2020-01-05 17:41:03
Angela Maria Casaril1,2 & Micaela Domingues1 & Suely Ribeiro Bampi1 & Darling de Andrade Lourenço1 &
Nathalia Batista Padilha3 & Eder João Lenardão3 & Mariana Sonego4 & Fabiana Kommling Seixas4 & Tiago Collares4 &
Cristina Wayne Nogueira5 & Robert Dantzer2 & Lucielli Savegnago1
Received: 21 August 2018 /Accepted: 11 December 2018 /Published online: 4 January 2019
# Springer-Verlag GmbH Germany, part of Springer Nature 2019
Introduction
Exposure to stress induces behavioral, emotional, and cognitive modifications that can increase the risk of progression of neuropsychiatric disorders such as major depressive disorder (Joëls et al. 2006). Although glucocorticoids (GCs) released by the adrenal cortex in response to stress are claimed to have immunosuppressive and anti-inflammatory properties, recent data indicate they can also have pro-inflammatory influence on the immune system (Elenkov 2008).
Stress can activate the innate immune system through the activation of Toll-like receptor 4 (TLR4) by damage-associated molecular patterns (DAMPs) that are generated from insults to tissues, or by pathogen-associated molecular patterns (PAMPs), which can be a result of bacterial translocation. Activated TLR4 promotes the activation of glycogen synthase kinase 3 beta (GSK-3β) (Jope et al. 2017), which can also be activated by increases in cortisol (Dobarro et al. 2013). Among its many actions, activation of GSK-3β by phosphorylation at tyrosine 216 residue prevents the translocation of nuclear factor erythroid 2-related factor 2 (Nrf2) from the cytosol to the nucleus (Kensler et al. 2007). Consequently, Nrf2 does not bind to the antioxidantresponse element (ARE), thereby reducing the transcription of antioxidant enzymes, such as heme oxygenase-1 (HO-1), superoxide dismutase (SOD), and catalase (CAT) (Kensler et al. 2007). Additionally, GSK-3β promotes the activation of transcription factors in the inflammatory response, such as nuclear factor kappa B (NF-κB), thereby increasing the production of pro-inflammatory cytokines in the central nervous system, especially by microglia and astrocytes (Jope et al. 2017). Proinflammatory cytokines are also responsible for the activation of the enzymes indoleamine-2,3-dioxygenase (IDO) (Schwarcz and Pellicciari 2002) and inducible nitric oxide synthase (iNOS), further contributing to impaired neurotransmission and oxidative imbalance by increased reactive species (RS) generation.
Excessive formation of free radicals and/or defects in the antioxidant defense can damage lipids, proteins, and nucleic acids,
which in turn are implicated in major depressive disorder (Maes et al. 2011), highlighting the importance of the antioxidant
balance in the central nervous system.
Selenium is an essential trace element for human health and exerts most of its action as integral constituent of selenoproteins that are widely implicated in redox signaling (Brigelius-Flohé and Flohé 2017). In turn, seleniumcontaining organic compounds have emerged as promising biologically active molecules (Casaril et al. 2015; Domingues et al., 2018 ; Singh et al. 2016; Masaki et al. 2016; Kil et al. 2017; Gandin et al. 2018; Sudati et al. 2018). Recently, Vieira et al. (2015) reported that the synthetic organoselenium compound 3-((4-chlorophenyl)selanyl)-1-methyl-1H-indole (CMI) has in vitro antioxidant activity, while Casaril et al. (2017b) showed that it potently inhibits inflammation-associated oxidative stress (i.e., hypochlorous acid, peroxynitrite, and hydrogen peroxide). In addition, Birmann et al. (2018) have demonstrated that the antinociceptive effect of CMI is mediated by the monoaminergic, opioidergic, and adenosinergic systems, pointing out the promising role of CMI in modulating different pathways for the treatment of pain and inflammation. Further studies revealed that CMI prevents the depressive-like behavior induced by lipopolysaccharide (LPS) in mice (Casaril et al. 2017a).
In light of the potential antioxidant and anti-inflammatory profile of CMI, we hypothesized that this organoselenium compound should be able to counteract stress-induced depression-like behavior in mice. We used for this purpose a model of acute restraint stress in mice that had already been validated (Pesarico et al. 2015; Thakare et al. 2016; Surkin et al. 2018).
We show here that CMI has antidepressant properties in this model that are associated with downregulation of stressinduced inflammation and oxidative stress.
Materials and methods
Drugs and reagents
CMI (Fig. 1A) was prepared and characterized at the Laboratory of Clean Organic Synthesis at the Federal University of Pelotas, according to Vieira et al. (2015). The RNA extraction reagent was purchased from Ambion (Life Technology, USA). The oligonucleotides were synthesized by Exxtend Biotecnologia Ltda (Campinas, Brazil). All other chemicals were of analytical grade and were obtained from Servylab and WF Científica (Brazil). CMI was dissolved in canola oil (a non-polar and inert substance) and administered intragastrically (i.g.) at a constant volume of 10 ml/kg body weight.
Animals
Behavioral experiments were performed in Swiss male mice (25–30 g; 10–13-week-old), maintained at 22–25 °C and 40–
60% relative humidity with free access to water and food, under a 12:12-h light/dark cycle (lights on at 7:00 a.m.). The studies were performed in accordance with protocols approved by the Committee on the Care and Use of Experimental Animal Resources at the Federal University of Pelotas, Brazil (4034-2017).
Acute restraint stress
The physical restraint was performed in mice as previously reported by Pesarico et al. (2015), with minor modifications. Briefly, mice were subjected to immobilization for 240 min using an individual rodent restraint device made of Plexiglas fenestrate, restraining all physical movement and causing minimum pain. Mice were deprived of food and water during
the physical stress. After the restraint stress, mice were put back in their home cage and received canola oil or CMI 10 min later. They were submitted to behavioral testing 30 min later and euthanized immediately after.
Experimental design
Animals were randomly divided into six experimental groups (n = 6/group) and the physical stress was performed for 240 min (Fig. 1B). The behavioral tasks—open field test (OFT), tail suspension test (TST), new object exploration test (NOET), and splash test—were carried out 280 min after the beginning of the acute restraint stress (ARS) protocol, and the vehicle or CMI (1 or 10 mg/kg, orally) was given 10 min after the physical restraint (Pesarico et al. 2015). Previous data also support the 30-min pre-treatment to evaluate the antidepressant-like activity of CMI (Casaril et al. 2017a) and other selenium-containing compounds (Martinez et al. 2014; Pinto Brod et al. 2016). All the observations were done by an observer blinded to the treatment and the behavioral tests were scored manually. Following the behavioral assessment, mice were anesthetized (inhalation of isoflurane) before blood collection by cardiac puncture (Parasuraman et al. 2010). After that, mice were killed by cervical dislocation, followed by brain removal (residual blood and blood cells can be present in the samples) and isolation of the prefrontal cortex (PFC) and total hippocampus (HC) for analysis (Sunkin et al. 2013).
Behavioral tests
Open field test
The open field test (OFT) was carried out before the other behavioral tests (Walsh and Cummins 1976) to assess the possible effect of the treatments on the locomotor activity. Mice were placed individually in the center of a box (30 ×
30 × 15 cm) divided into nine quadrants of equal areas, and observed for 5 min to report their locomotor (scored by the
number of segments crossed with the four paws) and exploratory activities (expressed by the number of time the mice
stood on rear limbs).
Tail suspension test
The total duration of immobility in the tail suspension test (TST) was measured according to the method described by
Steru et al. (1985). Mice that were both acoustically and visually isolated from each other were suspended 50 cm above the
floor by adhesive tape placed approximately 1 cm from the tip of their tail. During the first 2 min of habituation, the latency
time to immobility was recorded (i.e., time for the first immobility episode), and during the last 4 min, the immobility duration (defined as the absence of the escape attempt behavior) was observed.
New object exploration test
For this test, mice were allowed to explore a novel object for 15 min in an arena 18 × 25 cm2 surrounded by plastic walls (Strekalova et al. 2004). The object had a complex texture surface (artificial flower, 2.5 × 2.5 × 4 cm3) and it was fixed to the center of the arena. The total duration of time spent exploring the object was scored, and at the end of each test session, the arena was cleaned with a 70% ethanol solution.
Splash test
The grooming behavior of mice was observed as a measurement of motivational and self-care activities, which are considered to be deficient in depressive patients. A 10% sucrose solution was squirted on the dorsal coat of each mice and the grooming activity (including nose/face grooming, head washing, and body grooming) was recorded for 5 min (Freitas et al. 2013).
Biochemical evaluation
Tissue processing
The PFC and HC were separated in two hemispheres in order to submit each sample to all biochemical determinations (Casaril et al. 2017a). The right hemispheres were immersed in TRIzol, maintained at − 80 °C, and were submitted to the
quantitative real-time polymerase chain reaction (qRT-PCR). The left hemispheres were homogenized in 50 mM Tris-HCl,
pH 7.4 (1:10, w/v). The homogenate was centrifuged at 2500g for 10 min at 4 °C, and the supernatant fraction was used for
the determination of reactive oxygen species (ROS) formation, thiobarbituric acid reactive species (TBARS) levels, nitric
oxide metabolites (NOx), catalase (CAT), and superoxide dismutase (SOD) activities.
Plasma corticosterone assay
Determination of plasma corticosterone levels was performed according to Zenker and Bernstein (1958). Briefly, aliquots of
plasma were incubated with chloroform and centrifuged for 5 min at 2500 rpm, followed by addition of 0.1 M NaOH and
another round of centrifugation. After the addition of the fluorescence reagent (H2SO4 and ethanol 50%), samples were
centrifuged (5 min at 10,000g) and incubated at room temperature for 2 h. Fluorescence intensity emission, corresponding
to plasma corticosterone levels, was recorded at 540 nm (with 247-nm excitation) and corticosterone levels were expressed
as nanogram per milliliter.
Determination of the reactive oxygen species formation
Quantification of reactive oxygen species (ROS) levels in the PFC and HC of mice was performed according to Loetchutinat et al. (2005). Briefly, aliquots of the homogenate supernatant were incubated with 1 mM dichloro-dihydrofluorescein
diacetate (DCHF-DA) and 10 mM Tris-HCl pH 7.4. The oxidation of DCFH-DA to fluorescent dichlorofluorescein (DCF) is measured for the detection of intracellular ROS. The DCF fluorescence intensity emission was recorded at 520 nm (with 480-nm excitation) and ROS levels were expressed as arbitrary units (AU) of fluorescence.
Thiobarbituric acid reactive species assay
Lipid peroxidation in the PFC and HC was measured by the formation of thiobarbituric acid reactive species (TBARS) during an acid-heating reaction, as described by Ohkawa et al. (1979). An aliquot of the homogenate supernatant was incubated with 8.1% SDS, 0.8% TBA, and acetic acid/HCl (pH 3.4) at 95 °C during 2 h. Malondialdehyde (MDA) was used as a biomarker of lipid peroxidation. Absorbance was measured at 532 nm, and the results were expressed as nanomoles of MDA per gram of tissue.
Nitric oxide in neural tissue
The total nitric oxide (NO) metabolites, NOx, in the PFC and HC of mice were determined by the Griess reaction as an indicator of nitrate/nitrite production (Lima-Junior et al. 2013). Briefly, aliquots of the homogenate were incubated
with equal volume of the Griess reagent for 5 min at 25 °C, and the nitrite concentration was determined bymeasuring the
optical density at 550 nm in reference to a standard curve of NaNO2 solution. Results were expressed as nanomoles of
NOx per gram of tissue.
Evaluation of superoxide dismutase activity
The measurement of superoxide dismutase (SOD) activity is based on the capacity of SOD to inhibit autoxidation of adrenaline to adrenochrome, as described by Misra and Fridovich (1972). The color reaction was detected spectrophotometrically at 480 nmand the enzymatic activity was expressed as units per milligram of protein.
