2019-11-26 08:44:55
Wafaa S. Hamama 1, Moged A. Berghot 1, Eman A. Baz 1,2, Essam H. A. Hanashalshahaby 3,4 and Moustafa A. Gouda
Department of Chemistry, Faculty of Science, Mansoura University, Mansoura, 35516, Egypt
Department of Chemistry, Faculty of Science, Jazan University, Jazan, Kingdom of Saudi Arabia
Department of Chemistry, Faculty of Education, Sana'a University, Sana'a, Yemen
Department of Pharmacognosy, Faculty of Pharmacy, Ankara University, Ankara, Turkey
Department of Chemistry, Faculty of Science and Arts, Ulla, Taibah University, KSA
1. Introduction
Coumarins (II) are the simple compounds (I-V) belonging to a large class of molecules known as benzopyrones.1 Furthermore, coumarins and their derivatives form an elite class of compounds, occupying an important place in the realm of natural products and synthetic organic chemistry.1 They are widely used as additives in food, perfumes, cosmetics,2 pharmaceuticals, optical brighteners3(e.g. 7-diethylamino-4-methylcoumarin),4 dispersed fluorescent laser dyes,5 antithrombotic and anticoagulants6 (e.g. acenocoumarol),4 and in treatment of bronchial asthma (e.g. intal)4 and cancer.7
Also, coumarin derivatives are novel lipid-lowering agents, possessing moderate triglyceride lowering activity.8 Many coumarin derivatives can scavenge reactive oxygen species such as hydroxyl free radicals, superoxide radicals or hypochlorous acid to prevent free radical injury.9 While certain coumarin derivatives function as human immunodeficiency virus integrase inhibitors and are used in treatment of HIV infection,10 the others are used as anti-invasive compounds against some serine proteases and matrix metalloproteases (MMPs).11 Moreover, 6-nitro-7-hydroxycoumarin acts as a selective anti-proliferative agent.12, 13 Two naturally occurring coumarins have been isolated and shown to inhibit the polymerization of tubulin and arrest cells in mitotic phase by inhibiting microtubule formation.14 These coumarins act synergistically in inhibiting KB (human epidermoid carcinoma) cell proliferation.14
Coumarin derivatives usually occur as secondary metabolites in seeds, roots and leaves of many plant species via shikimate pathway. Their function includes waste products, plant growth regulators, fungistats and bacteriostats.15 Anthocyanins16 and flavones,17 grouped together, are known as flavonoids,18 and make up many flower pigments. Also, flavone and coumarin19 derivatives have marked toxic and other physiological properties in animals, though they have no part in normal metabolism of animals. The isomeric 2-banzopyrylium20 system is not naturally occurring; only a few isocoumarin derivatives21 occurs as natural products and, therefore, much less work on these has been described. Our review deals with the effective use of 3-acyl(aroyl)coumarin derivatives 1 in the synthesis of
different polyfunctional heterocyclic compounds.
2. Reactivity
3-Acyl(aroyl)coumarins are difunctional compounds possessing electrophilic and nucleophilic properties. Typical nucleophilic position is C10. Furthermore, C9 of C=O and C4 could act as an electrophile. These chemical properties have been used to design different heterocyclic moieties with different ring sizes such as oxazole, pyrazole, thiophene, thiazole, pyridine, diazepine, benzoxocin, benzoxonin, benzoxepin and pyrimidine.
2.1. Bromination
Halogenation of 1 with bromine in chloroform afforded 3-bromoacetyl coumarin derivatives 2.
Separately, La Pietra et. al prepared 3-bromoacetylcoumarin derivatives 2 via treating compound 1 with CuBr2 in CHCl3 / CH3COOEt mixture. The reaction of 3-bromoacetylcoumarin derivatives 2 with the appropriate arylamine 3 (aniline, 3-aminobenzoic acid, ethyl 3-aminobenzoate) in ethanol in the presence of NaHCO3 yielded compounds 4. Derivatives 5a-h were then obtained by treatment of compounds 4 with a large excess of ammonium thiocyanate in acetic acid.
Kurt, B. Z. et al., 26 carried out the reaction of 3-(bromoacetyl)-2H-chromen-2-one 2 in the presence of ethanol with thiourea 6, which yielded 3-(2-amino-1,3-thiazol-4-yl)-2H-chromen-2-one 7. This was reacted with sodium cyanate 8 in the presence of glacial acetic acid to produce N-[4-(2-oxo-2Hchromen-3-yl)-1,3-thiazol-2-yl]urea 9. Treatment of compound 9 with hydrazine hydrate 10 produced N-[4-(2-oxo-2H-chromen-3-yl)-1,3-thiazol-2-yl]hydrazinecarboxamide 11, which was condensed with different aromatic/ heteroaromatic aldehydes and ketones 12 to form (1E)-1-arylalkane-1-one-N-[4-(2-oxo-2Hchromen-2-yl)-1,3-thiazol-2-yl]semicarbazones 13a-w.
