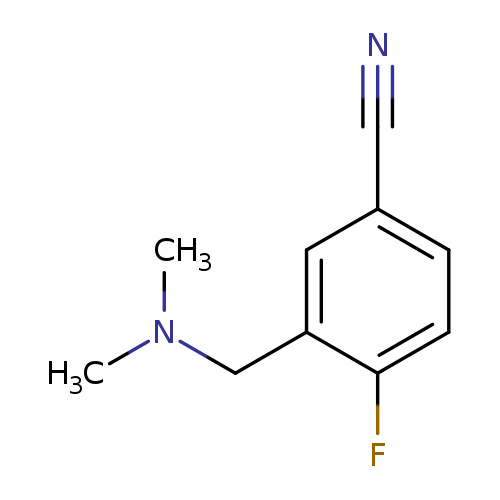
3-[(dimethylamino)methyl]-4-fluorobenzonitrileCatalog No.:AA01AHK1 CAS No.:1016834-55-1 MDL No.:MFCD09936472 MF:C10H11FN2 MW:178.2061 |
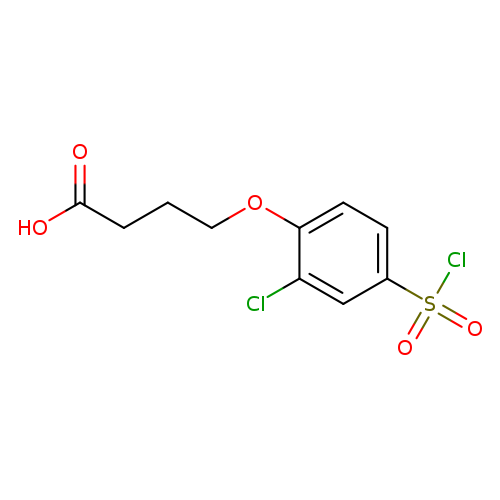
4-[2-chloro-4-(chlorosulfonyl)phenoxy]butanoic acidCatalog No.:AA01A8OT CAS No.:1016835-63-4 MDL No.:MFCD09950718 MF:C10H10Cl2O5S MW:313.1544 |
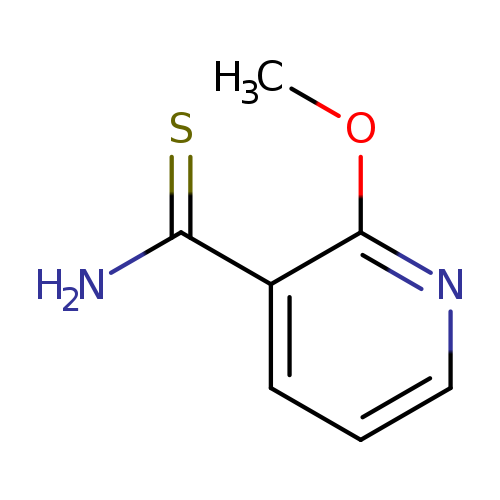
2-methoxypyridine-3-carbothioamideCatalog No.:AA01AAJE CAS No.:1016837-97-0 MDL No.:MFCD09947360 MF:C7H8N2OS MW:168.2162 |
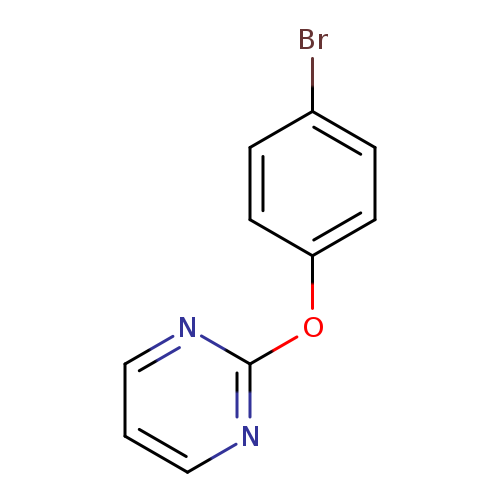
2-(4-Bromophenoxy)pyrimidineCatalog No.:AA00945R CAS No.:1016838-18-8 MDL No.:MFCD09943481 MF:C10H7BrN2O MW:251.0794 |
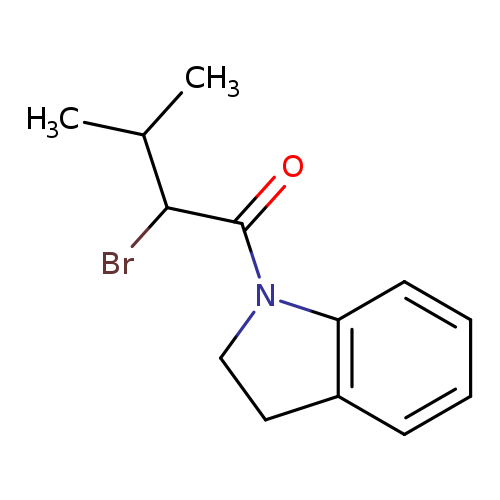
2-bromo-1-(2,3-dihydro-1H-indol-1-yl)-3-methylbutan-1-oneCatalog No.:AA019KWP CAS No.:1016838-76-8 MDL No.:MFCD09940757 MF:C13H16BrNO MW:282.1762 |
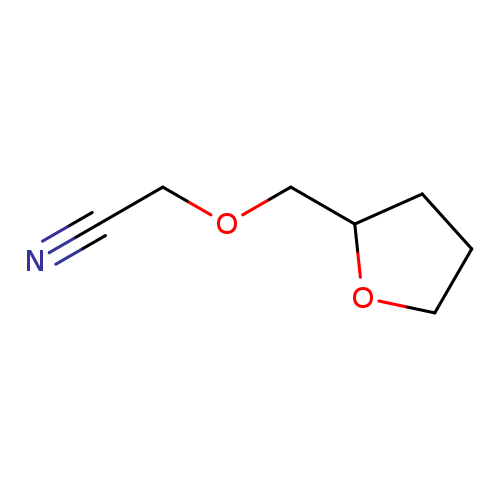
2-[(oxolan-2-yl)methoxy]acetonitrileCatalog No.:AA01AUCB CAS No.:1016838-93-9 MDL No.:MFCD09940794 MF:C7H11NO2 MW:141.1677 |
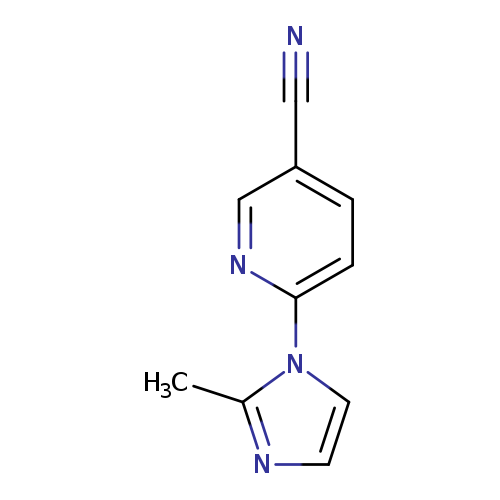
6-(2-methyl-1H-imidazol-1-yl)pyridine-3-carbonitrileCatalog No.:AA01A8HF CAS No.:1016839-05-6 MDL No.:MFCD09940807 MF:C10H8N4 MW:184.1973 |