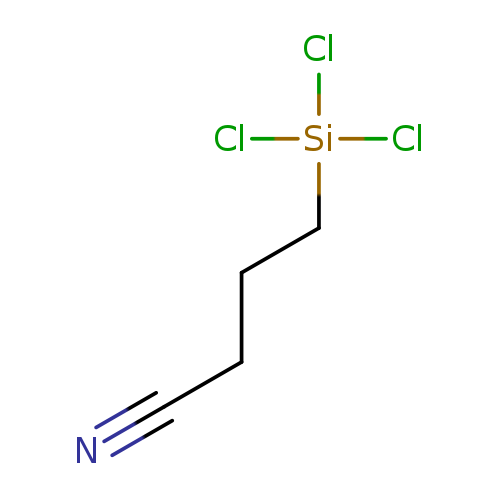
3-CYANOPROPYLTRICHLOROSILANECatalog No.:AA003J9P CAS No.:1071-27-8 MDL No.:MFCD00013832 MF:C4H6Cl3NSi MW:202.5416 |
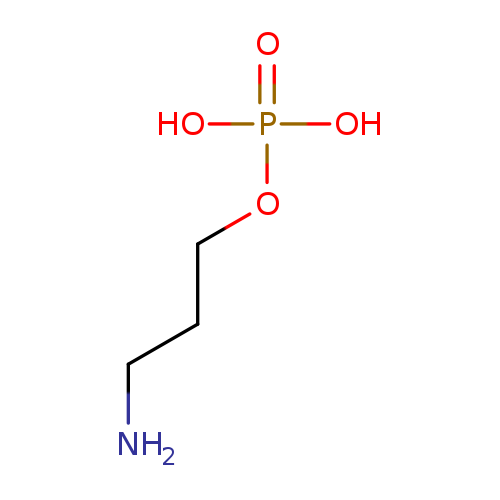
3-Aminopropyl dihydrogen phosphateCatalog No.:AA0082IO CAS No.:1071-28-9 MDL No.:MFCD01757086 MF:C3H10NO4P MW:155.0896 |
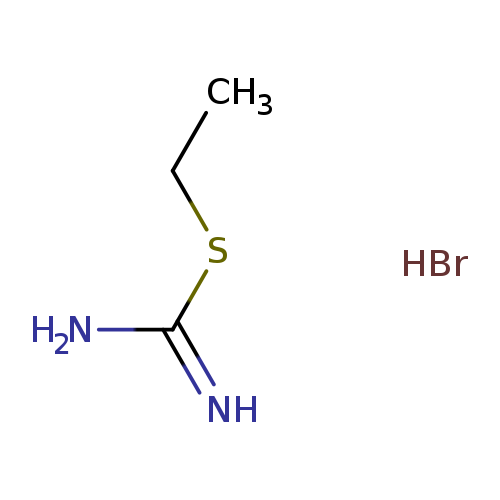
(ethylsulfanyl)methanimidamide hydrobromideCatalog No.:AA003U61 CAS No.:1071-37-0 MDL No.:MFCD00012585 MF:C3H9BrN2S MW:185.0860 |
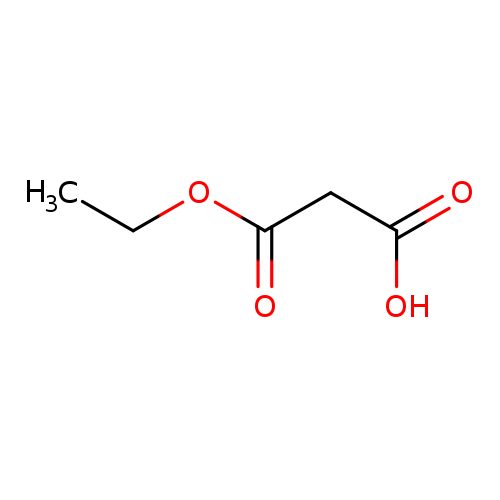
3-Ethoxy-3-oxopropanoic acidCatalog No.:AA003QCJ CAS No.:1071-46-1 MDL No.:MFCD00020490 MF:C5H8O4 MW:132.1146 |
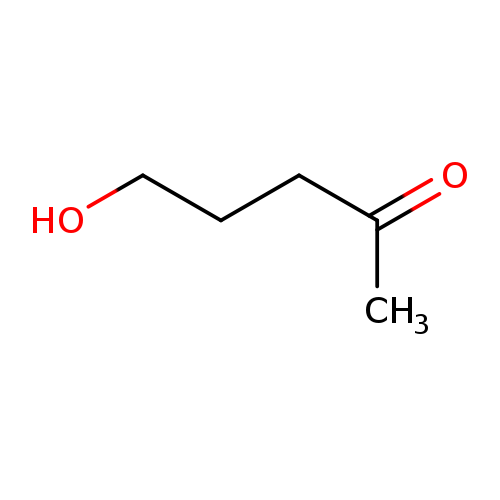
3-Acetyl-1-propanolCatalog No.:AA00347H CAS No.:1071-73-4 MDL No.:MFCD00002961 MF:C5H10O2 MW:102.1317 |
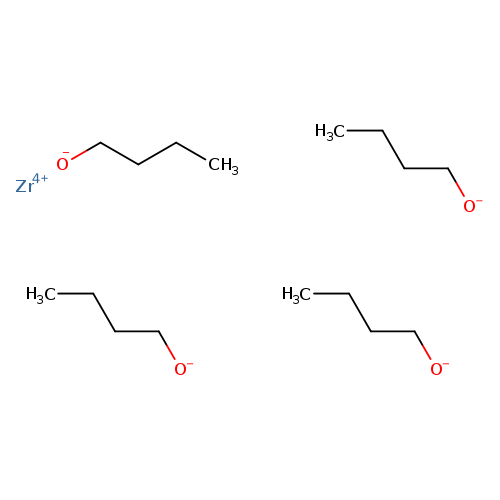
Zirconium(IV) TetrabutoxideCatalog No.:AA007T68 CAS No.:1071-76-7 MDL No.:MFCD00048765 MF:C16H36O4Zr MW:383.6786 |
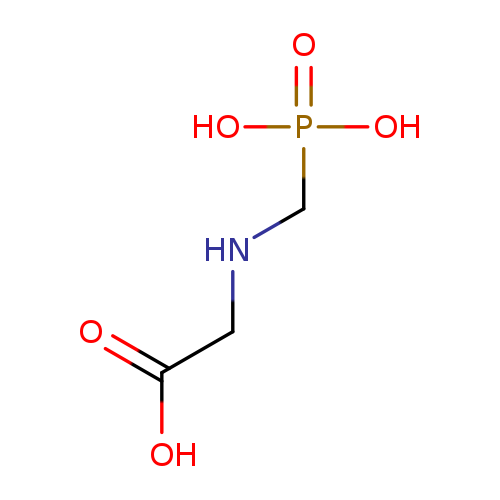
2-[(phosphonomethyl)amino]acetic acid, 40% aqueous solutionCatalog No.:AA0034WG CAS No.:1071-83-6 MDL No.:MFCD00055350 MF:C3H8NO5P MW:169.0731 |
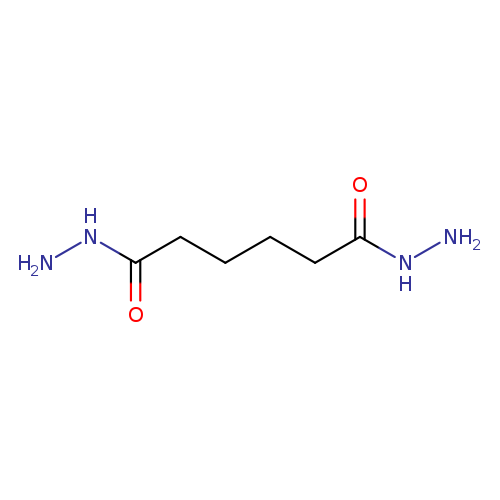
Adipic dihydrazideCatalog No.:AA003QU1 CAS No.:1071-93-8 MDL No.:MFCD00007614 MF:C6H14N4O2 MW:174.2010 |
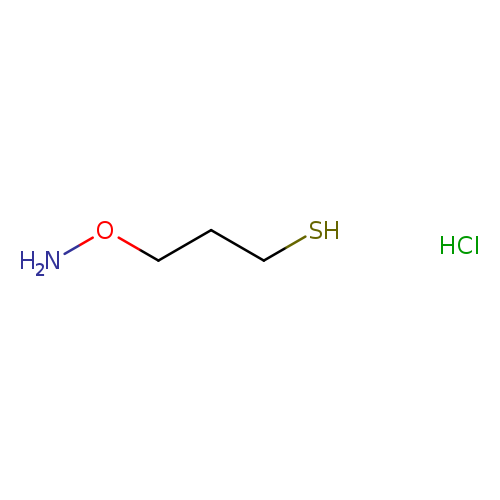
3-(Aminooxy)-1-propanethiol hydrochlorideCatalog No.:AA008WX0 CAS No.:1071-99-4 MDL No.:MFCD15144903 MF:C3H10ClNOS MW:143.6356 |
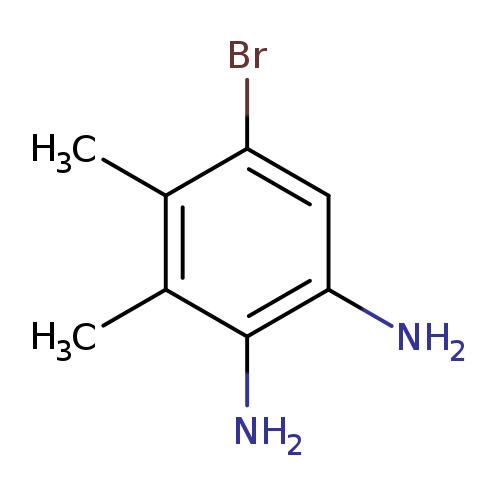
5-Bromo-3,4-dimethylbenzene-1,2-diamineCatalog No.:AA007B30 CAS No.:107100-16-3 MDL No.:MFCD00052696 MF:C8H11BrN2 MW:215.0903 |
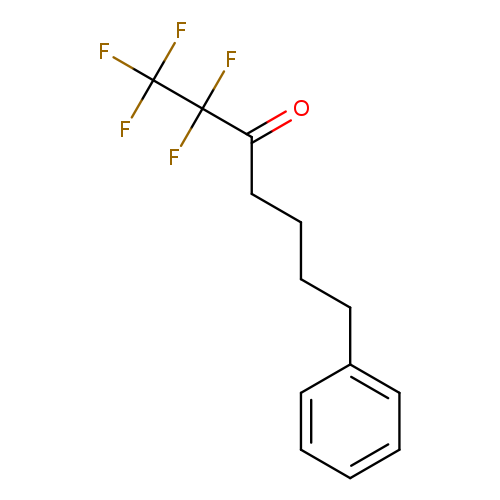
FKGK 11Catalog No.:AA008SP2 CAS No.:1071000-98-0 MDL No.:MFCD18382097 MF:C13H13F5O MW:280.2337 |
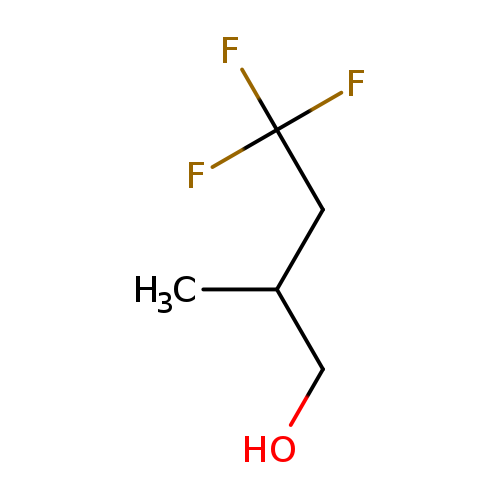
2-Methyl-4,4,4-trifluorobutanolCatalog No.:AA0082IS CAS No.:107103-95-7 MDL No.:MFCD00190639 MF:C5H9F3O MW:142.1196 |
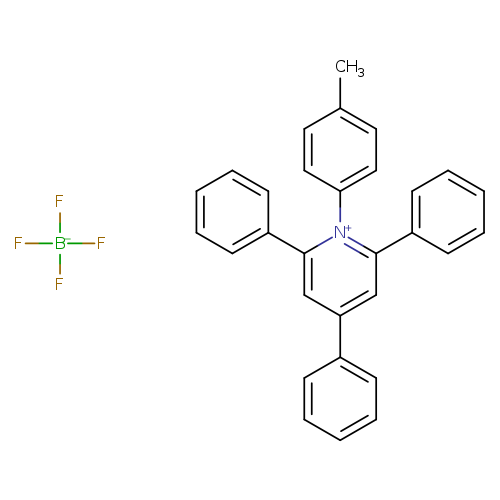
1-(4-Methylphenyl)-2,4,6-triphenylpyridinium tetrafluoroborateCatalog No.:AA01EQMA CAS No.:107108-26-9 MDL No.:MFCD00960926 MF:C30H24BF4N MW:485.3229 |
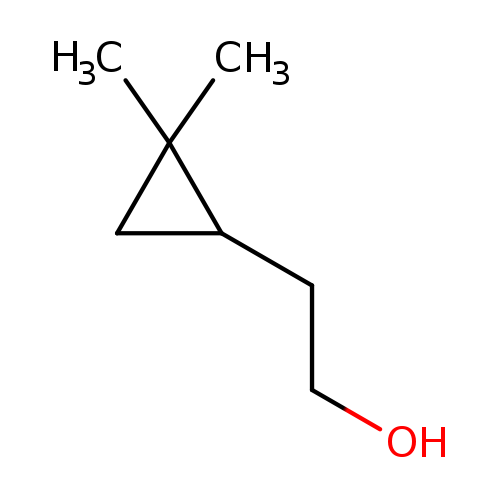
2-(2,2-dimethylcyclopropyl)ethan-1-olCatalog No.:AA01CAAJ CAS No.:1071087-95-0 MDL No.:MFCD29971681 MF:C7H14O MW:114.1855 |
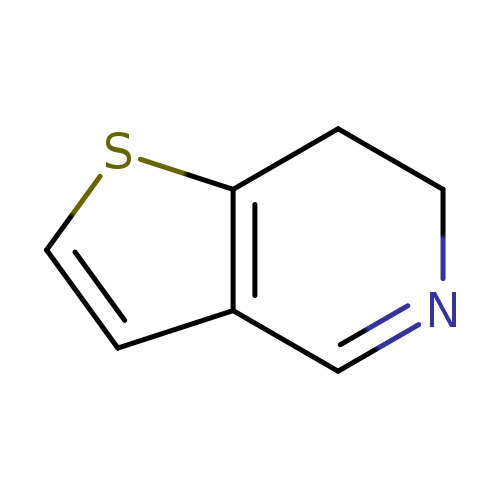
6,7-Dihydrothieno[3,2-C]-pyridineCatalog No.:AA008W7Q CAS No.:107112-93-6 MDL No.:MFCD30741464 MF:C7H7NS MW:137.2022 |
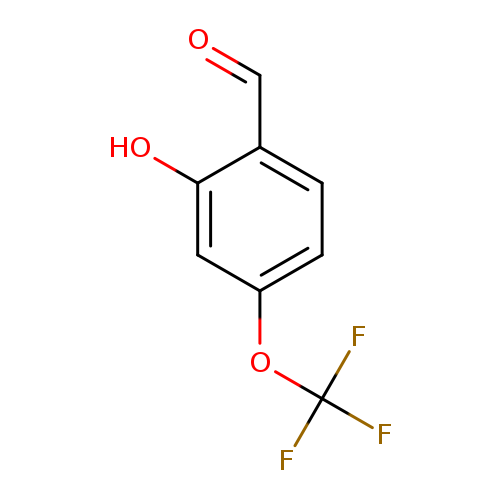
2-Hydroxy-4-(trifluoromethoxy)benzaldehydeCatalog No.:AA00HATX CAS No.:1071156-25-6 MDL No.:MFCD18397426 MF:C8H5F3O3 MW:206.1187 |
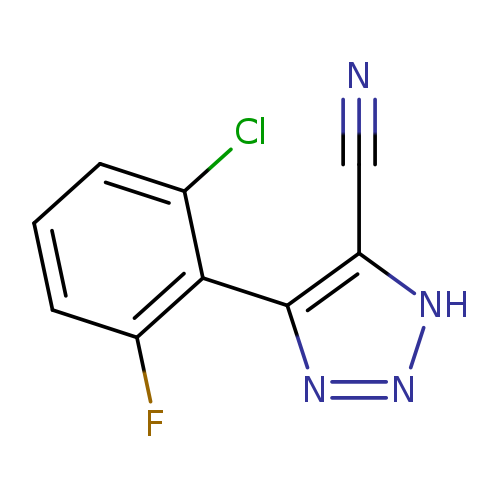
4-(2-chloro-6-fluorophenyl)-1H-1,2,3-triazole-5-carbonitrileCatalog No.:AA00ITI9 CAS No.:1071166-49-8 MDL No.:MFCD00245951 MF:C9H4ClFN4 MW:222.6063 |
