2020-02-03 12:15:13
Sundaravel Vivek Kumar1 · Gandhi Uma Rani1 · Manohar Divyalakshmi1 · Nattamai Bhuvanesh2 · Shanmugam Muthusubramanian1 · Subbu Perumal1
Introduction
1,3-Dipolar cycloaddition reaction provides avaluable platform for the synthesis of natural products with complex architectures involving high regio- and stereoselectivities. [1–6]. Recently, cycloadditions involving azomethine [7] have attracted much attention as they end up with spiroox- indole unit, which has present in many natural products like horsfiline, coerulescine, spirotryprostatins A and B, ptero- podine, isopteropodine, mitraphylline and isomitraphylline having significant biological activities [8]. The synthetic analogues of spirooxindole also exhibit significant biologi- cal properties [9–16]. In particular, MI-888 and NITD609 have entered in the preclinical evaluation of malaria and cancer [17]. Similarly, CFI-400945 and SAR405838 have been reported as promising candidates for the treatment of cancer and inhibition of MDM2-p53 interaction [18, 19].
Benzosuberone is an important and unique skeleton having a wide range of biological applications. Naturally occur- ring compounds bearing this unit—colchicine [20], theafla- vin [21, 22], bussealin E [23], demethylsalvicanol [24] and brussonol [25]—were clinically proven as anticancer agents [26, 27]. The related synthetic frameworks have also been found to exhibit anticancer, antimicrobial, antitubercular, anti-inflammatory, anti-HCV, anti-SSPE and antioxidant activities [28]. They act as dopamine and serotonin recep- tors, inhibitors of human aminopeptidase-N/CD13, metal- loaminopeptidase and p38α [29–34].
The individual biological significance of spirooxindole and benzosuberone prompted us to assemble them in a sin- gle framework anticipating valuable biological applications (Fig. 1). The reported methods of synthesising benzosuber- one-tethered spiroheterocycles include the cycloaddition reaction between diazomethane and arylidenebenzosuberone
[35] and the reaction of nitrile imines with arylidenebenzo- suberone [36]. Recently, Behbehani et al. [37] demonstrated the cycloaddition of arylidene benzosuberone with bis- hydrazonoyl chlorides to deliver the benzosuberone grafted spiropyrazolines in good yields (Scheme 1). However, these methods often suffer one or more shortcomings like limited substrate scope, selectivity and toxic solvent. Herein, we disclose the practical and efficient protocol for the regio- and stereoselective synthesis of novel benzosuberone-tethered spirooxindoles from a three-component 1,3-dipolar cycload- dition reaction of arylidene benzosuberone, isatin and sar- cosine in methanol (Scheme 1). Moderate-to-good yields, wide substrate scope and excellent selectivity are the major advantages of this method.
Results and discussion