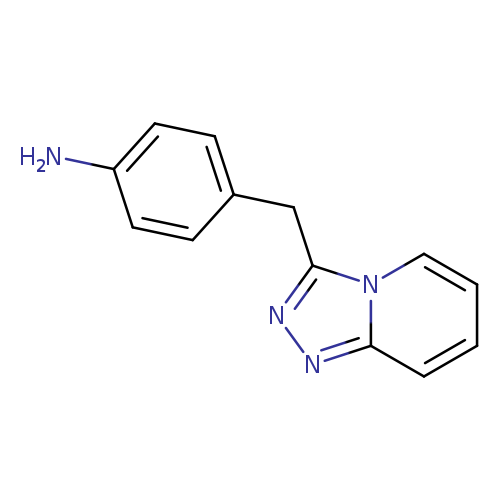
4-{[1,2,4]triazolo[4,3-a]pyridin-3-ylmethyl}anilineCatalog No.:AA01AHFJ CAS No.:1016839-14-7 MDL No.:MFCD09943606 MF:C13H12N4 MW:224.2612 |
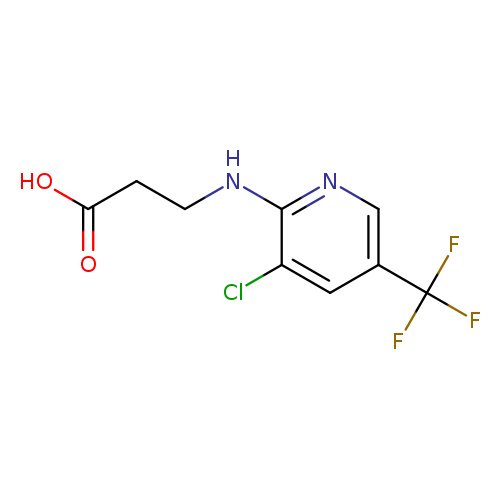
3-{[3-chloro-5-(trifluoromethyl)pyridin-2-yl]amino}propanoic acidCatalog No.:AA01AHNC CAS No.:1016839-74-9 MDL No.:MFCD09944771 MF:C9H8ClF3N2O2 MW:268.6202 |
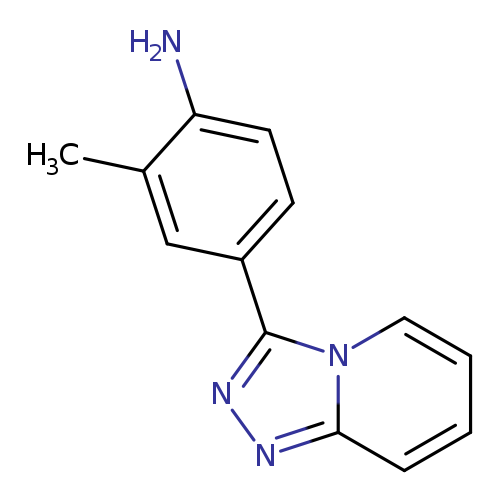
2-Methyl-4-([1,2,4]triazolo[4,3-a]pyridin-3-yl)anilineCatalog No.:AA01AALE CAS No.:1016839-90-9 MDL No.:MFCD09944785 MF:C13H12N4 MW:224.2612 |
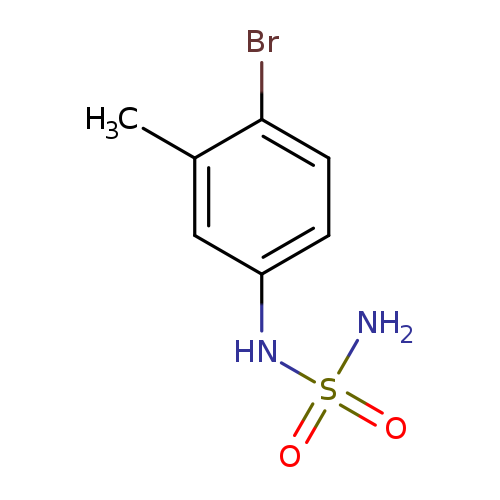
N-(4-Bromo-3-methylphenyl)aminosulfonamideCatalog No.:AA00H9GX CAS No.:1016840-61-1 MDL No.:MFCD09941017 MF:C7H9BrN2O2S MW:265.1276 |
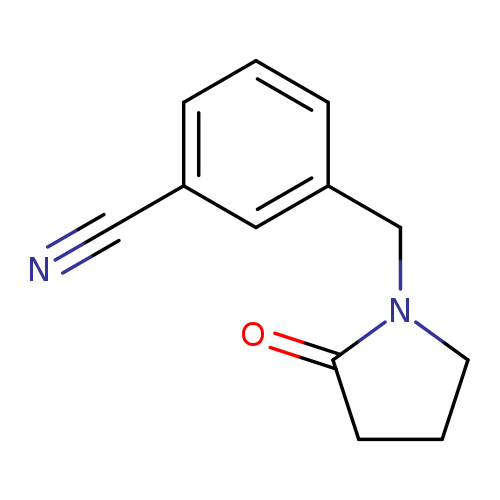
3-[(2-Oxopyrrolidin-1-yl)methyl]benzonitrileCatalog No.:AA01AHZP CAS No.:1016840-72-4 MDL No.:MFCD09941041 MF:C12H12N2O MW:200.2365 |
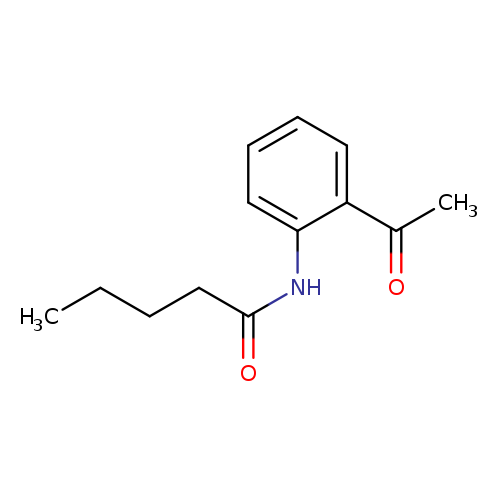
N-(2-Acetylphenyl)pentanamideCatalog No.:AA0099UX CAS No.:1016841-03-4 MDL No.:MFCD09948984 MF:C13H17NO2 MW:219.2796 |
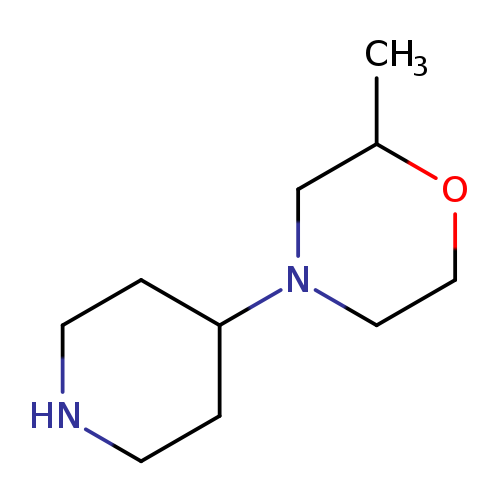
2-methyl-4-(piperidin-4-yl)morpholineCatalog No.:AA019WFS CAS No.:1016841-08-9 MDL No.:MFCD09941106 MF:C10H20N2O MW:184.2786 |
