2020-02-15 11:00:30
Saumen Hajra, Subrata Maity, Sayan Roy, Ramkrishna Maity, and Srikrishna Samanta
Introduction
A numerous synthetic strategy towards the construction of 3, 3-bisindole have been reported so far in literature. The examples of (i) Mukaiyama-aldol reaction of 3-(3-indolyl)-2-siloxy- indole with aldehyde,[4] (ii) acyl migration of indolyl carbon- ates,[5] (iii) Pd-catalysed allylic alkylation of 3-aryl-3-oxindoles with allenes,[6] (iv) organocatalytic conjugate addition of indoles to isatin derived nitroalkenes and α,ß-unsturated aldehydes, (v) α-alkylation of carbonyl compounds with 3-hydroxy-3-indol-3-yloxindoles,[8] (vi) Rh-catalysed multicomponent reaction of 3-diazooxindoles, indoles and aldehydes,[9] (vii) Cu-catalysed dearomatization of indoleacetamides with 3-indolylphenyliodo- nium salts[10] are worthy of attention. However the preparation of 3-aryl oxindole moiety is sparse in literature.[11–13] Only few reports are there such as (i) Hetero-Claisen reaction of nitrones with alkylarylketenes,[11] (ii) Cu catalysed dearomatisation of tryptamine derivative with aryl-iodonium salt,[12] (iii) Lewis acid catalysed F-C alkylation of 3-hydroxy-2-oxindole.[13] Thus construction of both these functionalities from a common precur- sor through a concise and divergent manner is very much necessary. A straightforward strategy for the synthesis of these sub- units could be a regioselective F-C reaction of easily accessible spiro-epoxyoxindole with heteroarenes and arenes. Our contin- uous research interest in exploring the reactivity of three mem- bered reactive intermediates[14] led us to envisage the Lewis acid catalysed F-C reaction of spiro-epoxyoxindoles with arenes as well as heteroarenes. After the affirmative presumption, re- cently, we have successfully reported the Lewis-acid catalysed efficient F-C reaction of spiro-epoxyoxindoles with indoles and arenes to obtain both the functionalities.
Thus, spiro-epoxyoxindole has been emerged as a common precursor for the generation of all carbon quaternary centre prior to the synthesis of various indole alkaloids or their core structures. After this initial success, we are again exploring alternative catalysts/solvents combination for the F-C reaction of spiro-epoxyoxindole. Eventually, we have observed that cata- lytic amount of Brønsted acid in organic solvent can also acti- vate the spiro-epoxide[16] and lead to regioselective ring open- ing at the tertiary centre by the carbon nucleophiles likely ind- oles and arenes. Moreover, we have recently reported catalyst free on-water ring opening reaction of spiro-aziridineoxindole and indole.[17] This result again encourages us to find an envi- ronment friendly greener approach for this ring-opening reac- tion. Herein we incorporated a detailed study of the Brønsted acid assisted regioselective F-C reaction of spiro-epoxides and comparison with the Lewis acid catalysed reactions along with its scopes and limitations.
Results and Discussion
As we mentioned earlier, epoxides are versatile and privileged framework to synthesis a various indole alkaloids with immense biological significance. Now regioselective ring opening at the tertiary centre of spiro-epoxides[15,18] with different carbon nu- cleophiles lead to the formation of all carbon quaternary centre. Friedel–Crafts reaction is one of such highly efficient atom-eco- nomic C-C bond forming reaction, although there are very few efficient approaches for intermolecular F-C reaction of epoxide.[19] Thus this method for the spiroepoxide demands thorough reconnaissance.
Reaction with Indoles
After initial success with Lewis acid, we tried to find out some other milder catalyst condition for this F-C reaction. During our research, we observed that the F-C reaction of spiro-epoxide was also proceeded well with the assistance of catalytic amount of Brønsted acid. To confirm it, we performed the model reac- tion between N-methyl spiro-epoxyoxindole 1a and indole em- ploying various Brønsted acids in different temperature. Our previous result with Lewis acid (Scheme 1) convinced us to per- form the reaction in DCE solvent. But unfortunately the reaction was not completed at 0 °C using 10 mol-% of ortho-phosphoric acid. Raising the temperature to 25 °C accomplished the F-C reaction with 81 % isolated yield of the regioselectively desired 3-(3-indolyl)-oxindole-3-methanol product (Table 1, entries 1 and 2). Next we changed the Brønsted acids to increase the yield of the desired product. But we were unfortunate to ac- quire significant amount of desired product with catalytic amount of benzoic acid, p-nitro benzoic acid, triflic acid (entries 3–5). Eventually, we were delighted to achieve the desired F-C product in presence of 10 mol-% of trifluoroacetic acid (TFA) with excellent isolated yield (92 %) within 6 h at 25 °C (entry 6). Further increase of the Brønsted acid did not affect the yield or time, on the contrary diminishing the amount of Brønsted acid decelerated the reaction with incomplete conversion. Thus 10 mol-% of TFA in DCE at 25 °C was the most accepted condition.
Method A: Optimisation with Brønsted Acids in Organic Solvent
After successful optimisation, we wished to generalise the method. With this intention, we executed the F-C reaction with an assemblage of spiro-epoxyoxindoles and indoles. Almost all the cases, we achieved similar yield to that of our previously mentioned Lewis acid condition. Only electron withdrawing group on both spiro-epoxide as well as indole demanded more time for complete conversion.
Not only unprotected indoles, we also performed the reaction with N-protected indoles (Scheme 3). With N-methyl as well as N-benzyl indoles, we obtained regioselectively desired prod- uct 3,3-bisindole along with 15–20 % of 3,2-bisindole as a side product. But when we performed the same reaction with elec- tron withdrawing protecting group, viz. N-benzoyl indole we gained exclusively C(3)-C(3) product with very good (78 % to 82 %) isolated yield.
Method B: Optimisation of on-Water Reaction
Water has a long history as a reaction medium in organic chem- istry.[20] As a solvent, it possesses a very interesting property due to its extensive H-bonding ability. According to Jung- Marcus theory,[21] approximately 25 % of water molecules hav- ing free OH groups at the interface are available for potential H-bonding with the substrate on the surface. Recently, we dis- covered catalyst-free on-water regio- and stereoselective ring opening of spiroaziridine oxindoles with indoles.[17] This led us to presume that water can also activate spiro-epoxyoxindole similar to that of spiro-aziridine. But the extent of H-bonding is less in case of spiro-epoxide compare to spiroaziridine.
This attributed to the incomplete reaction, alongwith sub- stantial amount of yield (Table 2, entry 1). To increase the H- bonding activity, we then planned to use different Brønsted acids as a promoter. We initiated our inspection with the model substrate N-methyl spiro-epoxyoxindole 1a with indole using TFA as a promoter in aqueous medium. While 0.5 equiv. of TFA yielded 41 % yield at 50 °C, 1.0 equiv. of TFA afforded up to 50–52 % yield (entries 2–4).
Employing ortho-phosphoric acid instead of TFA did not in- crease the yield (entries 5, 6). On the contrary, more acidic triflic acid reduced the yield of the desired product due to C3-proto- nation of indole (entry 7). Next we applied relatively less acidic acetic acid which led to the clean reaction profile. With 0.5 equiv. of acetic acid at 50 °C rendered 42 % yield, while increasing the temperature as well as amount of acetic acid up to 1.0 equiv. improved the yield up to 68 % (entries 8–10).
In contrast, a large excess of acetic acid (water/acetic acid = 9:1) led to messy reaction with reduced yield (entry 11). Thus
1.0 equiv. of acetic acid in water at 80 °C was the most suitable condition here.
With these optimised conditions in hand, we desired to ex- tend the substrate scope for the Brønsted acid promoted on- water ring opening reaction of spiro-epoxyoxindoles with indoles (Scheme 5). All the electron donating as well as electron withdrawing groups of spiro-epoxyoxindoles were well-toler- ated under optimised condition. All underwent smooth reac- tion rendering the desired 3, 3-bisindoles with moderate yield. It was also observed that all the spiro-epoxides irrespective of substitution or protection required longer times for completion than the previously described methods. Due to the nucleophilic character of water, we obtained a minor amount (15–20 %) of water adduct product in all the cases. Along with a minute amount of acetate adduct (<10 %) was also detected during the course of the reaction. These unwanted side reactions de- creased the overall yield of the desired 3,3 bisindole methanols 3.
We also unveiled the F-C reaction of spiro-epoxyoxindole with 3-substituted indole which might provide the bisindole with vicinal all-carbon quaternary centers. Consequently, com- pound 1c was treated with 3-methyl indole 4a under Brønsted acid catalysed condition in organic solvent (DCE) (Scheme 6). Interestingly, it afforded tetrahydrospirofuro-bisindole 5a hav- ing vicinal all- carbon quaternary centers as a diastereomeric mixture along with 2,3-bisindole 5a. It seemed epoxide open- ing with 3-methyl indole followed by intramolecular cyclization of the intermediate imine X afforded the tricyclic tetrahydro- furoindole core. Next we also examined the similar kind of reac- tion between N-benzyl spiro-epoxide 1b and 3-methyl N- benzoyl indole 4b under the same Method A. Consequently, both the reactions manifested identical outcome.