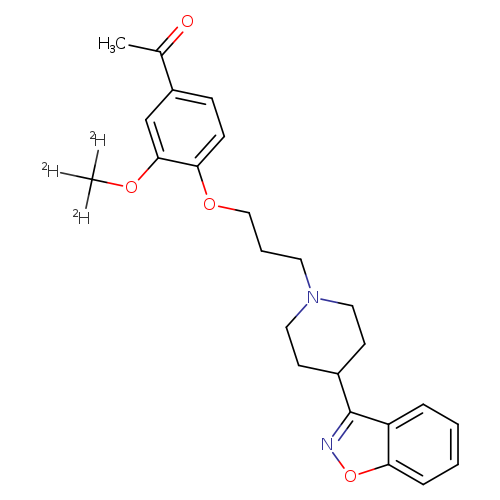
Iloperidone-d3Catalog No.:AA007B2Y CAS No.:1071167-49-1 MDL No.:MFCD30145898 MF:C24H25D3N2O4 MW:411.5086 |
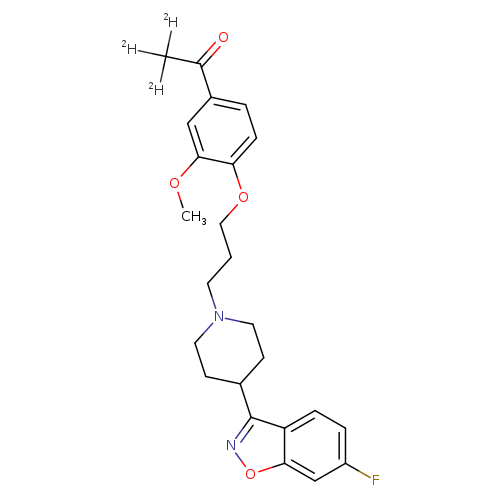
Iloperidone-d3Catalog No.:AA008WDG CAS No.:1071168-82-5 MDL No.: MF:C24H24D3FN2O4 MW:429.4991 |
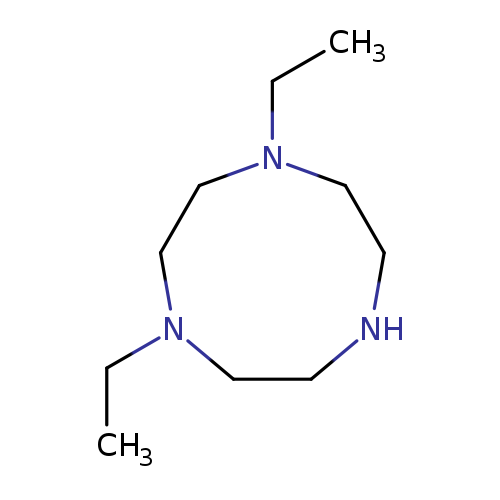
1,4-diethyl-1,4,7-triazonaneCatalog No.:AA01BU27 CAS No.:1071170-26-7 MDL No.:MFCD20683269 MF:C10H23N3 MW:185.3097 |
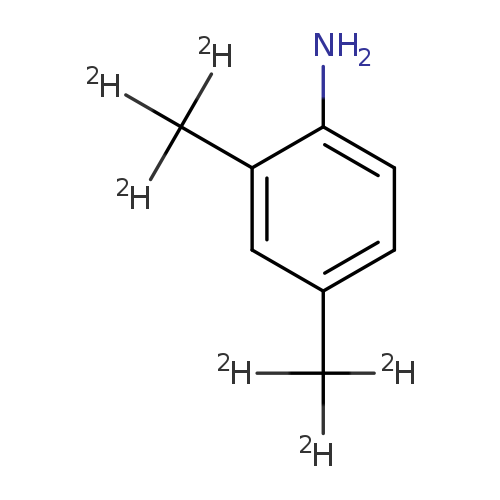
2,4-Xylidine-d6Catalog No.:AA00962J CAS No.:1071170-27-8 MDL No.:MFCD22565800 MF:C8H5D6N MW:127.2166 |
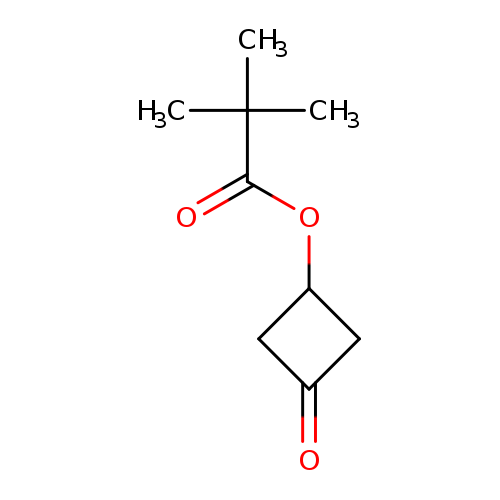
3-Oxocyclobutyl pivalateCatalog No.:AA008ZRL CAS No.:1071194-23-4 MDL No.:MFCD24690600 MF:C9H14O3 MW:170.2057 |
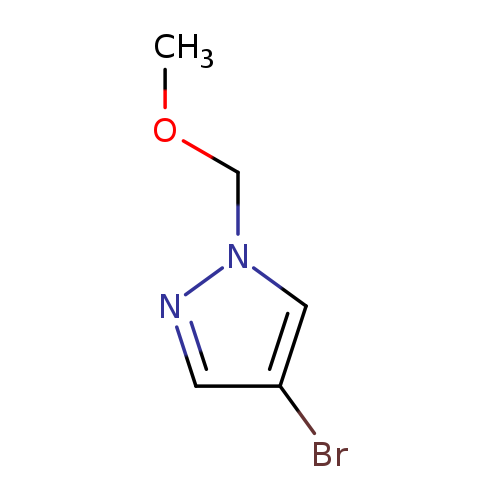
4-Bromo-1-(methoxymethyl)pyrazoleCatalog No.:AA00HATY CAS No.:1071200-42-4 MDL No.:MFCD18324047 MF:C5H7BrN2O MW:191.0259 |
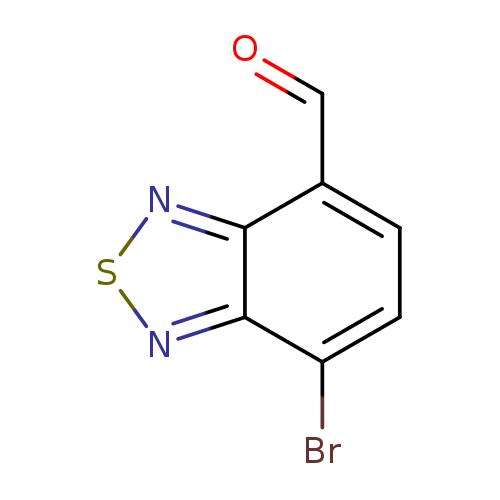
7-bromo-2,1,3-benzothiadiazole-4-carbaldehydeCatalog No.:AA0091WO CAS No.:1071224-34-4 MDL No.:MFCD23704428 MF:C7H3BrN2OS MW:243.0805 |
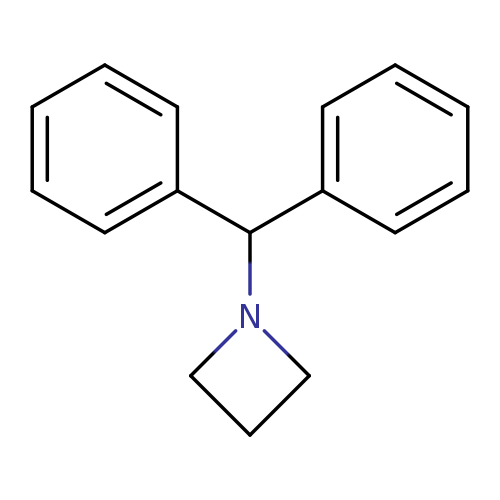
1-BenzhydrylazetidineCatalog No.:AA003DW4 CAS No.:107128-00-7 MDL No.:MFCD00192107 MF:C16H17N MW:223.3129 |
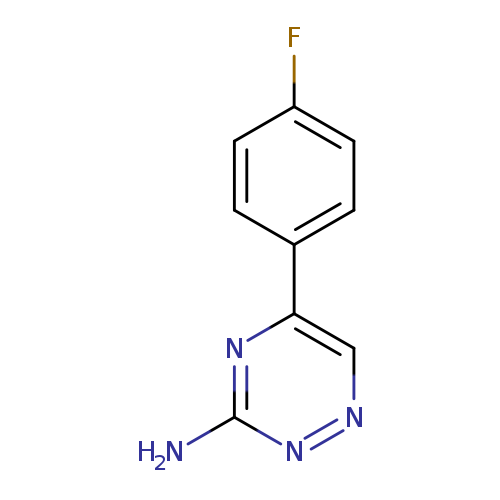
5-(4-Fluorophenyl)-1,2,4-triazin-3-amineCatalog No.:AA019X1L CAS No.:107128-47-2 MDL No.:MFCD06739746 MF:C9H7FN4 MW:190.1771 |
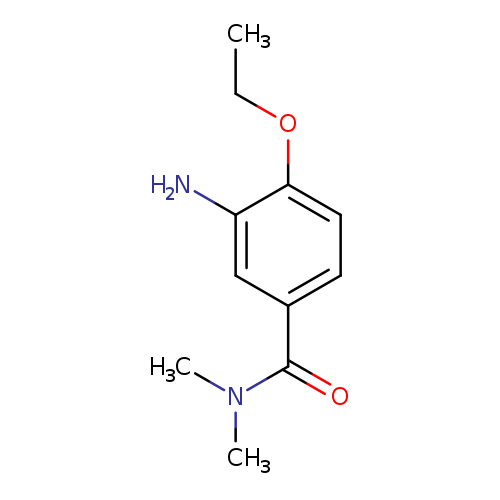
3-Amino-4-ethoxy-n,n-dimethylbenzamideCatalog No.:AA019YBL CAS No.:1071290-49-7 MDL No.:MFCD11695218 MF:C11H16N2O2 MW:208.2569 |
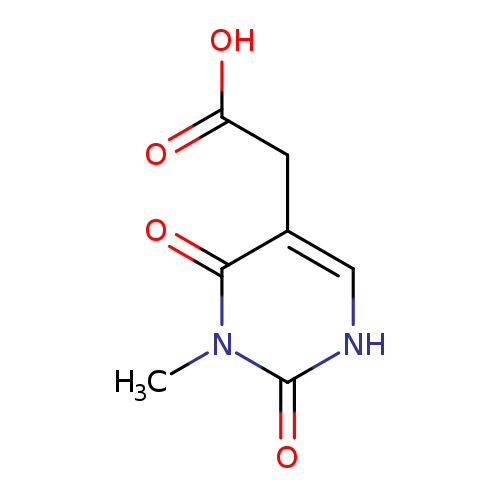
2-(3-Methyl-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-5-yl)acetic acidCatalog No.:AA008VI9 CAS No.:1071308-45-6 MDL No.:MFCD11695658 MF:C7H8N2O4 MW:184.1494 |
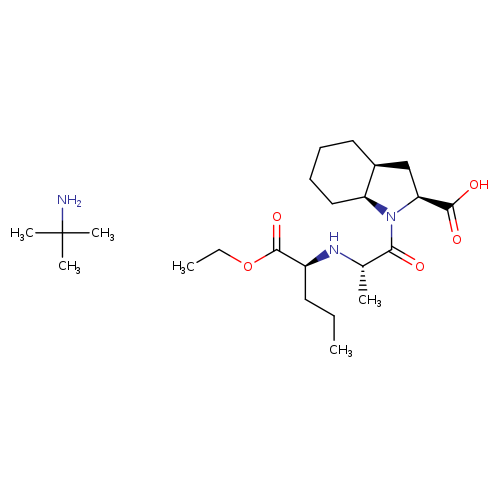
Perindopril erbumineCatalog No.:AA00IKH7 CAS No.:107133-36-8 MDL No.:MFCD02313824 MF:C23H43N3O5 MW:441.6046 |
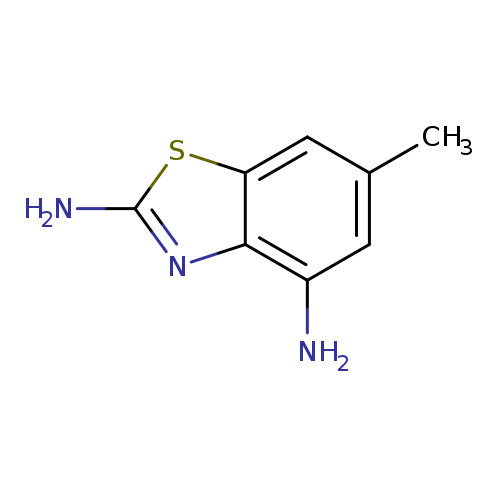
6-Methylbenzo[d]thiazole-2,4-diamineCatalog No.:AA008VBB CAS No.:1071346-94-5 MDL No.:MFCD11695879 MF:C8H9N3S MW:179.2422 |
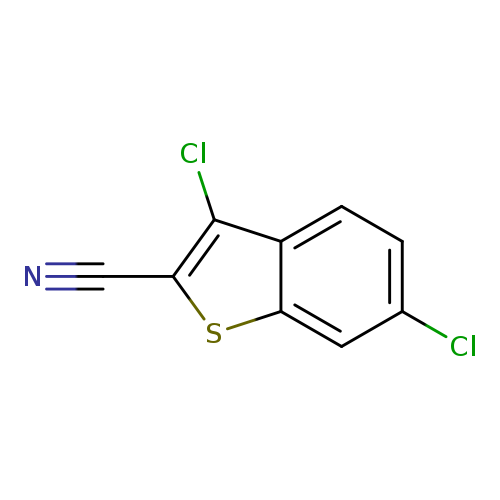
3,6-dichlorobenzo[b]thiophene-2-carbonitrileCatalog No.:AA01BU8M CAS No.:1071350-10-1 MDL No.:MFCD11696309 MF:C9H3Cl2NS MW:228.0978 |
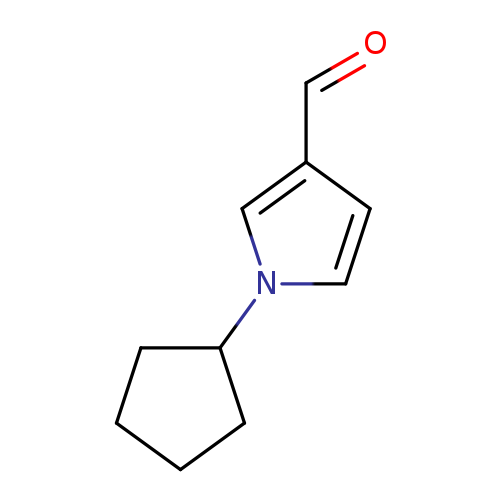
1-Cyclopentyl-1h-pyrrole-3-carbaldehydeCatalog No.:AA008V1F CAS No.:1071359-81-3 MDL No.:MFCD11695056 MF:C10H13NO MW:163.2163 |
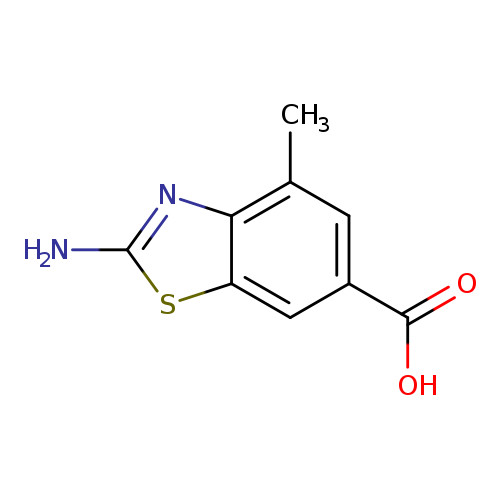
2-amino-4-methyl-1,3-benzothiazole-6-carboxylic acidCatalog No.:AA008VAW CAS No.:1071359-88-0 MDL No.:MFCD11695062 MF:C9H8N2O2S MW:208.2370 |
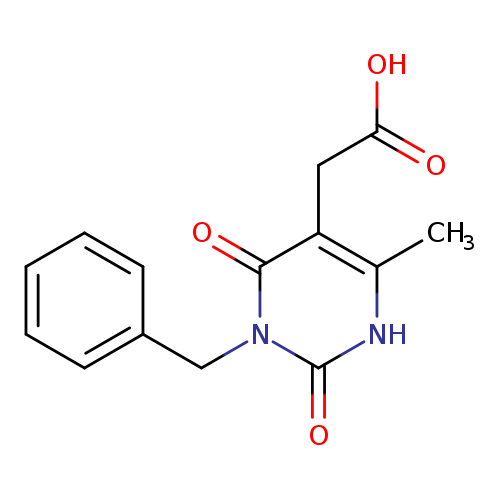
2-(3-benzyl-6-methyl-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-5-yl)acetic acidCatalog No.:AA01AQTJ CAS No.:1071381-42-4 MDL No.:MFCD11695554 MF:C14H14N2O4 MW:274.2720 |