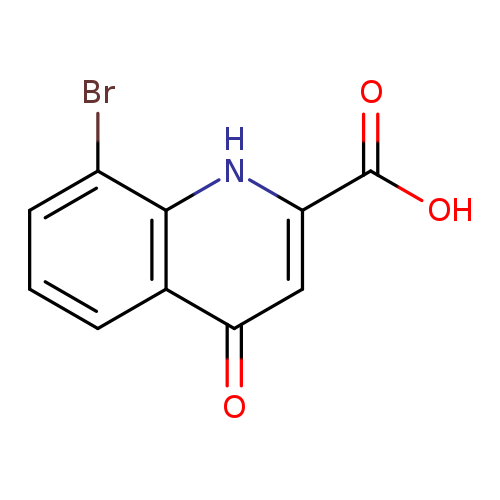
8-Bromo-4-oxo-1,4-dihydroquinoline-2-carboxylic acidCatalog No.:AA0096VF CAS No.:1016841-45-4 MDL No.:MFCD09937472 MF:C10H6BrNO3 MW:268.0635 |
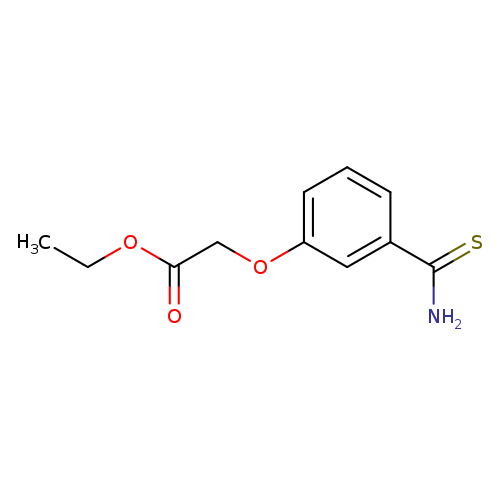
ethyl 2-(3-carbamothioylphenoxy)acetateCatalog No.:AA019TWV CAS No.:1016841-47-6 MDL No.:MFCD30342813 MF:C11H13NO3S MW:239.2908 |
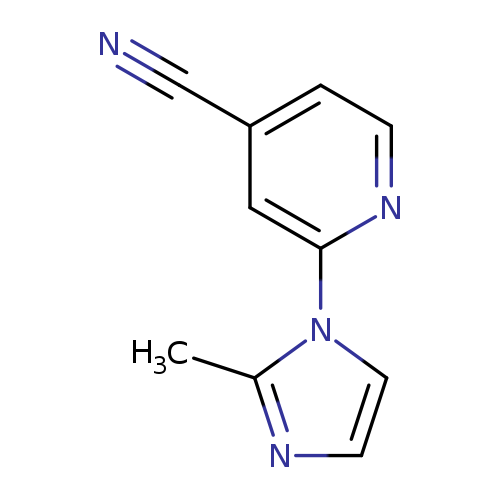
2-(2-methyl-1H-imidazol-1-yl)pyridine-4-carbonitrileCatalog No.:AA019MUD CAS No.:1016841-67-0 MDL No.:MFCD09937507 MF:C10H8N4 MW:184.1973 |
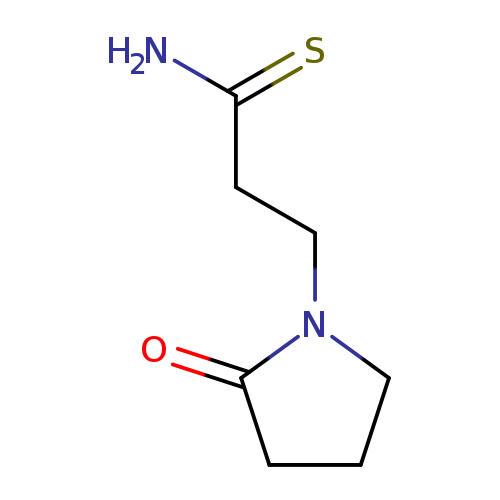
3-(2-oxopyrrolidin-1-yl)propanethioamideCatalog No.:AA019MLK CAS No.:1016841-80-7 MDL No.:MFCD09949111 MF:C7H12N2OS MW:172.2480 |
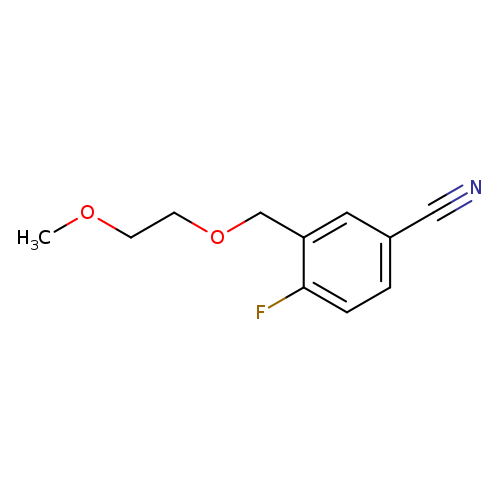
4-Fluoro-3-[(2-methoxyethoxy)methyl]benzonitrileCatalog No.:AA01B52D CAS No.:1016842-52-6 MDL No.:MFCD09949224 MF:C11H12FNO2 MW:209.2169 |
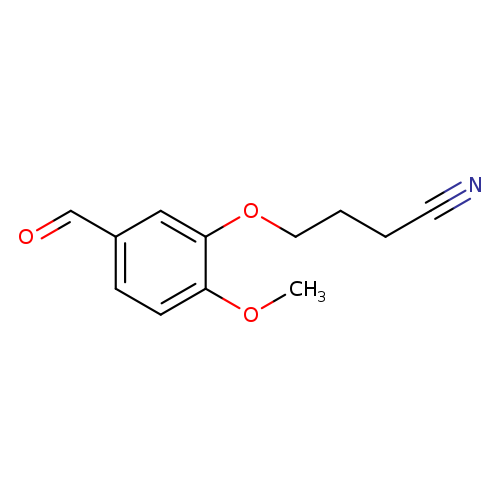
4-(5-formyl-2-methoxyphenoxy)butanenitrileCatalog No.:AA01A80N CAS No.:1016842-76-4 MDL No.:MFCD09947988 MF:C12H13NO3 MW:219.2365 |
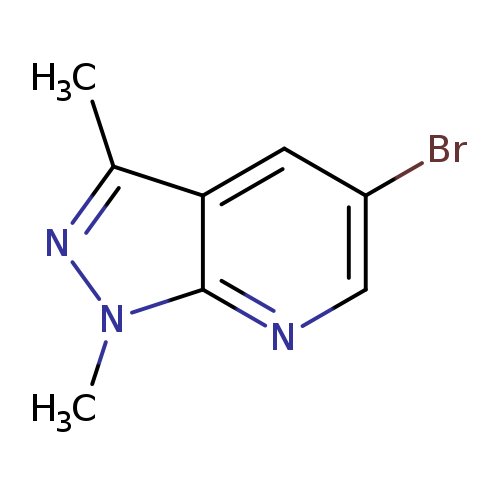
5-Bromo-1,3-dimethyl-1H-pyrazolo[3,4-b]pyridineCatalog No.:AA01AD4Q CAS No.:1016842-99-1 MDL No.:MFCD09948014 MF:C8H8BrN3 MW:226.0732 |