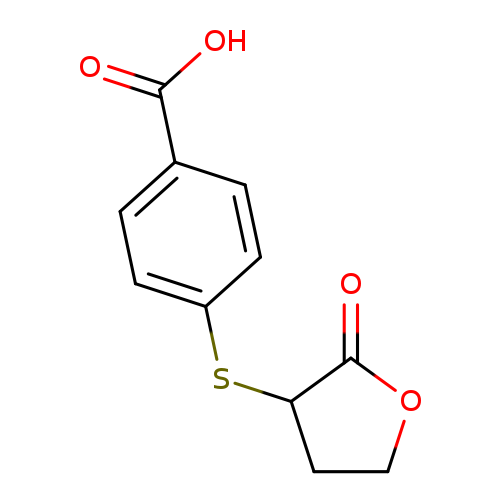
4-[(2-oxooxolan-3-yl)sulfanyl]benzoic acidCatalog No.:AA01A18J CAS No.:1098379-73-7 MDL No.:MFCD11647932 MF:C11H10O4S MW:238.2597 |
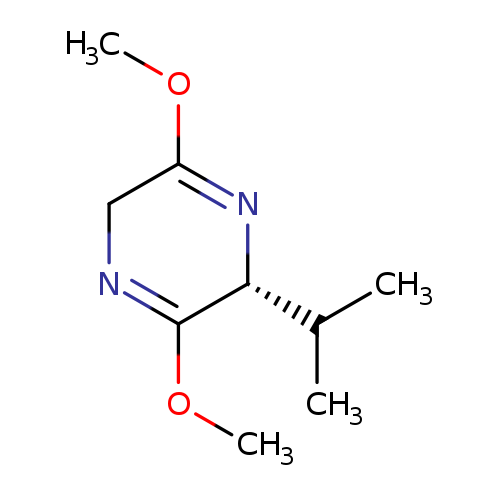
(R)-2,5-Dihydro-3,6-dimethoxy-2-isopropylpyrazineCatalog No.:AA0032BZ CAS No.:109838-85-9 MDL No.:MFCD00040565 MF:C9H16N2O2 MW:184.2355 |
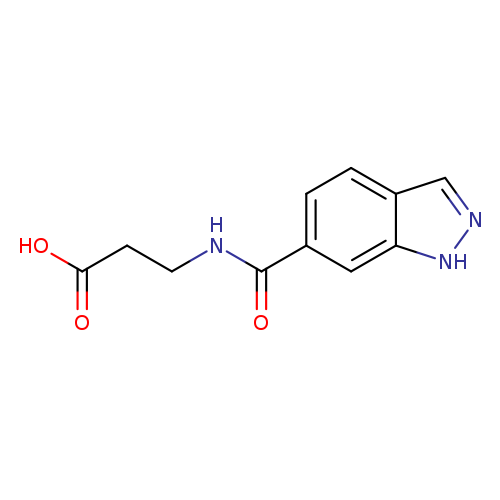
3-(1H-indazol-6-ylformamido)propanoic acidCatalog No.:AA01A7TF CAS No.:1098380-06-3 MDL No.:MFCD11648448 MF:C11H11N3O3 MW:233.2233 |
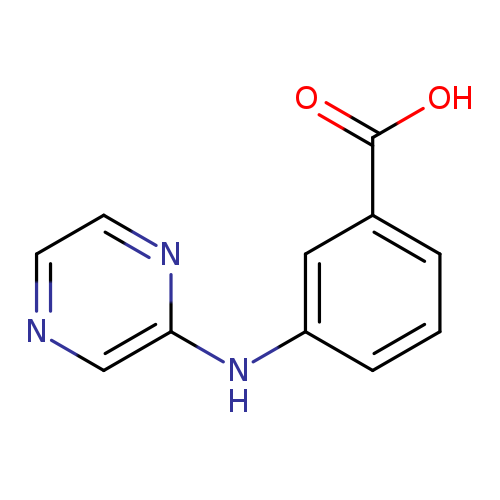
3-[(pyrazin-2-yl)amino]benzoic acidCatalog No.:AA01AJV3 CAS No.:1098383-88-0 MDL No.:MFCD11649237 MF:C11H9N3O2 MW:215.2081 |
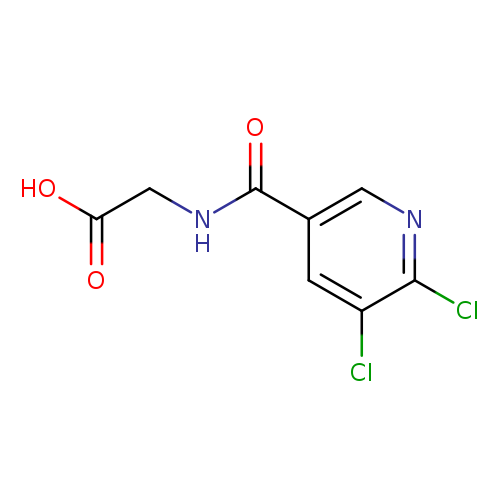
2-[(5,6-dichloropyridin-3-yl)formamido]acetic acidCatalog No.:AA01B6UB CAS No.:1098384-79-2 MDL No.:MFCD11646713 MF:C8H6Cl2N2O3 MW:249.0508 |
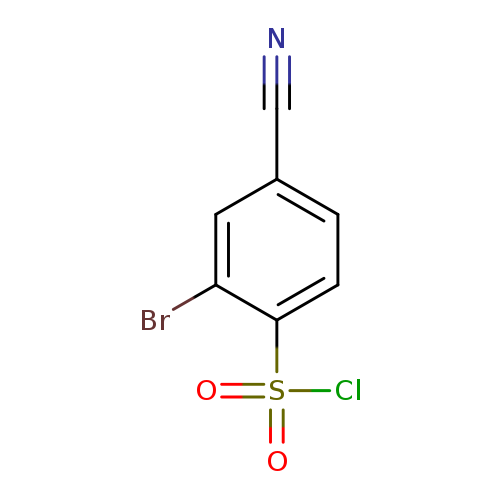
2-bromo-4-cyanobenzene-1-sulfonyl chlorideCatalog No.:AA019LC5 CAS No.:1098387-29-1 MDL No.:MFCD13634254 MF:C7H3BrClNO2S MW:280.5262 |
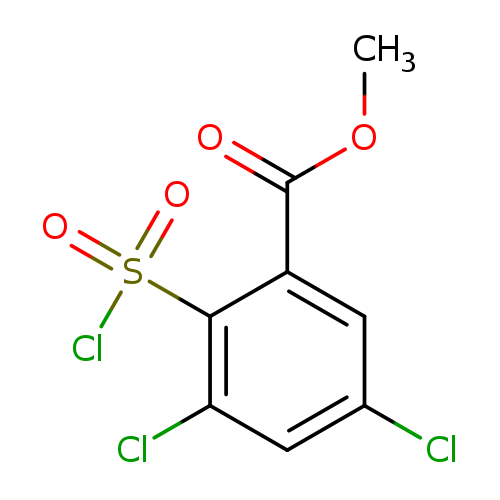
methyl 3,5-dichloro-2-(chlorosulfonyl)benzoateCatalog No.:AA01B9VO CAS No.:1098387-68-8 MDL No.:MFCD11650112 MF:C8H5Cl3O4S MW:303.5469 |
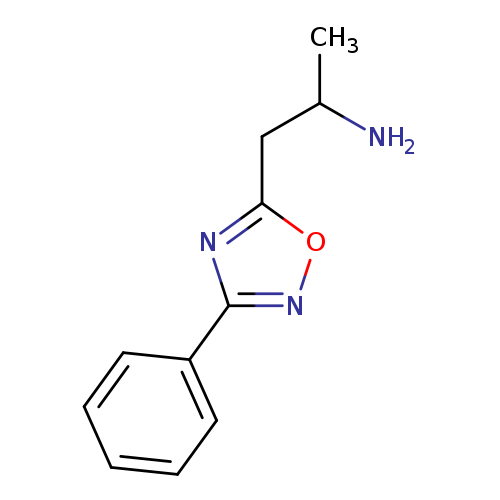
1-(3-phenyl-1,2,4-oxadiazol-5-yl)propan-2-amineCatalog No.:AA01A34A CAS No.:1098387-73-5 MDL No.:MFCD11649110 MF:C11H13N3O MW:203.2404 |
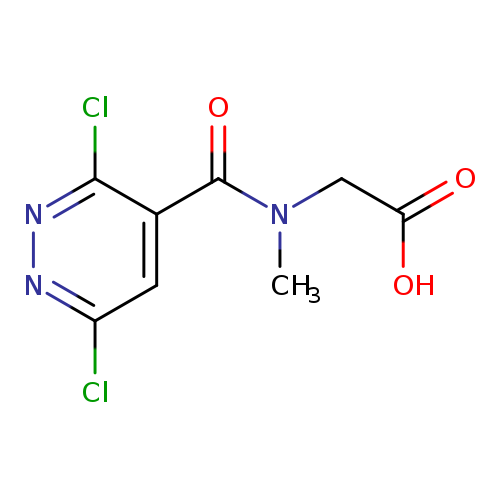
2-[1-(3,6-Dichloropyridazin-4-yl)-n-methylformamido]acetic acidCatalog No.:AA019PPV CAS No.:1098389-56-0 MDL No.:MFCD11648827 MF:C8H7Cl2N3O3 MW:264.0655 |
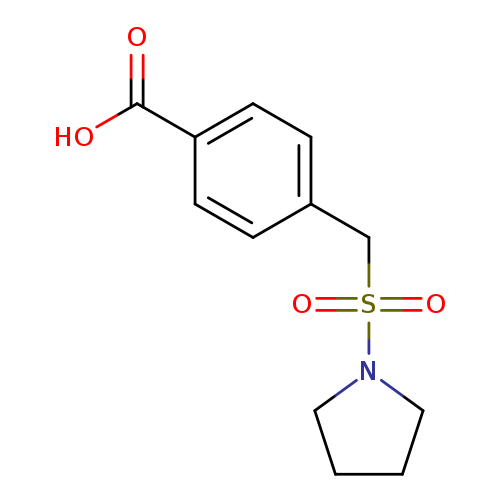
4-[(Pyrrolidin-1-ylsulfonyl)methyl]benzoic acidCatalog No.:AA00HBJS CAS No.:1098391-16-2 MDL No.:MFCD11549006 MF:C12H15NO4S MW:269.3168 |
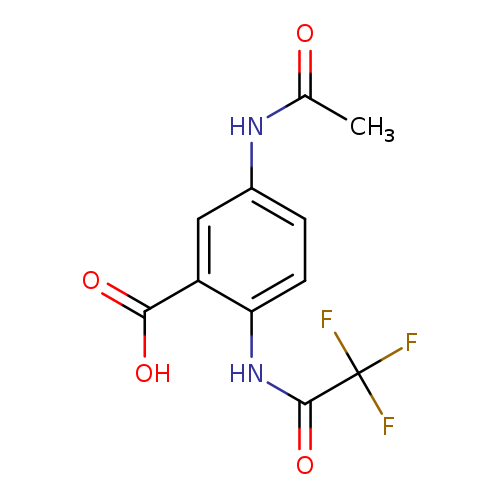
5-acetamido-2-(trifluoroacetamido)benzoic acidCatalog No.:AA01BTXV CAS No.:1098397-49-9 MDL No.:MFCD30723944 MF:C11H9F3N2O4 MW:290.1954 |
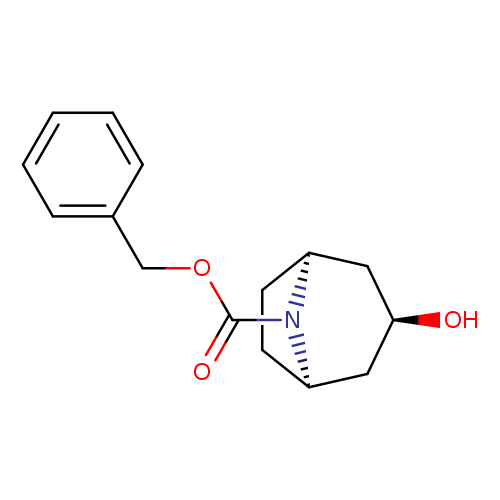
endo-Benzyl 3-hydroxy-8-azabicyclo[3.2.1]octane-8-carboxylateCatalog No.:AA003STG CAS No.:109840-91-7 MDL No.:MFCD09991642 MF:C15H19NO3 MW:261.3163 |
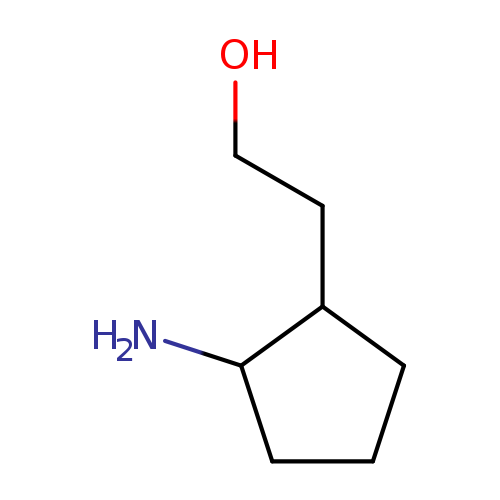