Initially, the model reaction of 6-(4-chlorobenzylidene)- 6,7,8,9-tetrahydro-5H-benzo[7] annulen-5-one 1b, isatin 2 and sarcosine 3 has been investigated. The reaction has been carried out in methanol under reflux (Scheme 1), which furnished a single diastereoisomer of 4′-(4-chlorophenyl)- 1′-methyl-8,9-dihydrodispiro[benzo[7]annulene-6,3′- pyrrolidine-2′,3″-indoline]-2″,5(7H)-dione 4b in 76% yield. In order to identify the best solvent for this trans- formation, extensive optimization with different solvents like EtOH, i-PrOH, CH3CN, 1,4-dioxane and toluene was done (Table 1). From the data listed in Table 1, it has been found that methanol emerges as the preferred solvent for this cycloaddition reaction to attain maximum yield.
After establishing the best reaction conditions, the sub- strate scope of the cycloaddition was extended involving a series of 1 with 2 and 3 yielding different 4. When electron- demanding group was introduced at the 4-position of the aryl ring (1a, 1b, 1c and 1h), the reaction underwent smoothly to afford 4a–4c and 4h in 74–78% yield. The same trend was observed with electron-releasing substituents as well (1e, 1f and 1g). Presence of fluoro group (1j) at the third position or chloro or methoxy at the second position (1k and 1l) is well tolerated furnishing 4j–l in 67–70% yields. In addition, the reactivity of some di- and tri-substituted systems (1m–1t) has also been checked under this condition to produce 4m–4t in 62–76% yield (Table 2). In all the cases, this cycloaddi- tion proceeded yielding a single diastereomer. The presence of sterically bulky groups like naphthyl or 2,6-dichlorophe- nyl impeded the reaction completely.
The structure of 4 was deduced from one- and two- dimensional NMR spectroscopic data, as detailed for 4b as a representative example (Fig. 2). The H-4′ appears as a triplet at 4.56 ppm (J = 9.0 Hz), which shows HMBCs with C-5, C-5′ and C-6 at 211.1, 58.4 and 67.5 ppm, respec- tively. The diastereotopic protons H-5′ appear as triplets at 3.50 (J = 12.0 Hz) and 3.90 (J = 9.3 Hz) ppm, which show HMBCs with C-2′ and C-3′ at 78.2 and 67.5 ppm, respec- tively. The N–Me protons appear as a singlet at 2.16 ppm, which shows HMB correlations with C-2′ at 78.2 ppm. NH peak appears as a broad singlet at 7.36 ppm. The diaste- reotopic protons H-7 appear as multiplets at 1.75–1.83 and 2.01–2.11 ppm, which show HMBCs with C-3′ and C-5 at 67.5 and 211.1 ppm, respectively. Finally, the complete structure and stereochemistry of the compounds were une- quivocally assigned by X-ray diffraction study (for details, see SI) of a single crystal of 4h (Fig. 3).