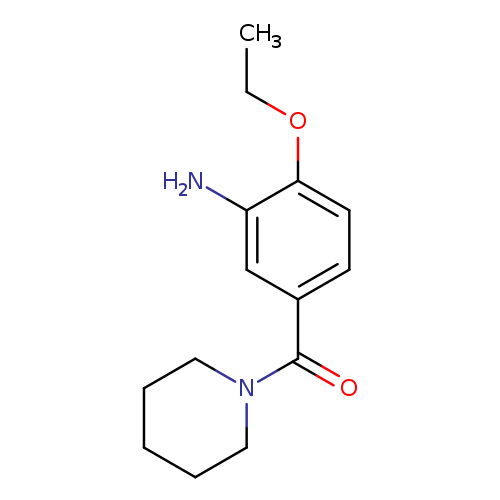
2-Ethoxy-5-[(piperidin-1-yl)carbonyl]anilineCatalog No.:AA019YE6 CAS No.:1071395-78-2 MDL No.:MFCD11695230 MF:C14H20N2O2 MW:248.3208 |
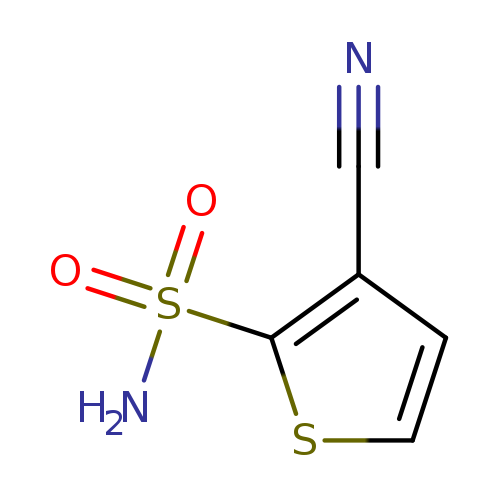
3-Cyanothiophene-2-sulfonamideCatalog No.:AA009T9V CAS No.:107142-12-1 MDL No.:MFCD18817149 MF:C5H4N2O2S2 MW:188.2275 |
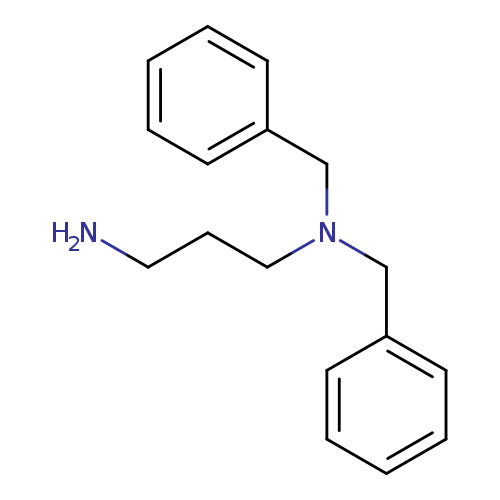
(3-Aminopropyl)dibenzylamineCatalog No.:AA00HAUL CAS No.:107142-94-9 MDL No.:MFCD12569920 MF:C17H22N2 MW:254.3700 |

Methyl 1-Methyl-1H-indazole-4-carboxylateCatalog No.:AA007B19 CAS No.:1071428-42-6 MDL No.:MFCD00509937 MF:C10H10N2O2 MW:190.1986 |
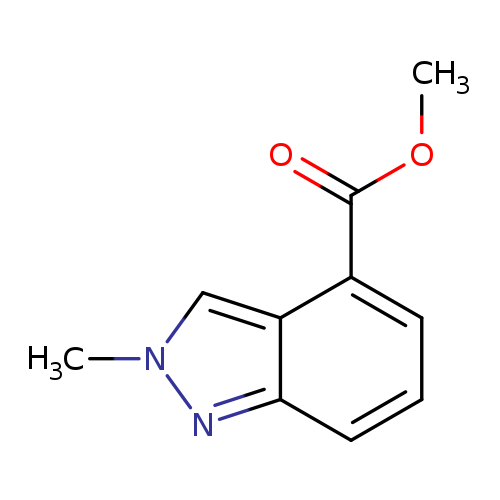
Methyl 2-methyl-2H-indazole-4-carboxylateCatalog No.:AA008U27 CAS No.:1071428-43-7 MDL No.:MFCD11109405 MF:C10H10N2O2 MW:190.1986 |
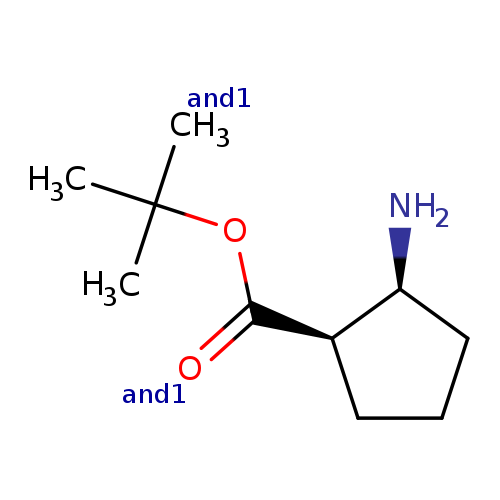
rac-tert-butyl (1R,2S)-2-aminocyclopentane-1-carboxylate, cisCatalog No.:AA01CA4Q CAS No.:1071428-75-5 MDL No.:MFCD31617830 MF:C10H19NO2 MW:185.2634 |
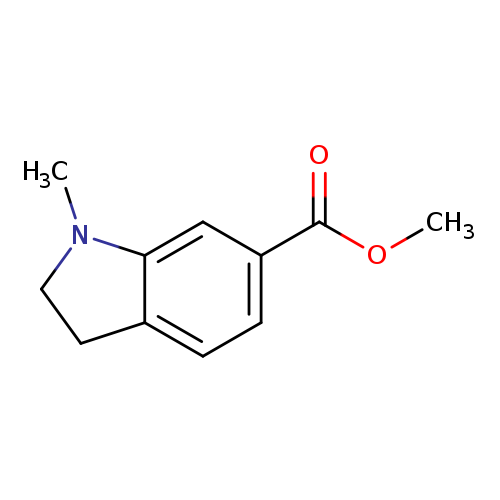
Methyl 1-methyl-2,3-dihydro-1h-indole-6-carboxylateCatalog No.:AA01A6H8 CAS No.:1071432-28-4 MDL No.:MFCD25970327 MF:C11H13NO2 MW:191.2264 |
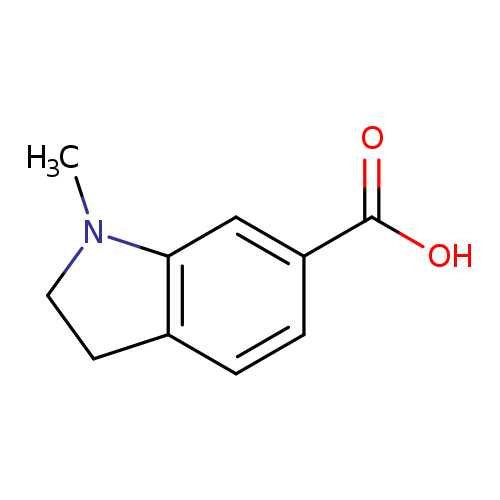
1-Methylindoline-6-carboxylic acidCatalog No.:AA01A668 CAS No.:1071432-99-9 MDL No.:MFCD20660457 MF:C10H11NO2 MW:177.1998 |
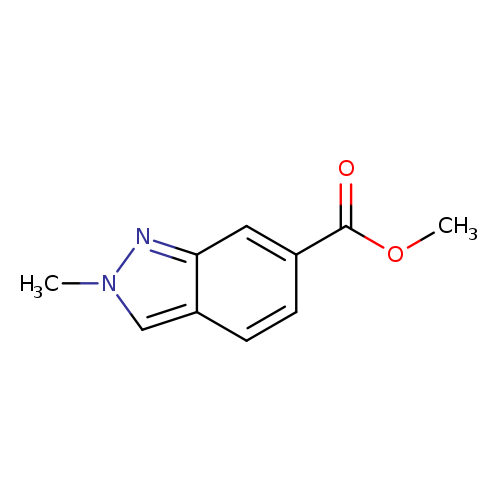
2-Methyl-2h-indazole-6-carboxylic acid methyl esterCatalog No.:AA008TO1 CAS No.:1071433-01-6 MDL No.:MFCD11109407 MF:C10H10N2O2 MW:190.1986 |
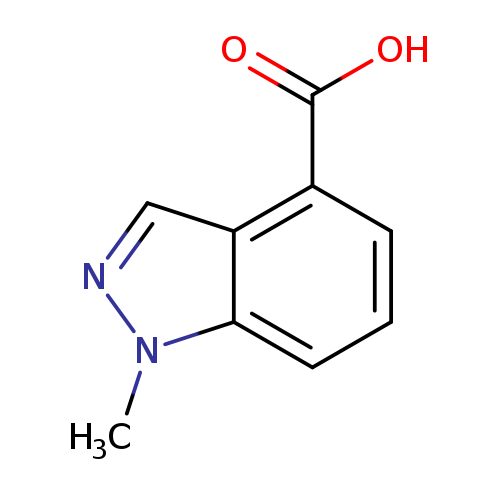
1-Methyl-1h-indazole-4-carboxylic acidCatalog No.:AA008TWW CAS No.:1071433-05-0 MDL No.:MFCD15071442 MF:C9H8N2O2 MW:176.1720 |
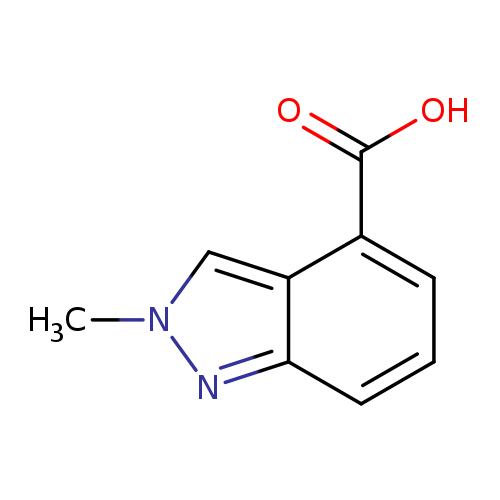
2-Methyl-2h-indazole-4-carboxylic acidCatalog No.:AA008U4G CAS No.:1071433-06-1 MDL No.:MFCD15071443 MF:C9H8N2O2 MW:176.1720 |
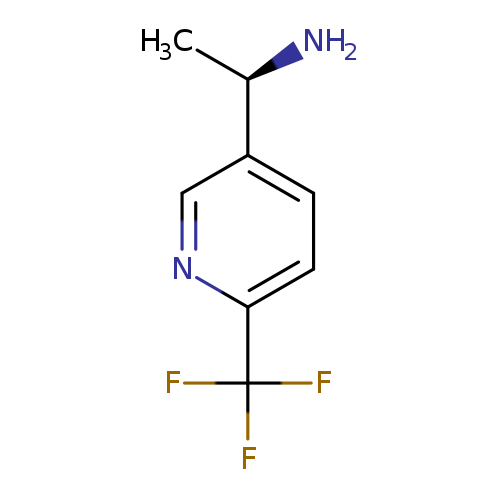
(1R)-1-[6-(Trifluoromethyl)(3-pyridyl)]ethylamineCatalog No.:AA00968Y CAS No.:1071435-61-4 MDL No.:MFCD09256746 MF:C8H9F3N2 MW:190.1657 |
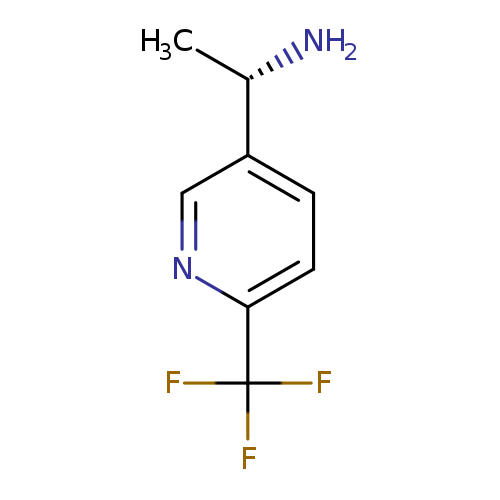
(S)-1-(6-(Trifluoromethyl)pyridin-3-yl)ethanamineCatalog No.:AA009690 CAS No.:1071435-62-5 MDL No.:MFCD09256747 MF:C8H9F3N2 MW:190.1657 |
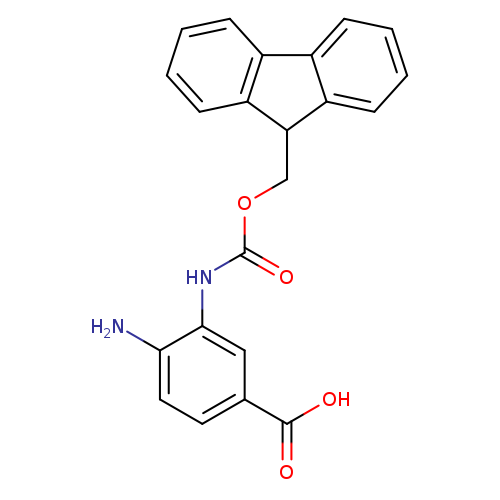
3-Fmoc-4-diaminobenzoic acidCatalog No.:AA009KAG CAS No.:1071446-05-3 MDL No.:MFCD22989433 MF:C22H18N2O4 MW:374.3893 |
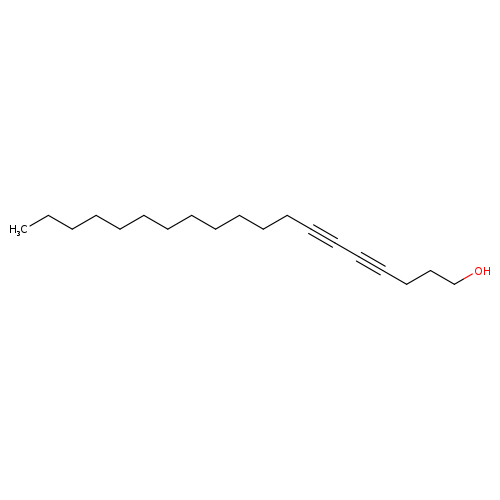
4,6-Nonadecadiyn-1-olCatalog No.:AA008XXW CAS No.:1071447-01-2 MDL No.:MFCD06797150 MF:C19H32O MW:276.4568 |
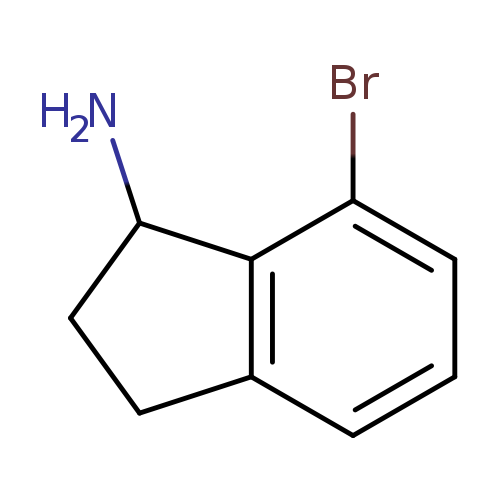
7-Bromo-2,3-dihydro-1H-inden-1-amineCatalog No.:AA009663 CAS No.:1071449-08-5 MDL No.:MFCD17215685 MF:C9H10BrN MW:212.0864 |
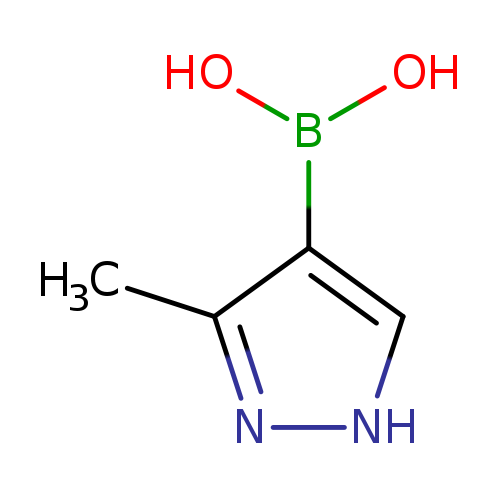