2-(2-aminocyclopentyl)ethan-1-olCatalog No.:AA00K9O5 CAS No.:109842-93-5 MDL No.:MFCD24448482 MF:C7H15NO MW:129.2001 |
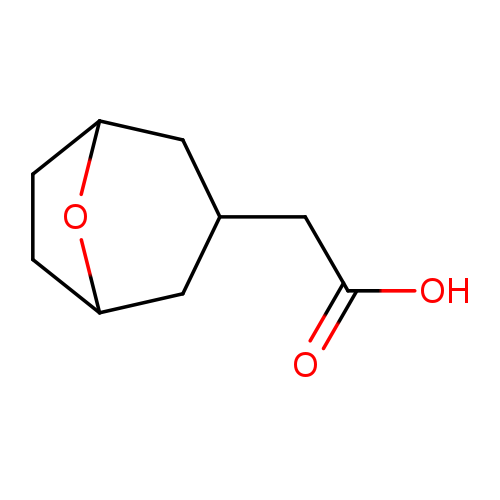
2-(8-Oxabicyclo[3.2.1]octan-3-yl)acetic acidCatalog No.:AA00HBJT CAS No.:1098432-95-1 MDL No.:MFCD27988076 MF:C9H14O3 MW:170.2057 |
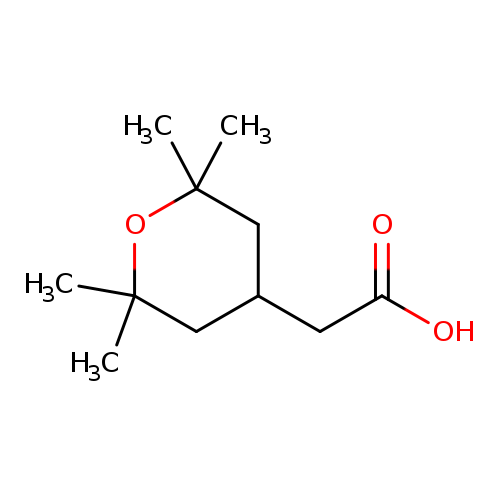
2-(2,2,6,6-tetramethyloxan-4-yl)acetic acidCatalog No.:AA0091F3 CAS No.:1098432-96-2 MDL No.:MFCD31691286 MF:C11H20O3 MW:200.2747 |
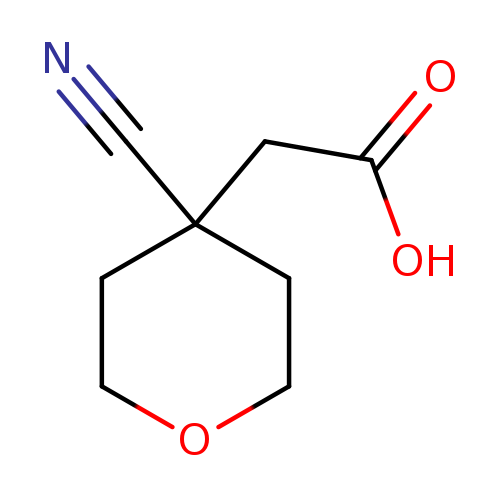
2-(4-cyanooxan-4-yl)acetic acidCatalog No.:AA01B4TR CAS No.:1098432-97-3 MDL No.:MFCD30342649 MF:C8H11NO3 MW:169.1778 |
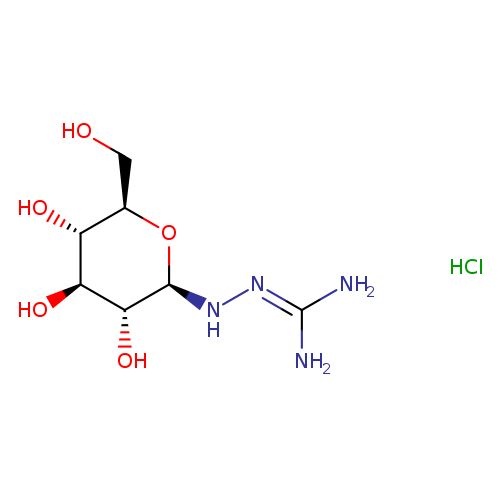
N1-Beta-d-glucopyranosylamino-guanidine hclCatalog No.:AA007WAV CAS No.:109853-81-8 MDL No.:MFCD08703738 MF:C7H17ClN4O5 MW:272.6867 |
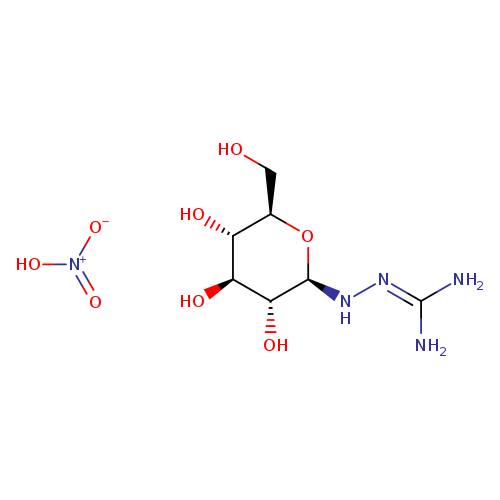
N1-b-D-Glucopyranosylamino-guanidine HNO3Catalog No.:AA007WAU CAS No.:109853-83-0 MDL No.:MFCD08703738 MF:C7H17N5O8 MW:299.2386 |
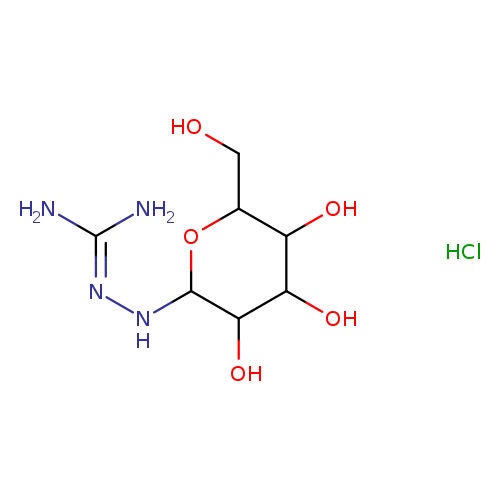
N1-Beta-d-galactopyranosyl amino-guanidine hclCatalog No.:AA008XE2 CAS No.:109853-84-1 MDL No.:MFCD08704124 MF:C7H17ClN4O5 MW:272.6867 |
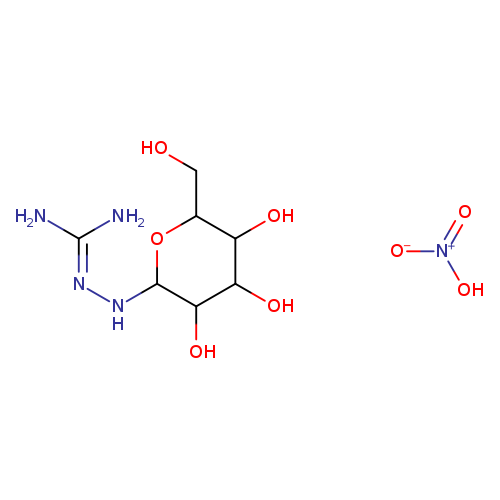
N1-BETA-D-GALACTOPYRANOSYL AMINO-GUANIDINE HNO3Catalog No.:AA008XEP CAS No.:109853-86-3 MDL No.:MFCD08704123 MF:C7H17N5O8 MW:299.2386 |
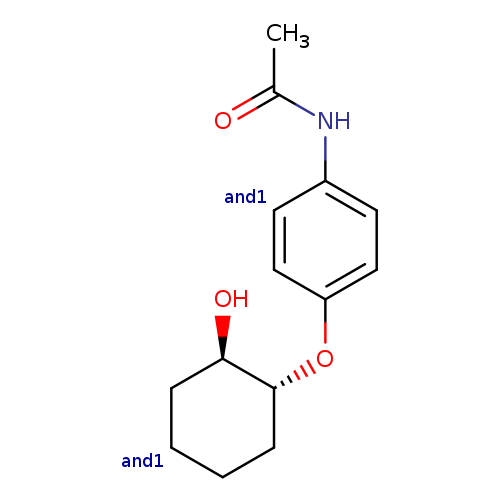
rac-N-(4-{[(1R,2R)-2-hydroxycyclohexyl]oxy}phenyl)acetamide, transCatalog No.:AA01EMH4 CAS No.:109859-10-1 MDL No.:MFCD31617821 MF:C14H19NO3 MW:249.3056 |
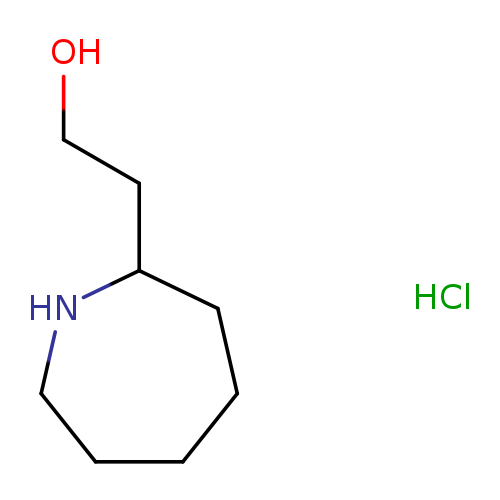
2-(Azepan-2-yl)ethan-1-ol hydrochlorideCatalog No.:AA01B9A0 CAS No.:109859-93-0 MDL No.:MFCD28383817 MF:C8H18ClNO MW:179.6876 |
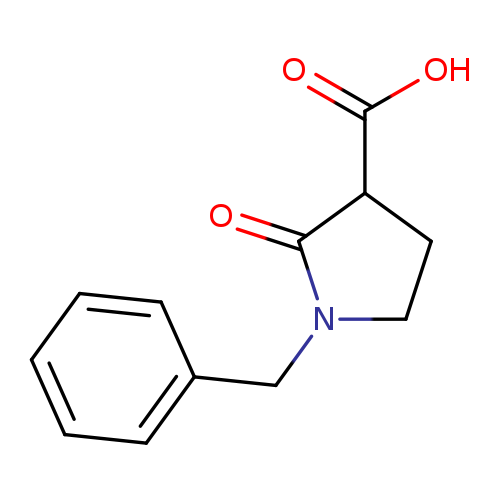
1-Benzyl-2-oxopyrrolidine-3-carboxylic acidCatalog No.:AA00HBJW CAS No.:109859-99-6 MDL No.:MFCD10003729 MF:C12H13NO3 MW:219.2365 |
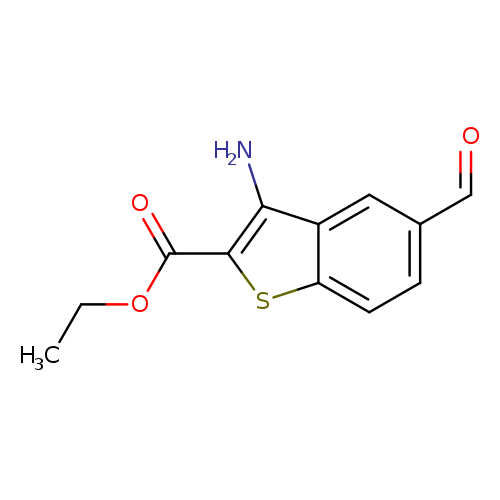
Ethyl 3-amino-5-formylbenzo[b]thiophene-2-carboxylateCatalog No.:AA00IZ90 CAS No.:1098608-18-4 MDL No.:MFCD18803525 MF:C12H11NO3S MW:249.2856 |
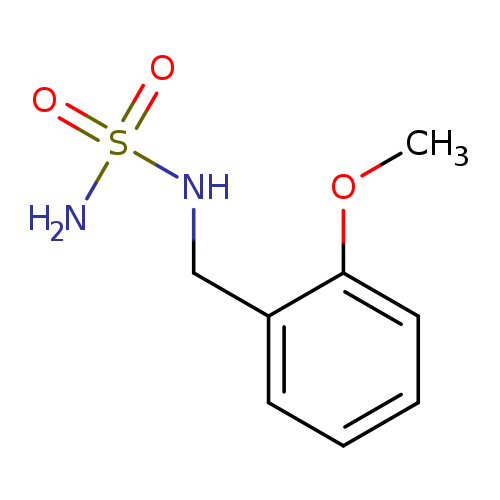
N-[(2-methoxyphenyl)methyl]aminosulfonamideCatalog No.:AA01B63X CAS No.:1098609-11-0 MDL No.:MFCD25978519 MF:C8H12N2O3S MW:216.2575 |
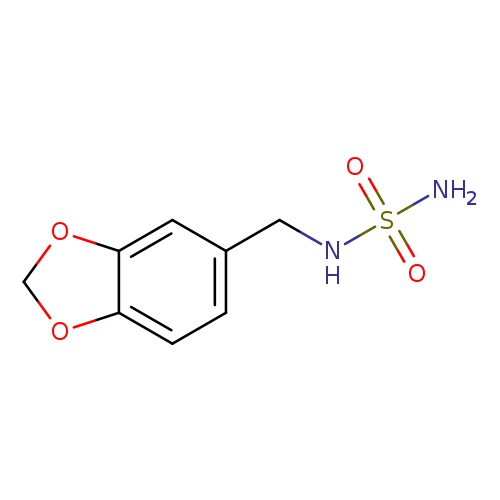