A series of coumarylthiazole derivatives containing arylurea/thiourea groups 17 and 18, respectively, were obtained by the reactions of 2 with thiourea 6, which was followed by treatment of the formed aminothiazole 7 with arylisocyanates 14 in THF and arylisothiocyanates 15 in DMF, respectively.
Razi, et. al. prepared several thiazolylamine derivatives 19 by treating of compound 7 with the corresponding 2-hydroxy-3-methoxybenzaldehyde 18 in a basic ethanol solution. The Pd(II) and Pt(II) complexes 20 were synthesized by complexation of thiazolylamine derivatives 19 with Pd(II) and Pt(II), respectively.
The reaction of 2 with P(OR)3 in refluxing toluene in the presence of p-toluene sulfonic acid was completely different to that of acetic acid,52 giving new 1,4-addition products 90 along with the expected
enol phosphates 83.
Furthermore, when 3-bromoacetylcoumarin 2 was reacted with dialkylphosphites in refluxing toluene, a complicated reaction mixture was obtained, i.e. 3-acetylcoumarin 1 (2-5 %), enol phosphates 83 (~ 20%) and 84, which are the products of 1,4-additions of dialkylphosphites to 3-acetylcoumarin 1. Compound 90 formed via the following mechanism.
2.2. Reactions of trialkylphosphites
Reactions of trialkylphosphites 82 with 3-acetylcoumarin 1 as well as with 3-benzoylcoumarin 91 and 3-ethoxycarbonylcoumarin 92, in the presence of p-toluenesulfonic acid under ultrasound irradiation gave the corresponding 4-dialkylphosphono-2-oxocoumarin derivatives 84/ 85, 93/ 94 and 95,respectively, in 60 to 95% yields.
2.3.Chlorosulfonation
3-Acetyl-8-methoxycoumarin 1 was subjected to chlorosulfonation reaction using excess chlorosulfonic acid 96 to give the corresponding 8-hydroxy-2-oxo-2H-chromen-3,5,7-trisulfonamide 97.
2.4. Reduction
3-Acetylcoumarin 1 was agitated with palladium-charcoal and hydrogen at 60 Ibs./ sq. at room temperature to give an excellent yield of 3-acetyl-3,4-dihydrocoumarin as a keto-enol mixture 98. The keto form was isolated giving a negative ferric reaction, while the enol form was obtained as a sole product via acetylation of the mixture 98 to give the acetate 99. Acetylation of 3-acetylcoumarin 1 with acetic acid and acetic anhydride produced the corresponding 3-acetylcoumarin derivative 100 (Scheme 19). Liu et al. reported the selective reduction of endocyclic double bond of the 3-substituted coumarin
derivatives 1 by using Hantzsch 1,4-dihydropyridine (HEH) 101 as a reducing agent, which yielded 3,4-dihydrocoumarin derivatives 102 (Scheme 19). 56 Chemo-selective reduction of the endocyclic double
bond in 3-substituted coumarin derivatives 1 took place by o-phenylenediamine and benzaldehyde to generate in situ 2-phenyl benzimidazoline.57 Reduction of 3-acetyl and 3-benzoyl coumarin derivatives 1 occurred with sodium borohydride in alcohol to give the corresponding ethyl-2-(2-hydroxy benzyl)-3-oxo(butanoate) and 3-phenyl propanoate 105.
2.5. Photoreduction
Photo reduction of 3-acetylcoumarin 1 in i-propyl alcohol 106 gave the dihydro dimer 3,3'-diacetyl-4,4'-bichroman-2,2'-dione 107. Cyclobutanes 109 were formed by [2+2] cycloaddition of cyclohexene 108 with 3-acetylcoumarin 1 upon UV irradiation in benzene (Scheme 20). 59 Furthermore, photo [2+2] cycloaddition of olefins 110 with 3-acetylcoumarin 1 gave 1-exo-substituted 1,2,2a,8b-tetrahydro-3Hbenzo[b]cyclobuta[d]pyran-3-one derivatives 111. Endo-substituted 1,2,2a,8b-tetrahydro-3Hbenzo[b]cyclobuta[d]pyran-3-one derivatives 114 were prepared by photo [2+2] cycloaddition of 3- acetylcoumarin 1 with acetylenes 112, which was followed by hydrogenation of the formed 2a,8bdihydro-3H-benzo[b]cyclobuta[d]pyran-3-one derivatives 113 over Pd-C.