A plausible mechanism for the formation of benzosu- berone-tethered spirooxindoles is illustrated in Scheme 2. Initially, the azomethine ylide is generated by the reaction between isatin and sarcosine via decarboxylation. Then, the dipole 5 reacts with the dipolarophile 1 giving a single diastereomer through cycloaddition. In order to explain the origin of the diastereoselectivity in spite of the presence of three stereo centers, the most acceptable approaching mode is depicted in Scheme 2. Likely, the cycloaddition follows the track 1 rather than the track 2. In track 1, the pyrrolidine part and carbonyl of the benzosuberone system are trans to each other to avoid steric interaction. Simi- larly, to avoid the electrostatic repulsion, the two carbonyls of the products are found trans to each other. The other regioisomer 4′ is not at all formed even in traces as con- firmed by the 1H NMR spectra of crude reaction mixtures.
Conclusion
In summary, we have developed a three-component 1,3 dipolar cycloadditions reaction of arylidene benzosuberone, isatin and sarcosine in refluxing methanol to afford novel benzosuberone- tethered spirooxindoles in good yields. This protocol offers wide substrate scope, good functional group tolerance, mild reaction condition and high regio and stereoselectivities.
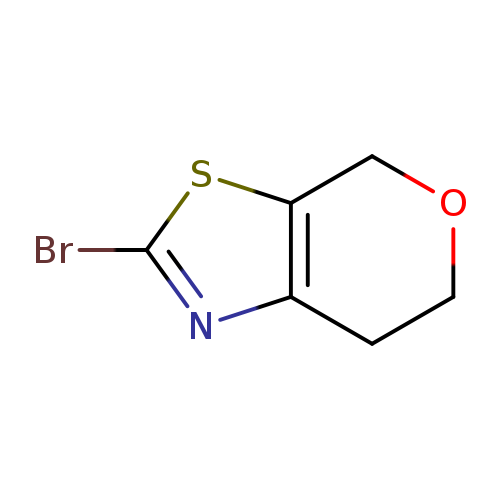
2-Bromo-6,7-dihydro-4H-pyrano[4,3-d]thiazoleCatalog No.:AA008Z0F CAS No.:1093107-56-2 MDL No.:MFCD22565668 MF:C6H6BrNOS MW:220.0869 |
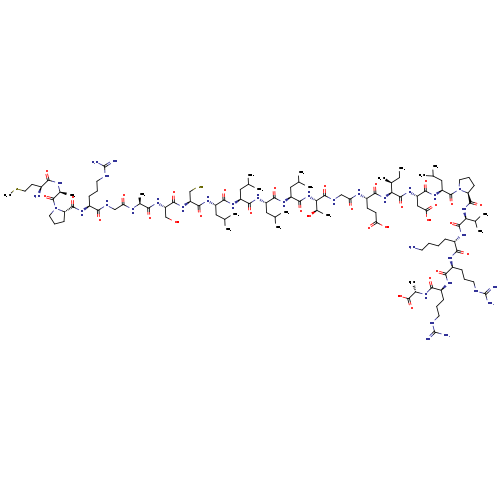
HNGF6ACatalog No.:AA01ENJR CAS No.:1093111-54-6 MDL No.:MFCD28133401 MF:C112H198N34O31S2 MW:2581.1097 |
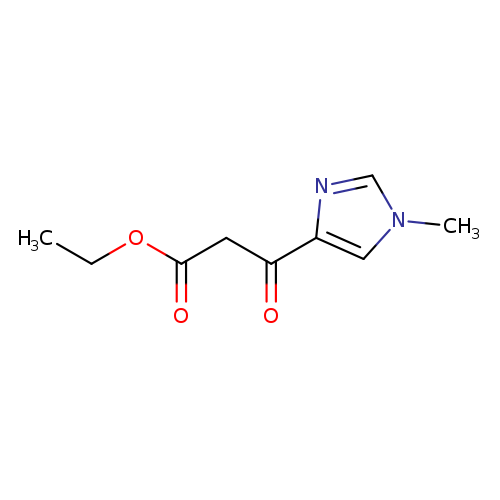
ethyl 3-(1-methyl-1H-imidazol-4-yl)-3-oxopropanoateCatalog No.:AA01B6ET CAS No.:1093114-85-2 MDL No.:MFCD30344916 MF:C9H12N2O3 MW:196.2032 |
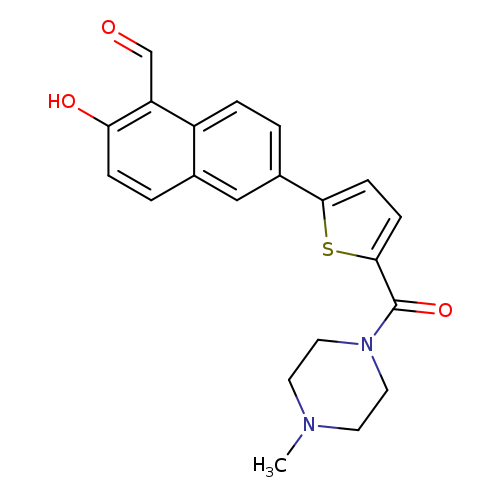
MKC3946Catalog No.:AA01EAYE CAS No.:1093119-54-0 MDL No.:MFCD30496700 MF:C21H20N2O3S MW:380.4601 |
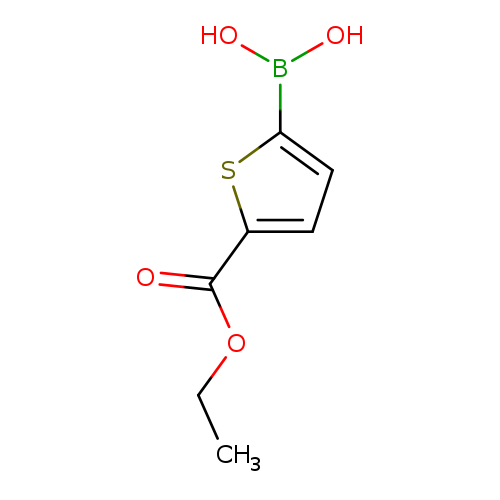
(5-(Ethoxycarbonyl)thiophen-2-yl)boronic acidCatalog No.:AA0099AR CAS No.:1093120-64-9 MDL No.:MFCD09952062 MF:C7H9BO4S MW:200.0200 |
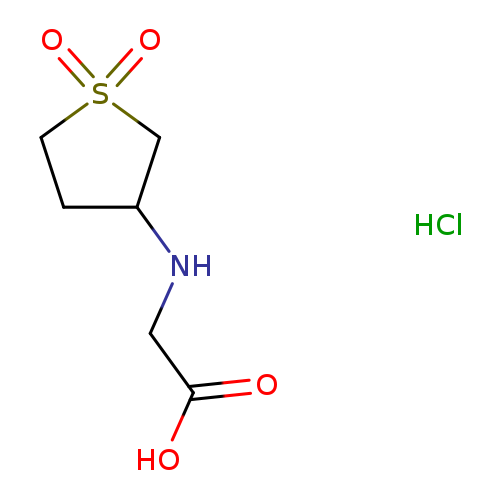
[(1,1-Dioxidotetrahydrothien-3-yl)amino]acetic acid hydrochlorideCatalog No.:AA01C4HG CAS No.:1093123-73-9 MDL No.:MFCD00456684 MF:C6H12ClNO4S MW:229.6818 |
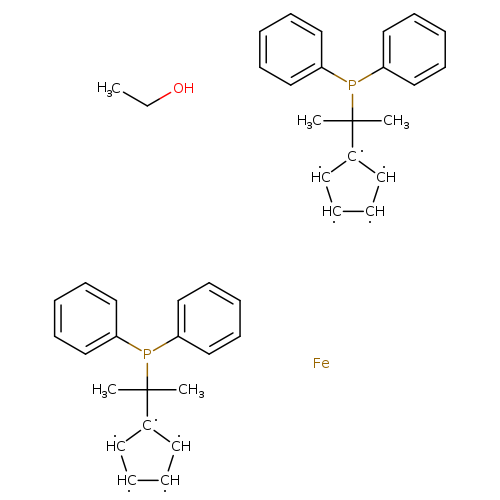
1,1'-Bis(1-diphenylphosphino-1-methylethyl)ferroceneCatalog No.:AA008VV8 CAS No.:109313-83-9 MDL No.:MFCD28144554 MF:C42H46FeOP2 MW:684.6066 |
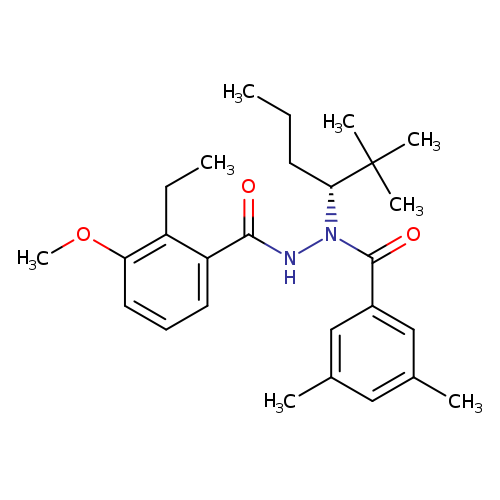
VeledimexCatalog No.:AA01EODX CAS No.:1093130-72-3 MDL No.:MFCD28963985 MF:C27H38N2O3 MW:438.6022 |
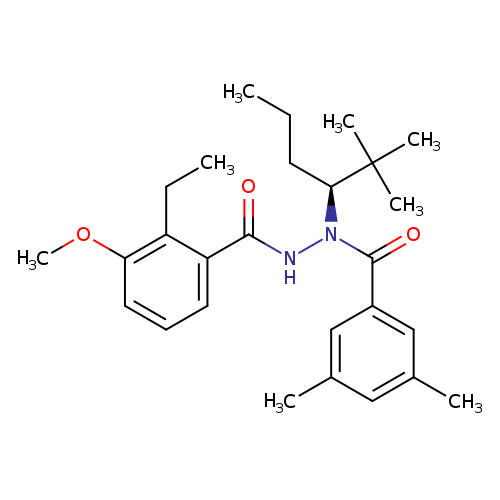
Veledimex S enantiomerCatalog No.:AA01EODU CAS No.:1093131-03-3 MDL No.:MFCD31382195 MF:C27H38N2O3 MW:438.6022 |
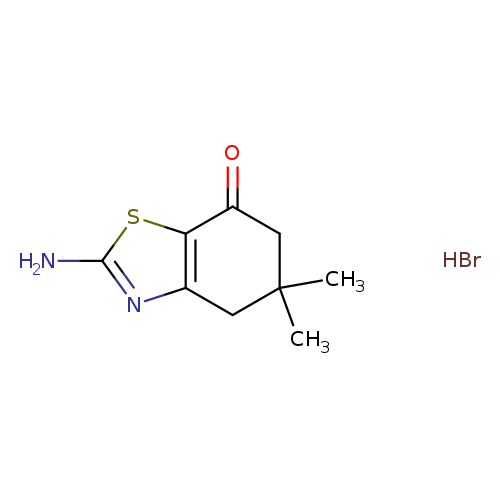
2-Amino-5,5-dimethyl-5,6-dihydro-4h-benzothiazol-7-one HBrCatalog No.:AA0093H8 CAS No.:109317-52-4 MDL No.:MFCD19441990 MF:C9H13BrN2OS MW:277.1813 |
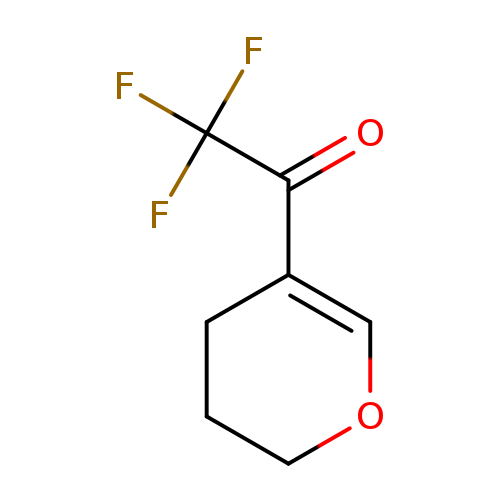