N-[(2H-1,3-benzodioxol-5-yl)methyl]aminosulfonamideCatalog No.:AA01B2QU CAS No.:1098609-12-1 MDL No.:MFCD26144724 MF:C8H10N2O4S MW:230.2410 |
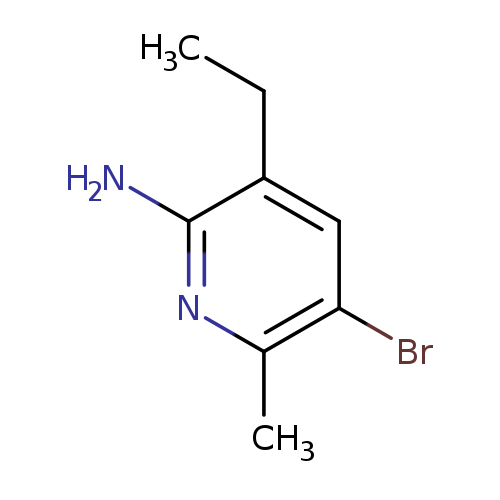
5-bromo-3-ethyl-6-methylpyridin-2-amineCatalog No.:AA01B2HT CAS No.:1098652-28-8 MDL No.:MFCD11043703 MF:C8H11BrN2 MW:215.0903 |
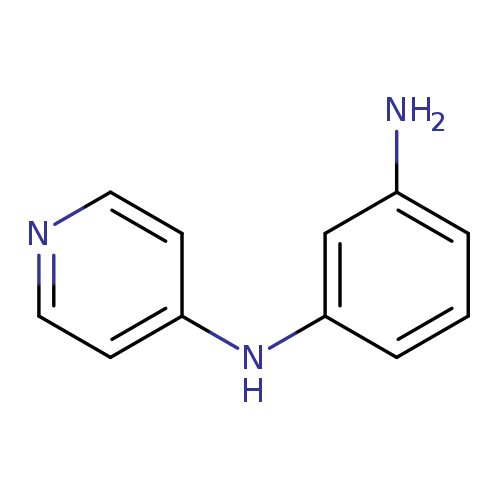
1-N-(pyridin-4-yl)benzene-1,3-diamineCatalog No.:AA019Y5B CAS No.:109871-19-4 MDL No.:MFCD09935428 MF:C11H11N3 MW:185.2251 |
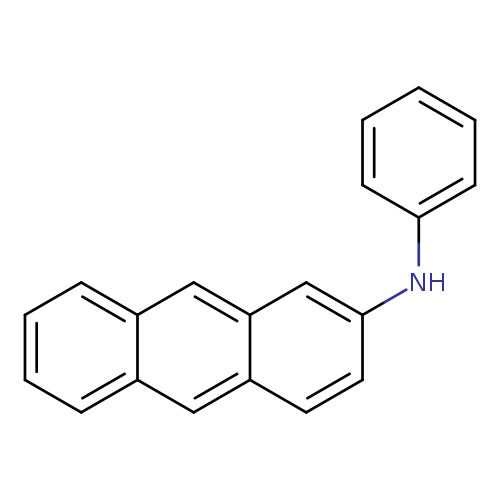
N-Phenyl-2-anthramineCatalog No.:AA003GHJ CAS No.:109871-20-7 MDL No.:MFCD08276408 MF:C20H15N MW:269.3398 |
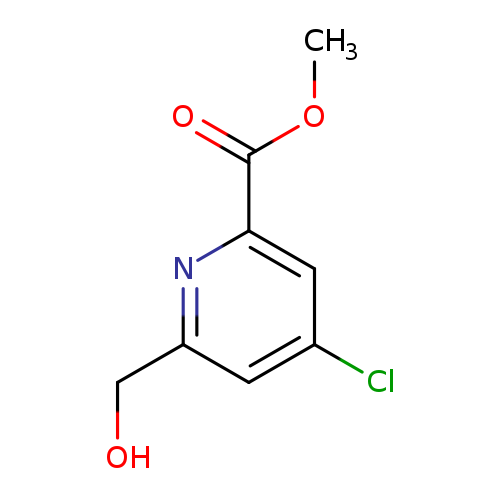
Methyl 4-chloro-6-(hydroxymethyl)picolinateCatalog No.:AA0084LQ CAS No.:109880-43-5 MDL No.:MFCD09263974 MF:C8H8ClNO3 MW:201.6070 |
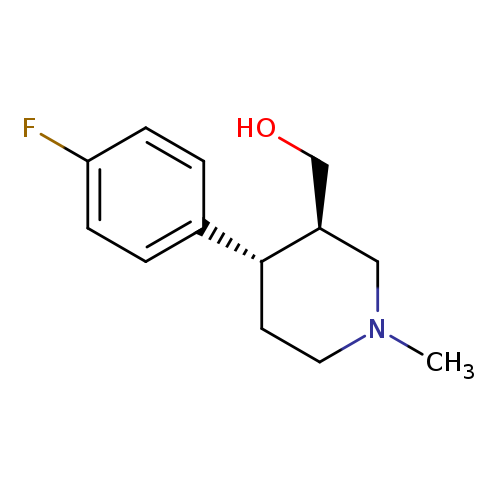
(+/-)-Trans-4-(fluorophenyl)-3-hydroxymethyl-1-methylpiperidineCatalog No.:AA007EFM CAS No.:109887-53-8 MDL No.:MFCD02093092 MF:C13H18FNO MW:223.2865 |
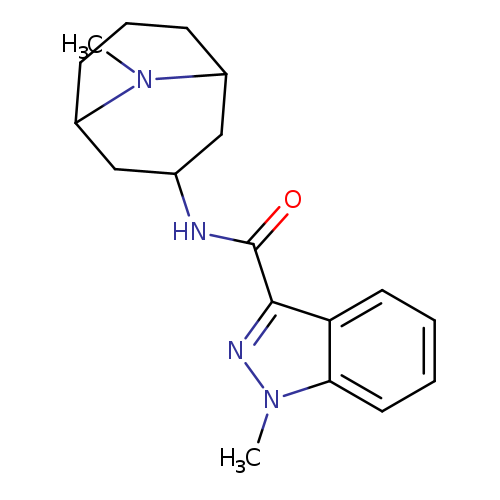
GranisetronCatalog No.:AA007EFL CAS No.:109889-09-0 MDL No.:MFCD00865550 MF:C18H24N4O MW:312.4094 |
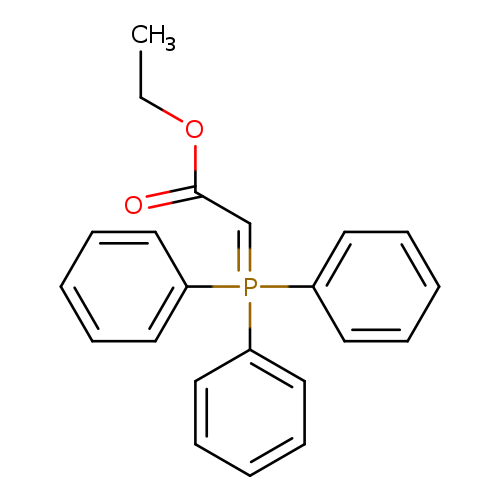
Ethyl (triphenylphosphoranylidene) acetateCatalog No.:AA0034R2 CAS No.:1099-45-2 MDL No.:MFCD00009183 MF:C22H21O2P MW:348.3747 |
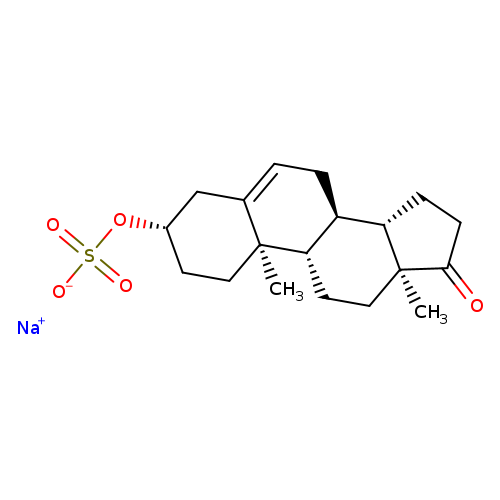
Androst-5-en-17-one, 3-(sulfooxy)-, sodium salt, (3b)-Catalog No.:AA00HBK5 CAS No.:1099-87-2 MDL No.:MFCD00010470 MF:C19H27NaO5S MW:390.4694 |
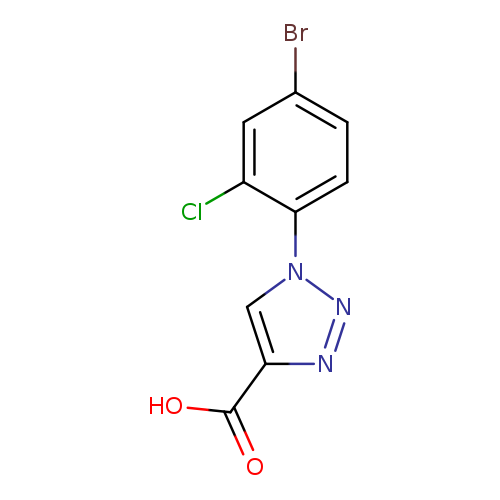
1-(4-bromo-2-chlorophenyl)-1H-1,2,3-triazole-4-carboxylic acidCatalog No.:AA01AIHZ CAS No.:1099026-08-0 MDL No.:MFCD11644680 MF:C9H5BrClN3O2 MW:302.5119 |
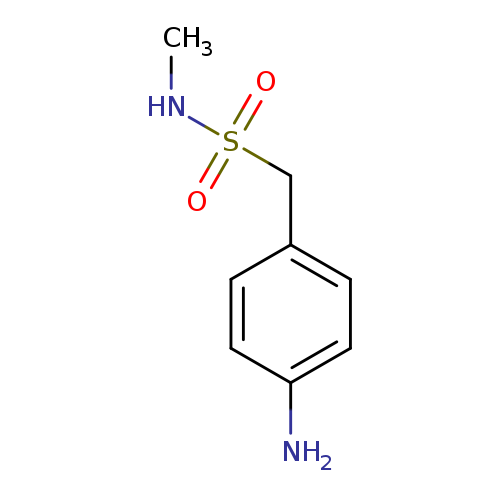
4-Amino-n-methylbenzenemethanesulfonamide hydrochlorideCatalog No.:AA0033TY CAS No.:109903-35-7 MDL No.:MFCD00671697 MF:C8H12N2O2S MW:200.2581 |
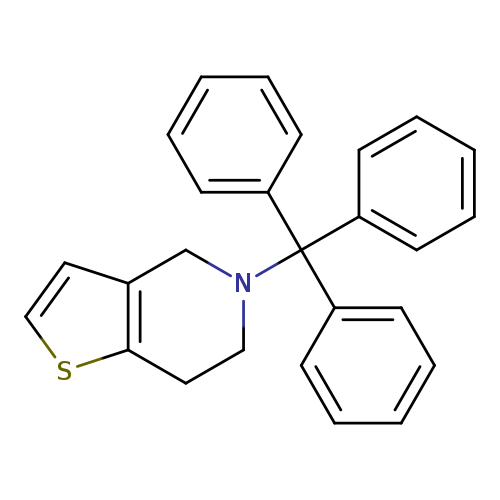
5-Trityl-4,5,6,7-tetrahydrothieno[3,2-c]pyridineCatalog No.:AA007W4A CAS No.:109904-25-8 MDL No.:MFCD11655877 MF:C26H23NS MW:381.5325 |
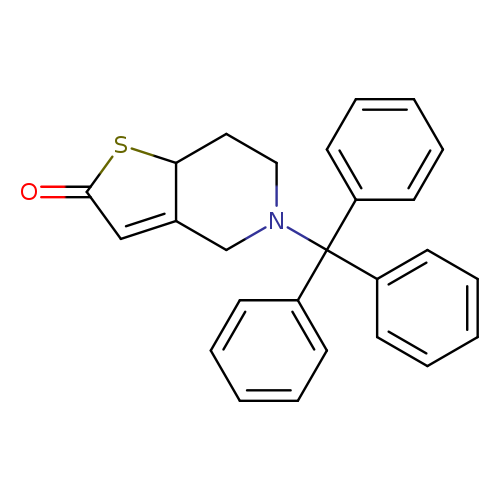
5-Trityl-5,6,7,7a-tetrahydrothieno[3,2-c]pyridin-2(4h)-oneCatalog No.:AA003M6A CAS No.:109904-26-9 MDL No.:MFCD12024648 MF:C26H23NOS MW:397.5319 |