Treatment of 3-acetylcoumarin 1 with dimethylformamide in the presence of phosphorus oxychloride or HClO4 yielded chloropropeniminium salts 115 and aldehydes 116, respectively.
2.6. Mannich reaction
Mannich base 119 of 3-acetylcoumarin 1 was prepared via condensation of the corresponding acetylcoumarin 1 with paraformaldehyde 117 and dimethylamine 118 in the presence of conc. HCl. The reaction of 119 with substituted phenacylpyridiniumbromide salts 120 in the presence of ammonium acetate in refluxing acetic acid gave the corresponding 3-(6-arylpyridin-2-yl) 121 in moderate to good yields.
2.7. Reaction with hydrazine hydrate and its derivatives
It was reported that the reaction of hydrazine hydrate 10 with 3-acetylcoumarin 1 led to fission of the coumarin ring giving salicaldazine 122. On the other hand, refluxing of 3-acetylcoumarin 1 with phenylhydrazine 123a 64 or (2,4,6-trichlorophenyl)hydrazine 123b 65 in ethanol gave the corresponding hydrazones 124 and 125, respectively. Oxidation of 125 with tertbutylhypochlorite yielded chloroalkylazo 126. When 126 was treated with antimonypentachloride at 60 °C in dichloromethane, an orange precipitate 127 was formed. On addition of acetonitrile at room temperature 1H-triazolium salt 131 was afforded in 71% yield. The formation of 131 is assumed to take place via the formation of non-isolable acyclic intermediate 129, followed by cyclization to afford the non-isolable tiazole 130. This underwent Wagner-Meerwein type [1,2] shift of a methyl group to furnish the 1Htriazolium salts 131.
Microwave irradiation of 3-hydrazinylquinoxalin-2(1H)-one 132 and 6-substituted acetylcoumarin 1 in dry DMF at 400 W for 1 min afforded the corresponding hydrazones 133. Furthermore, treatment of substituted acetylcoumarins 1 with phenylhydrazine 124a gave the hydrazone 134. Fischer indole synthesis of 134 in the presence of Eaton’s reagent produced substituted 3-(1HIndol-2-yl)chromen-2-ones 136. Compounds 136 were allowed to undergo benzylation with beznyl chloride 137 and Vilsmeyer–Haack formylation to yield substituted 3-(1-benzyl-1H-indol-2-yl)-2Hchromen-2-ones 138 and 2(2-oxo-2H-chromen-3-yl)-1H-indole-3-carbaldehydes 139. Oxidation of 139 in the presence of potassium permanganate afforded 2-(2-oxo-2H-chromen-3-yl)-1H-indole-3-carboxylic acids 140.
Sixteen novel fluoro-substituted coumarin hydrazones were synthesized from a series of ethyl 2-hydroxy-2-(trifluoromethyl)-2H-chromene-3-carboxylates, using supported acid catalyst under microwave-assisted one-pot and solvent free conditions. The reaction was carried out in two steps under solid acid and microwave conditions. In step1, fluoro-substituted coumarin esters 139 were transformed into fluoro-substituted coumarin ketones 140. Then, the ketones, 140, which were not isolated from the mixture, were directly reacted with arylhydrazine to give the hydrazones 142.
3-[(IE)-2-aza-I-methyl-2-[(methylthiothioxomethyl]vinyl]-2H-chromen- 2-one 144, which prepared through the reaction of 3-acetylcoumarin 1 with methyl hydrazinecarbodithioate 143 in 2-propenol, was reacted with 145 to afford ethyl 2-[(2Z)-1,2-diaza-3-(2-oxo(2H-chromen-3-yl)but-2-enylidene]-3-phenyll,3,4-tbiadiazoline-5-carboxylate 146.