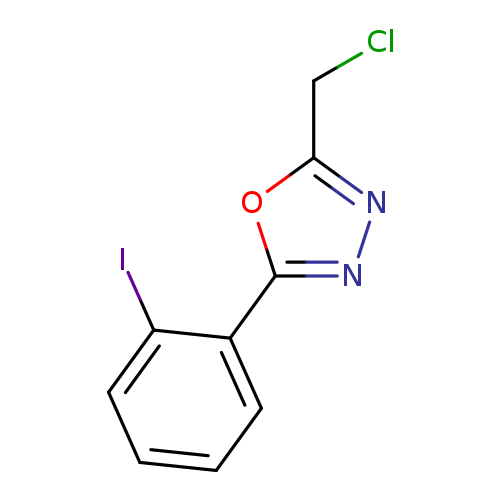
2-(Chloromethyl)-5-(2-iodophenyl)-1,3,4-oxadiazoleCatalog No.:AA00H9GY CAS No.:1016843-20-1 MDL No.:MFCD09948050 MF:C9H6ClIN2O MW:320.5142 |
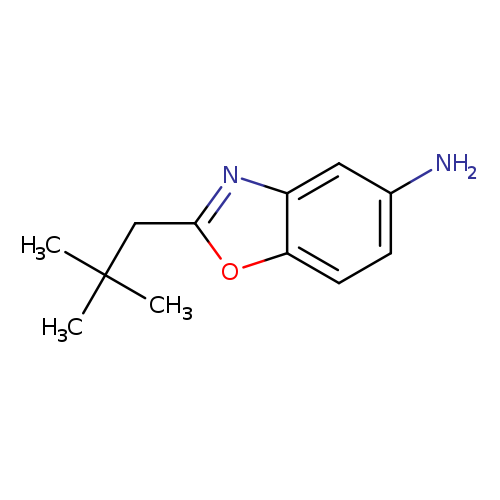
2-(2,2-dimethylpropyl)-1,3-benzoxazol-5-amineCatalog No.:AA01AGR7 CAS No.:1016843-36-9 MDL No.:MFCD09941677 MF:C12H16N2O MW:204.2682 |
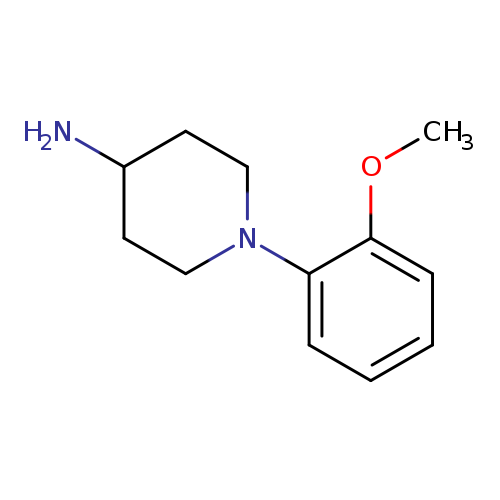
1-(2-methoxyphenyl)piperidin-4-amineCatalog No.:AA01A806 CAS No.:1016843-47-2 MDL No.:MFCD01681683 MF:C12H18N2O MW:206.2841 |
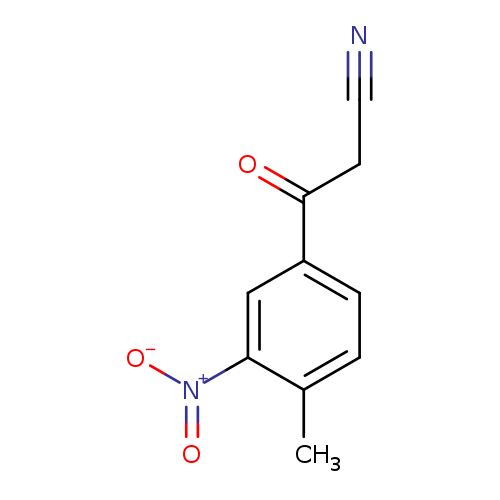
Benzenepropanenitrile, 4-methyl-3-nitro-β-oxo-Catalog No.:AA00054R CAS No.:1016843-83-6 MDL No.:MFCD09941734 MF:C10H8N2O3 MW:204.1821 |
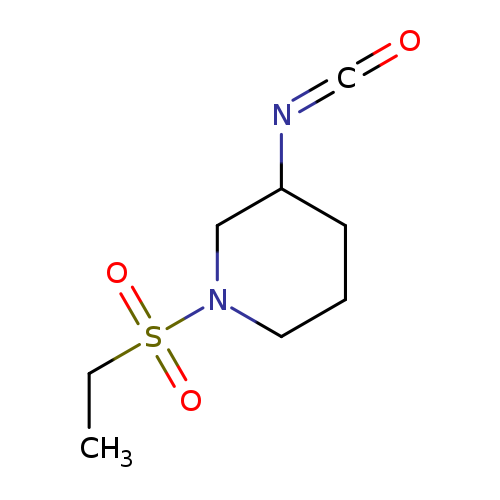
1-(ethanesulfonyl)-3-isocyanatopiperidineCatalog No.:AA01A9CG CAS No.:1016844-38-4 MDL No.:MFCD09941793 MF:C8H14N2O3S MW:218.2734 |
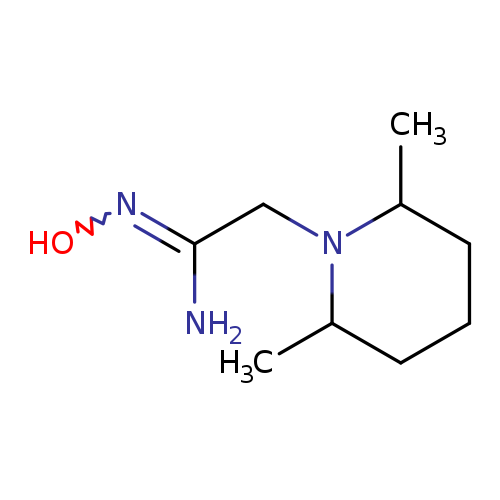
2-(2,6-dimethylpiperidin-1-yl)-N'-hydroxyethanimidamideCatalog No.:AA019UY5 CAS No.:1016846-03-9 MDL No.:MFCD09938216 MF:C9H19N3O MW:185.2667 |
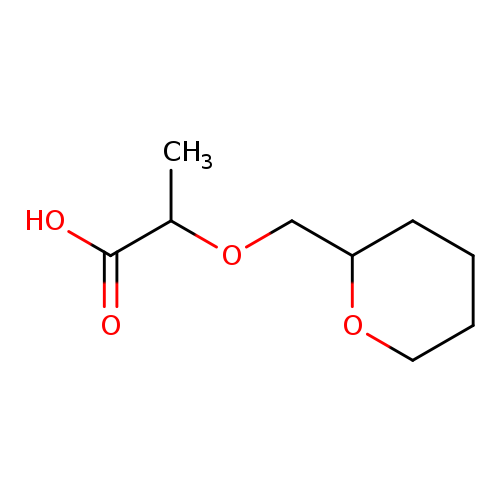
2-(oxan-2-ylmethoxy)propanoic acidCatalog No.:AA019Y16 CAS No.:1016846-06-2 MDL No.:MFCD09949864 MF:C9H16O4 MW:188.2209 |