1-(3,4-Dihydro-2h-pyran-5-yl)-2,2,2-trifluoroethan-1-oneCatalog No.:AA008UGR CAS No.:109317-74-0 MDL No.:MFCD00975077 MF:C7H7F3O2 MW:180.1245 |
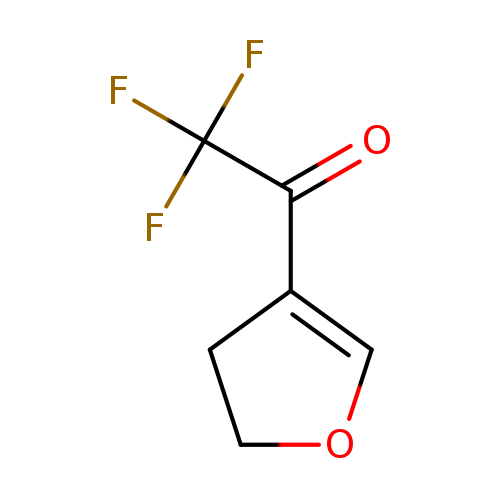
1-(4,5-Dihydrofuran-3-yl)-2,2,2-trifluoroethanoneCatalog No.:AA008S64 CAS No.:109317-75-1 MDL No.:MFCD00728712 MF:C6H5F3O2 MW:166.0979 |
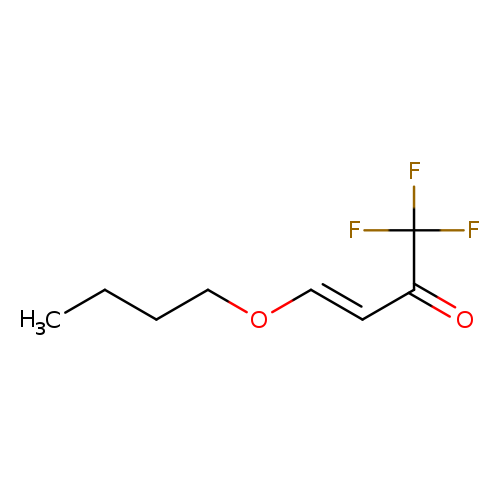
3-Buten-2-one, 4-butoxy-1,1,1-trifluoro-, (3E)-Catalog No.:AA007T9W CAS No.:109317-78-4 MDL No.:MFCD09259041 MF:C8H11F3O2 MW:196.1669 |
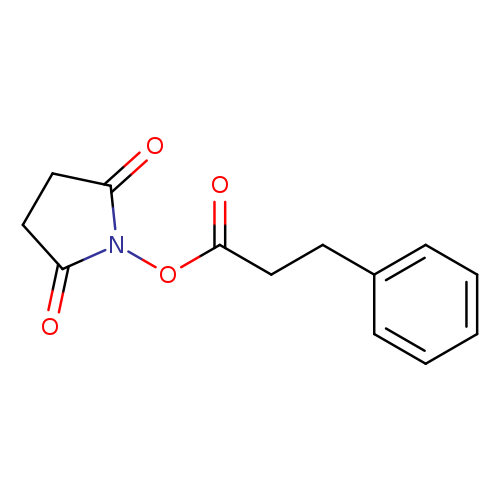
Hydrocinnamic acid n-hydroxysuccinimide esterCatalog No.:AA008WDH CAS No.:109318-10-7 MDL No.:MFCD09840689 MF:C13H13NO4 MW:247.2466 |
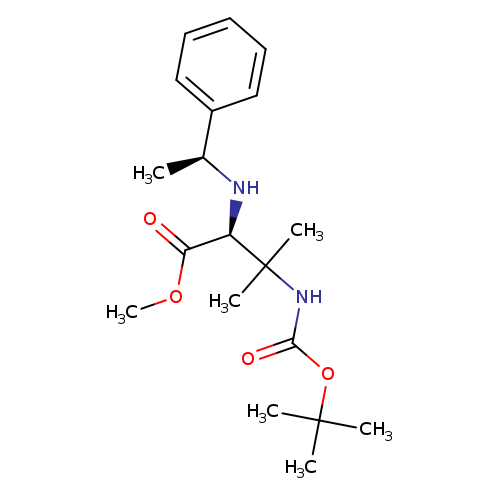
(S)-Methyl 3-(tert-butoxycarbonylamino)-3-methyl-2-((s)-1-phenylethylamino)butanoateCatalog No.:AA00HBCM CAS No.:1093192-06-3 MDL No.:MFCD27992079 MF:C19H30N2O4 MW:350.4525 |
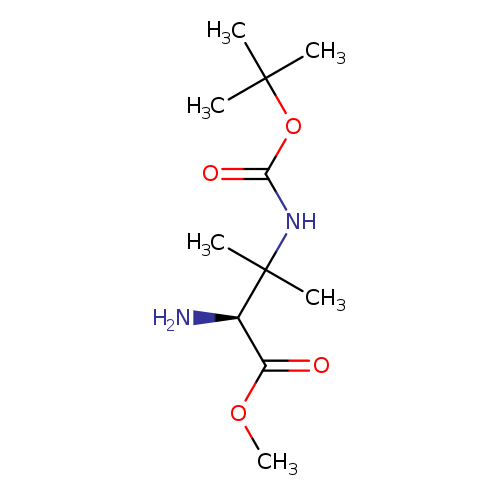
(S)-Methyl 2-amino-3-(tert-butoxycarbonylamino)-3-methylbutanoateCatalog No.:AA0095MO CAS No.:1093192-07-4 MDL No.:MFCD16293742 MF:C11H22N2O4 MW:246.3034 |
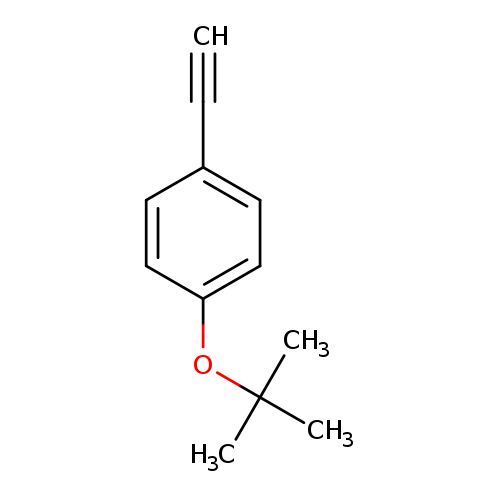
Benzene, 1-(1,1-diMethylethoxy)-4-ethynyl-Catalog No.:AA0093RW CAS No.:1093192-41-6 MDL No.:MFCD06797761 MF:C12H14O MW:174.2390 |
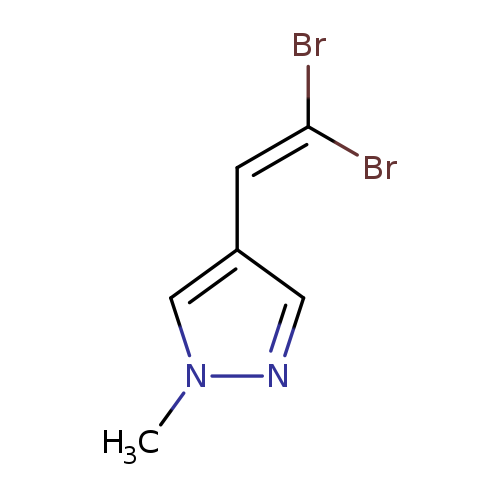
4-(2,2-Dibromoethenyl)-1-methylpyrazoleCatalog No.:AA00HBCN CAS No.:1093193-15-7 MDL No.:MFCD23703723 MF:C6H6Br2N2 MW:265.9332 |
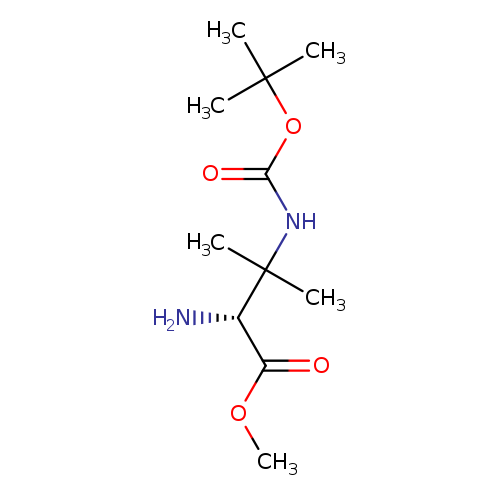
(R)-Methyl 2-amino-3-(tert-butoxycarbonylamino)-3-methylbutanoateCatalog No.:AA00HBCO CAS No.:1093198-33-4 MDL No.:MFCD28991774 MF:C11H22N2O4 MW:246.3034 |
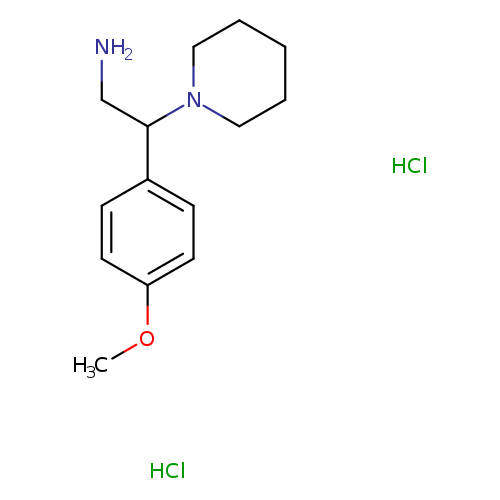
2-(4-Methoxy-phenyl)-2-piperidin-1-yl-ethylamine dihydrochlorideCatalog No.:AA01FP8B CAS No.:1093199-31-5 MDL No.:MFCD09698502 MF:C14H24Cl2N2O MW:307.2592 |
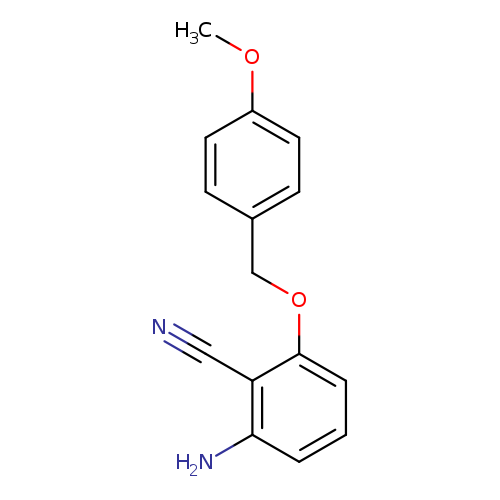
2-Amino-6-(4-methoxy-benzyloxy)-benzonitrileCatalog No.:AA0096Y1 CAS No.:1093203-81-6 MDL No.:MFCD30529883 MF:C15H14N2O2 MW:254.2839 |
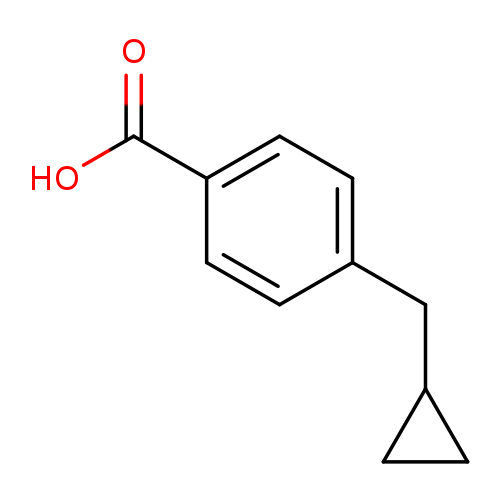
4-(cyclopropylmethyl)benzoic acidCatalog No.:AA01EK51 CAS No.:1093214-56-2 MDL No.:MFCD20660132 MF:C11H12O2 MW:176.2118 |
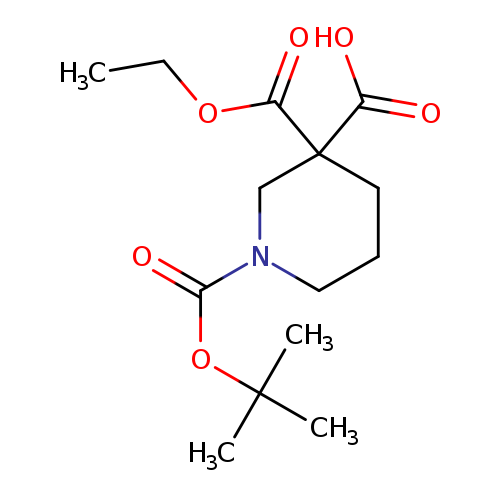
1-[(tert-butoxy)carbonyl]-3-(ethoxycarbonyl)piperidine-3-carboxylic acidCatalog No.:AA01EKAP CAS No.:1093214-91-5 MDL No.:MFCD24470819 MF:C14H23NO6 MW:301.3355 |
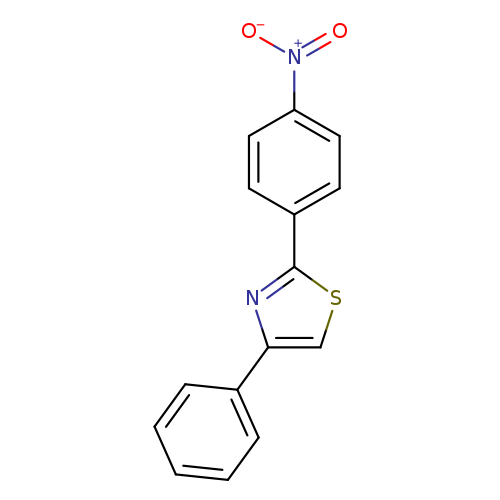
2-(4-Nitrophenyl)-4-phenylthiazoleCatalog No.:AA01FDYR CAS No.:1093215-83-8 MDL No.:MFCD27978262 MF:C15H10N2O2S MW:282.3171 |
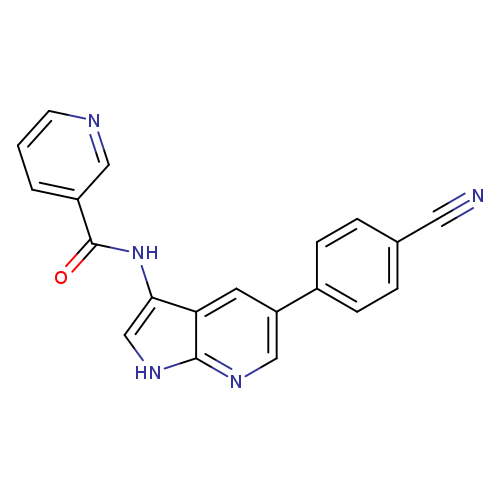
N-[5-(4-Cyanophenyl)-1H-pyrrolo[2,3-b]pyridin-3-yl]-3-pyridinecarboxamideCatalog No.:AA008TEL CAS No.:1093222-27-5 MDL No.:MFCD15528944 MF:C20H13N5O MW:339.3501 |
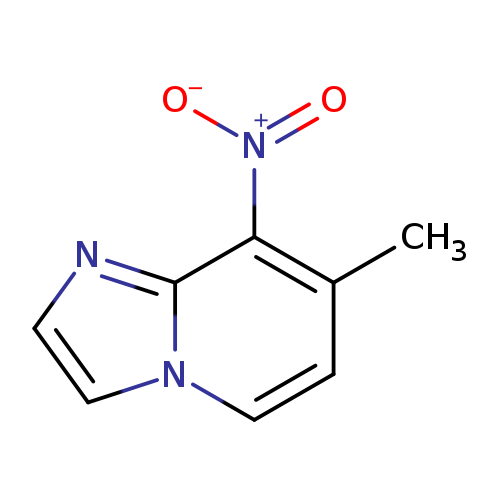
7-Methyl-8-nitroimidazo[1,2-a]pyridineCatalog No.:AA0096WL CAS No.:1093239-74-7 MDL No.:MFCD21336516 MF:C8H7N3O2 MW:177.1601 |
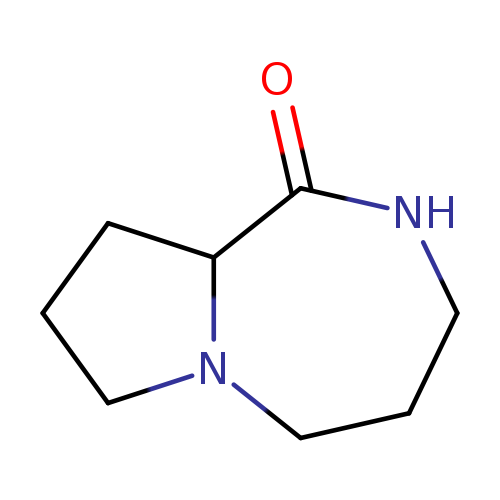