3-Methylpyrazole-4-boronic acidCatalog No.:AA00388V CAS No.:1071455-14-5 MDL No.:MFCD12546507 MF:C4H7BN2O2 MW:125.9216 |
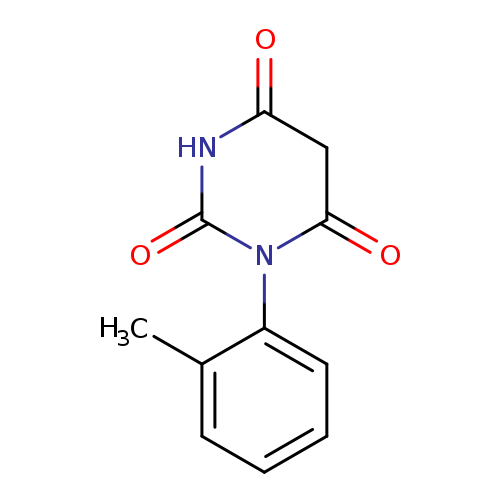
1-(2-Methylphenyl)-2,4,6(1h,3h,5h)-pyrimidinetrioneCatalog No.:AA01APXL CAS No.:107147-53-5 MDL No.:MFCD00552519 MF:C11H10N2O3 MW:218.2087 |
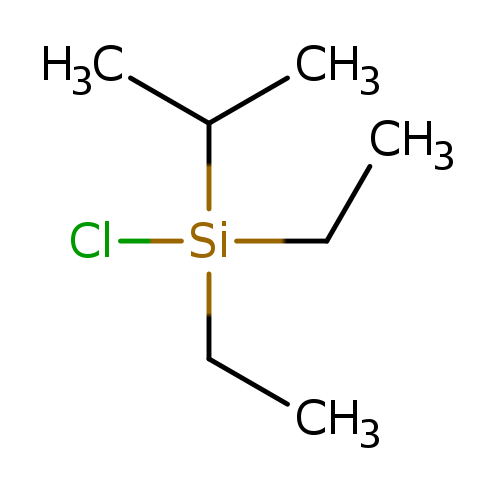
DIETHYLISOPROPYLSILYL CHLORIDECatalog No.:AA003PFG CAS No.:107149-56-4 MDL No.:MFCD00191631 MF:C7H17ClSi MW:164.7484 |
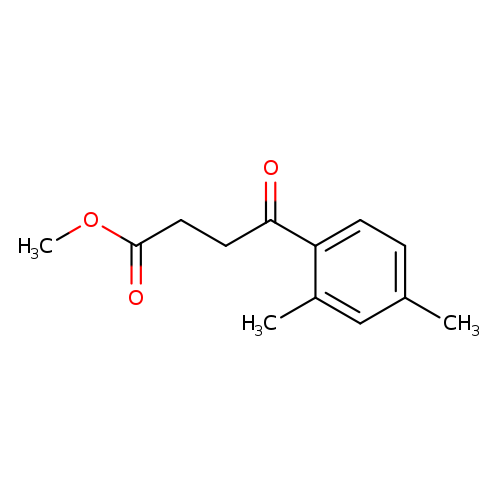
Methyl 4-(2,4-dimethylphenyl)-4-oxobutanoateCatalog No.:AA01FAGM CAS No.:107151-32-6 MDL No.:MFCD16068125 MF:C13H16O3 MW:220.2643 |
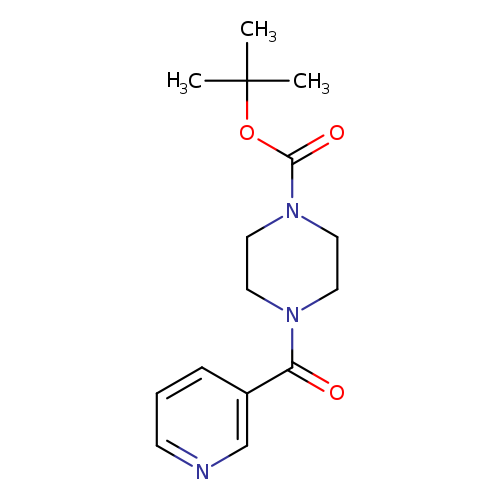
1-Boc-4-(3-pyridinylcarbonyl)piperazineCatalog No.:AA00HAUU CAS No.:1071521-50-0 MDL No.:MFCD16620803 MF:C15H21N3O3 MW:291.3455 |
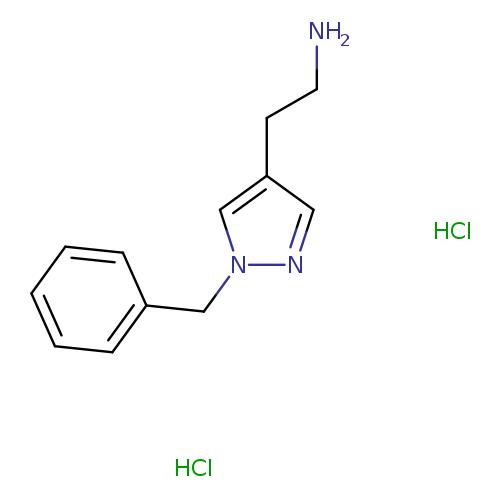
2-(1-benzyl-1H-pyrazol-4-yl)ethan-1-amine dihydrochlorideCatalog No.:AA019ZFV CAS No.:1071537-11-5 MDL No.:MFCD22369966 MF:C12H17Cl2N3 MW:274.1895 |
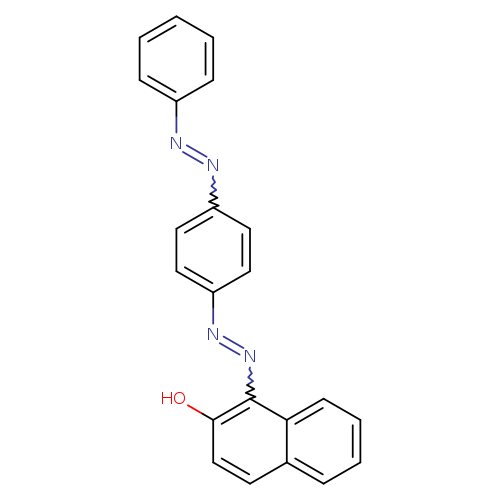
1-[4-(Phenylazo)phenylazo]-2-naphtholCatalog No.:AA01EMHK CAS No.:1071538-45-8 MDL No.:MFCD00003905 MF:C22H16N4O MW:352.3886 |
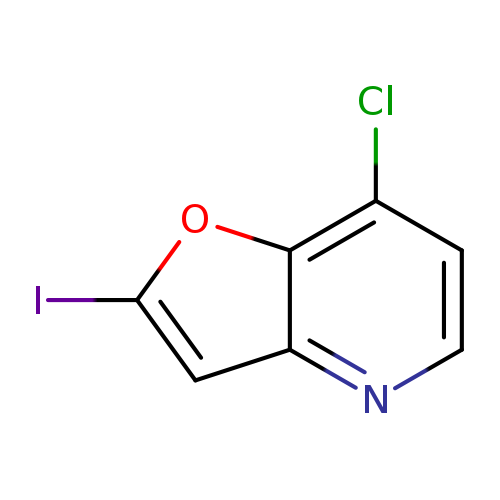
7-Chloro-2-iodofuro[3,2-b]pyridineCatalog No.:AA0094OC CAS No.:1071540-54-9 MDL No.:MFCD17016060 MF:C7H3ClINO MW:279.4623 |
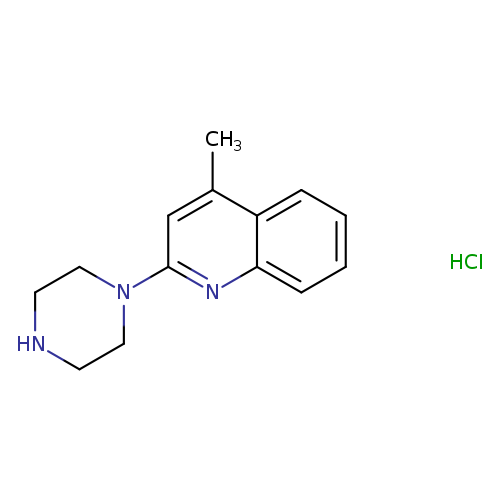
4-methyl-2-(1-piperazinyl)quinoline hydrochlorideCatalog No.:AA00J0SK CAS No.:1071545-91-9 MDL No.:MFCD07202007 MF:C14H18ClN3 MW:263.7658 |
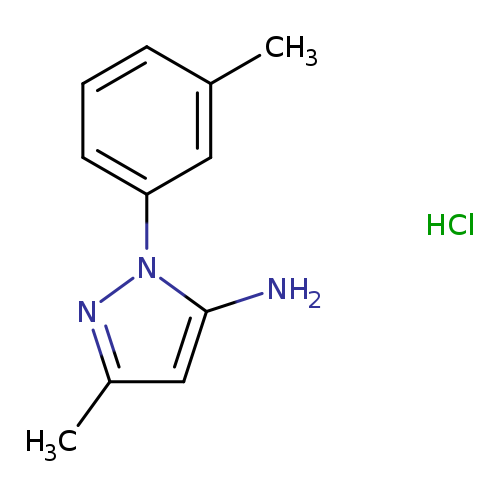
3-methyl-1-(3-methylphenyl)-1H-pyrazol-5-amine hydrochlorideCatalog No.:AA019H0Y CAS No.:1071548-24-7 MDL No.:MFCD08447169 MF:C11H14ClN3 MW:223.7020 |
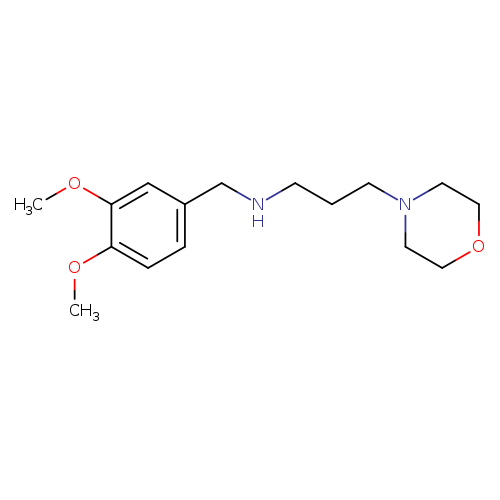
N-(3,4-Dimethoxybenzyl)-3-morpholinopropan-1-amineCatalog No.:AA018XWV CAS No.:107155-56-6 MDL No.:MFCD01475400 MF:C16H26N2O3 MW:294.3892 |
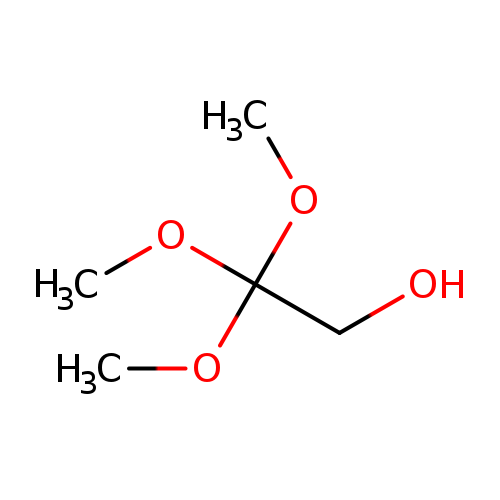
2,2,2-TRIMETHOXYETHAN-1-OLCatalog No.:AA01DX73 CAS No.:107155-72-6 MDL No.:MFCD12403548 MF:C5H12O4 MW:136.1464 |
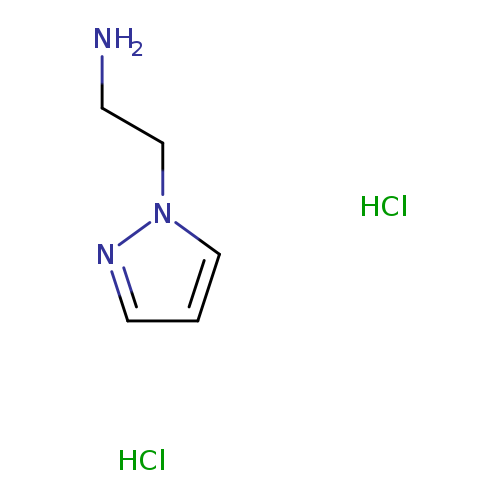
[2-(1H-pyrazol-1-yl)ethyl]amine dihydrochlorideCatalog No.:AA00J0QY CAS No.:1071550-46-3 MDL No.:MFCD06801243 MF:C5H11Cl2N3 MW:184.0669 |
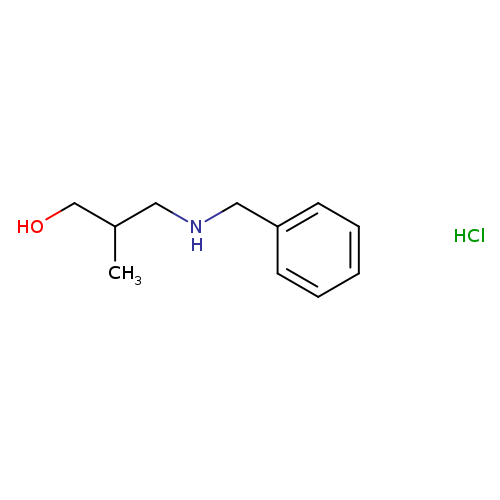
3-(Benzylamino)-2-methylpropan-1-ol hydrochlorideCatalog No.:AA00J3VE CAS No.:1071567-87-7 MDL No.:MFCD11179446 MF:C11H18ClNO MW:215.7197 |
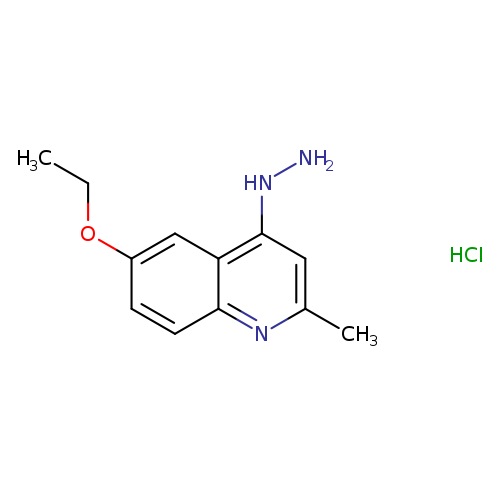
6-Ethoxy-4-hydrazino-2-methylquinoline hydrochlorideCatalog No.:AA007TC0 CAS No.:1071572-37-6 MDL No.:MFCD09787829 MF:C12H16ClN3O MW:253.7279 |
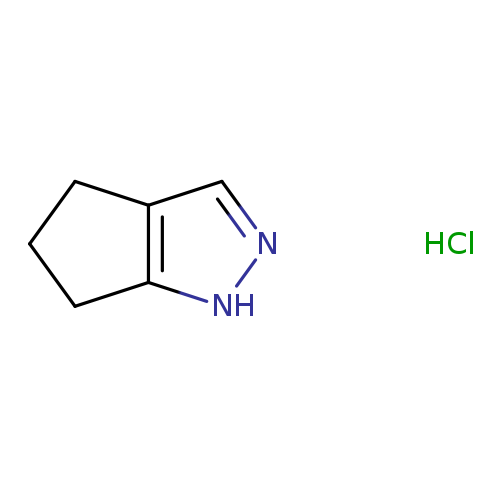
1,4,5,6-Tetrahydrocyclopenta[c]pyrazole hydrochlorideCatalog No.:AA00J1ZY CAS No.:1071575-85-3 MDL No.:MFCD11983586 MF:C6H9ClN2 MW:144.6021 |
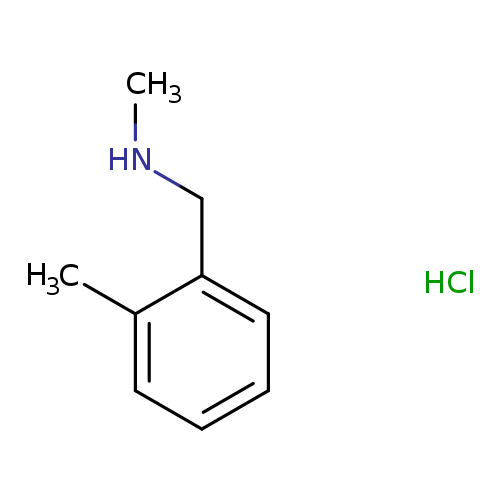