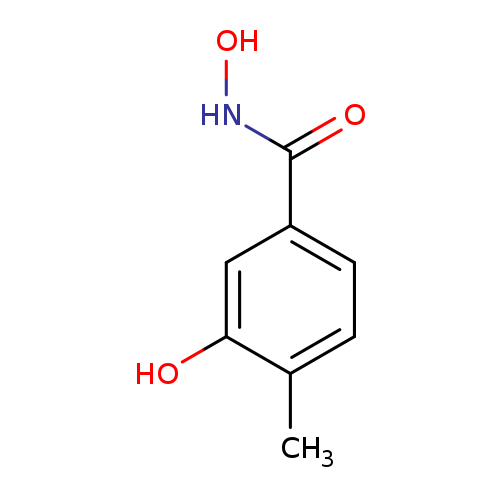
N,3-dihydroxy-4-methylbenzamideCatalog No.:AA01AKLW CAS No.:1016847-61-2 MDL No.:MFCD09938400 MF:C8H9NO3 MW:167.1620 |
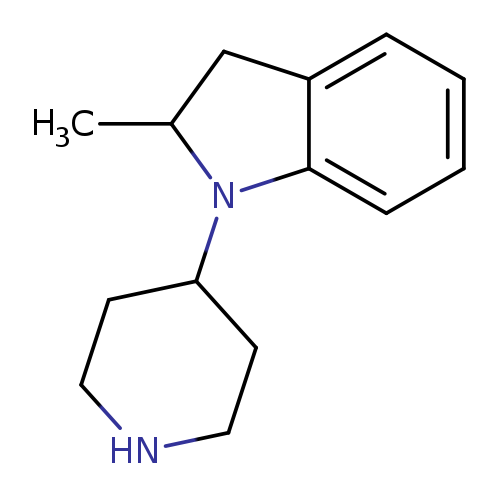
2-methyl-1-(piperidin-4-yl)-2,3-dihydro-1H-indoleCatalog No.:AA01A77T CAS No.:1016847-63-4 MDL No.:MFCD09946214 MF:C14H20N2 MW:216.3220 |
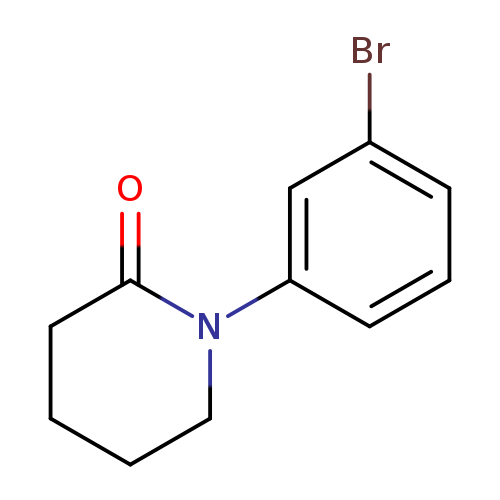
1-(3-Bromophenyl)piperidin-2-oneCatalog No.:AA00054Q CAS No.:1016847-66-7 MDL No.:MFCD04037063 MF:C11H12BrNO MW:254.1231 |
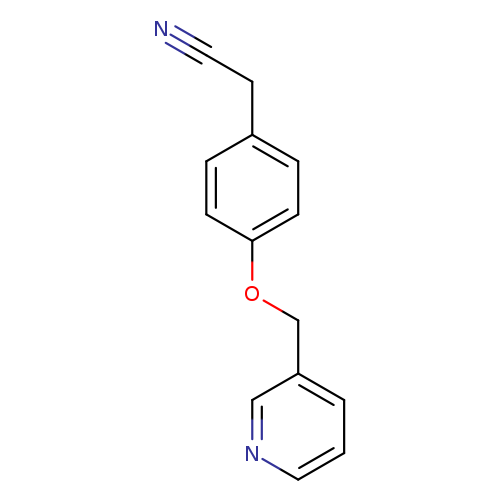
2-[4-(pyridin-3-ylmethoxy)phenyl]acetonitrileCatalog No.:AA019V59 CAS No.:1016850-10-4 MDL No.:MFCD09946536 MF:C14H12N2O MW:224.2579 |
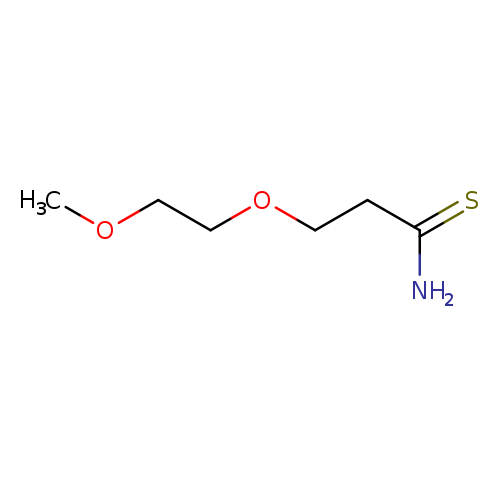
3-(2-methoxyethoxy)propanethioamideCatalog No.:AA01C3PV CAS No.:1016850-44-4 MDL No.:MFCD09946573 MF:C6H13NO2S MW:163.2379 |
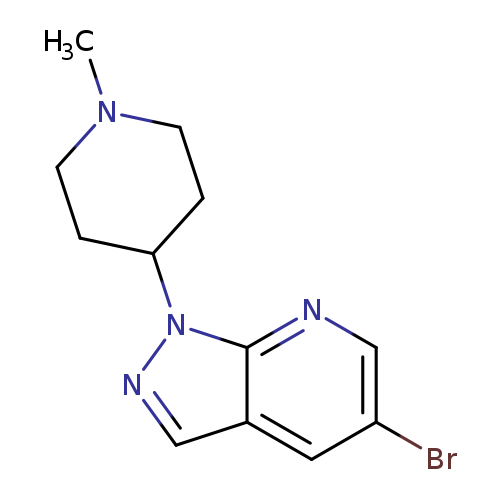
4-{5-bromo-1H-pyrazolo[3,4-b]pyridin-1-yl}-1-methylpiperidineCatalog No.:AA019XPS CAS No.:1016850-95-5 MDL No.:MFCD09946632 MF:C12H15BrN4 MW:295.1783 |
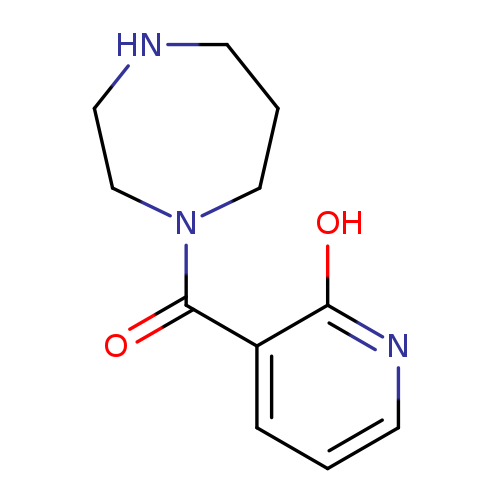
3-(1,4-diazepane-1-carbonyl)pyridin-2-olCatalog No.:AA019WOT CAS No.:1016851-38-9 MDL No.:MFCD09950428 MF:C11H15N3O2 MW:221.2557 |
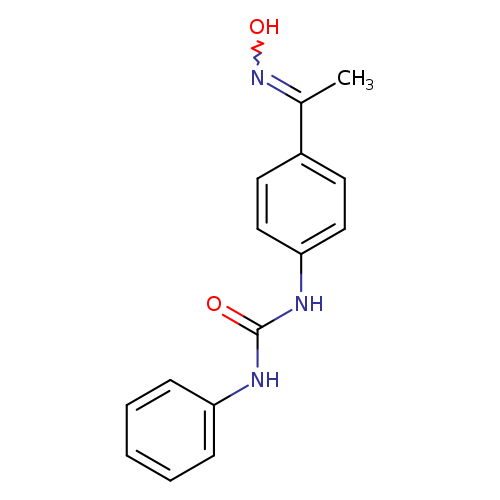
