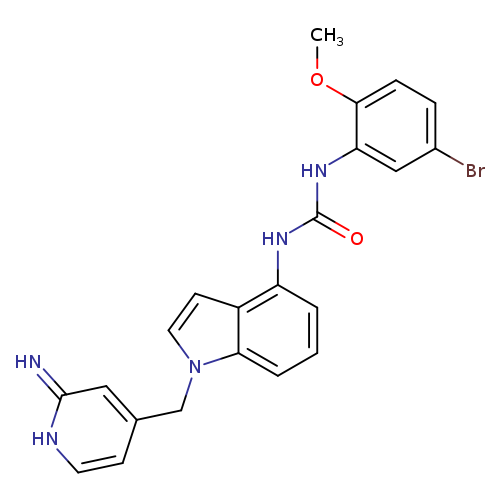
Title: A strategy for extending the applicability of a validated plasma calibration curve to quantitative measurements in multiple tissue homogenate samples: a case study from a rat tissue distribution study of JI-101, a triple kinase inhibitor.
Journal: Biomedical chromatography : BMC 20120401
Title: Highly sensitive method for the determination of JI-101, a multi-kinase inhibitor in human plasma and urine by LC-MS/MS-ESI: method validation and application to a clinical pharmacokinetic study.
Journal: Biomedical chromatography : BMC 20120201
Title: Pharmacokinetics, tissue distribution and identification of putative metabolites of JI-101 - a novel triple kinase inhibitor in rats.
Journal: Arzneimittel-Forschung 20120101
Title: Highly sensitive method for the determination of a novel triple kinase inhibitor with anti-cancer activity, JI-101, in rat plasma by liquid chromatography-electrospray ionization tandem mass spectrometry: application to a pharmacokinetic study.
Journal: Biomedical chromatography : BMC 20110701
Title: Gurav SD, et al. Pharmacokinetics, tissue distribution and identification of putative metabolites of JI-101 - a novel triple kinase inhibitor in rats. Arzneimittelforschung. 2012 Jan;62(1):27-34.