2,3,4,5-Tetrahydro-1H-pyrrolo[1,2-a][1,4]diazepin-1-oneCatalog No.:AA0094M5 CAS No.:109324-81-4 MDL No.:MFCD09832104 MF:C8H14N2O MW:154.2096 |
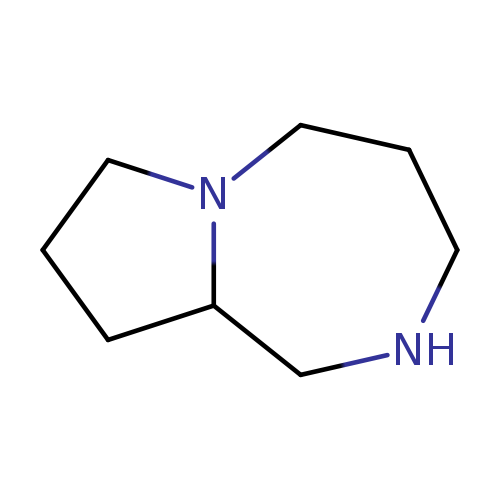
Octahydro-1h-pyrrolo[1,2-a][1,4]diazepineCatalog No.:AA008RL9 CAS No.:109324-83-6 MDL No.:MFCD09832105 MF:C8H16N2 MW:140.2260 |
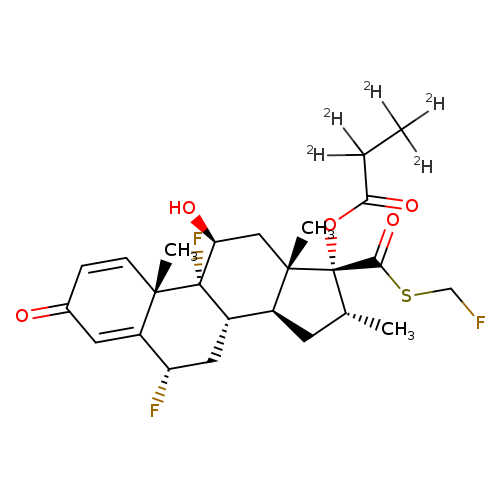
Fluticasone propionate-d5Catalog No.:AA008WZP CAS No.:1093258-28-6 MDL No.:MFCD08064194 MF:C25H26D5F3O5S MW:505.6017 |
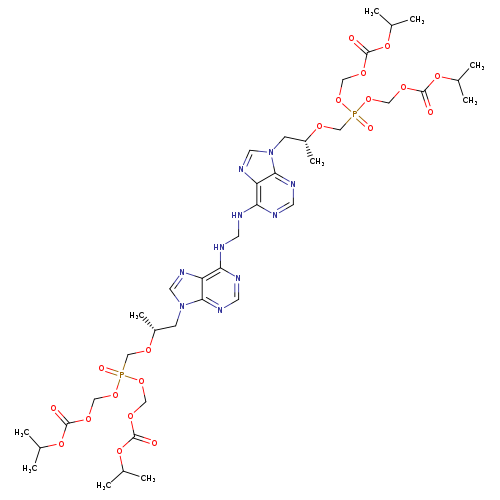
Tenofovir Disoproxil DimerCatalog No.:AA008W7C CAS No.:1093279-76-5 MDL No.:MFCD21363857 MF:C39H60N10O20P2 MW:1050.8962 |
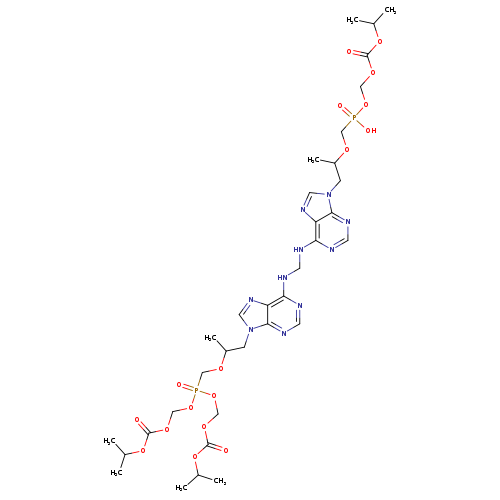
2,4,6,8-Tetraoxa-5-phosphanonanedioic acid, 5-[[(1R)-2-[6-[[[[9-[(2R)-5-hydroxy-2,11-diMethyl-5-oxido-9-oxo-3,6,8,10-tetraoxa-5-phosphadodec-1-yl]-9H-purin-6-yl]aMino]Methyl]aMino]-9H-purin-9-yl]-1-Methylethoxy]Methyl]-, 1,9-bis(1-Methylethyl) ester, 5-oxCatalog No.:AA008W0E CAS No.:1093279-77-6 MDL No.:MFCD28016539 MF:C34H52N10O17P2 MW:934.7810 |
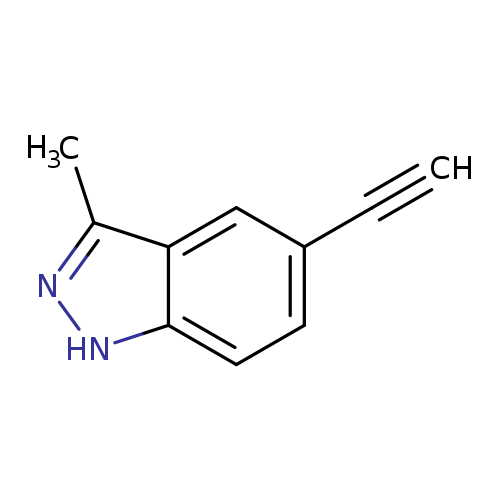
5-Ethynyl-3-methyl-1h-indazoleCatalog No.:AA0082H7 CAS No.:1093307-29-9 MDL No.:MFCD14584652 MF:C10H8N2 MW:156.1839 |
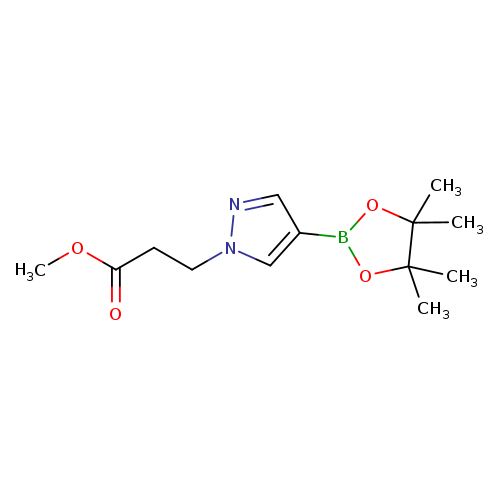
Methyl 3-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1h-pyrazol-1-yl)propanoateCatalog No.:AA007T4R CAS No.:1093307-33-5 MDL No.:MFCD16660987 MF:C13H21BN2O4 MW:280.1278 |
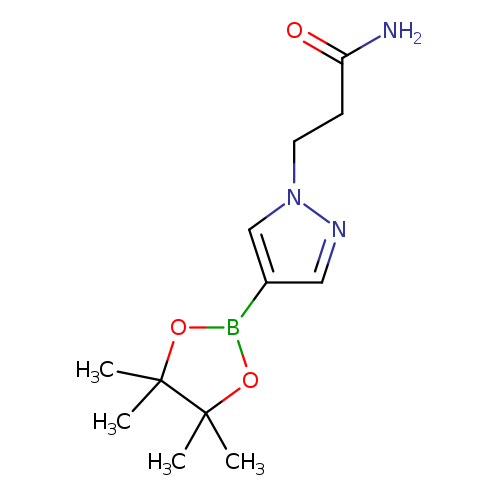
3-(4-(4,4,5,5-Tetramethyl-1,3,2-dioxaborolan-2-yl)-1h-pyrazol-1-yl)propanamideCatalog No.:AA007AY7 CAS No.:1093307-34-6 MDL No.:MFCD16660238 MF:C12H20BN3O3 MW:265.1165 |
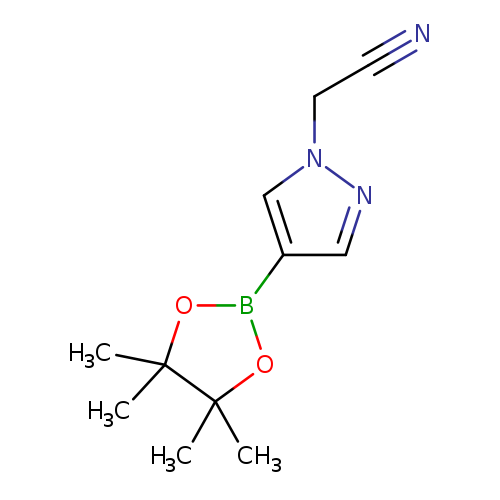
2-(4-(4,4,5,5-Tetramethyl-1,3,2-dioxaborolan-2-yl)-1h-pyrazol-1-yl)acetonitrileCatalog No.:AA003A3K CAS No.:1093307-35-7 MDL No.:MFCD17011852 MF:C11H16BN3O2 MW:233.0746 |
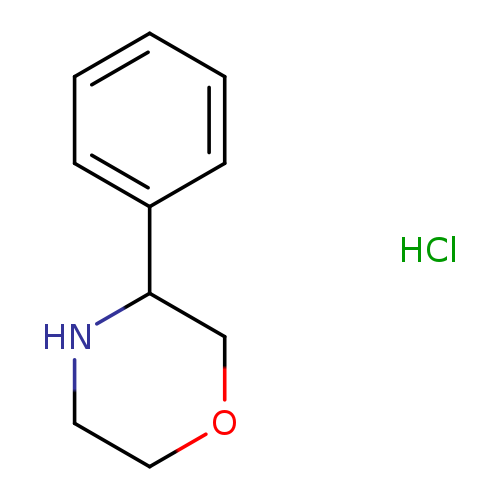
3-Phenylmorpholine, HClCatalog No.:AA00964A CAS No.:1093307-44-8 MDL No.:MFCD03840086 MF:C10H14ClNO MW:199.6773 |
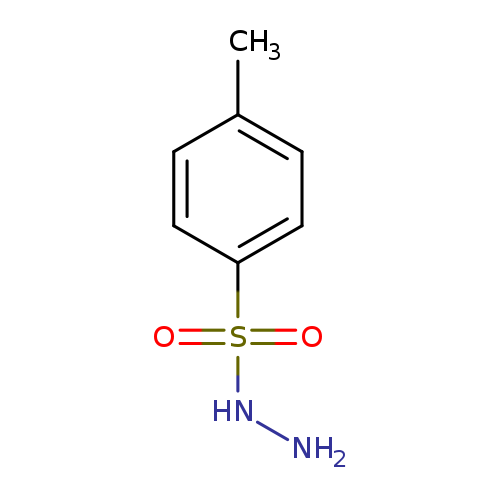
P-TOLUENESULFONYLHYDRAZIDE-N,N,N-D3Catalog No.:AA008SY7 CAS No.:109333-73-5 MDL No.:MFCD03428206 MF:C7H10N2O2S MW:186.2315 |
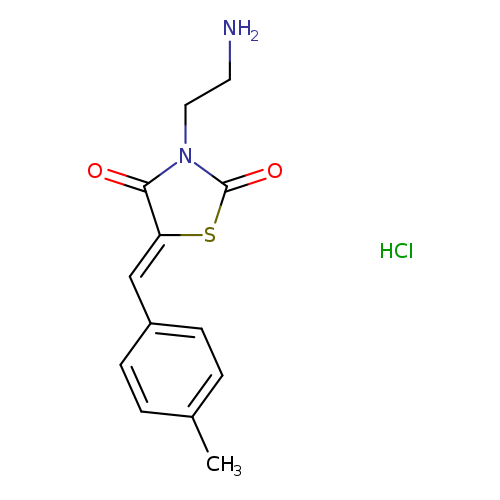
(5Z)-3-(2-aminoethyl)-5-[(4-methylphenyl)methylidene]-1,3-thiazolidine-2,4-dione hydrochlorideCatalog No.:AA019HSR CAS No.:1093344-89-8 MDL No.:MFCD08445272 MF:C13H15ClN2O2S MW:298.7884 |

Methyl 2,3,5-tri-O-benzoyl-b-D-arabinofuranosideCatalog No.:AA01EAU2 CAS No.:1093344-99-0 MDL No.: MF: MW: |
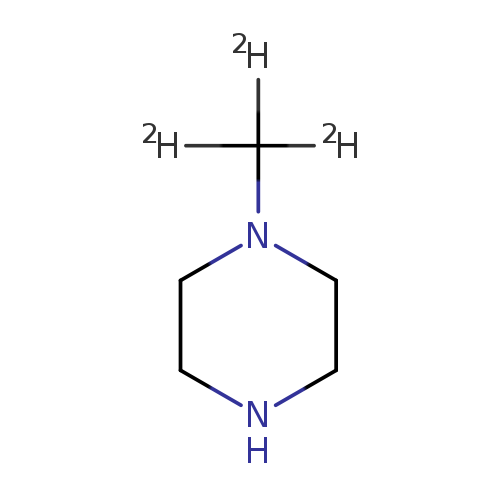
N-METHYL-D3-PIPERAZINECatalog No.:AA008SYE CAS No.:1093380-08-5 MDL No.:MFCD03428305 MF:C5H9D3N2 MW:103.1807 |
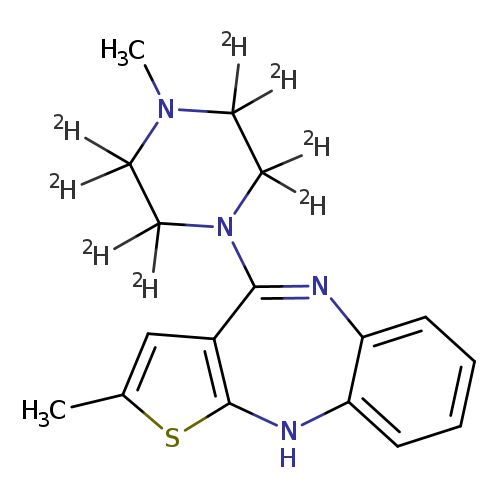
2-Methyl-4-(4-methyl-1-piperazinyl-2,2,3,3,5,5,6,6-d8)-10H-thieno[2,3-b][1,5]benzodiazepineCatalog No.:AA01ENIZ CAS No.:1093380-13-2 MDL No.:MFCD11977864 MF:C17H12D8N4S MW:320.4818 |