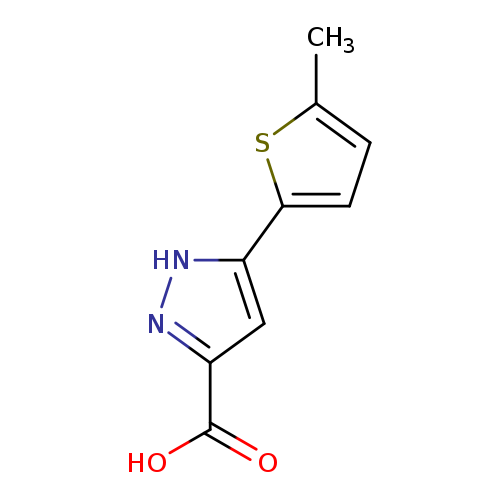
3-(5-Methylthiophen-2-yl)-1H-pyrazole-5-carboxylic acidCatalog No.:AA00J0YN CAS No.:1025010-00-7 MDL No.:MFCD03030191 MF:C9H8N2O2S MW:208.2370 |
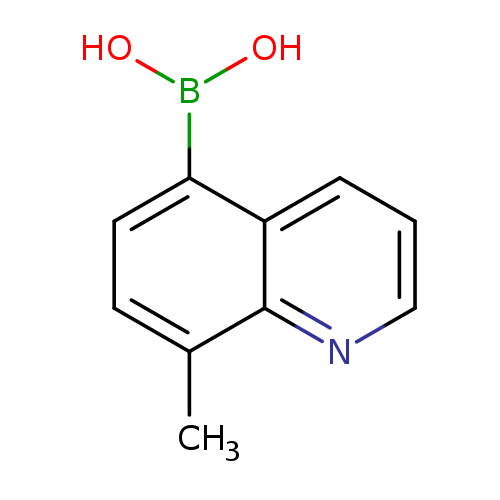
8-Methylquinoline-5-boronic acidCatalog No.:AA0007IE CAS No.:1025010-58-5 MDL No.:MFCD13193789 MF:C10H10BNO2 MW:187.0029 |
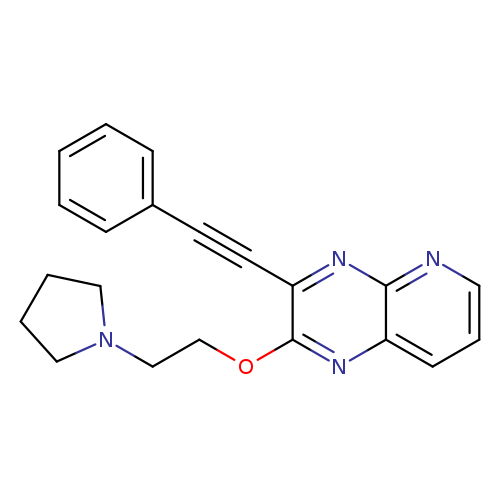
GK921Catalog No.:AA008TDO CAS No.:1025015-40-0 MDL No.:MFCD28167818 MF:C21H20N4O MW:344.4097 |
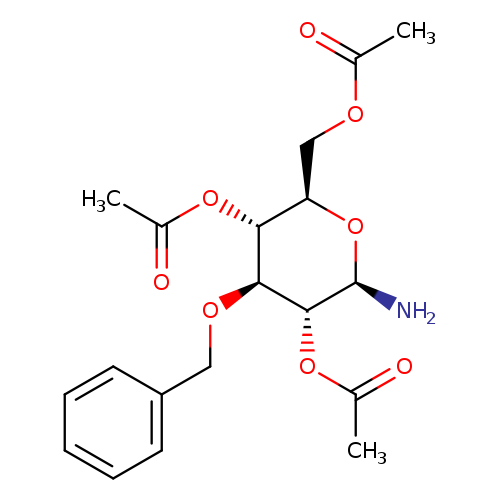
(2R,3R,4S,5R,6R)-2-(Acetoxymethyl)-6-amino-4-(benzyloxy)tetrahydro-2h-pyran-3,5-diyl diacetateCatalog No.:AA01DG36 CAS No.:1025019-40-2 MDL No.:MFCD31803746 MF:C19H25NO8 MW:395.4037 |
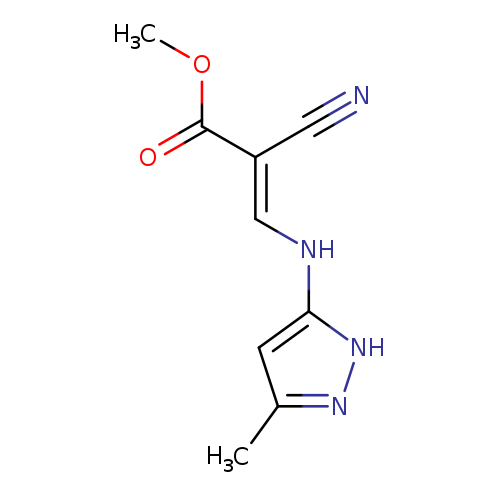
methyl (2E)-2-cyano-3-[(3-methyl-1H-pyrazol-5-yl)amino]prop-2-enoateCatalog No.:AA00IQIN CAS No.:1025019-81-1 MDL No.:MFCD03012800 MF:C9H10N4O2 MW:206.2013 |