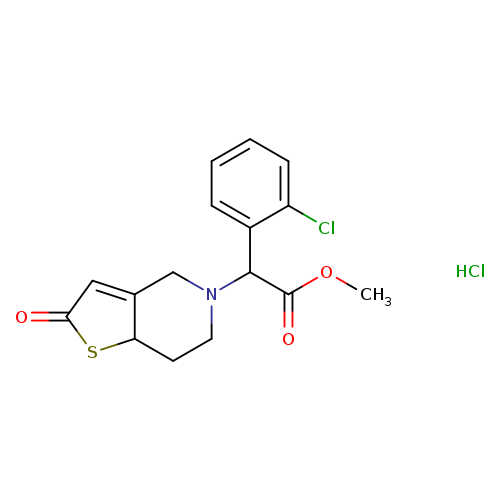
2-Oxo Clopidogrel Hydrochloride(Mixture of Diastereomers)Catalog No.:AA007W49 CAS No.:109904-27-0 MDL No.:MFCD16036308 MF:C16H17Cl2NO3S MW:374.2821 |
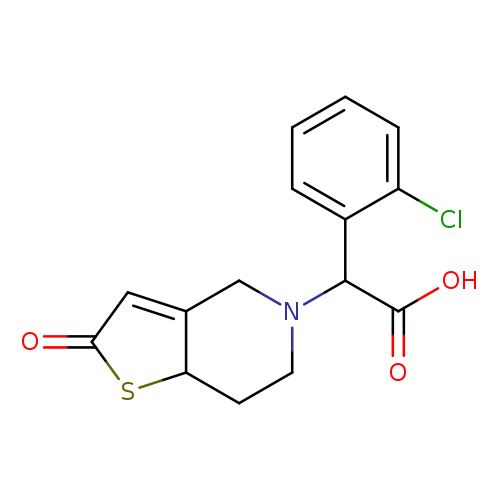
α-(2-Chlorophenyl)-2,6,7,7a-tetrahydro-2-oxothieno[3,2-c]pyridine-5(4H)-acetic AcidCatalog No.:AA008W57 CAS No.:109904-36-1 MDL No.: MF:C15H14ClNO3S MW:323.7946 |
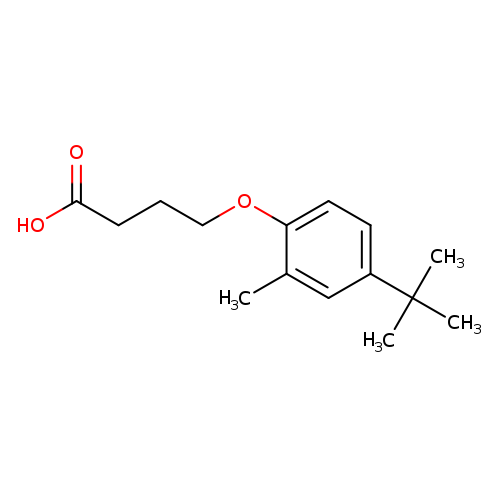
4-(4-tert-butyl-2-methylphenoxy)butanoic acidCatalog No.:AA01A0U5 CAS No.:1099046-07-7 MDL No.:MFCD11549133 MF:C15H22O3 MW:250.3334 |
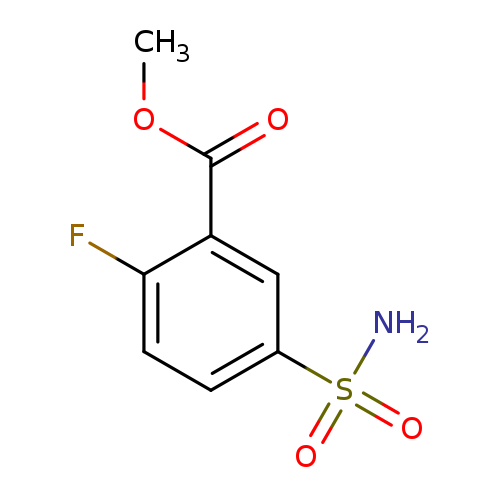
methyl 2-fluoro-5-sulfamoylbenzoateCatalog No.:AA019KT2 CAS No.:1099052-48-8 MDL No.:MFCD11649977 MF:C8H8FNO4S MW:233.2168 |
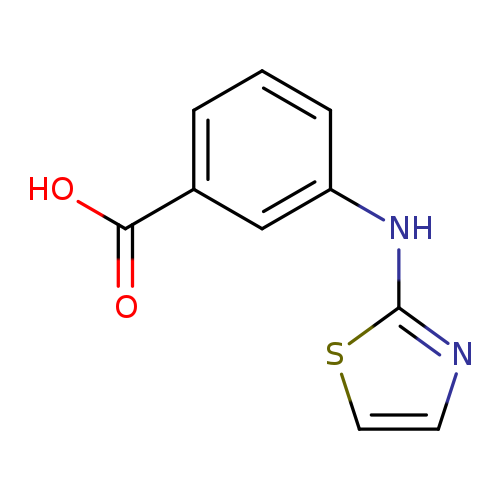
3-[(1,3-thiazol-2-yl)amino]benzoic acidCatalog No.:AA019URH CAS No.:1099057-19-8 MDL No.:MFCD11649236 MF:C10H8N2O2S MW:220.2477 |
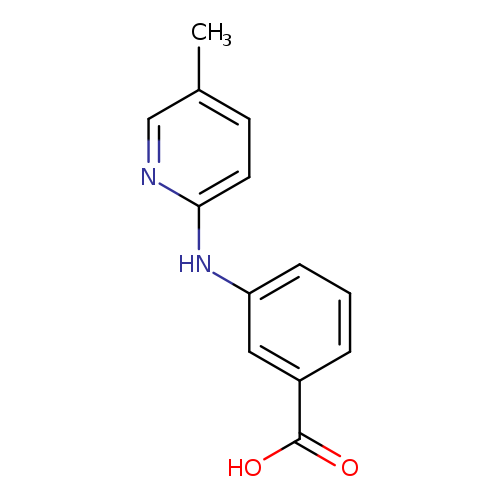
3-((5-methylpyridin-2-yl)amino)benzoic acidCatalog No.:AA01BRI2 CAS No.:1099057-20-1 MDL No.:MFCD14699343 MF:C13H12N2O2 MW:228.2466 |
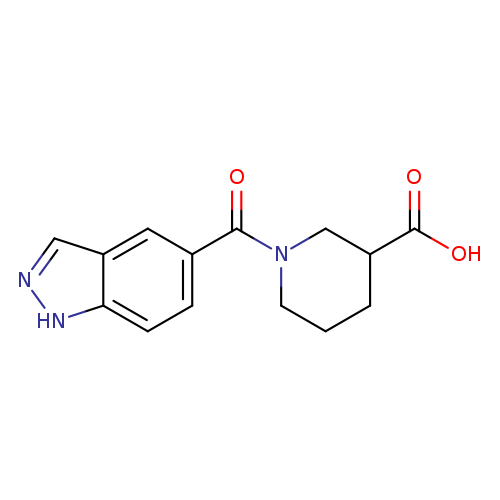
1-(1H-indazole-5-carbonyl)piperidine-3-carboxylic acidCatalog No.:AA01A984 CAS No.:1099058-83-9 MDL No.:MFCD13668527 MF:C14H15N3O3 MW:273.2872 |
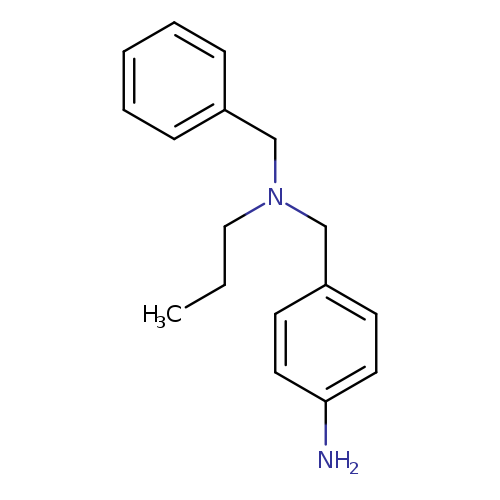
4-{[benzyl(propyl)amino]methyl}anilineCatalog No.:AA01B244 CAS No.:1099073-00-3 MDL No.:MFCD12579141 MF:C17H22N2 MW:254.3700 |
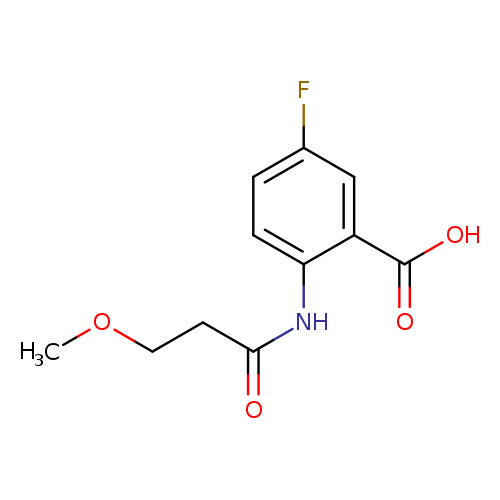
5-fluoro-2-(3-methoxypropanamido)benzoic acidCatalog No.:AA019W2A CAS No.:1099091-63-0 MDL No.:MFCD11649320 MF:C11H12FNO4 MW:241.2157 |
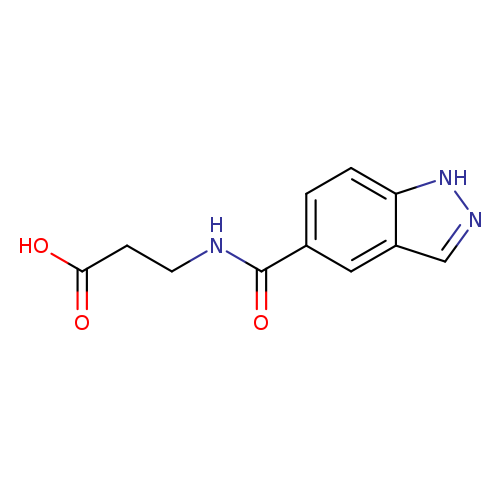
3-(1H-indazol-5-ylformamido)propanoic acidCatalog No.:AA01A8MU CAS No.:1099107-57-9 MDL No.:MFCD11648430 MF:C11H11N3O3 MW:233.2233 |
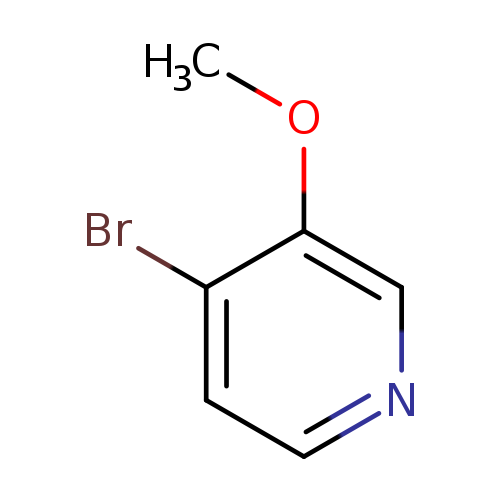
4-Bromo-3-methoxypyridineCatalog No.:AA0084LH CAS No.:109911-38-8 MDL No.:MFCD08234926 MF:C6H6BrNO MW:188.0219 |
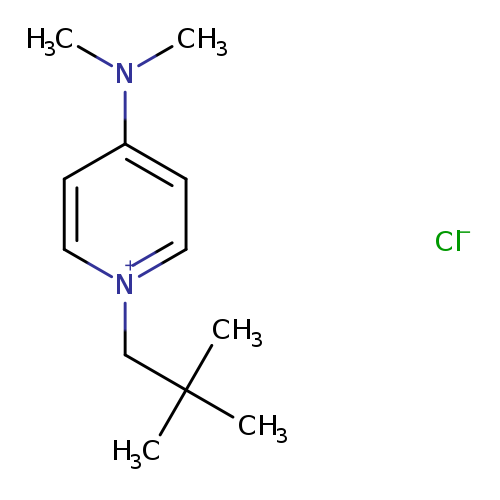
4-(Dimethylamino)-1-neopentylpyridin-1-ium chlorideCatalog No.:AA003L7M CAS No.:109911-77-5 MDL No.:MFCD00060050 MF:C12H21ClN2 MW:228.7615 |
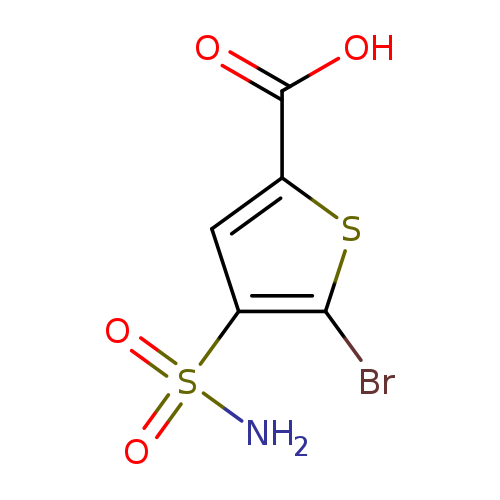