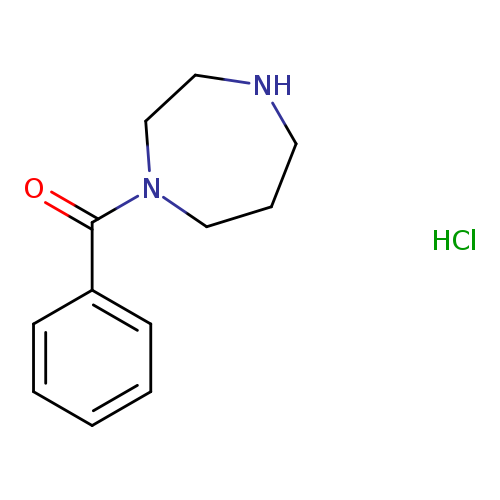
1-Benzoyl-1,4-diazepane hydrochlorideCatalog No.:AA019LA6 CAS No.:1093380-30-3 MDL No.:MFCD09863847 MF:C12H17ClN2O MW:240.7292 |
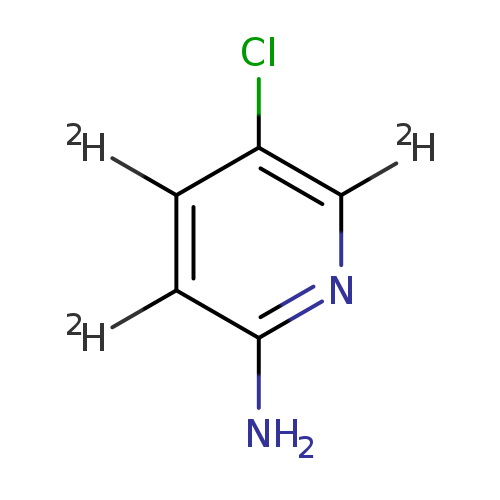
2-AMino-5-chloropyridine--d3Catalog No.:AA0094S0 CAS No.:1093384-99-6 MDL No.:MFCD08705155 MF:C5H2ClD3N2 MW:131.5781 |

ESZOPICLONE-D8Catalog No.:AA0094AV CAS No.:1093385-24-0 MDL No.: MF: MW: |
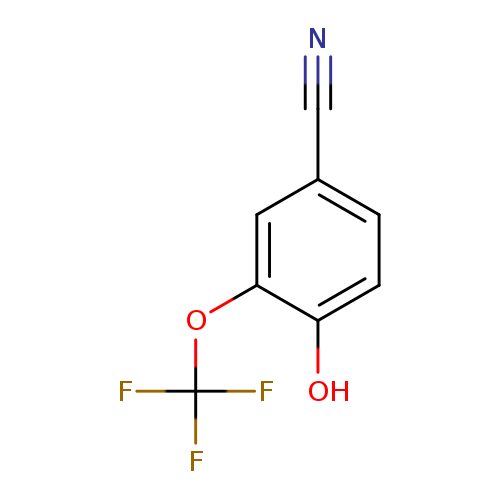
4-Hydroxy-3-(trifluoromethoxy)benzonitrileCatalog No.:AA00HBCU CAS No.:1093397-72-8 MDL No.:MFCD18397473 MF:C8H4F3NO2 MW:203.1181 |
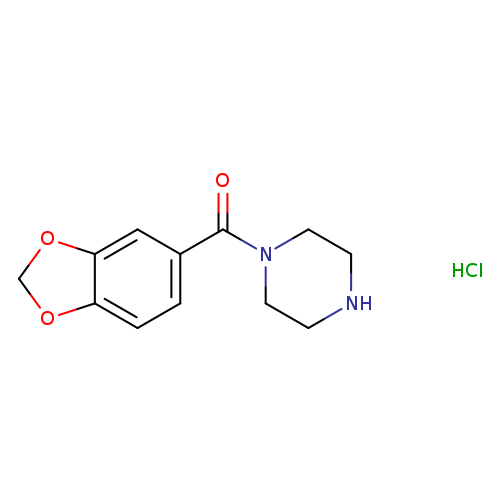
Benzo[d][1,3]dioxol-5-yl(piperazin-1-yl)methanone hydrochlorideCatalog No.:AA008ZOQ CAS No.:1093402-61-9 MDL No.:MFCD10534024 MF:C12H15ClN2O3 MW:270.7121 |
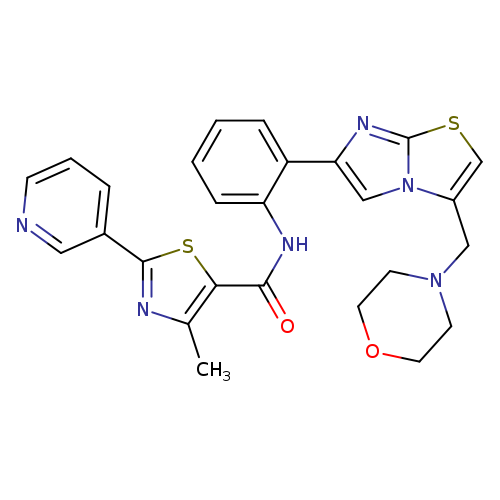
4-Methyl-N-(2-(3-(morpholinomethyl)imidazo[2,1-b]thiazol-6-yl)phenyl)-2-(pyridin-3-yl)thiazole-5-carboxamideCatalog No.:AA008TGQ CAS No.:1093403-33-8 MDL No.:MFCD22572733 MF:C26H24N6O2S2 MW:516.6378 |
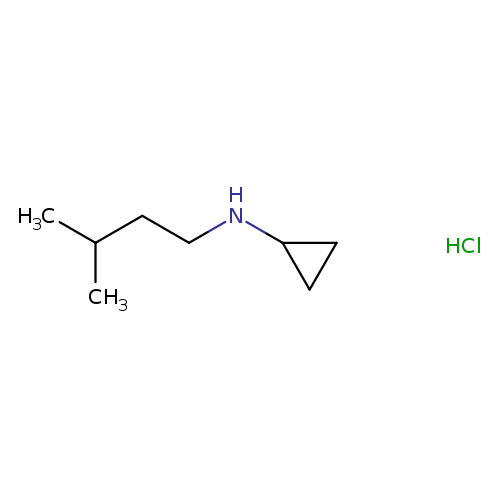
N-(3-methylbutyl)cyclopropanamine hydrochlorideCatalog No.:AA00J1UA CAS No.:1093403-65-6 MDL No.:MFCD09971442 MF:C8H18ClN MW:163.6882 |
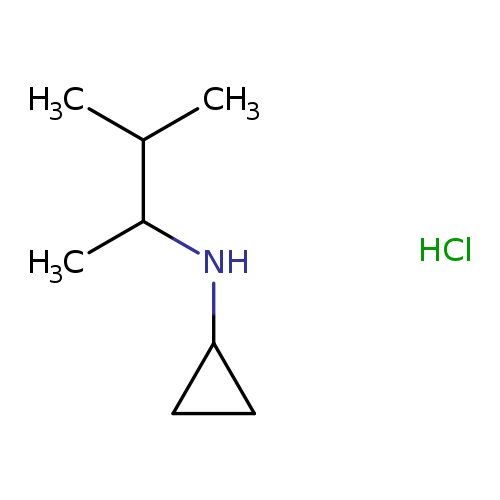
N-(3-methylbutan-2-yl)cyclopropanamine hydrochlorideCatalog No.:AA019LDX CAS No.:1093403-67-8 MDL No.:MFCD09971421 MF:C8H18ClN MW:163.6882 |
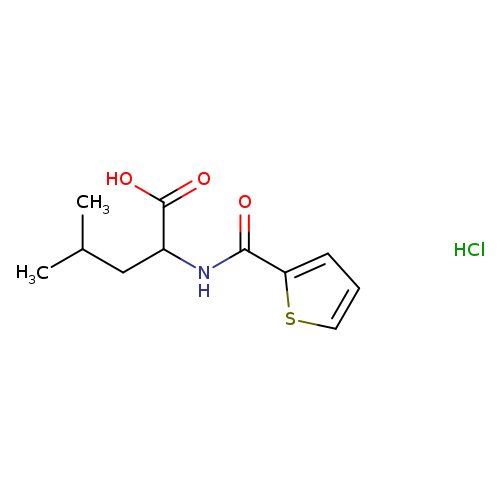
N-(2-Thienylcarbonyl)leucine hydrochlorideCatalog No.:AA01EHM8 CAS No.:1093404-71-7 MDL No.:MFCD09907575 MF:C11H16ClNO3S MW:277.7676 |
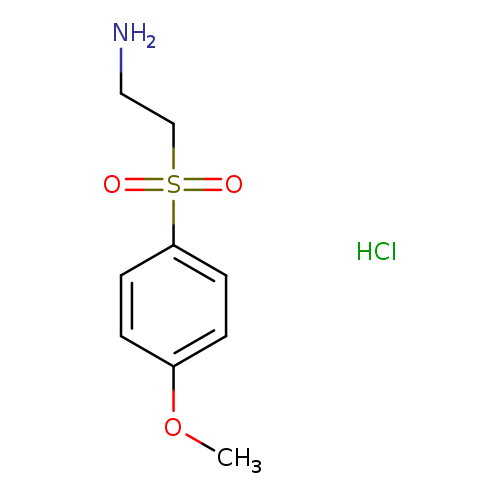
2-((4-Methoxyphenyl)sulfonyl)ethanamine hydrochlorideCatalog No.:AA01APHI CAS No.:1093405-08-3 MDL No.:MFCD09907570 MF:C9H14ClNO3S MW:251.7304 |
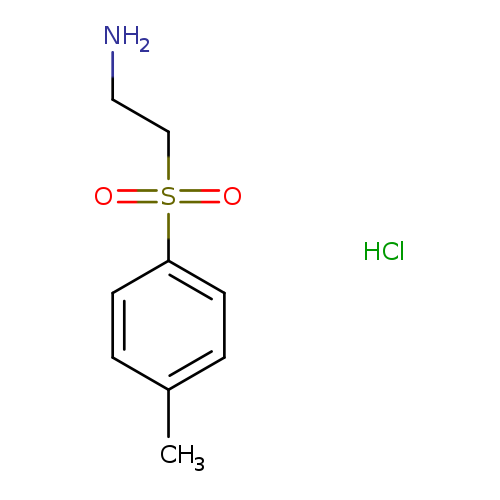
2-(4-Methyl-benzenesulfonyl)ethylamine, HClCatalog No.:AA009990 CAS No.:1093405-12-9 MDL No.:MFCD03840167 MF:C9H14ClNO2S MW:235.7310 |
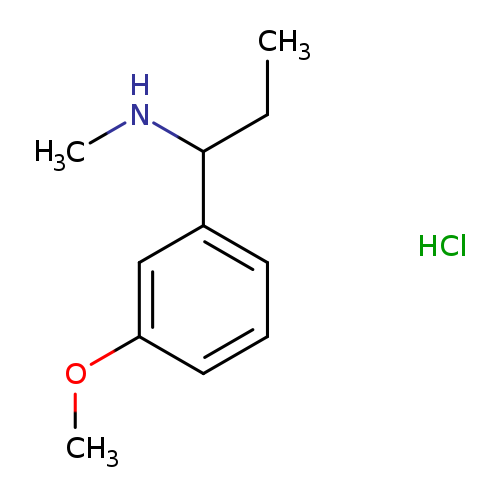
[1-(3-Methoxyphenyl)propyl]methylamine hydrochlorideCatalog No.:AA01BBMQ CAS No.:1093405-43-6 MDL No.:MFCD10083097 MF:C11H18ClNO MW:215.7197 |
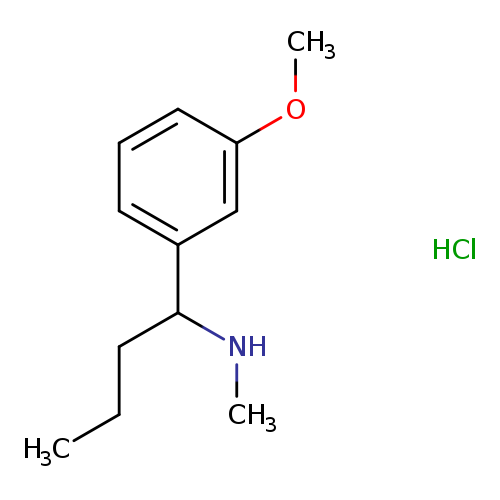
[1-(3-Methoxyphenyl)butyl]methylamine hydrochlorideCatalog No.:AA01APHJ CAS No.:1093405-61-8 MDL No.:MFCD09907573 MF:C12H20ClNO MW:229.7463 |
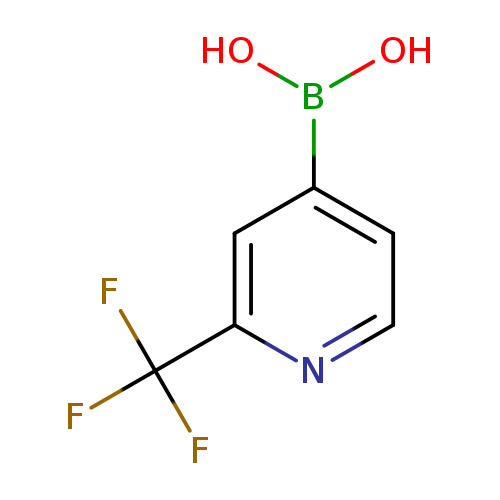
2-(Trifluoromethyl)pyridine-4-boronic acidCatalog No.:AA003BHI CAS No.:1093407-58-9 MDL No.:MFCD11616522 MF:C6H5BF3NO2 MW:190.9156 |
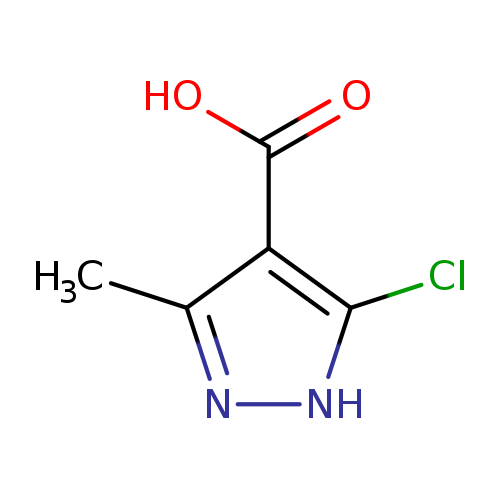
5-chloro-3-methyl-1H-pyrazole-4-carboxylic acidCatalog No.:AA01E8Z3 CAS No.:1093414-62-0 MDL No.:MFCD00995442 MF:C5H5ClN2O2 MW:160.5584 |
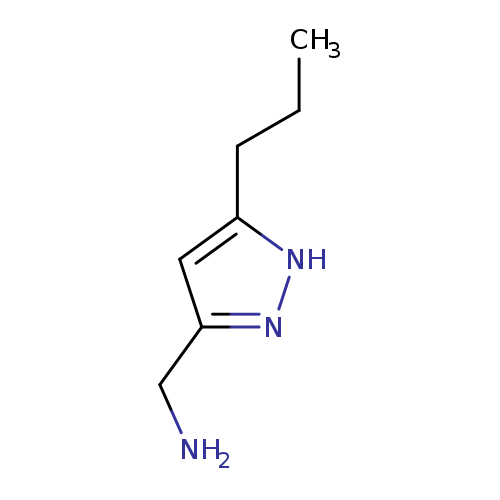
(5-Propyl-1h-pyrazol-3-yl)methanamineCatalog No.:AA003I3F CAS No.:1093415-70-3 MDL No.:MFCD08700610 MF:C7H13N3 MW:139.1982 |
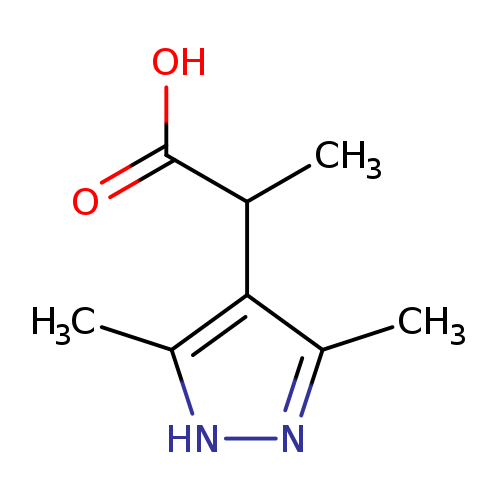