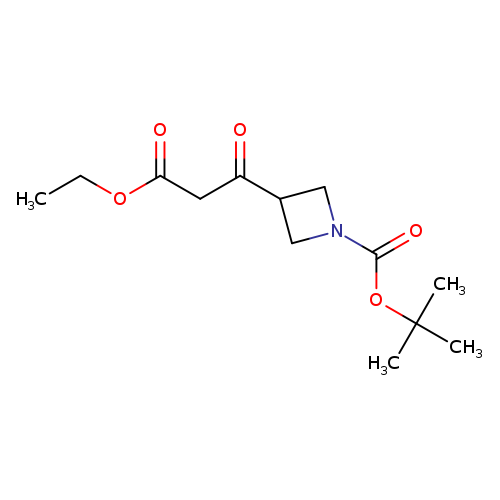
1-Boc-3-(3-ethoxy-3-oxopropanoyl)azetidineCatalog No.:AA0007J5 CAS No.:1025029-60-0 MDL No.:MFCD11974391 MF:C13H21NO5 MW:271.3095 |
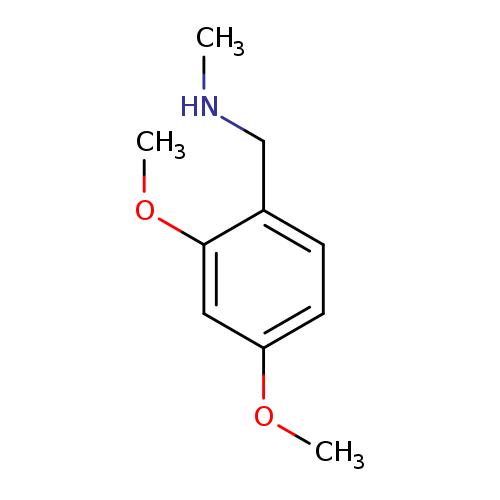
N-(2,4-Dimethoxybenzyl)-N-methylamineCatalog No.:AA0007JP CAS No.:102503-23-1 MDL No.:MFCD04633426 MF:C10H15NO2 MW:181.2316 |
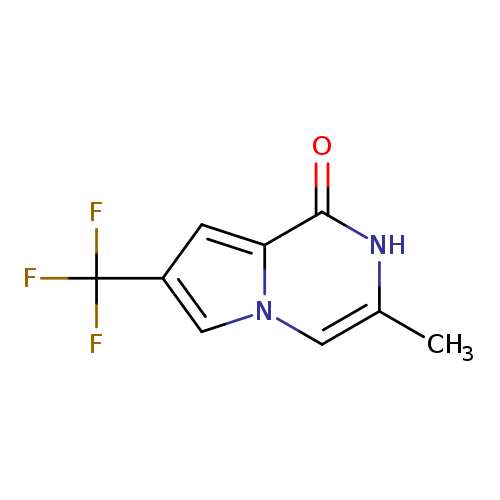
1,2-Dihydro-3-methyl-1-oxo-7-(trifluoromethyl)pyrrolo[1,2-a]pyrazineCatalog No.:AA00H9Q8 CAS No.:1025054-82-3 MDL No.:MFCD19982827 MF:C9H7F3N2O MW:216.1599 |
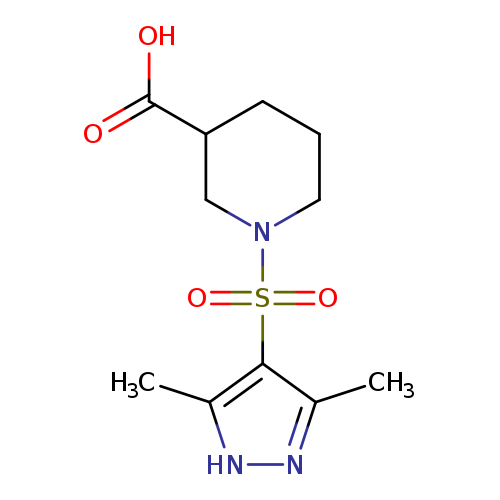
1-[(3,5-dimethyl-1H-pyrazol-4-yl)sulfonyl]piperidine-3-carboxylic acidCatalog No.:AA01EKHM CAS No.:1025058-85-8 MDL No.:MFCD07366391 MF:C11H17N3O4S MW:287.3354 |
1-CHLORO-4-[CHLORO(4-CHLOROPHENYL)METHYL]BENZENECatalog No.:AA008U4H CAS No.:1025058-88-1 MDL No.:MFCD08691091 MF:C11H14ClNO6S2 MW:355.8150 |