5-bromo-4-sulfamoylthiophene-2-carboxylic acidCatalog No.:AA01A7HW CAS No.:1099110-57-2 MDL No.:MFCD11647661 MF:C5H4BrNO4S2 MW:286.1236 |
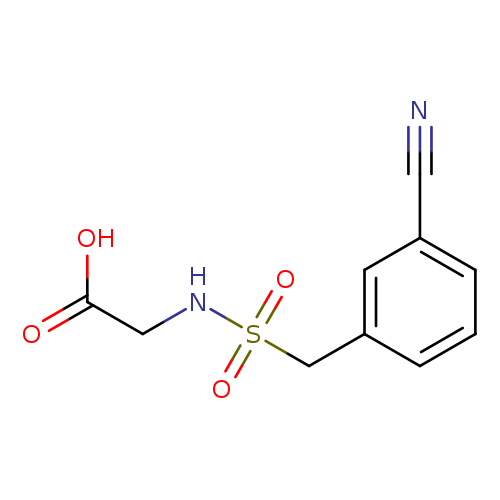
2-[(3-cyanophenyl)methanesulfonamido]acetic acidCatalog No.:AA01AJX9 CAS No.:1099113-52-6 MDL No.:MFCD03423096 MF:C10H10N2O4S MW:254.2624 |
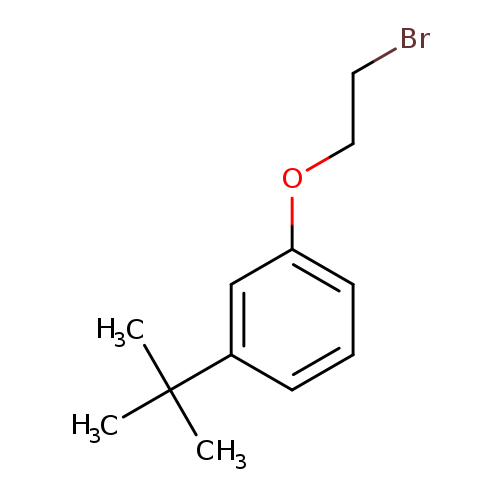
1-(2-bromoethoxy)-3-tert-butylbenzeneCatalog No.:AA01AI0W CAS No.:1099115-94-2 MDL No.:MFCD11647764 MF:C12H17BrO MW:257.1668 |
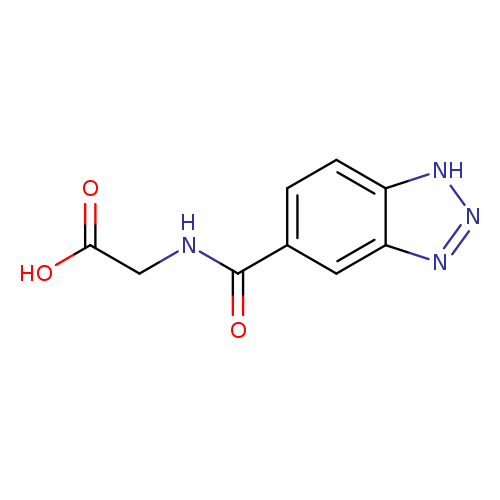
2-(1H-1,2,3-Benzotriazol-5-ylformamido)acetic acidCatalog No.:AA01EKBC CAS No.:1099119-95-5 MDL No.:MFCD11648323 MF:C9H8N4O3 MW:220.1848 |
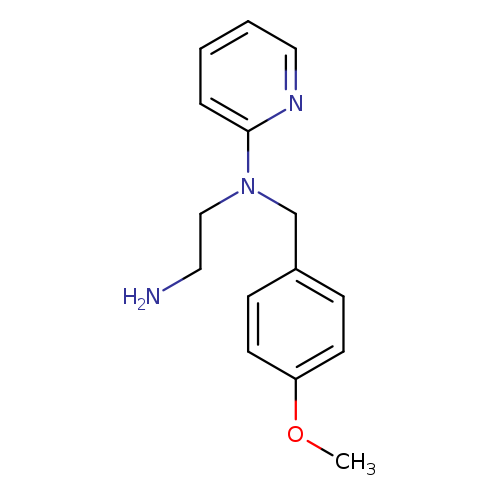
N1-(4-Methoxybenzyl)-N1-(pyridin-2-yl)ethane-1,2-diamineCatalog No.:AA007EF4 CAS No.:109912-28-9 MDL No.:MFCD04039588 MF:C15H19N3O MW:257.3309 |
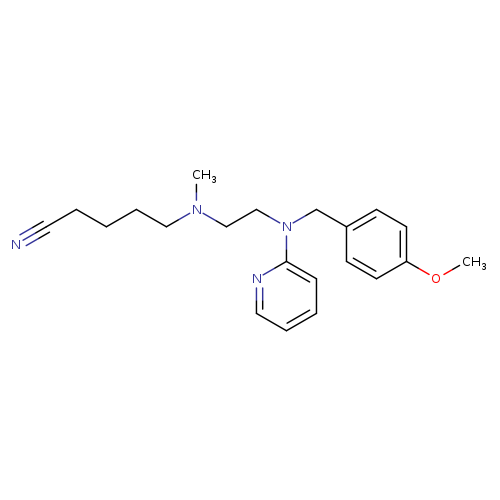
N'-(4-Cyanobutyl)-N-(4-methoxybenzyl)-N'-methyl-N-2-pyridinyl-1,2-ethanediamineCatalog No.:AA0084LE CAS No.:109912-34-7 MDL No.:MFCD04039487 MF:C21H28N4O MW:352.4732 |
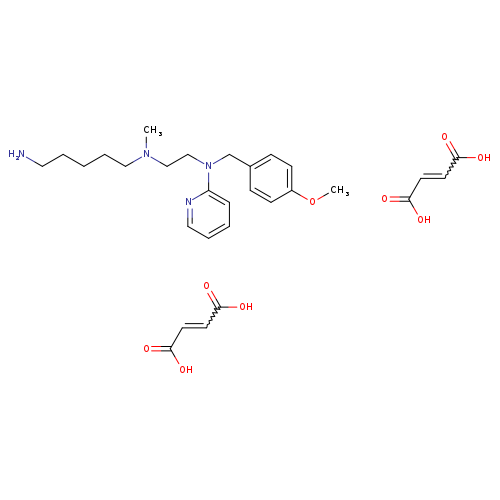
N'-(5-AMINOPENTYL)-N-(4-METHOXYBENZYL)-N'-METHYL-N-2-PYRIDINYL-1,2-ETHANEDIAMINE, DIMALEATE SALTCatalog No.:AA008VUV CAS No.:109912-42-7 MDL No.:MFCD04116105 MF:C29H40N4O9 MW:588.6493 |
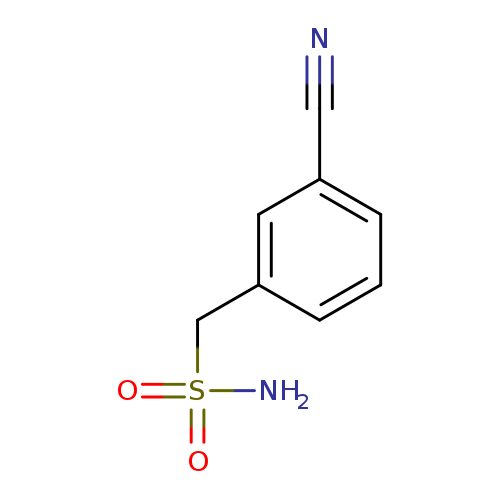
(3-Cyanophenyl)methanesulfonamideCatalog No.:AA00HBKB CAS No.:1099122-39-0 MDL No.:MFCD11647643 MF:C8H8N2O2S MW:196.2263 |
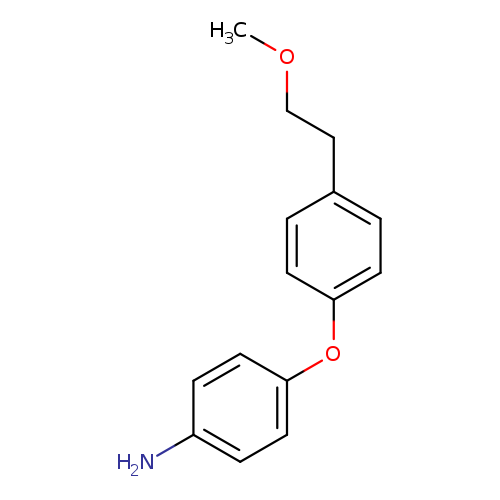
4-[4-(2-methoxyethyl)phenoxy]anilineCatalog No.:AA01AGPM CAS No.:1099130-23-0 MDL No.:MFCD11549248 MF:C15H17NO2 MW:243.3010 |
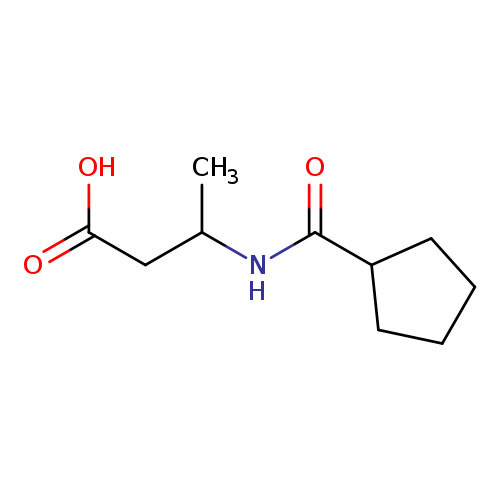
3-(cyclopentylformamido)butanoic acidCatalog No.:AA01A9Q3 CAS No.:1099132-22-5 MDL No.:MFCD11647081 MF:C10H17NO3 MW:199.2469 |
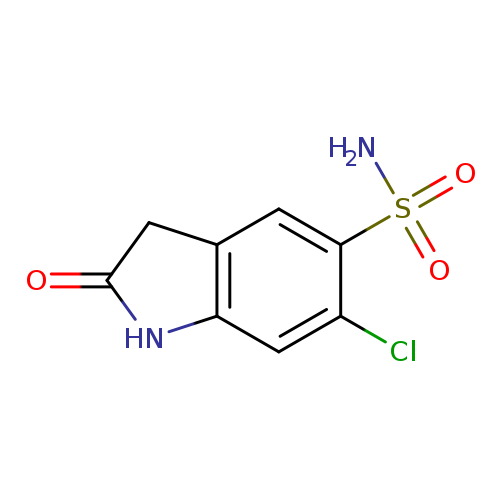
6-chloro-2-oxo-2,3-dihydro-1H-indole-5-sulfonamideCatalog No.:AA01B2R8 CAS No.:1099139-86-2 MDL No.:MFCD11647579 MF:C8H7ClN2O3S MW:246.6708 |
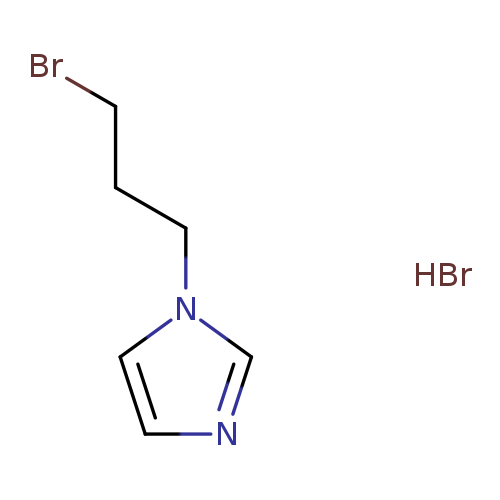
1-(3-bromopropyl)-1H-imidazole hydrobromideCatalog No.:AA00J25K CAS No.:109914-45-6 MDL No.:MFCD18917307 MF:C6H10Br2N2 MW:269.9650 |
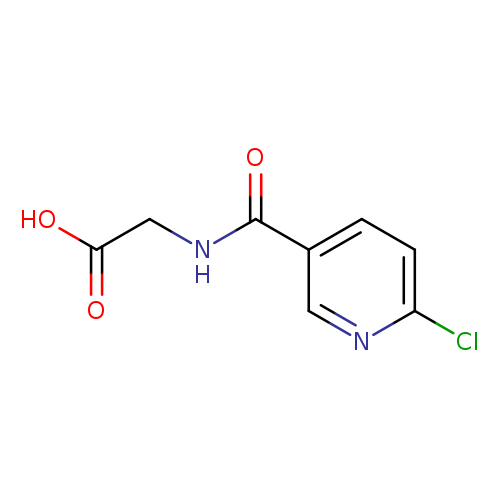
2-[(6-chloropyridin-3-yl)formamido]acetic acidCatalog No.:AA01FLZ7 CAS No.:1099143-66-4 MDL No.:MFCD11646710 MF:C8H7ClN2O3 MW:214.6058 |