2-(3,5-Dimethyl-1h-pyrazol-4-yl)propanoic acidCatalog No.:AA01AGK3 CAS No.:1093416-51-3 MDL No.:MFCD08700814 MF:C8H12N2O2 MW:168.1931 |
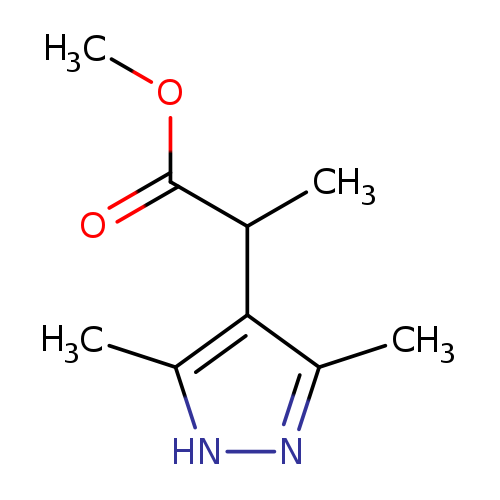
methyl 2-(3,5-dimethyl-1H-pyrazol-4-yl)propanoateCatalog No.:AA019ZDH CAS No.:1093416-52-4 MDL No.:MFCD08700815 MF:C9H14N2O2 MW:182.2197 |
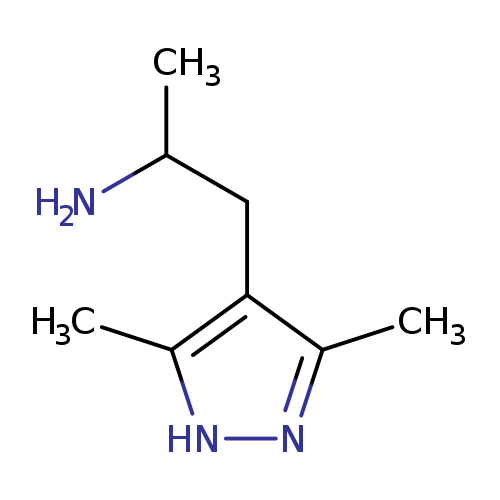
1-(3,5-dimethyl-1H-pyrazol-4-yl)propan-2-amineCatalog No.:AA01C46G CAS No.:1093416-63-7 MDL No.:MFCD08700861 MF:C8H15N3 MW:153.2248 |
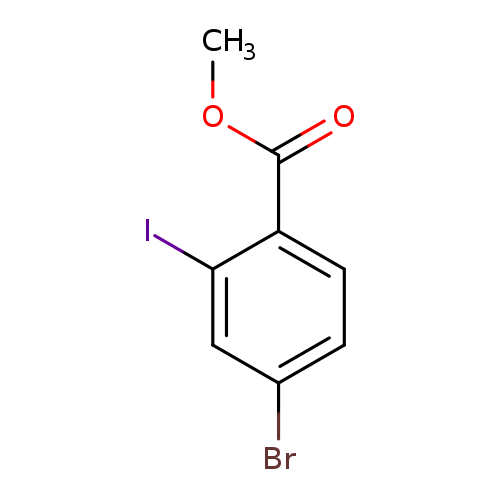
Methyl 4-bromo-2-iodobenzoateCatalog No.:AA0093IU CAS No.:1093418-75-7 MDL No.:MFCD16877107 MF:C8H6BrIO2 MW:340.9405 |
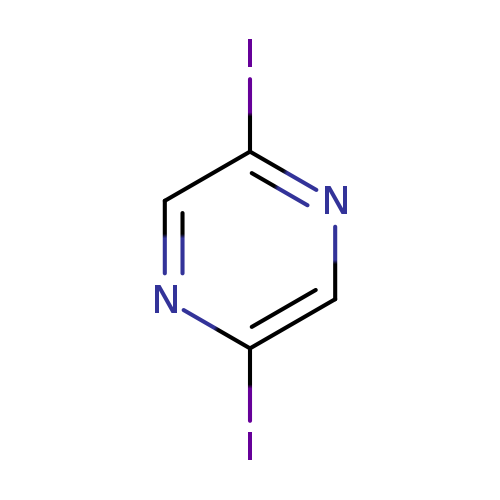
2,5-DiiodopyrazineCatalog No.:AA008WTM CAS No.:1093418-77-9 MDL No.:MFCD23143964 MF:C4H2I2N2 MW:331.8810 |
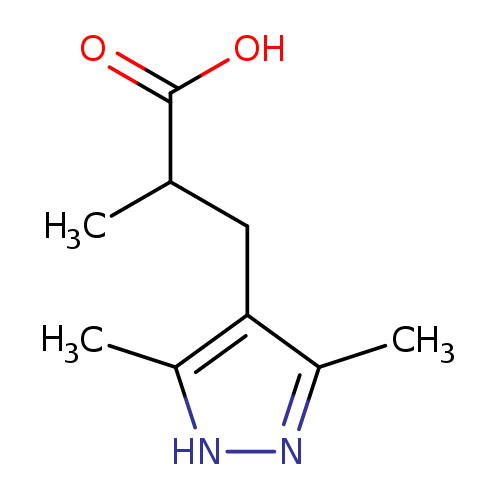
3-(3,5-dimethyl-1H-pyrazol-4-yl)-2-methylpropanoic acidCatalog No.:AA019PO3 CAS No.:1093430-18-2 MDL No.:MFCD08700961 MF:C9H14N2O2 MW:182.2197 |
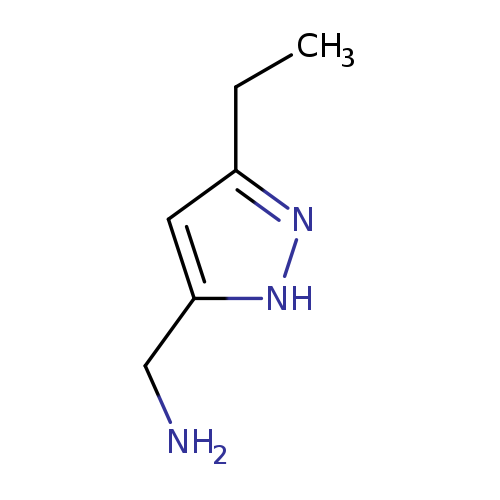
(3-ethyl-1H-pyrazol-5-yl)methanamineCatalog No.:AA01AHN0 CAS No.:1093430-33-1 MDL No.:MFCD08701010 MF:C6H11N3 MW:125.1716 |
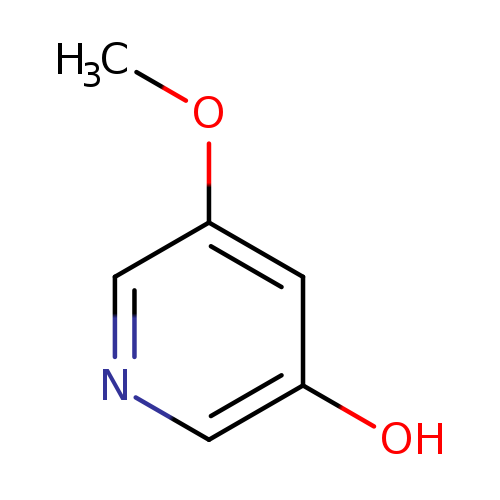
5-Methoxypyridin-3-olCatalog No.:AA003MRP CAS No.:109345-94-0 MDL No.:MFCD06797672 MF:C6H7NO2 MW:125.1253 |
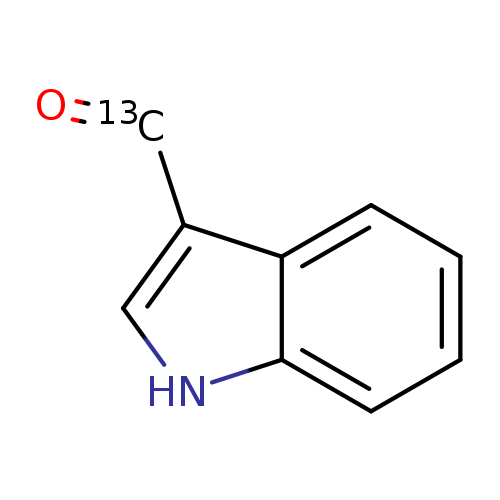
Indole-3-carboxaldehyde-13CCatalog No.:AA003QXG CAS No.:1093452-52-8 MDL No.:MFCD19443430 MF:C9H7NO MW:146.1506 |
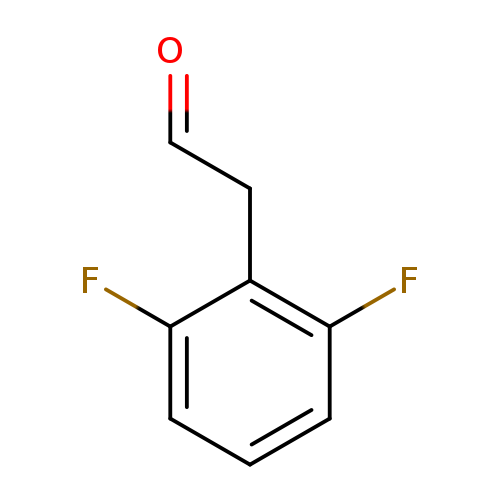
2-(2,6-Difluorophenyl)acetaldehydeCatalog No.:AA0091ZZ CAS No.:109346-83-0 MDL No.:MFCD11110139 MF:C8H6F2O MW:156.1294 |
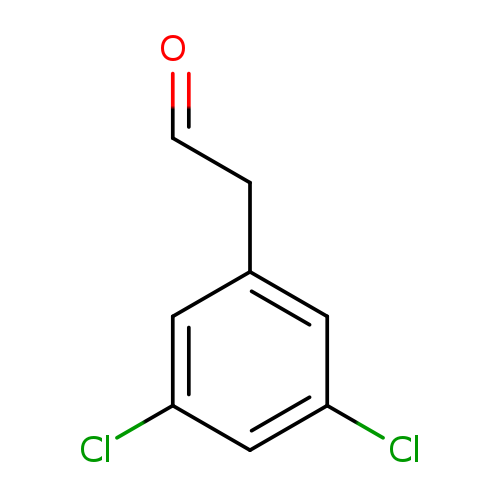
2-(3,5-Dichlorophenyl)acetaldehydeCatalog No.:AA009NR2 CAS No.:109346-95-4 MDL No.:MFCD02261742 MF:C8H6Cl2O MW:189.0386 |
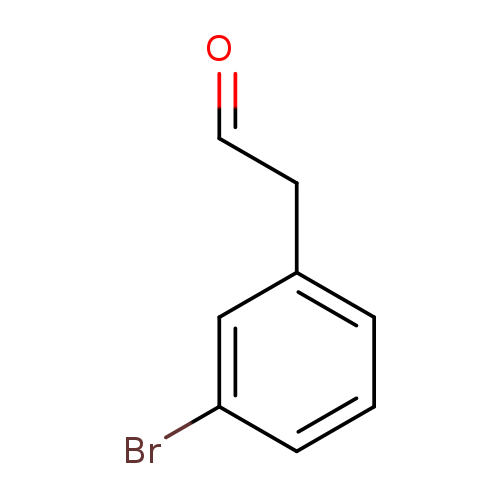
2-(3-Bromophenyl)acetaldehydeCatalog No.:AA008S03 CAS No.:109347-40-2 MDL No.:MFCD02261727 MF:C8H7BrO MW:199.0446 |
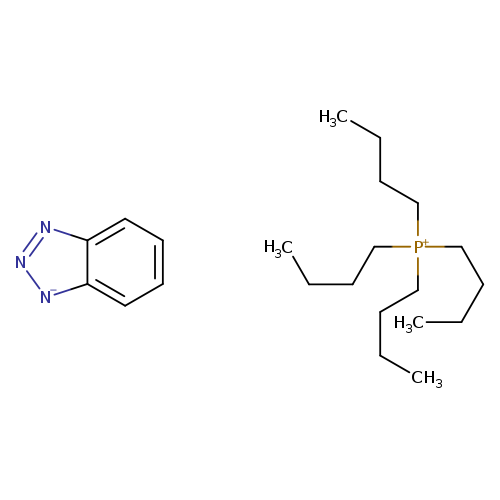
Tetrabutylphosphonium benzotriazolateCatalog No.:AA003UK9 CAS No.:109348-55-2 MDL No.:MFCD04038148 MF:C22H40N3P MW:377.5469 |
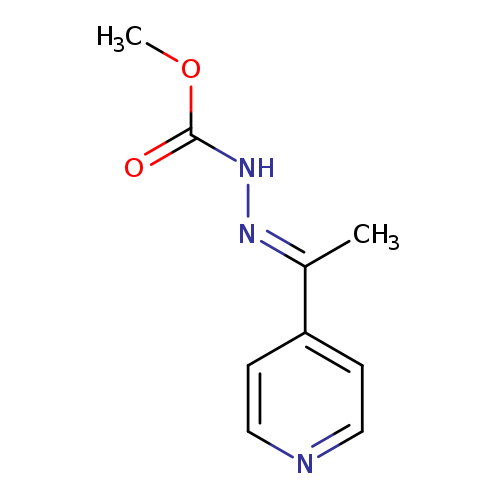
N'-[(1E)-1-(pyridin-4-yl)ethylidene]methoxycarbohydrazideCatalog No.:AA00IPIE CAS No.:109352-76-3 MDL No.:MFCD00792962 MF:C9H11N3O2 MW:193.2025 |
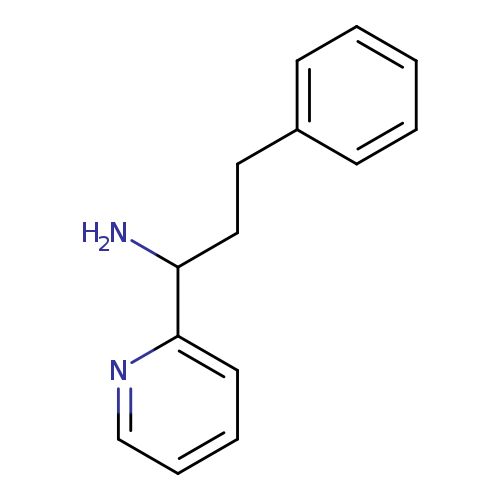
3-phenyl-1-(pyridin-2-yl)propan-1-amineCatalog No.:AA01AU21 CAS No.:1093602-21-1 MDL No.:MFCD12153919 MF:C14H16N2 MW:212.2902 |
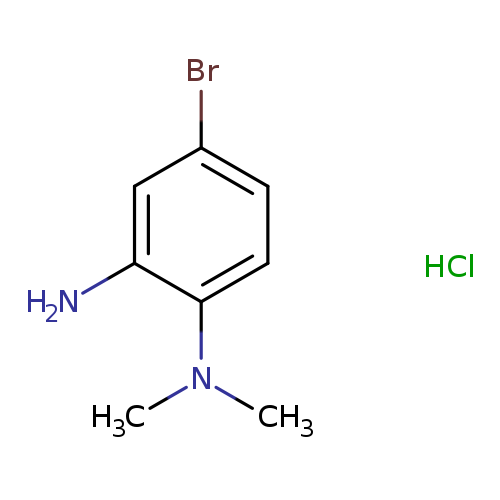
(2-amino-4-bromophenyl)dimethylamine hydrochlorideCatalog No.:AA00J1TT CAS No.:1093619-91-0 MDL No.:MFCD10565005 MF:C8H12BrClN2 MW:251.5513 |
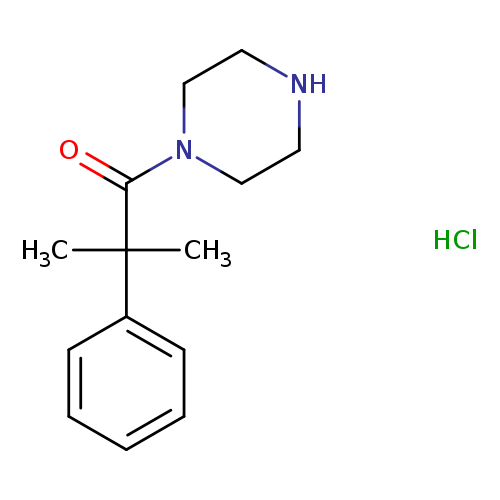
2-methyl-2-phenyl-1-(piperazin-1-yl)propan-1-one hydrochlorideCatalog No.:AA019L77 CAS No.:1093619-94-3 MDL No.:MFCD09863368 MF:C14H21ClN2O MW:268.7823 |