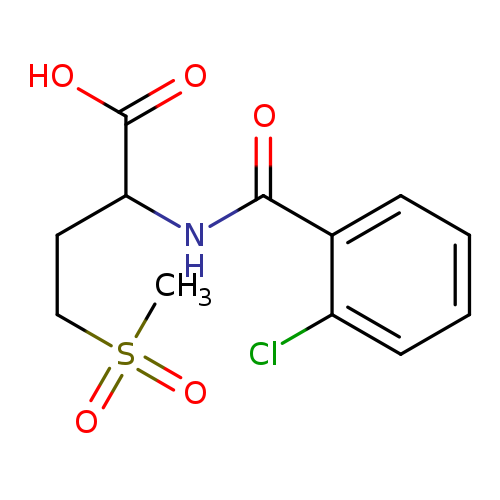
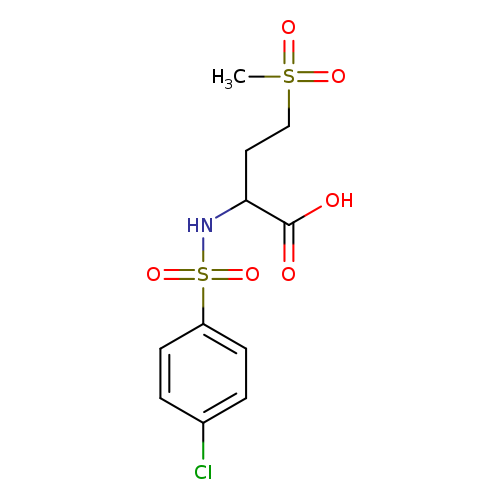
2-(2-Chlorobenzamido)-4-(methylsulfonyl)butanoic acidCatalog No.:AA008U0B CAS No.:1025058-89-2 MDL No.:MFCD08691092 MF:C12H14ClNO5S MW:319.7613 |
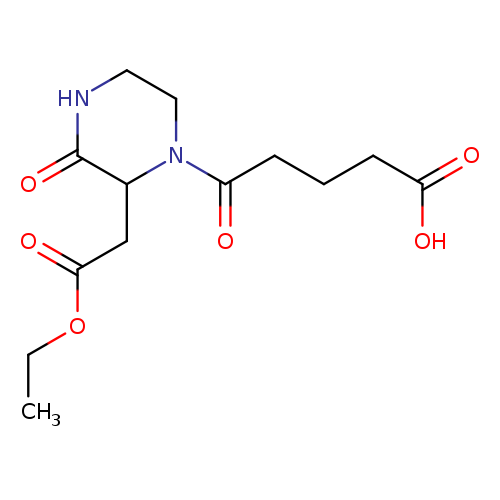
5-[2-(2-Ethoxy-2-oxoethyl)-3-oxo-1-piperazinyl]-5-oxopentanoic acidCatalog No.:AA01FO88 CAS No.:1025063-22-2 MDL No.: MF:C13H20N2O6 MW:300.3077 |
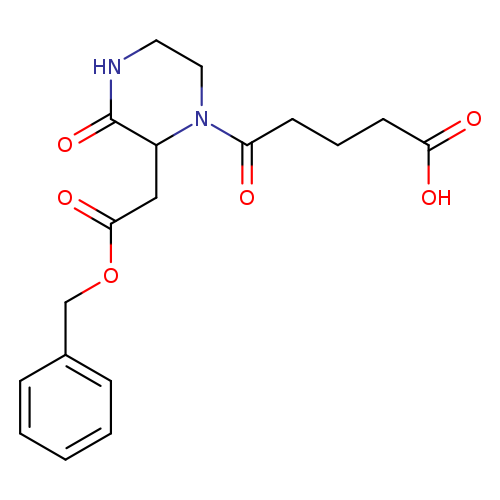
5-{2-[2-(Benzyloxy)-2-oxoethyl]-3-oxo-1-piperazinyl}-5-oxopentanoic acidCatalog No.:AA01FO85 CAS No.:1025063-23-3 MDL No.: MF:C18H22N2O6 MW:362.3771 |
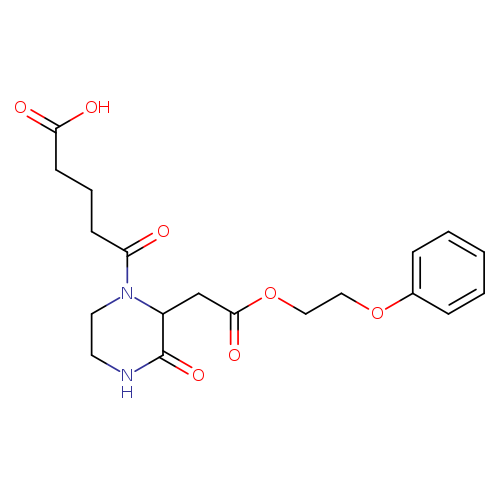
5-Oxo-5-{3-oxo-2-[2-oxo-2-(2-phenoxyethoxy)ethyl]-1-piperazinyl}pentanoic acidCatalog No.:AA01FO8D CAS No.:1025063-24-4 MDL No.: MF:C19H24N2O7 MW:392.4031 |
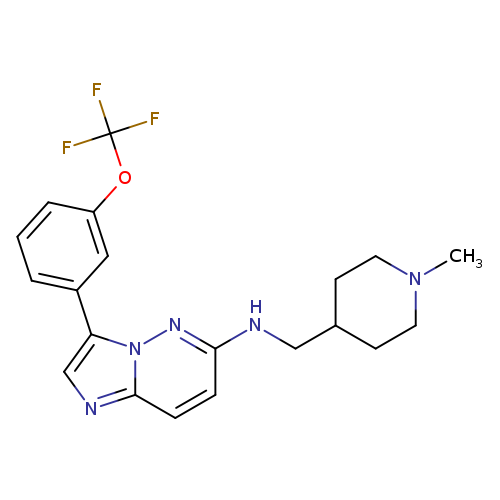