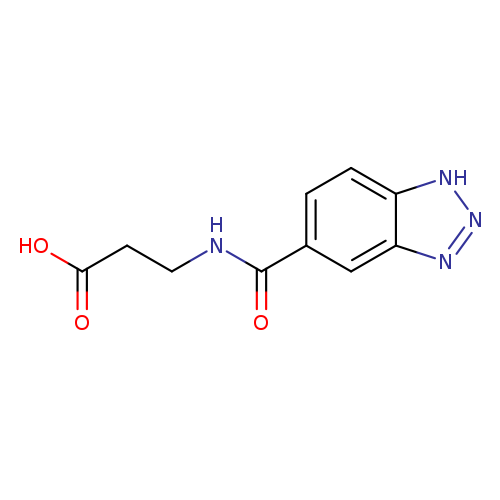
3-(1H-1,2,3-benzotriazol-5-ylformamido)propanoic acidCatalog No.:AA019ZGU CAS No.:1099152-06-3 MDL No.:MFCD11648479 MF:C10H10N4O3 MW:234.2114 |
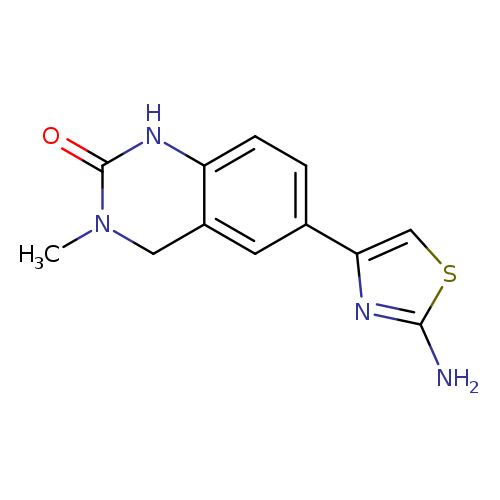
6-(2-amino-1,3-thiazol-4-yl)-3-methyl-1,2,3,4-tetrahydroquinazolin-2-oneCatalog No.:AA01BHM7 CAS No.:109916-22-5 MDL No.:MFCD25772257 MF:C12H12N4OS MW:260.3149 |
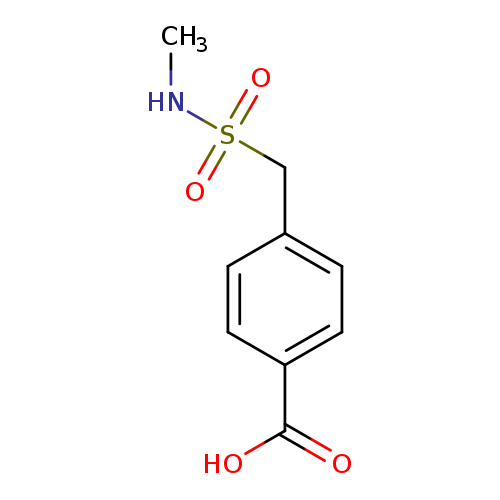
4-[(Methylsulfamoyl)methyl]benzoic acidCatalog No.:AA01EH2R CAS No.:1099184-20-9 MDL No.:MFCD11649779 MF:C9H11NO4S MW:229.2529 |
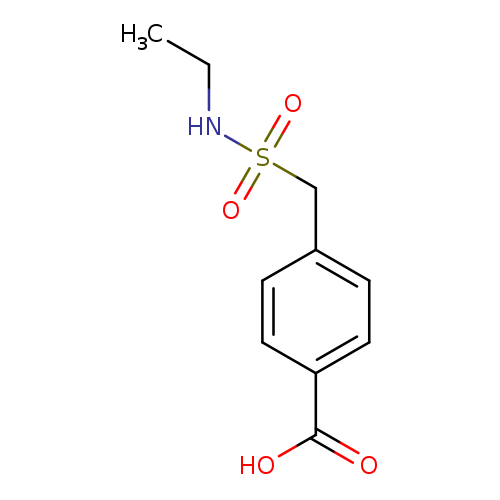
4-[(Ethylsulfamoyl)methyl]benzoic acidCatalog No.:AA01EH47 CAS No.:1099184-28-7 MDL No.:MFCD11649782 MF:C10H13NO4S MW:243.2795 |
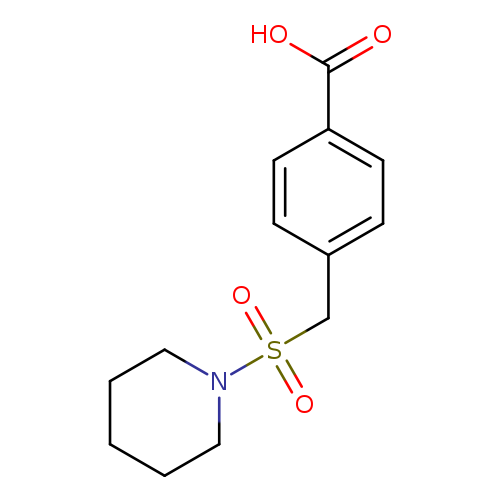
4-[(Piperidin-1-ylsulfonyl)methyl]benzoic acidCatalog No.:AA00HBKC CAS No.:1099184-76-5 MDL No.:MFCD12754214 MF:C13H17NO4S MW:283.3434 |
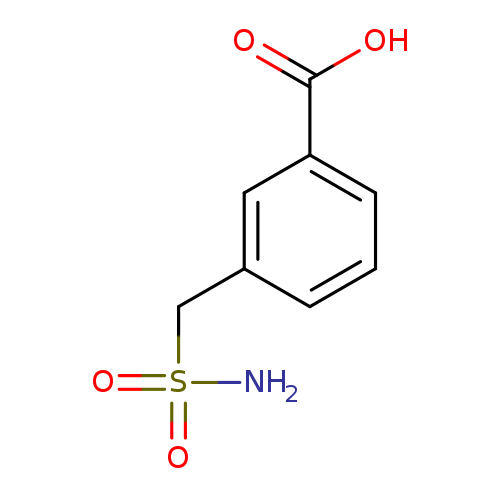
3-(Sulfamoylmethyl)benzoic acidCatalog No.:AA01A88O CAS No.:1099184-83-4 MDL No.:MFCD11649786 MF:C8H9NO4S MW:215.2264 |
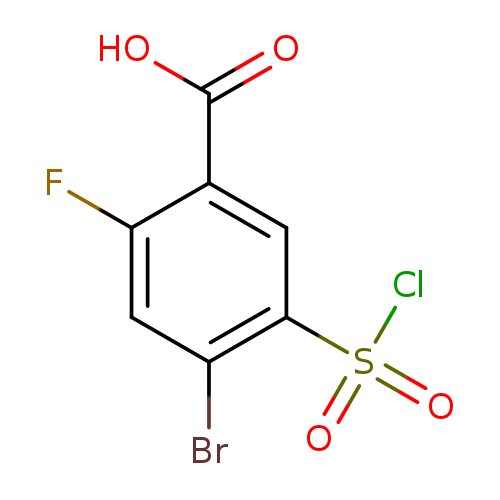
4-bromo-5-(chlorosulfonyl)-2-fluorobenzoic acidCatalog No.:AA01A179 CAS No.:1099186-78-3 MDL No.:MFCD11650163 MF:C7H3BrClFO4S MW:317.5167 |
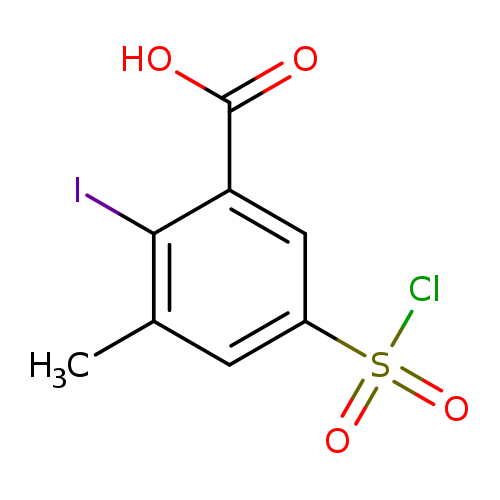
5-(chlorosulfonyl)-2-iodo-3-methylbenzoic acidCatalog No.:AA01B2S9 CAS No.:1099186-84-1 MDL No.:MFCD11650167 MF:C8H6ClIO4S MW:360.5533 |
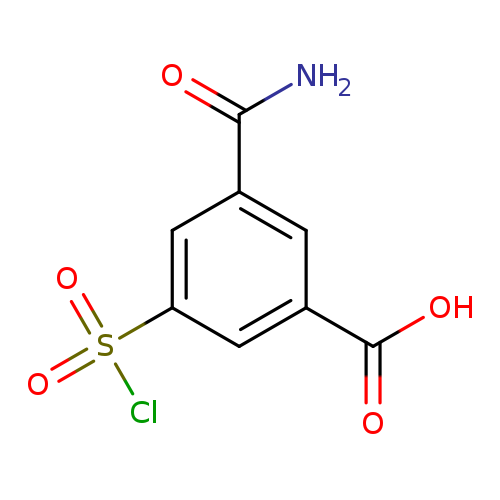
3-carbamoyl-5-(chlorosulfonyl)benzoic acidCatalog No.:AA01BCC1 CAS No.:1099186-87-4 MDL No.:MFCD11650170 MF:C8H6ClNO5S MW:263.6549 |
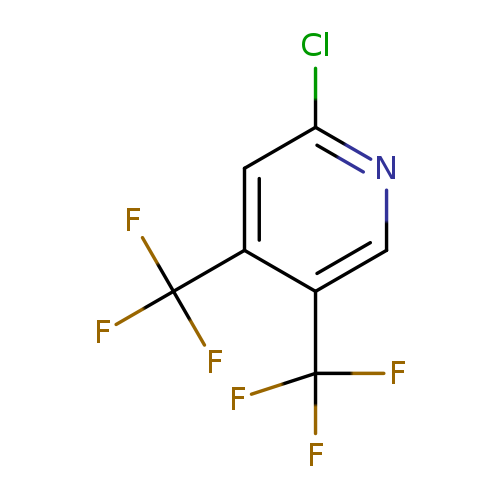
Pyridine,2-chloro-4,5-bis(trifluoromethyl)-Catalog No.:AA007W43 CAS No.:109919-25-7 MDL No.:MFCD00082556 MF:C7H2ClF6N MW:249.5409 |
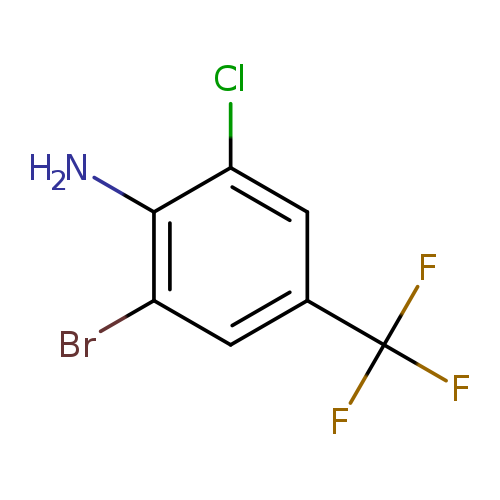
2-Bromo-6-chloro-4-(trifluoromethyl)anilineCatalog No.:AA003B3S CAS No.:109919-26-8 MDL No.:MFCD00042180 MF:C7H4BrClF3N MW:274.4656 |
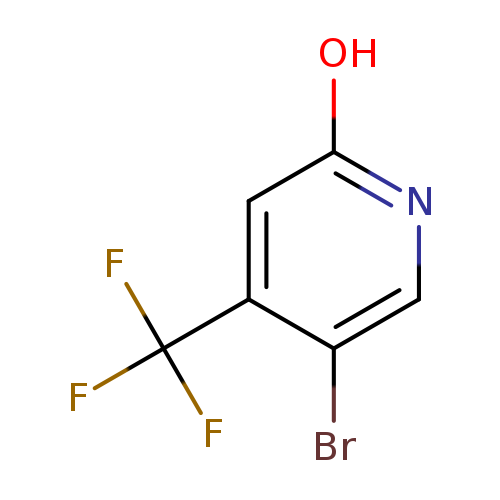
5-Bromo-2-hydroxy-4-trifluoromethylpyridineCatalog No.:AA007ECF CAS No.:109919-32-6 MDL No.:MFCD11227189 MF:C6H3BrF3NO MW:241.9933 |
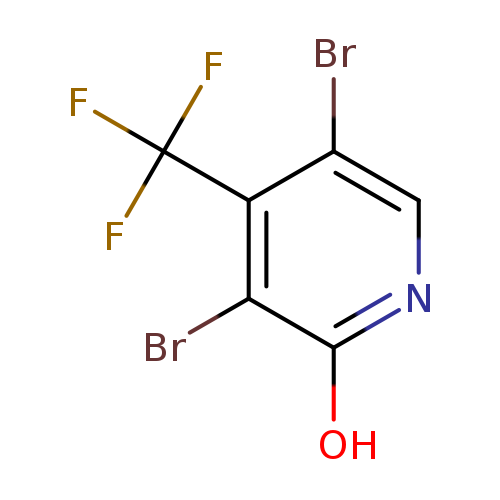