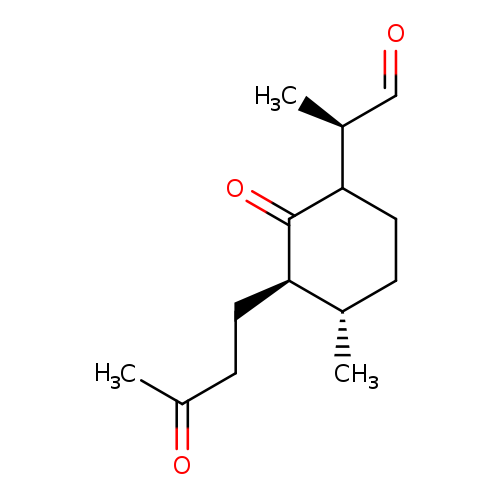
(2S,3R,6RS)-2-(3-Oxobutyl)-3-Methyl-6-[(R)-2-propanal]cyclohexanoneCatalog No.:AA008WNL CAS No.:1093625-96-7 MDL No.: MF:C14H22O3 MW:238.3227 |
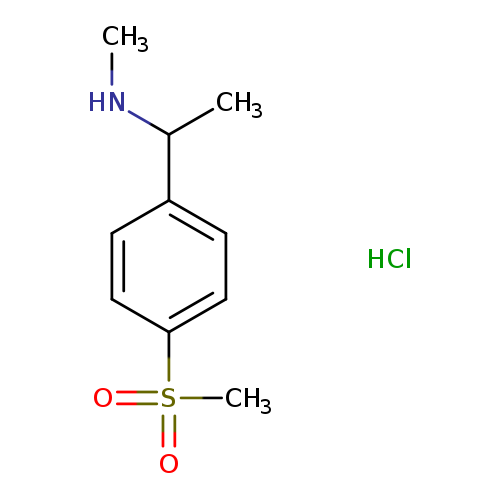
N-Methyl-n-(1-[4-(methylsulfonyl)phenyl]ethyl)amine hydrochlorideCatalog No.:AA019L2F CAS No.:1093630-40-0 MDL No.:MFCD09863318 MF:C10H16ClNO2S MW:249.7575 |
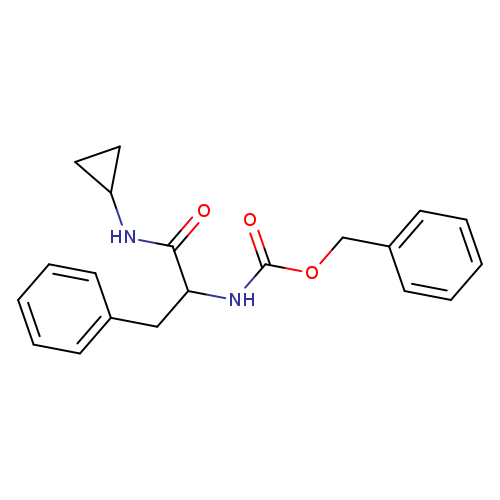
N-Cyclopropyl L-Z-PhenylalaninamideCatalog No.:AA00948C CAS No.:1093631-63-0 MDL No.:MFCD10402053 MF:C20H22N2O3 MW:338.4003 |
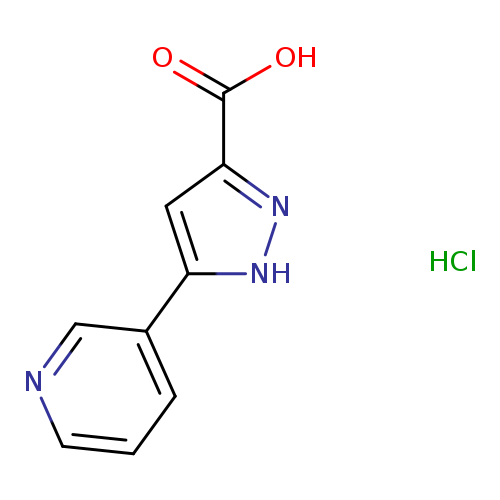
5-(Pyridin-3-yl)-1H-pyrazole-3-carboxylic acid hydrochlorideCatalog No.:AA009A2S CAS No.:1093636-79-3 MDL No.:MFCD09997651 MF:C9H8ClN3O2 MW:225.6317 |
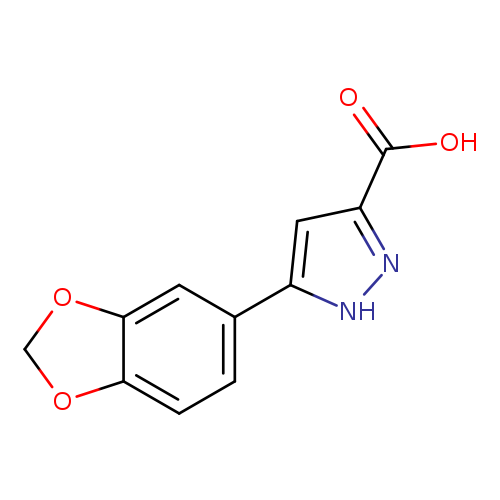
5-(1,3-benzodioxol-5-yl)-1H-pyrazole-3-carboxylic acidCatalog No.:AA00JEKE CAS No.:1093642-04-6 MDL No.:MFCD04969779 MF:C11H8N2O4 MW:232.1922 |
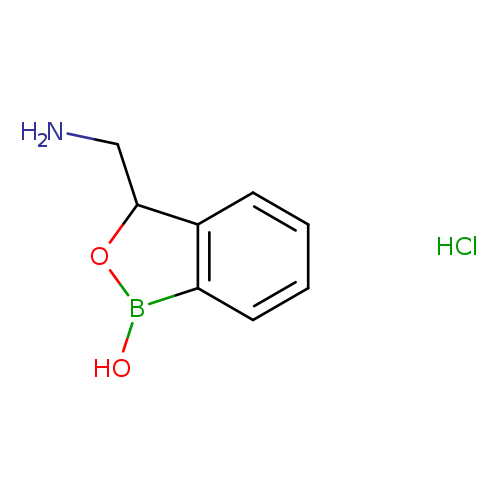
3-(aminomethyl)-1,3-dihydro-2,1-benzoxaborol-1-ol hydrochlorideCatalog No.:AA01B207 CAS No.:1093642-78-4 MDL No.:MFCD29907297 MF:C8H11BClNO2 MW:199.4424 |
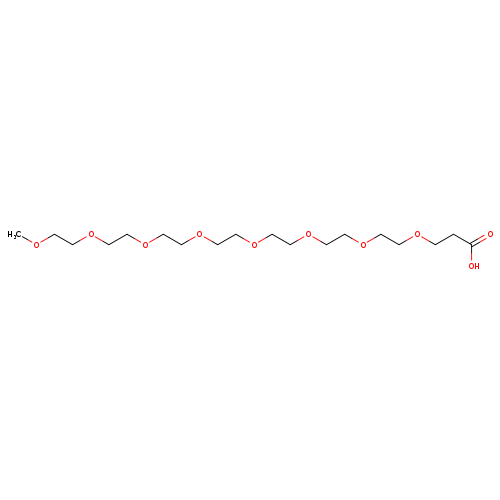
m-PEG8-acidCatalog No.:AA008U6O CAS No.:1093647-41-6 MDL No.:MFCD13184957 MF:C18H36O10 MW:412.4724 |
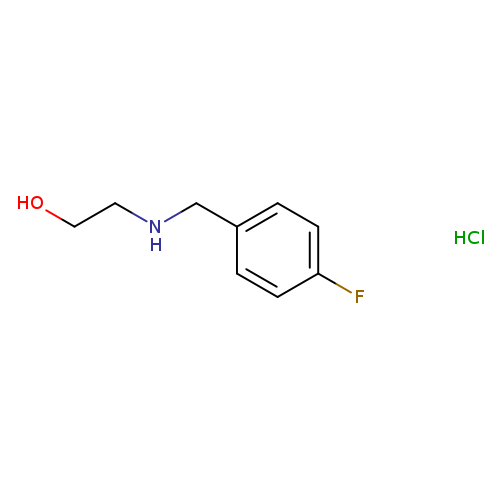
2-[(4-Fluorobenzyl)amino]ethanol hydrochlorideCatalog No.:AA0090VD CAS No.:1093648-13-5 MDL No.:MFCD13184430 MF:C9H13ClFNO MW:205.6570 |
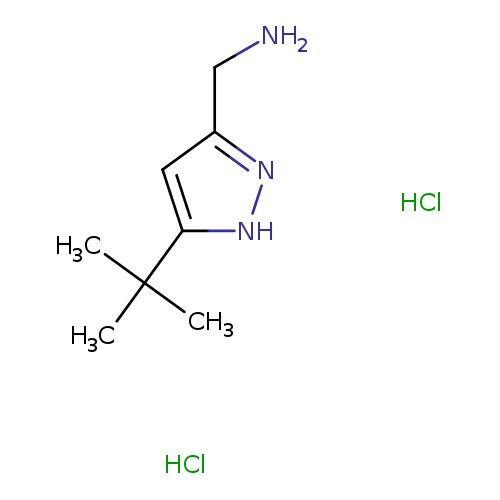
[(5-tert-Butyl-1h-pyrazol-3-yl)methyl]amine dihydrochlorideCatalog No.:AA0090UH CAS No.:1093649-71-8 MDL No.:MFCD31008931 MF:C8H17Cl2N3 MW:226.1467 |
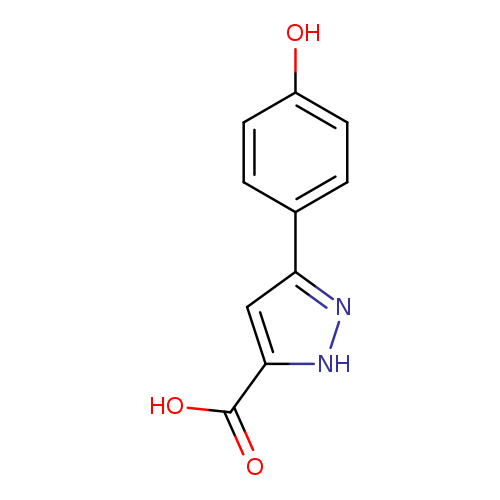
3-(4-Hydroxyphenyl)-1H-pyrazole-5-carboxylic acidCatalog No.:AA00HBD0 CAS No.:1093649-88-7 MDL No.:MFCD08282768 MF:C10H8N2O3 MW:204.1821 |
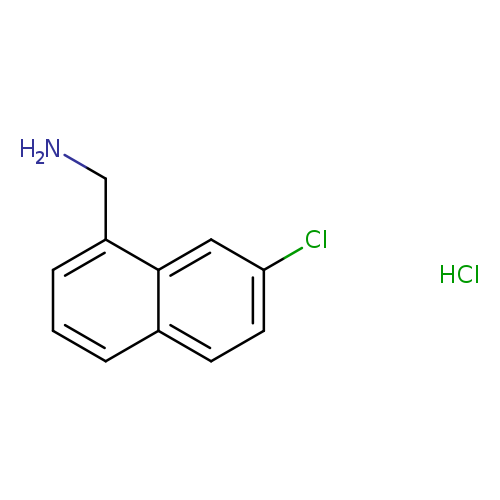
(7-chloronaphthalen-1-yl)methanamine hydrochlorideCatalog No.:AA01DUW1 CAS No.:1093651-77-4 MDL No.:MFCD31617675 MF:C11H11Cl2N MW:228.1177 |
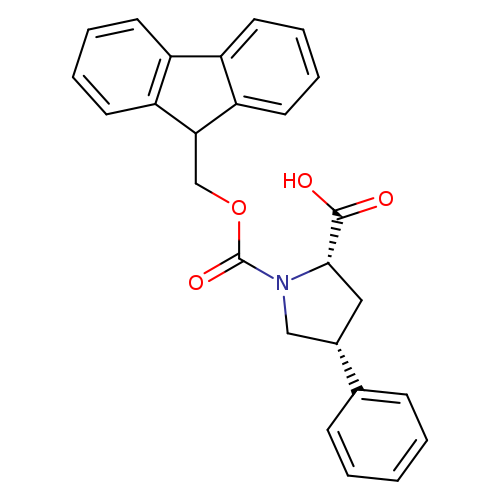
(2S,4R)-Fmoc-4-phenyl-pyrrolidine-2-carboxylic acidCatalog No.:AA00HBD1 CAS No.:1093651-96-7 MDL No.:MFCD06656464 MF:C26H23NO4 MW:413.4651 |
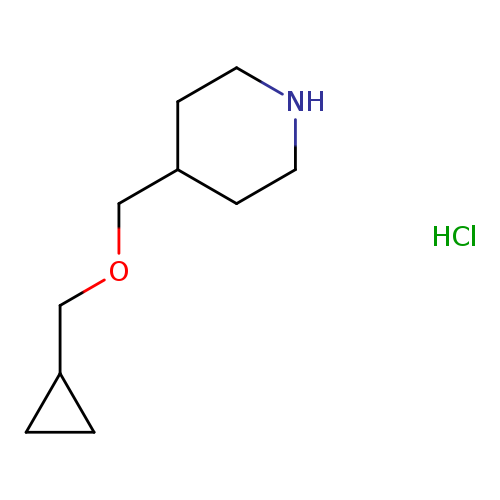
4-[(CYCLOPROPYLMETHOXY)METHYL]PIPERIDINEHYDROCHLORIDECatalog No.:AA008ZQJ CAS No.:1093652-85-7 MDL No.:MFCD09859629 MF:C10H20ClNO MW:205.7249 |
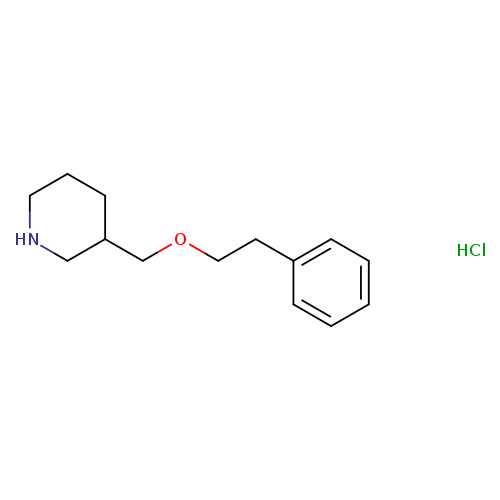
3-[(Phenethyloxy)methyl]piperidinehydrochlorideCatalog No.:AA01FO3U CAS No.:1093653-12-3 MDL No.: MF:C14H22ClNO MW:255.7836 |
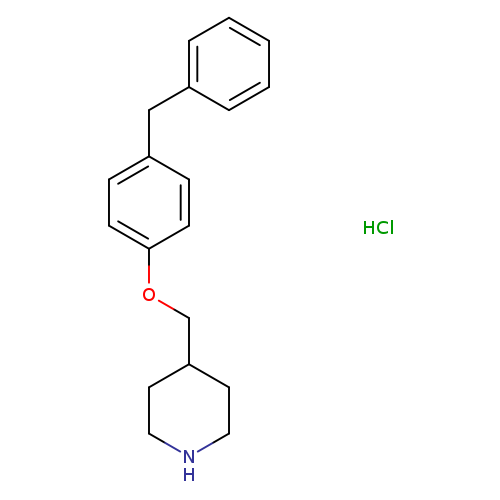
4-[(4-Benzylphenoxy)methyl]piperidinehydrochlorideCatalog No.:AA01FO8S CAS No.:1093653-13-4 MDL No.: MF:C19H24ClNO MW:317.8530 |
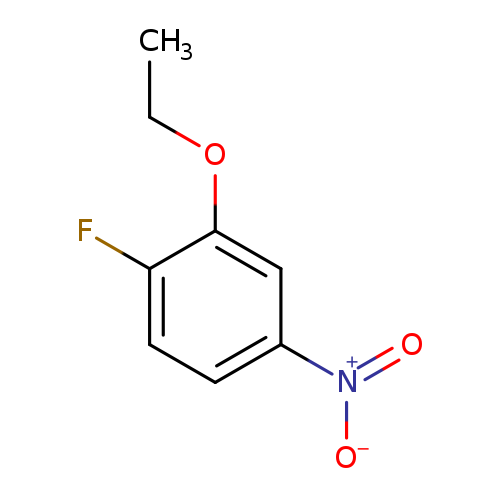
2-Ethoxy-1-fluoro-4-nitrobenzeneCatalog No.:AA009477 CAS No.:1093656-34-8 MDL No.:MFCD20133797 MF:C8H8FNO3 MW:185.1524 |