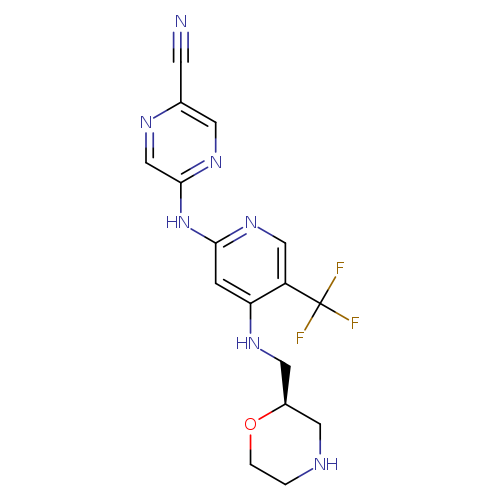
[1]CurrentPatentAssignee:CANCERRESEARCHUK-WO2013/171470,2013,A1
[2]Osborne,JamesD.;Matthews,ThomasP.;McHardy,Tatiana;Proisy,Nicolas;Cheung,Kwai-MingJ.;Lainchbury,Michael;Brown,Nathan;Walton,MichaelI.;Eve,PaulD.;Boxall,KatherineJ.;Hayes,Angela;Henley,AlanT.;Valenti,MelanieR.;DeHavenBrandon,AlexisK.;Box,Gary;Jamin,Yann;Robinson,SimonP.;Westwood,IsaacM.;VanMontfort,RobL.M.;Leonard,PhilipM.;Lamers,MariekeB.A.C.;Reader,JohnC.;Aherne,G.Wynne;Raynaud,FlorenceI.;Eccles,SuzanneA.;Garrett,MichelleD.;Collins,Ian[JournalofMedicinalChemistry,2016,vol.59,#11,p.5221-5237]

[1]Patent:WO2013/171470,2013,A1
 113305-94-5
113305-94-5

[1]Patent:WO2013/171470,2013,A1
[1]CurrentPatentAssignee:CANCERRESEARCHUK-WO2013/171470,2013,A1Locationinpatent:Page/Pagecolumn42-43
[2]Osborne,JamesD.;Matthews,ThomasP.;McHardy,Tatiana;Proisy,Nicolas;Cheung,Kwai-MingJ.;Lainchbury,Michael;Brown,Nathan;Walton,MichaelI.;Eve,PaulD.;Boxall,KatherineJ.;Hayes,Angela;Henley,AlanT.;Valenti,MelanieR.;DeHavenBrandon,AlexisK.;Box,Gary;Jamin,Yann;Robinson,SimonP.;Westwood,IsaacM.;VanMontfort,RobL.M.;Leonard,PhilipM.;Lamers,MariekeB.A.C.;Reader,JohnC.;Aherne,G.Wynne;Raynaud,FlorenceI.;Eccles,SuzanneA.;Garrett,MichelleD.;Collins,Ian[JournalofMedicinalChemistry,2016,vol.59,#11,p.5221-5237]
[3]CurrentPatentAssignee:GLAXOSMITHKLINEPLC-WO2021/207210,2021,A1Locationinpatent:Paragraph00247;00252;00254-00256
[1]Osborne,JamesD.;Matthews,ThomasP.;McHardy,Tatiana;Proisy,Nicolas;Cheung,Kwai-MingJ.;Lainchbury,Michael;Brown,Nathan;Walton,MichaelI.;Eve,PaulD.;Boxall,KatherineJ.;Hayes,Angela;Henley,AlanT.;Valenti,MelanieR.;DeHavenBrandon,AlexisK.;Box,Gary;Jamin,Yann;Robinson,SimonP.;Westwood,IsaacM.;VanMontfort,RobL.M.;Leonard,PhilipM.;Lamers,MariekeB.A.C.;Reader,JohnC.;Aherne,G.Wynne;Raynaud,FlorenceI.;Eccles,SuzanneA.;Garrett,MichelleD.;Collins,Ian[JournalofMedicinalChemistry,2016,vol.59,#11,p.5221-5237]
Title: Multiparameter Lead Optimization to Give an Oral Checkpoint Kinase 1 (CHK1) Inhibitor Clinical Candidate: (R)-5-((4-((Morpholin-2-ylmethyl)amino)-5-(trifluoromethyl)pyridin-2-yl)amino)pyrazine-2-carbonitrile (CCT245737).
Journal: Journal of medicinal chemistry 20160609
Title: The clinical development candidate CCT245737 is an orally active CHK1 inhibitor with preclinical activity in RAS mutant NSCLC and Eµ-MYC driven B-cell lymphoma.
Journal: Oncotarget 20160119
Title: Osborne JD, et al. Multiparameter Lead Optimization to Give an Oral Checkpoint Kinase 1 (CHK1) Inhibitor Clinical Candidate: (R)-5-((4-((Morpholin-2-ylmethyl)amino)-5-(trifluoromethyl)pyridin-2-yl)amino)pyrazine-2-carbonitrile (CCT245737). J Med Chem. 2016 Jun 9;59(11):5221-37.
Title: Walton MI, et al. The clinical development candidate CCT245737 is an orally active CHK1 inhibitor with preclinical activity in RAS mutant NSCLC and Eμ-MYC driven B-cell lymphoma.