N-methyl-1-(2-methylphenyl)methanamine hydrochlorideCatalog No.:AA00J1RL CAS No.:1071580-83-0 MDL No.:MFCD07110180 MF:C9H14ClN MW:171.6672 |
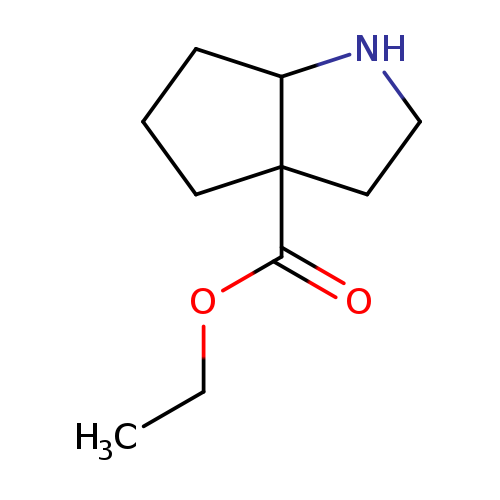
ethyl octahydrocyclopenta[b]pyrrole-3a-carboxylateCatalog No.:AA01BNAU CAS No.:1071585-99-3 MDL No.:MFCD24518617 MF:C10H17NO2 MW:183.2475 |
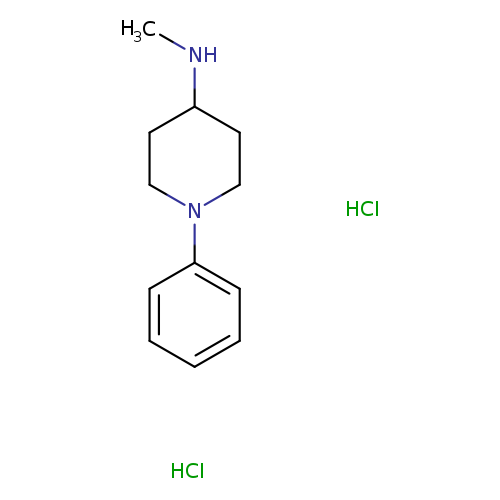
N-Methyl-1-phenylpiperidin-4-amine dihydrochlorideCatalog No.:AA01BH74 CAS No.:1071590-88-9 MDL No.:MFCD28139558 MF:C12H20Cl2N2 MW:263.2066 |
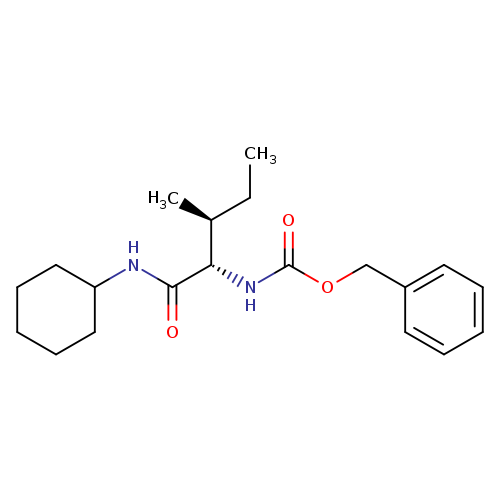
N-Cyclohexyl L-Z-isoleucinamideCatalog No.:AA009484 CAS No.:1071594-16-5 MDL No.:MFCD23135925 MF:C20H30N2O3 MW:346.4638 |
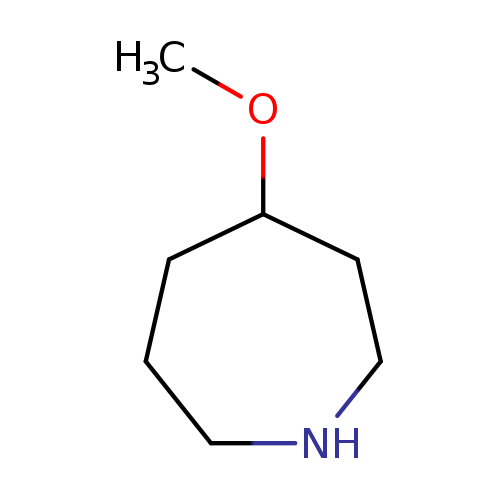
4-MethoxyazepaneCatalog No.:AA00995Y CAS No.:1071594-49-4 MDL No.:MFCD17015864 MF:C7H15NO MW:129.2001 |
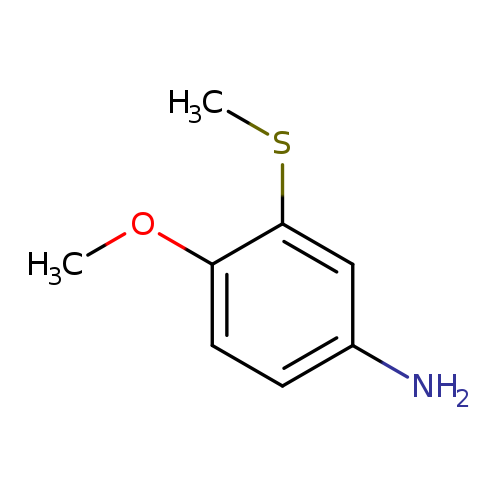
4-methoxy-3-(methylsulfanyl)anilineCatalog No.:AA01C2GR CAS No.:1071602-10-2 MDL No.:MFCD22489025 MF:C8H11NOS MW:169.2440 |
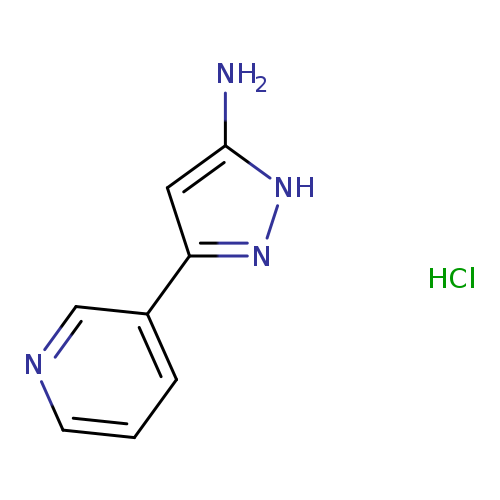
5-(Pyridin-3-yl)-1H-pyrazol-3-amine hydrochlorideCatalog No.:AA01A7SD CAS No.:1071623-05-6 MDL No.:MFCD28252264 MF:C8H9ClN4 MW:196.6369 |
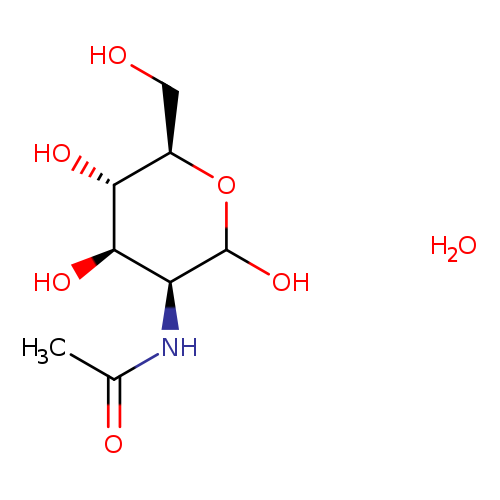
N-Acetyl-D-mannosamine monohydrateCatalog No.:AA01FDYP CAS No.:1071625-31-4 MDL No.:MFCD00149493 MF:C8H17NO7 MW:239.2231 |
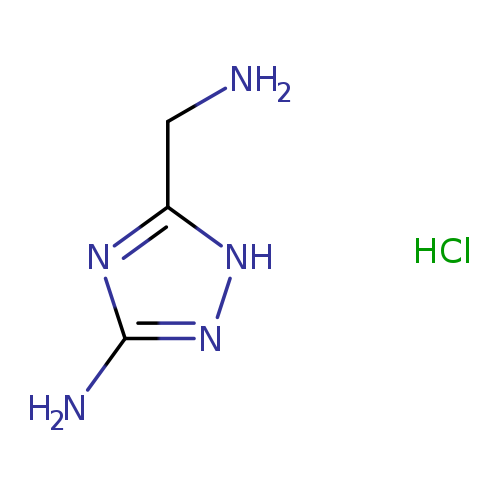
3-(Aminomethyl)-1H-1,2,4-triazol-5-amine hydrochlorideCatalog No.:AA008UAC CAS No.:1071625-99-4 MDL No.:MFCD19691792 MF:C3H8ClN5 MW:149.5821 |
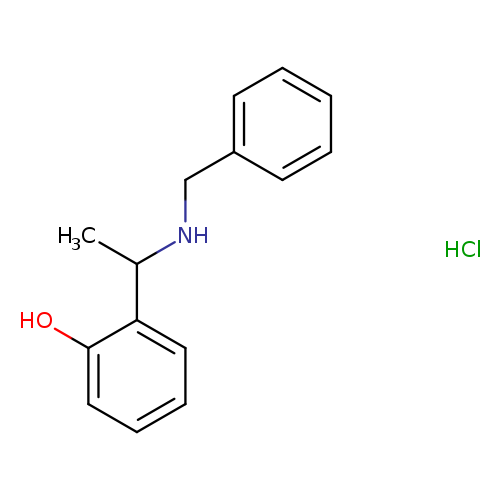
2-(1-(Benzylamino)ethyl)phenol hydrochlorideCatalog No.:AA00HAUY CAS No.:1071628-77-7 MDL No.:MFCD26959779 MF:C15H18ClNO MW:263.7625 |
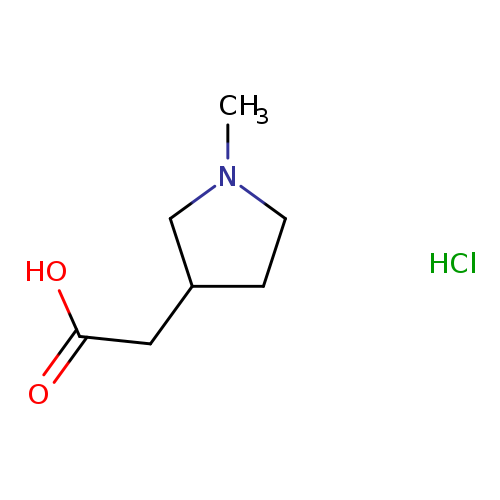
2-(1-Methylpyrrolidin-3-yl)acetic acid hydrochlorideCatalog No.:AA007TBZ CAS No.:1071634-26-8 MDL No.:MFCD23135758 MF:C7H14ClNO2 MW:179.6446 |
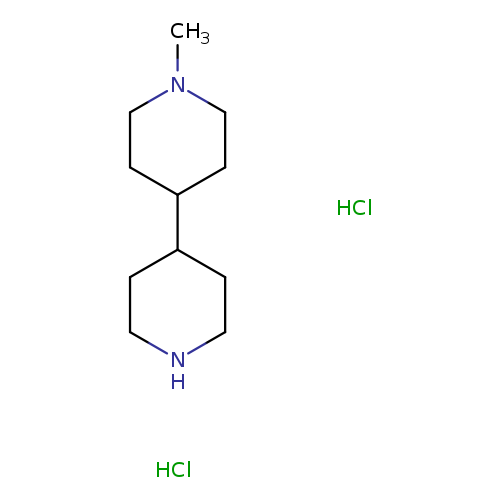
1-Methyl-4,4'-bipiperidine hydrochlorideCatalog No.:AA0092B0 CAS No.:1071634-39-3 MDL No.:MFCD09759210 MF:C11H24Cl2N2 MW:255.2277 |
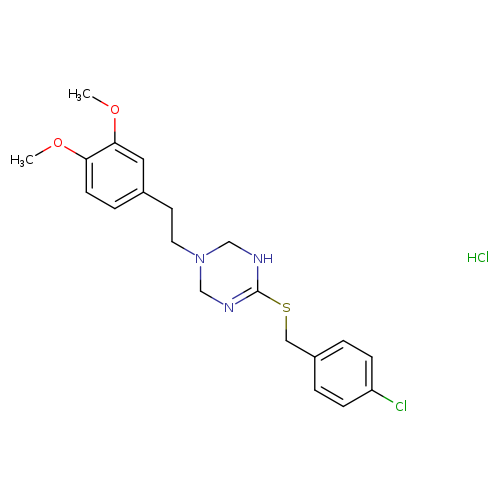
Mac13243Catalog No.:AA008TEV CAS No.:1071638-38-4 MDL No.:MFCD18782745 MF:C20H25Cl2N3O2S MW:442.4024 |
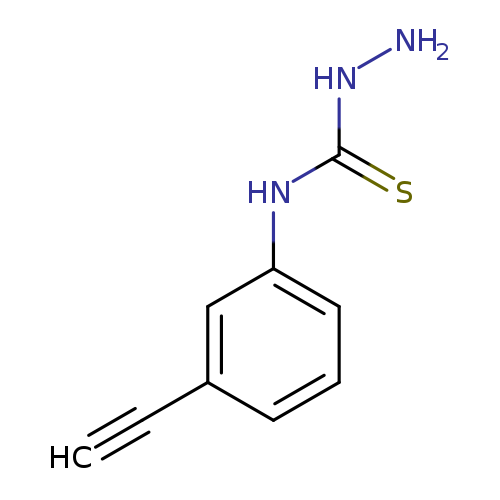
3-amino-1-(3-ethynylphenyl)thioureaCatalog No.:AA01A9WT CAS No.:1071638-49-7 MDL No.:MFCD16817510 MF:C9H9N3S MW:191.2529 |
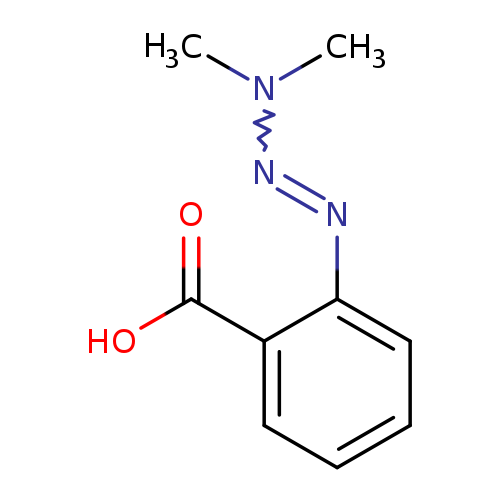
2-(dimethyltriaz-1-en-1-yl)benzoic acidCatalog No.:AA019OWL CAS No.:1071638-58-8 MDL No.:MFCD00013975 MF:C9H11N3O2 MW:193.2025 |
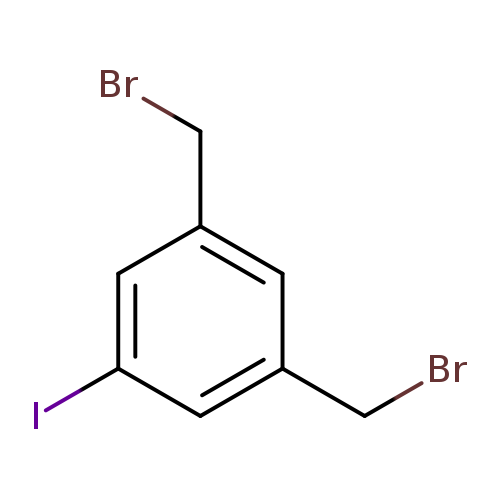
1,3-bis(bromomethyl)-5-iodobenzeneCatalog No.:AA01BTIB CAS No.:107164-93-2 MDL No.:MFCD22492791 MF:C8H7Br2I MW:389.8537 |
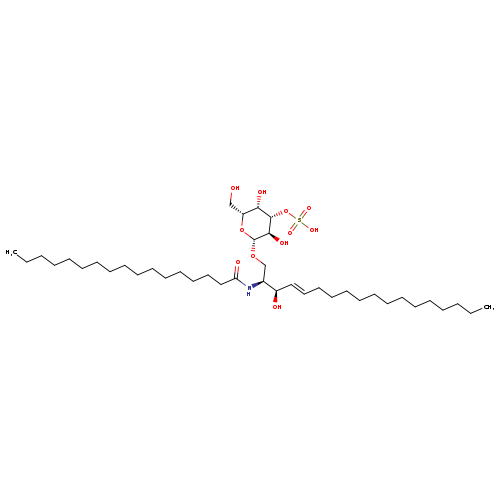