3-{4-[1-(hydroxyimino)ethyl]phenyl}-1-phenylureaCatalog No.:AA019N10 CAS No.:1016852-21-3 MDL No.:MFCD09946796 MF:C15H15N3O2 MW:269.2985 |
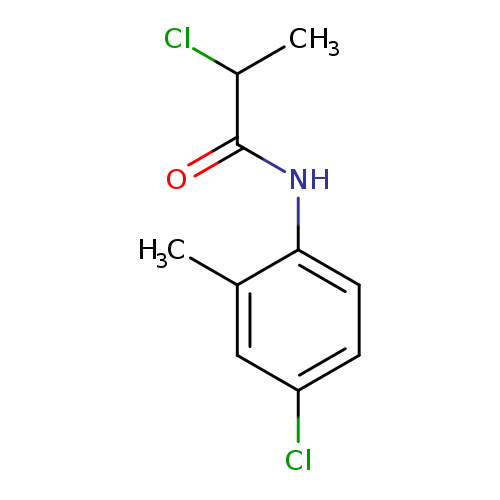
2-chloro-N-(4-chloro-2-methylphenyl)propanamideCatalog No.:AA019KMQ CAS No.:1016852-56-4 MDL No.:MFCD09950580 MF:C10H11Cl2NO MW:232.1064 |
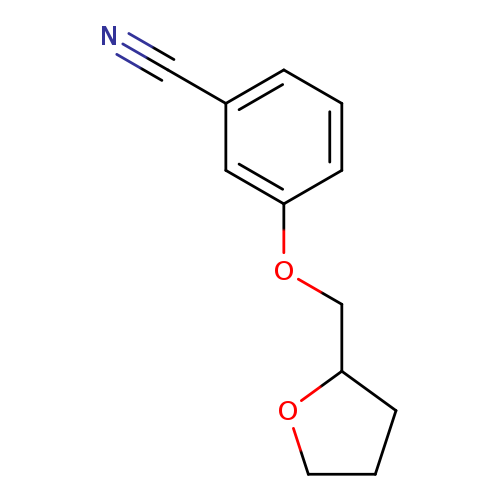
3-(Oxolan-2-ylmethoxy)benzonitrileCatalog No.:AA00948L CAS No.:1016853-20-5 MDL No.:MFCD09946912 MF:C12H13NO2 MW:203.2371 |
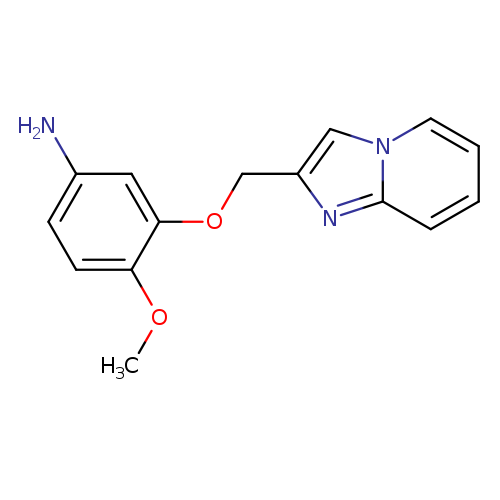
3-(Imidazo[1,2-a]pyridin-2-ylmethoxy)-4-methoxyanilineCatalog No.:AA019VXF CAS No.:1016854-05-9 MDL No.:MFCD09947036 MF:C15H15N3O2 MW:269.2985 |
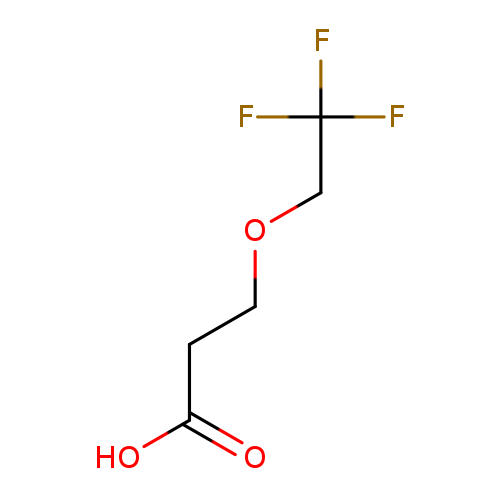
3-(2,2,2-trifluoroethoxy)propanoic acidCatalog No.:AA019LIY CAS No.:1016854-20-8 MDL No.:MFCD09940500 MF:C5H7F3O3 MW:172.1025 |
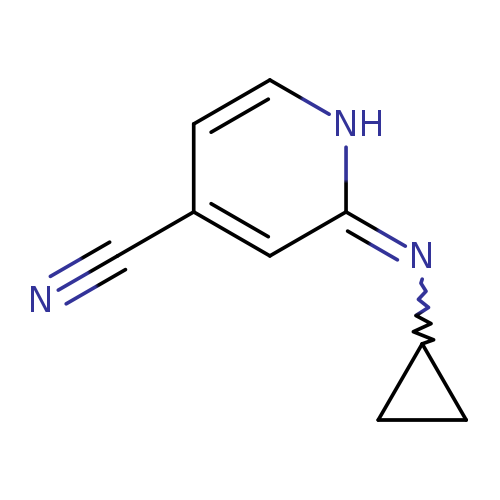
2-(cyclopropylimino)-1,2-dihydropyridine-4-carbonitrileCatalog No.:AA01E968 CAS No.:1016855-14-3 MDL No.:MFCD09943385 MF:C9H9N3 MW:159.1879 |
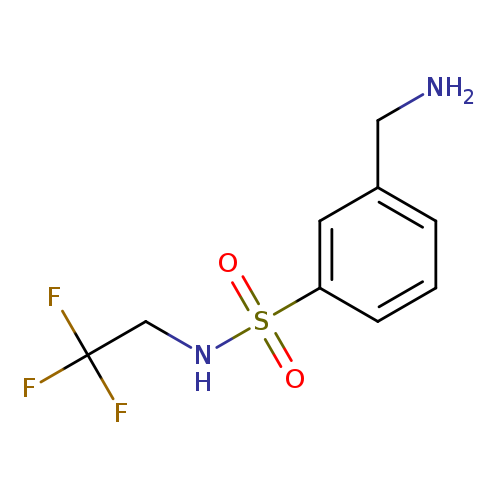
3-(aminomethyl)-N-(2,2,2-trifluoroethyl)benzene-1-sulfonamideCatalog No.:AA01AMY6 CAS No.:1016856-86-2 MDL No.:MFCD09936905 MF:C9H11F3N2O2S MW:268.2560 |
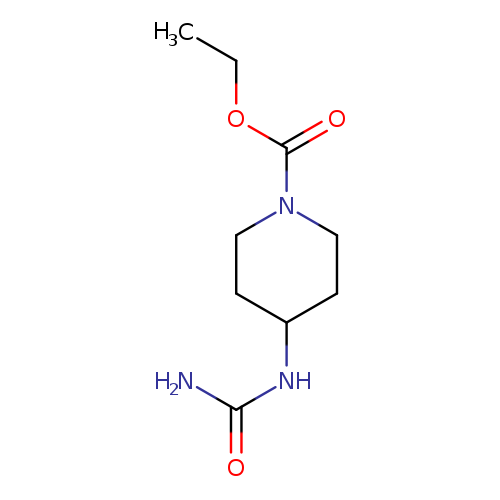
