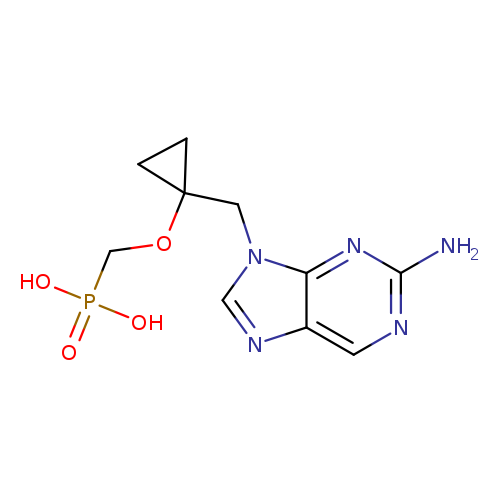
[1]CurrentPatentAssignee:SICHUANKELUNPHARMACEUTICALCOMPANYLIMITED-CN106432330,2017,ALocationinpatent:Paragraph0219;0220;0221;0222
[2]CurrentPatentAssignee:GUANGZHOUYUEMEIMEDICINETECH-CN108997429,2018,ALocationinpatent:Paragraph0060;'0061
[1]CurrentPatentAssignee:HAISCOPHARMACEUTICALGROUPCO.,LTD.-CN106977548,2017,ALocationinpatent:Paragraph0082;0083;0084
[1]Patent:CN106977548,2017,A.Locationinpatent:Paragraph0066;0072
[1]CurrentPatentAssignee:HAISCOPHARMACEUTICALGROUPCO.,LTD.-CN106977548,2017,ALocationinpatent:Paragraph0066;0072
[1]CurrentPatentAssignee:HAISCOPHARMACEUTICALGROUPCO.,LTD.-CN106977548,2017,ALocationinpatent:Paragraph0066;0072
Title: Jung JA, et al. Pharmacokinetic comparison of the maleate and free base formulations of LB80380, a novel nucleotide analog, in healthy male volunteers. Int J Clin Pharmacol Ther. 2012 Sep;50(9):657-64.
Title: Yuen MF, et al. A randomized placebo-controlled, dose-finding study of oral LB80380 in HBeAg-positive patients with chronic hepatitis B. Antivir Ther. 2006;11(8):977-83.