N-[(1S,2R,3E)-2-hydroxy-1-[[(3-O-sulfo-β-D-galactopyranosyl)oxy]methyl]-3-heptadecen-1-yl]-heptadecanamideCatalog No.:AA01EPUS CAS No.:1071654-84-6 MDL No.: MF:C41H79NO11S MW:794.1311 |
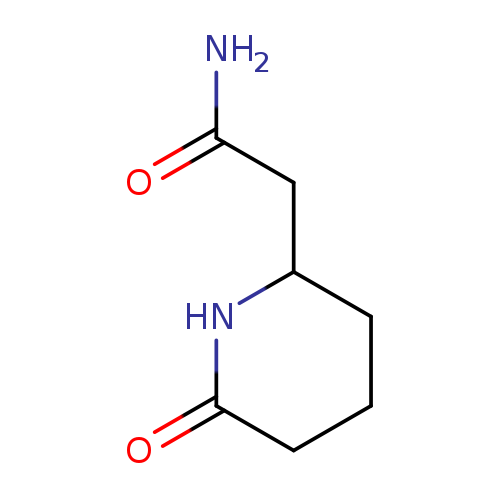
2-(6-Oxopiperidin-2-yl)acetamideCatalog No.:AA01AJ5Y CAS No.:1071661-20-5 MDL No.:MFCD19382158 MF:C7H12N2O2 MW:156.1824 |
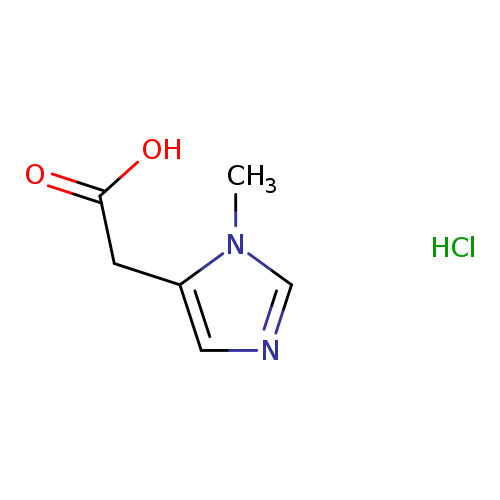
2-(1-Methyl-1h-imidazol-5-yl)acetic acid hydrochlorideCatalog No.:AA01AF3X CAS No.:1071661-55-6 MDL No.:MFCD27500744 MF:C6H9ClN2O2 MW:176.6009 |
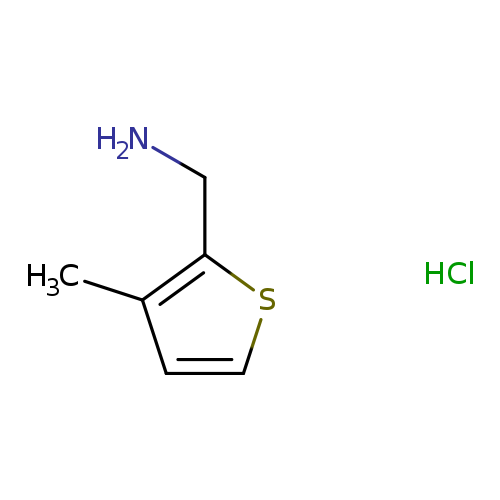
[(3-Methyl-2-thienyl)methyl]amine hydrochlorideCatalog No.:AA00HAV0 CAS No.:1071674-80-0 MDL No.:MFCD09971550 MF:C6H10ClNS MW:163.6683 |
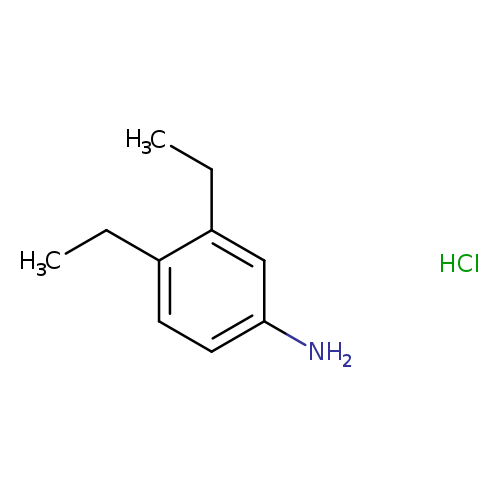
3,4-diethylaniline hydrochlorideCatalog No.:AA01A4II CAS No.:1071678-12-0 MDL No.:MFCD18827535 MF:C10H16ClN MW:185.6937 |
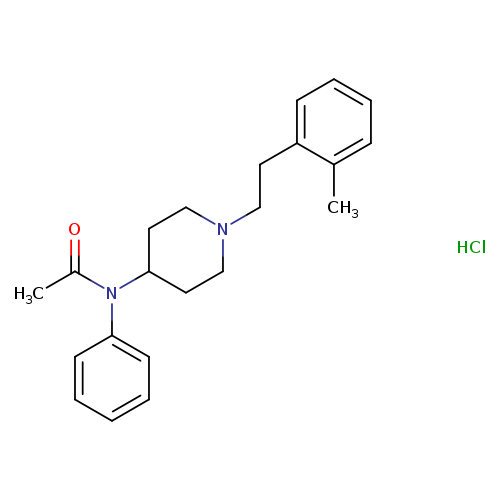
N-[1-[2-(2-methylphenyl)ethyl]-4-piperidinyl]-N-phenyl-acetamide,monohydrochlorideCatalog No.:AA01EQ9T CAS No.:1071703-90-6 MDL No.: MF:C22H29ClN2O MW:372.9315 |
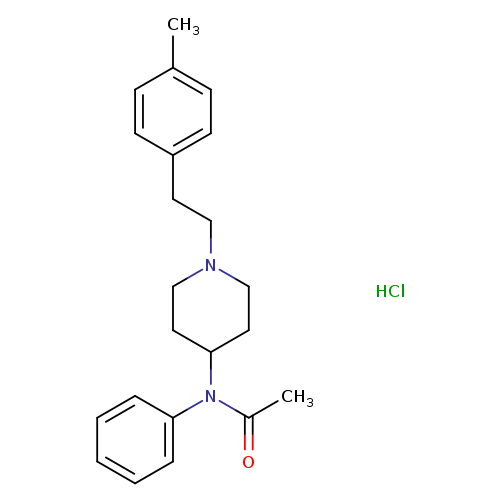
N-(1-(4-Methylphenethyl)piperidin-4-yl)-N-phenylacetamideHydrochlorideCatalog No.:AA01DZFI CAS No.:1071703-95-1 MDL No.:MFCD30186655 MF:C22H29ClN2O MW:372.9315 |
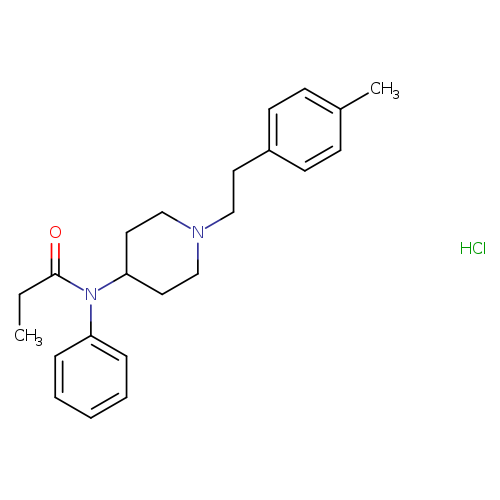
N-[1-[2-(4-methylphenyl)ethyl]-4-piperidinyl]-N-phenyl-propanamide,monohydrochlorideCatalog No.:AA01EPP7 CAS No.:1071703-97-3 MDL No.: MF:C23H31ClN2O MW:386.9580 |
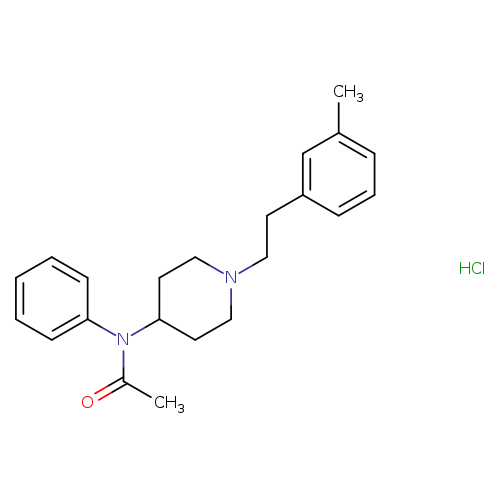
N-[1-[2-(3-methylphenyl)ethyl]-4-piperidinyl]-N-phenyl-acetamide,monohydrochlorideCatalog No.:AA01EQ9P CAS No.:1071704-02-3 MDL No.: MF:C22H29ClN2O MW:372.9315 |
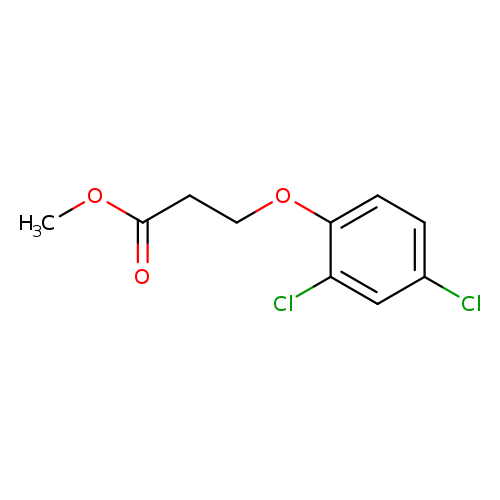
methyl 3-(2,4-dichlorophenoxy)propanoateCatalog No.:AA019Z2T CAS No.:1071715-08-6 MDL No.:MFCD00152418 MF:C10H10Cl2O3 MW:249.0906 |
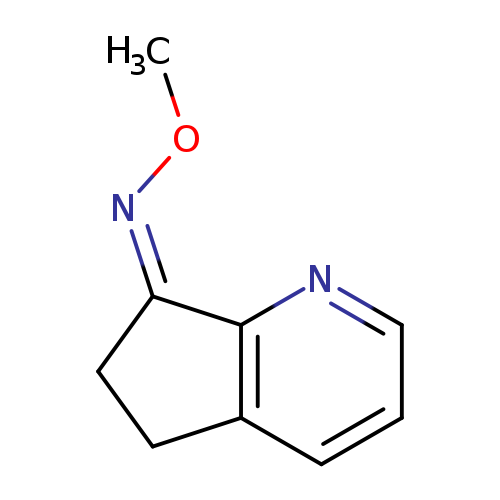
(Z)-5H-Cyclopenta[b]pyridin-7(6h)-one o-methyl oximeCatalog No.:AA0096KM CAS No.:1071727-78-0 MDL No.:MFCD28975314 MF:C9H10N2O MW:162.1885 |
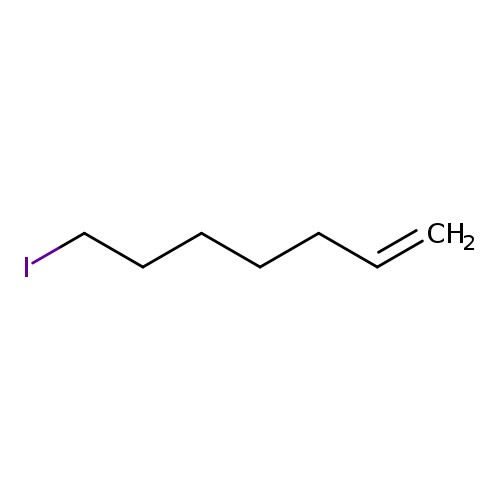
7-iodohept-1-eneCatalog No.:AA00KG7E CAS No.:107175-49-5 MDL No.:MFCD10570159 MF:C7H13I MW:224.0826 |
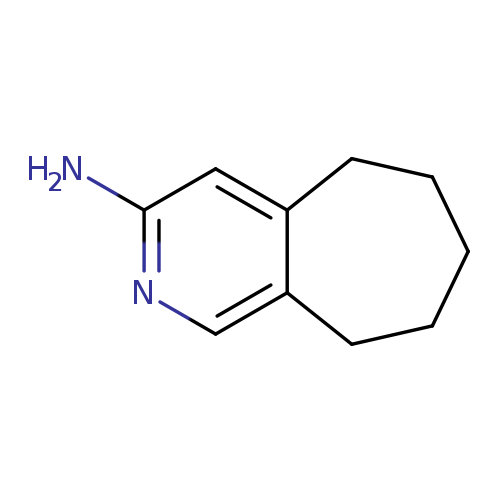
5H,6H,7H,8H,9H-cyclohepta[c]pyridin-3-amineCatalog No.:AA01DQQ9 CAS No.:1071763-80-8 MDL No.:MFCD22550643 MF:C10H14N2 MW:162.2316 |
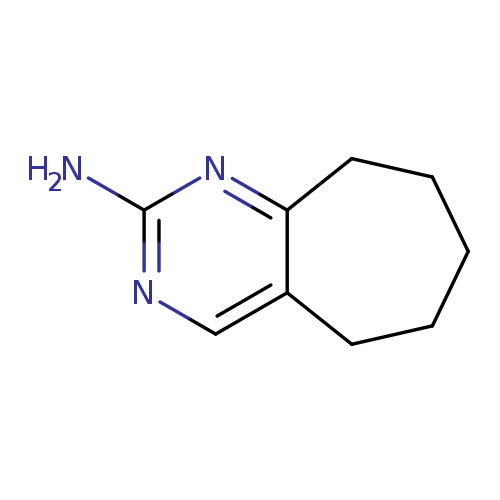
6,7,8,9-Tetrahydro-5h-cyclohepta[d]pyrimidin-2-amineCatalog No.:AA00HAV3 CAS No.:1071763-85-3 MDL No.:MFCD18361690 MF:C9H13N3 MW:163.2196 |
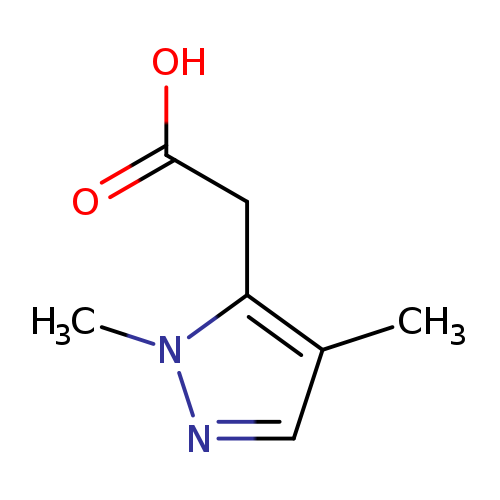
2-(1,4-dimethyl-1H-pyrazol-5-yl)acetic acidCatalog No.:AA01EK33 CAS No.:1071814-38-4 MDL No.:MFCD20660111 MF:C7H10N2O2 MW:154.1665 |
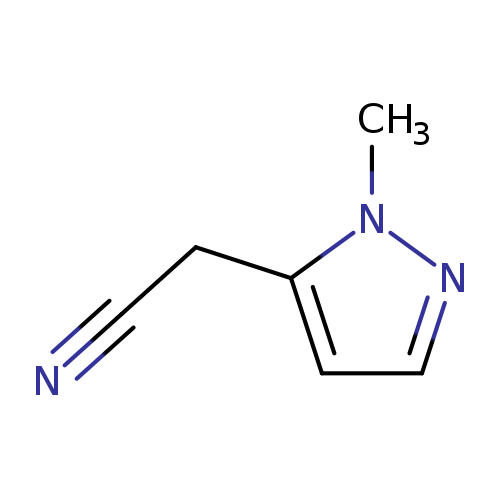
(1-Methyl-1h-pyrazol-5-yl)acetonitrileCatalog No.:AA00397V CAS No.:1071814-43-1 MDL No.:MFCD09859321 MF:C6H7N3 MW:121.1399 |