Sgi-1776Catalog No.:AA0007J1 CAS No.:1025065-69-3 MDL No.:MFCD16659064 MF:C20H22F3N5O MW:405.4168 |
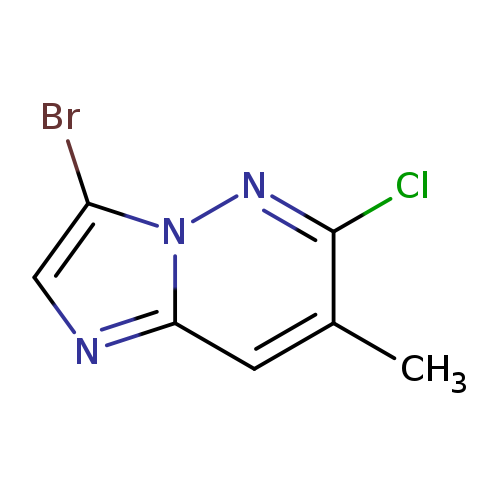
3-bromo-6-chloro-7-methylimidazo[1,2-b]pyridazineCatalog No.:AA01DTOO CAS No.:1025066-16-3 MDL No.:MFCD30471268 MF:C7H5BrClN3 MW:246.4917 |
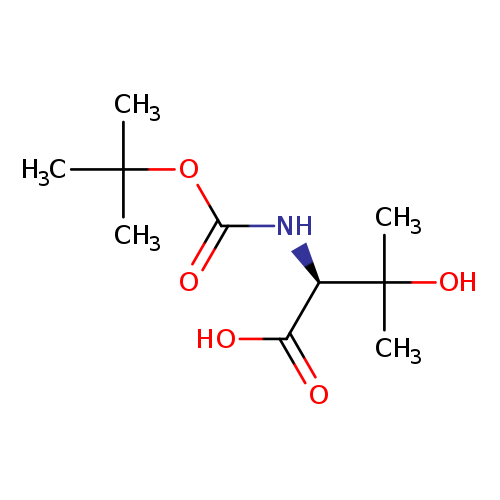
N-Boc-(s)-2-amino-3-hydroxy-3-methylbutanoic acidCatalog No.:AA0007JE CAS No.:102507-13-1 MDL No.:MFCD03094792 MF:C10H19NO5 MW:233.2616 |
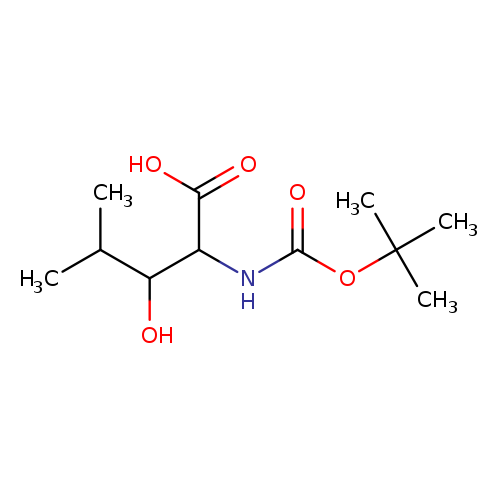
(2S,3S)-2-((TERT-BUTOXYCARBONYL)AMINO)-3-HYDROXY-4-METHYLPENTANOIC ACIDCatalog No.:AA01DLCZ CAS No.:102507-16-4 MDL No.:MFCD25541908 MF:C11H21NO5 MW:247.2881 |
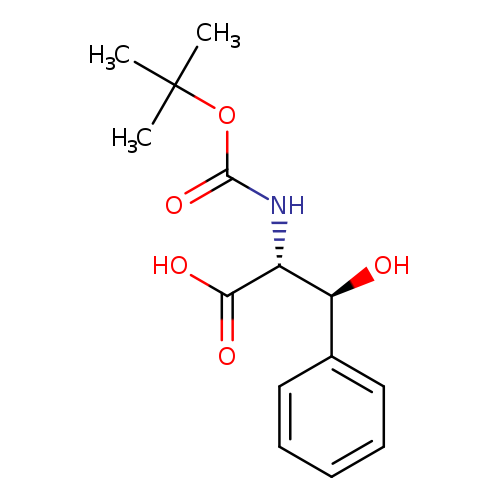
(2R, 3S)/(2S, 3R)-Racemic boc-beta-hydroxyphenylalanineCatalog No.:AA0007JD CAS No.:102507-18-6 MDL No.:MFCD06656444 MF:C14H19NO5 MW:281.3044 |