ethyl 4-(carbamoylamino)piperidine-1-carboxylateCatalog No.:AA019NDQ CAS No.:1016857-27-4 MDL No.:MFCD09947244 MF:C9H17N3O3 MW:215.2496 |
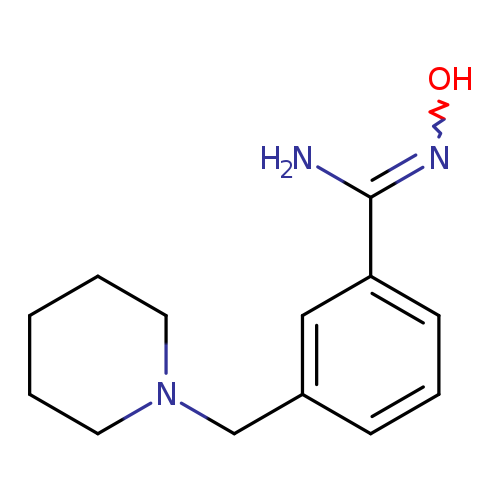
N'-hydroxy-3-(piperidin-1-ylmethyl)benzene-1-carboximidamideCatalog No.:AA019XSB CAS No.:1016857-30-9 MDL No.:MFCD09936978 MF:C13H19N3O MW:233.3095 |
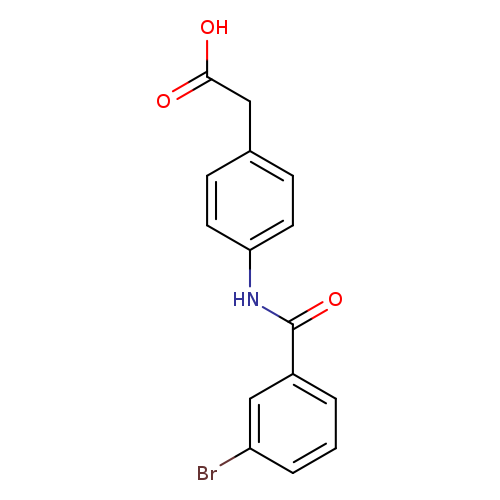
2-(4-[(3-Bromobenzene)amido]phenyl)acetic acidCatalog No.:AA019V2K CAS No.:1016857-62-7 MDL No.:MFCD09937023 MF:C15H12BrNO3 MW:334.1647 |
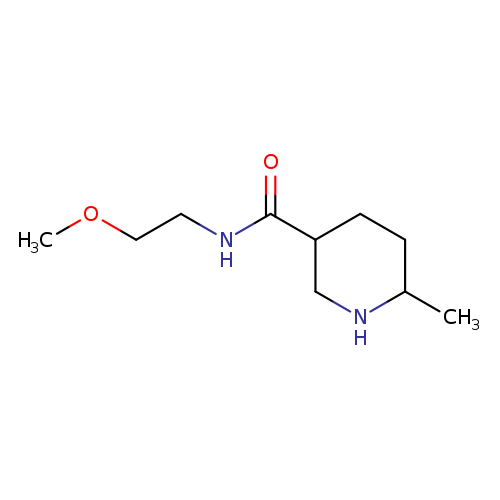
N-(2-methoxyethyl)-6-methylpiperidine-3-carboxamideCatalog No.:AA019WSY CAS No.:1016857-64-9 MDL No.:MFCD09939803 MF:C10H20N2O2 MW:200.2780 |
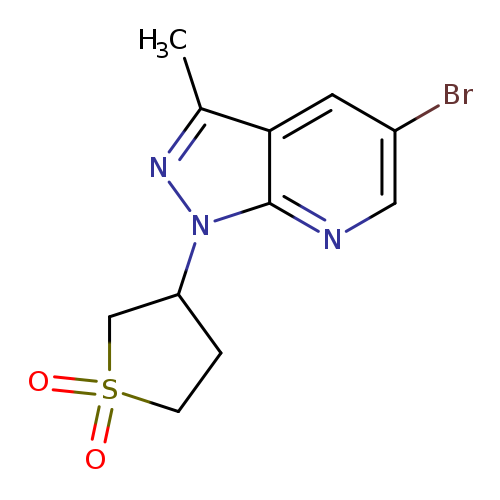
3-{5-bromo-3-methyl-1H-pyrazolo[3,4-b]pyridin-1-yl}-1lambda6-thiolane-1,1-dioneCatalog No.:AA01A6IN CAS No.:1016857-76-3 MDL No.:MFCD09939819 MF:C11H12BrN3O2S MW:330.2009 |
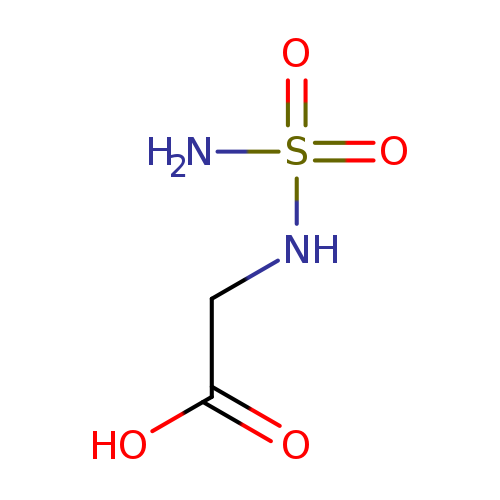
2-(SULFAMOYLAMINO)ACETIC ACIDCatalog No.:AA01C08O CAS No.:1016857-92-3 MDL No.:MFCD09947345 MF:C2H6N2O4S MW:154.1450 |
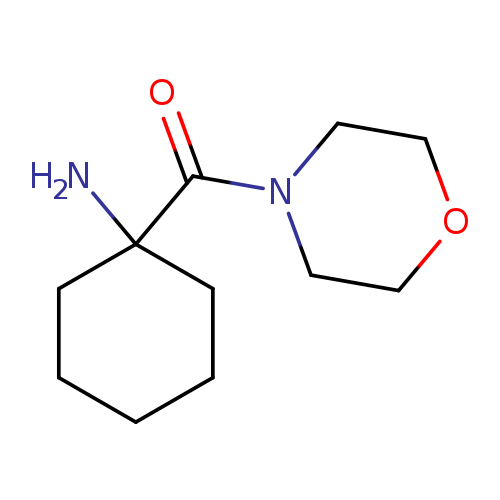
1-(morpholine-4-carbonyl)cyclohexan-1-amineCatalog No.:AA01A78Y CAS No.:1016857-95-6 MDL No.:MFCD09937079 MF:C11H20N2O2 MW:212.2887 |