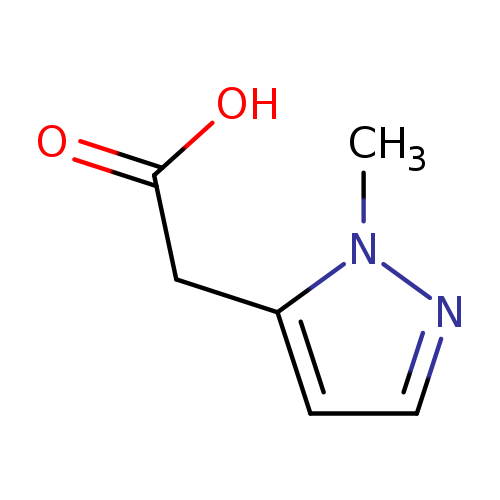
2-(1-Methyl-1h-pyrazol-5-yl)acetic acidCatalog No.:AA0096FO CAS No.:1071814-44-2 MDL No.:MFCD18262290 MF:C6H8N2O2 MW:140.1399 |
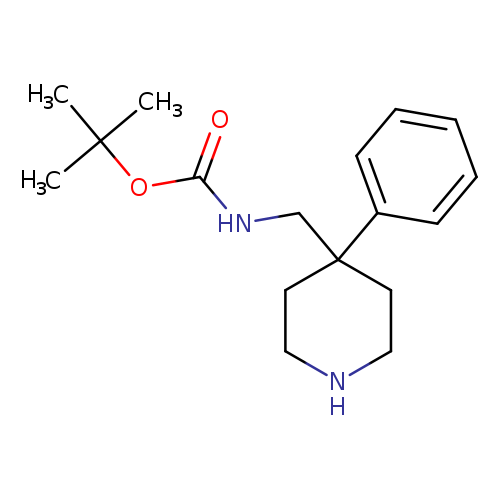
(4-Phenyl-piperidin-4-ylmethyl)-carbamic acid tert-butyl esterCatalog No.:AA00HAV4 CAS No.:1071866-01-7 MDL No.:MFCD09753905 MF:C17H26N2O2 MW:290.4005 |
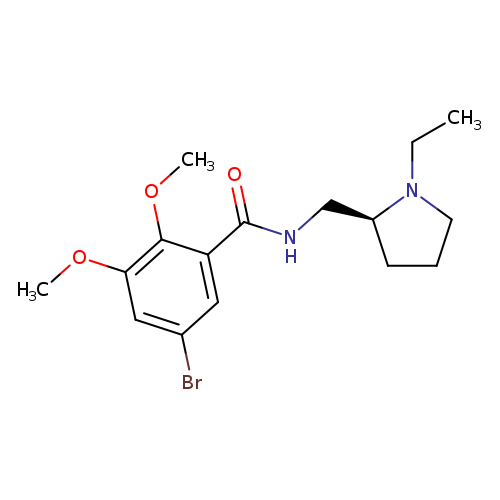
(S)-5-Bromo-N-((1-ethylpyrrolidin-2-yl)methyl)-2,3-dimethoxybenzamideCatalog No.:AA007T64 CAS No.:107188-74-9 MDL No.:MFCD00894744 MF:C16H23BrN2O3 MW:371.2694 |
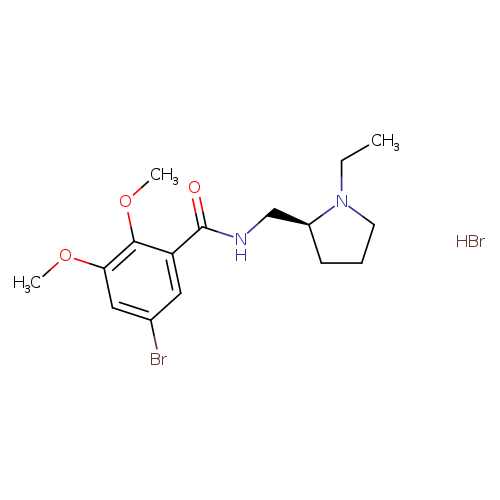
FLB 457 HydrobromideCatalog No.:AA007ARR CAS No.:107188-92-1 MDL No.:MFCD18252334 MF:C16H24Br2N2O3 MW:452.1814 |
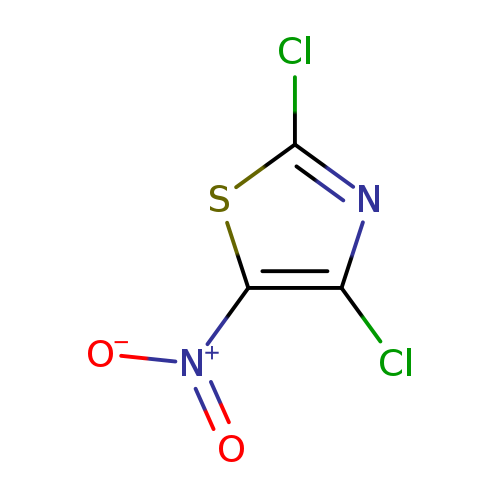
2,4-Dichloro-5-nitrothiazoleCatalog No.:AA009TAV CAS No.:107190-42-1 MDL No.:MFCD11131237 MF:C3Cl2N2O2S MW:199.0153 |
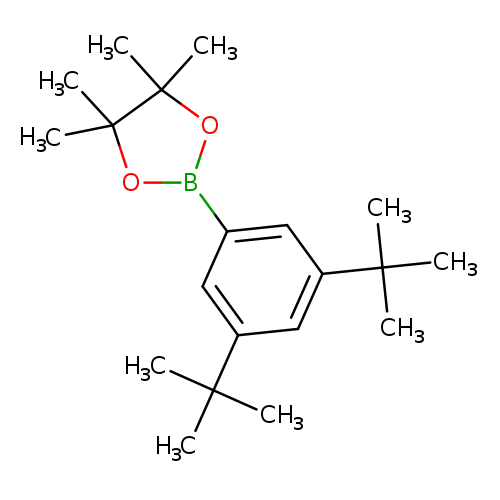
2-(3,5-Di-tert-butylphenyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolaneCatalog No.:AA0094NL CAS No.:1071924-13-4 MDL No.:MFCD24039797 MF:C20H33BO2 MW:316.2858 |
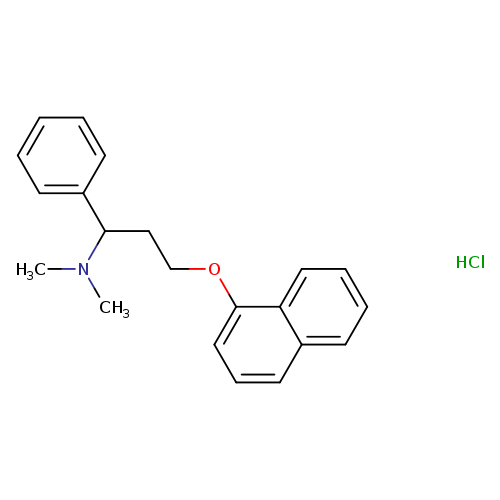
Dl-dapoxetine hclCatalog No.:AA008XLC CAS No.:1071929-03-7 MDL No.:MFCD20922882 MF:C21H24ClNO MW:341.8744 |
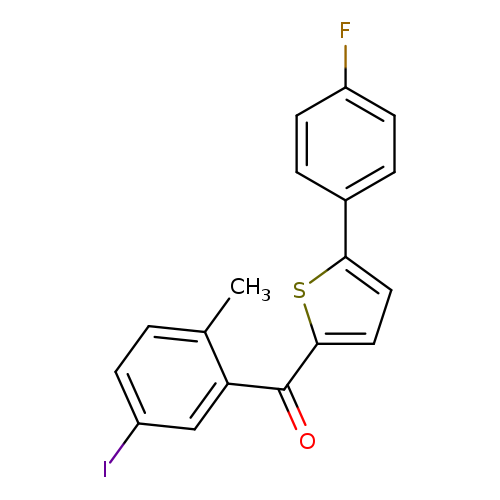
(5-(4-Fluorophenyl)thiophen-2-yl)(5-iodo-2-methylphenyl)methanoneCatalog No.:AA00HAV5 CAS No.:1071929-08-2 MDL No.:MFCD12405594 MF:C18H12FIOS MW:422.2552 |
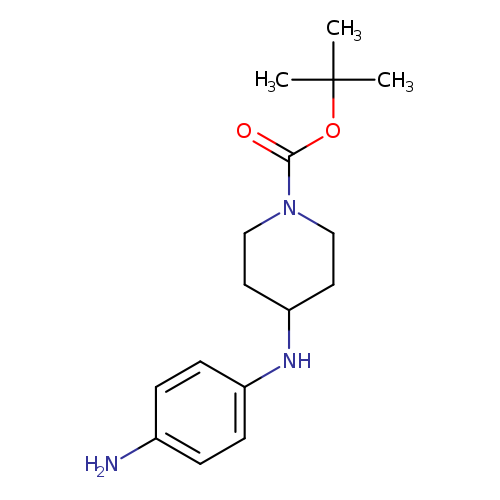
4-(4-Amino-phenylamino)-piperidine-1-carboxylic acid tert-butyl esterCatalog No.:AA007T63 CAS No.:1071932-29-0 MDL No.:MFCD12026437 MF:C16H25N3O2 MW:291.3886 |
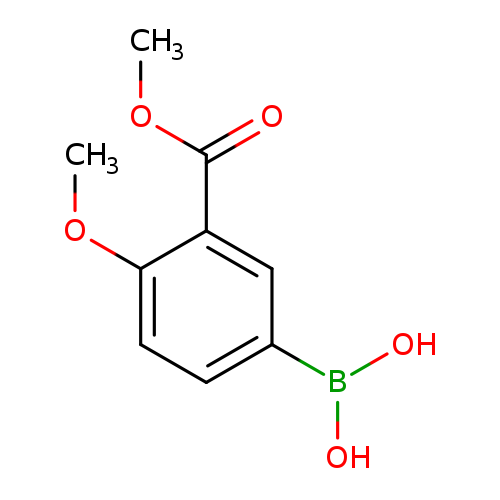
4-methoxy-3-(methoxycarbonyl)phenylboronic acidCatalog No.:AA00HAV6 CAS No.:1071958-96-7 MDL No.:MFCD23380361 MF:C9H11BO5 MW:209.9916 |
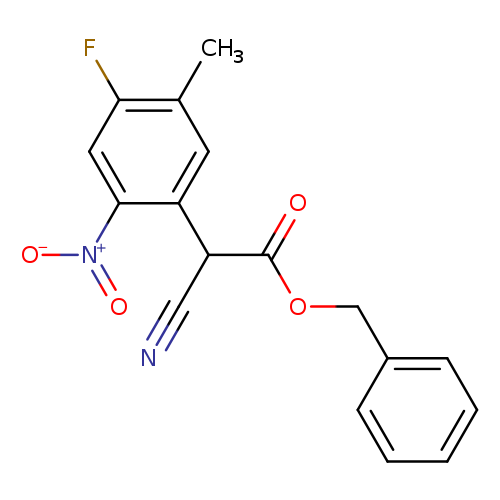
Benzyl 2-cyano-2-(4-fluoro-5-methyl-2-nitrophenyl)acetateCatalog No.:AA0096L1 CAS No.:1071974-01-0 MDL No.:MFCD28369447 MF:C17H13FN2O4 MW:328.2945 |
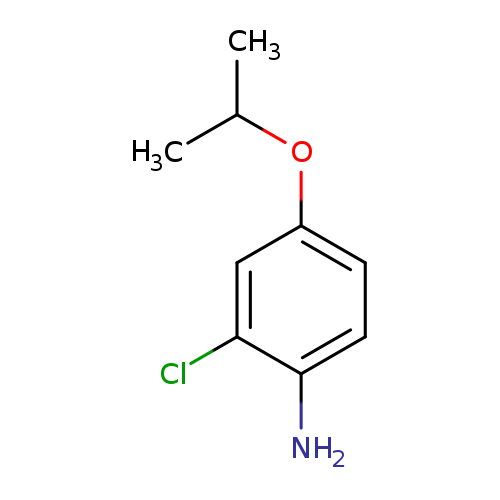
2-chloro-4-(propan-2-yloxy)anilineCatalog No.:AA01AG32 CAS No.:1071989-24-6 MDL No.:MFCD16680491 MF:C9H12ClNO MW:185.6507 |
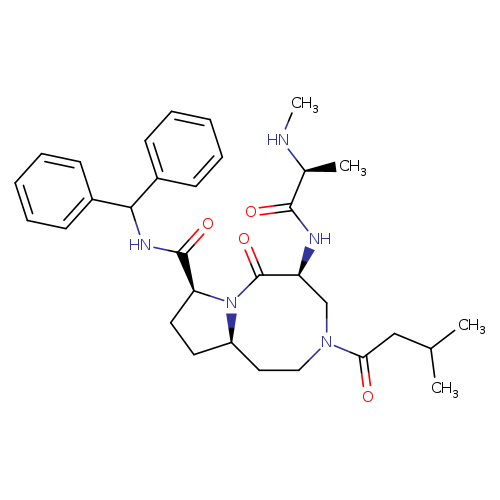
AT406Catalog No.:AA008WG6 CAS No.:1071992-99-8 MDL No.:MFCD22124467 MF:C32H43N5O4 MW:561.7149 |
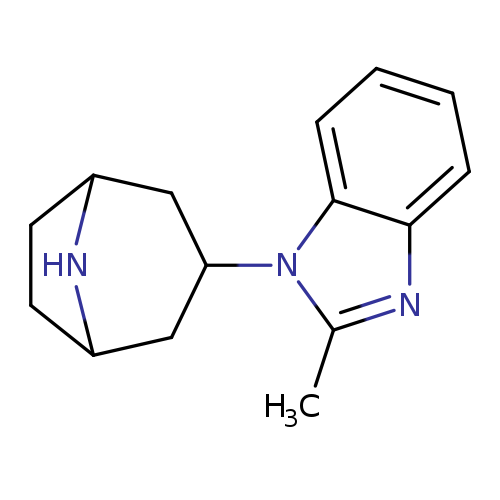
1-(8-Azabicyclo[3.2.1]oct-3-yl)-2-methyl-1h-benzimidazoleCatalog No.:AA00906Q CAS No.:1071993-26-4 MDL No.:MFCD08064258 MF:C15H19N3 MW:241.3315 |
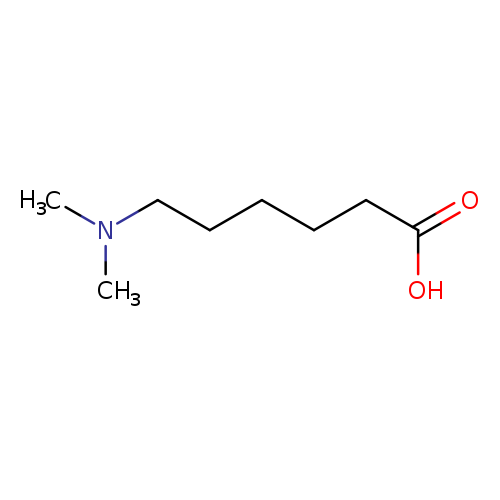
6-(dimethylamino)hexanoic acidCatalog No.:AA01EK2X CAS No.:1072-09-9 MDL No.:MFCD20318750 MF:C8H17NO2 MW:159.2261 |
Silane, hexyl-Catalog No.:AA007T5T CAS No.:1072-14-6 MDL No.:MFCD00027291 MF:C6H16Si MW:116.2767 |