3,5-Dibromo-2-hydroxy-4-(trifluoromethyl)pyridineCatalog No.:AA003II7 CAS No.:109919-34-8 MDL No.:MFCD11227186 MF:C6H2Br2F3NO MW:320.8894 |
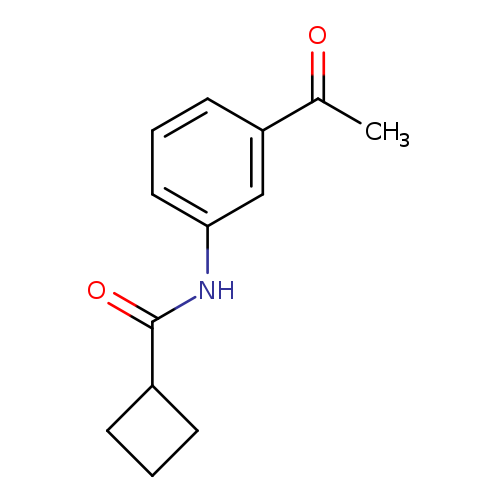
N-(3-Acetylphenyl)cyclobutanecarboxamideCatalog No.:AA0093IA CAS No.:109920-59-4 MDL No.:MFCD02176923 MF:C13H15NO2 MW:217.2637 |
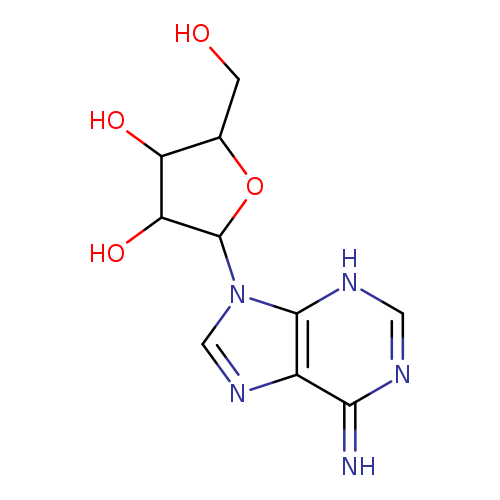
ADENOSINE-2-D1Catalog No.:AA008SW9 CAS No.:109923-50-4 MDL No.: MF:C10H13N5O4 MW:267.2413 |
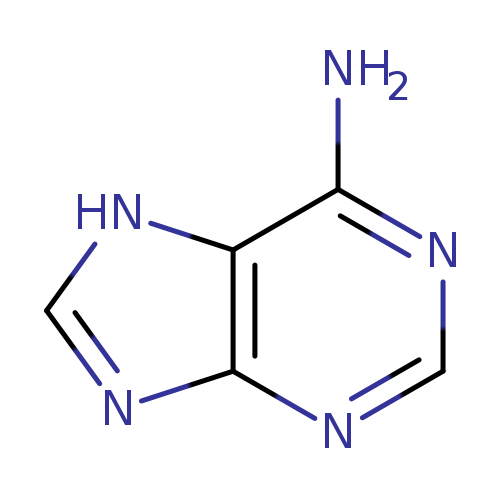
ADENINE-2-D1Catalog No.:AA008SWB CAS No.:109923-52-6 MDL No.:MFCD06658867 MF:C5H4DN5 MW:136.1329 |
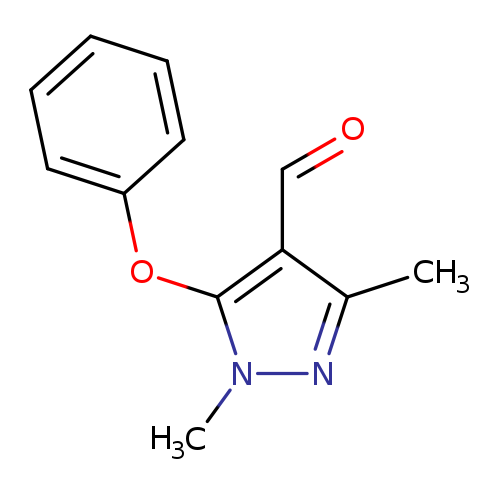
1,3-Dimethyl-5-phenoxy-1H-pyrazole-4-carbaldehydeCatalog No.:AA007ECD CAS No.:109925-10-2 MDL No.:MFCD07698583 MF:C12H12N2O2 MW:216.2359 |
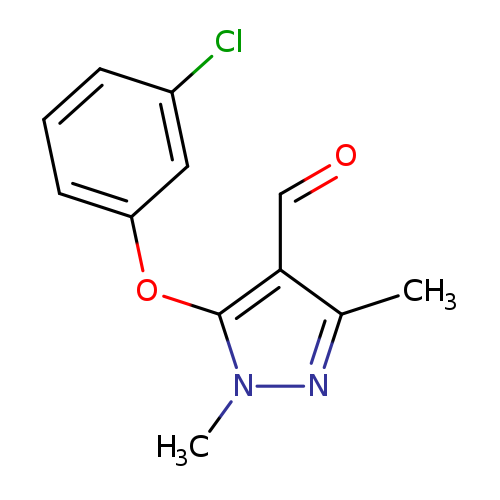
5-(3-chlorophenoxy)-1,3-dimethyl-1H-pyrazole-4-carbaldehydeCatalog No.:AA00IU9Y CAS No.:109925-12-4 MDL No.:MFCD00215393 MF:C12H11ClN2O2 MW:250.6809 |
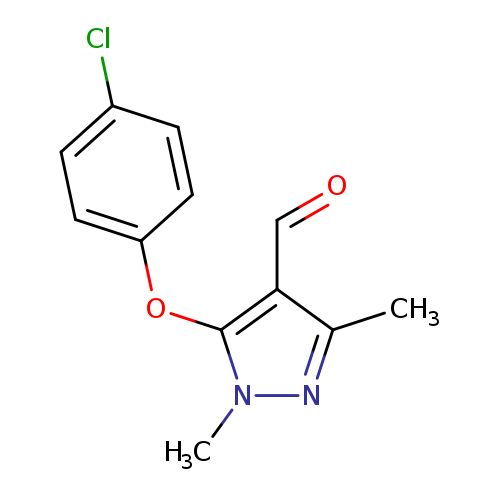
5-(4-Chlorophenoxy)-1,3-dimethyl-1h-pyrazole-4-carbaldehydeCatalog No.:AA008RYH CAS No.:109925-13-5 MDL No.:MFCD00215390 MF:C12H11ClN2O2 MW:250.6809 |
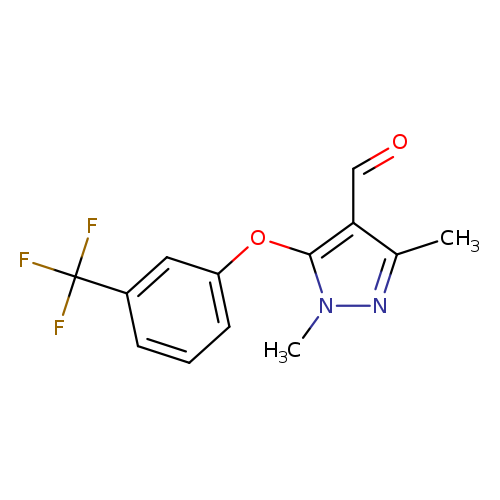
1,3-Dimethyl-5-[3-(trifluoromethyl)phenoxy]-1h-pyrazole-4-carbaldehydeCatalog No.:AA009OBH CAS No.:109925-28-2 MDL No.:MFCD00215392 MF:C13H11F3N2O2 MW:284.2338 |
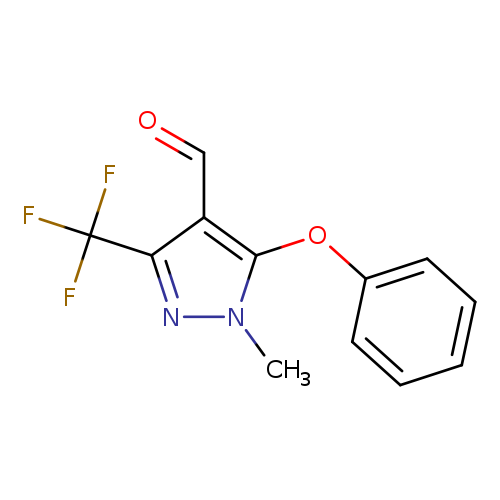
1-Methyl-5-phenoxy-3-(trifluoromethyl)-1H-pyrazole-4-carbaldehydeCatalog No.:AA007ECB CAS No.:109925-42-0 MDL No.:MFCD01936001 MF:C12H9F3N2O2 MW:270.2073 |
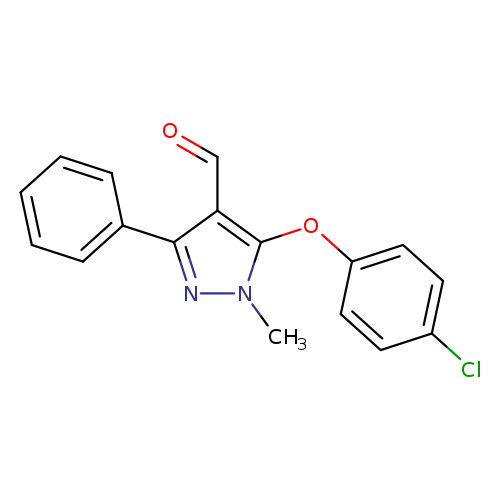
5-(4-chlorophenoxy)-1-methyl-3-phenyl-1H-pyrazole-4-carbaldehydeCatalog No.:AA00IQX9 CAS No.:109925-44-2 MDL No.:MFCD01314899 MF:C17H13ClN2O2 MW:312.7503 |
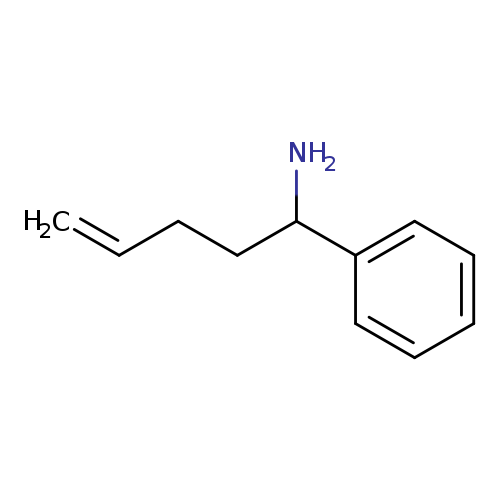
1-phenylpent-4-en-1-amineCatalog No.:AA01A1G8 CAS No.:109925-99-7 MDL No.:MFCD20291162 MF:C11H15N MW:161.2435 |
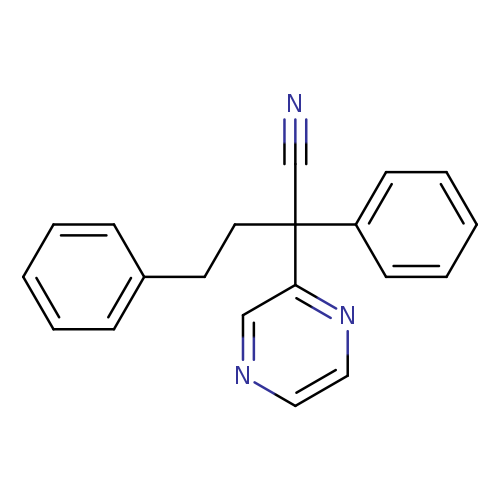
Pyrazineacetonitrile, a-phenyl-a-(2-phenylethyl)-Catalog No.:AA01FLS4 CAS No.:109929-60-4 MDL No.:MFCD00959318 MF:C20H17N3 MW:299.3691 |
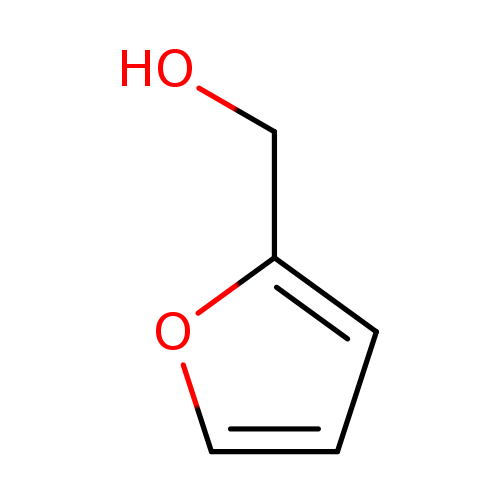
FURFURYLALCOHOL-D2Catalog No.:AA008VRV CAS No.:109930-25-8 MDL No.:MFCD09840649 MF:C5H6O2 MW:98.0999 |