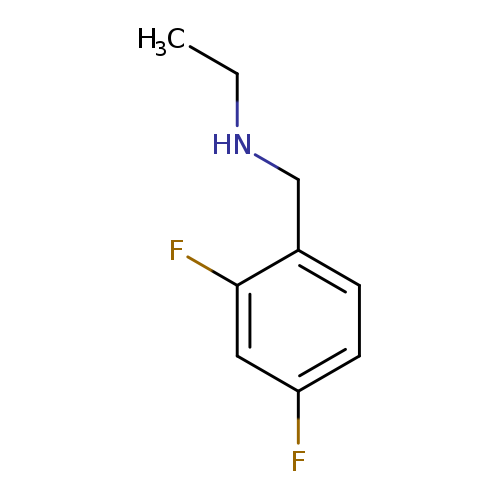
[(2,4-difluorophenyl)methyl](ethyl)amineCatalog No.:AA019LNN CAS No.:1016859-58-7 MDL No.:MFCD09943740 MF:C9H11F2N MW:171.1871 |
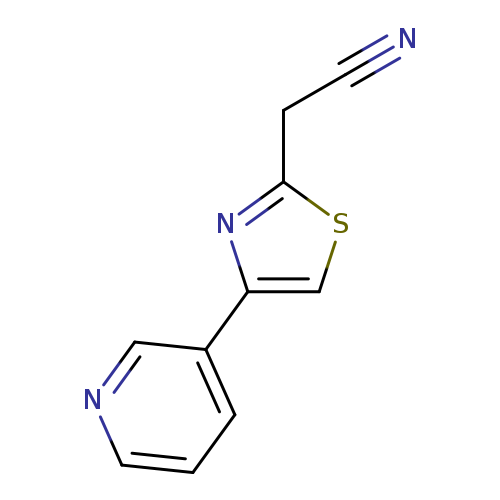
2-(4-(Pyridin-3-yl)thiazol-2-yl)acetonitrileCatalog No.:AA01FBOJ CAS No.:1016859-62-3 MDL No.:MFCD09943745 MF:C10H7N3S MW:201.2477 |
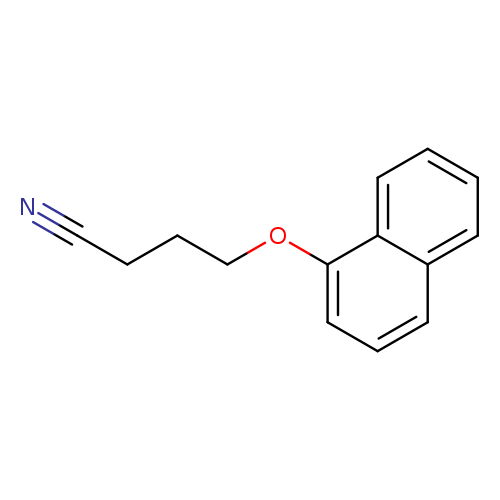
4-(naphthalen-1-yloxy)butanenitrileCatalog No.:AA01A9WA CAS No.:1016860-12-0 MDL No.:MFCD09944811 MF:C14H13NO MW:211.2591 |
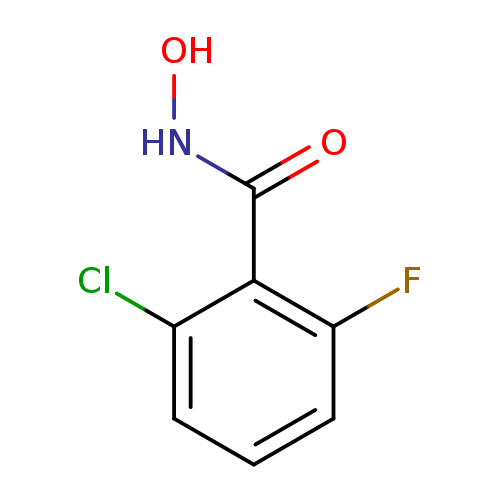
2-Chloro-6-fluoro-n-hydroxybenzamideCatalog No.:AA01AAN1 CAS No.:1016860-39-1 MDL No.:MFCD09941079 MF:C7H5ClFNO2 MW:189.5715 |
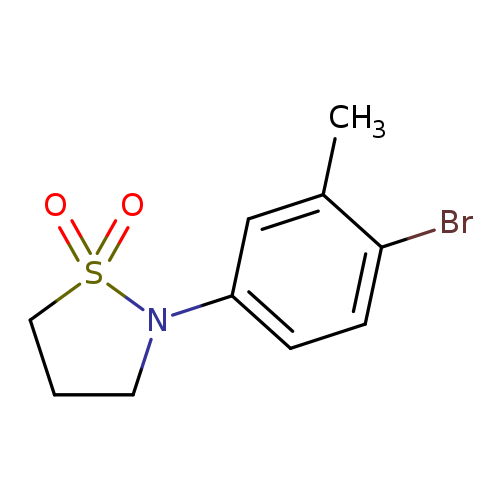
N-(4-Bromo-3-methylphenyl)-1,3-propanesultamCatalog No.:AA00054P CAS No.:1016860-62-0 MDL No.:MFCD09936816 MF:C10H12BrNO2S MW:290.1768 |
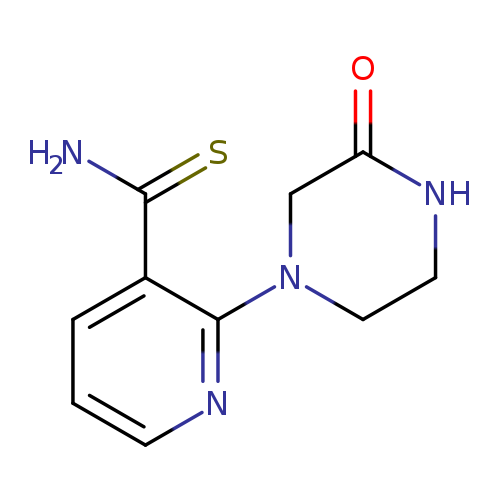
2-(3-oxopiperazin-1-yl)pyridine-3-carbothioamideCatalog No.:AA01A3PD CAS No.:1016861-56-5 MDL No.:MFCD09936950 MF:C10H12N4OS MW:236.2935 |
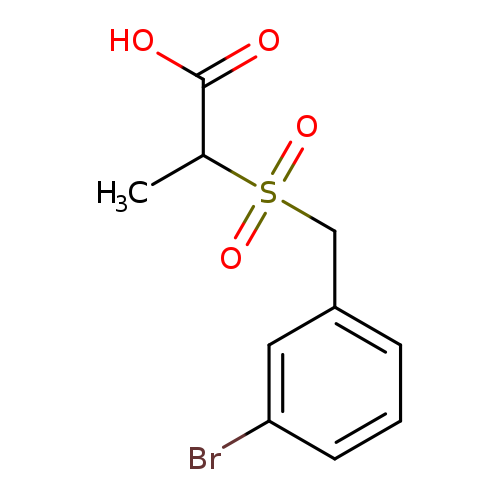
2-[(3-bromophenyl)methanesulfonyl]propanoic acidCatalog No.:AA019NAB CAS No.:1016862-04-6 MDL No.:MFCD09936998 MF:C10H11BrO4S MW:307.1609 |