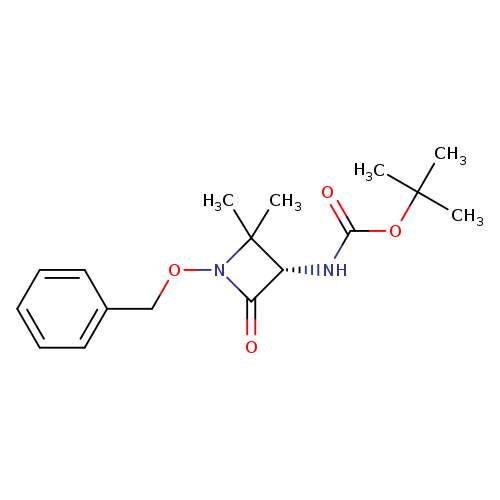
tert-butyl N-[(3S)-1-(benzyloxy)-2,2-dimethyl-4-oxoazetidin-3-yl]carbamateCatalog No.:AA01DEE1 CAS No.:102507-25-5 MDL No.:MFCD29483702 MF:C17H24N2O4 MW:320.3835 |
4-(4-bromophenyl)-2-{[3-(diethylamino)propyl]amino}-4-oxobutanoic acidCatalog No.:AA00IZL8 CAS No.:1025085-92-0 MDL No.:MFCD00954927 MF:C17H25BrN2O3 MW:385.2960 |
[1-Methyl-2-(5-methyl-1h-pyrazol-3-yl)ethyl]amineCatalog No.:AA008RUG CAS No.:1025087-55-1 MDL No.:MFCD05022480 MF:C7H13N3 MW:139.1982 |
4-Butyl-N-(diphenylphosphino)benzenesulfonamideCatalog No.:AA008VKR CAS No.:1025096-61-0 MDL No.:MFCD18802283 MF:C22H24NO2PS MW:397.4702 |
CadazolidCatalog No.:AA01EOPJ CAS No.:1025097-10-2 MDL No.:MFCD26961092 MF:C29H29F2N3O8 MW:585.5527 |
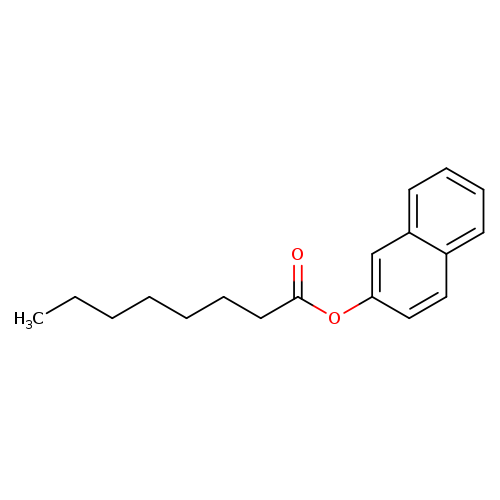
2-Naphthyl caprylateCatalog No.:AA0007KD CAS No.:10251-17-9 MDL No.:MFCD00046465 MF:C18H22O2 MW:270.3661 |
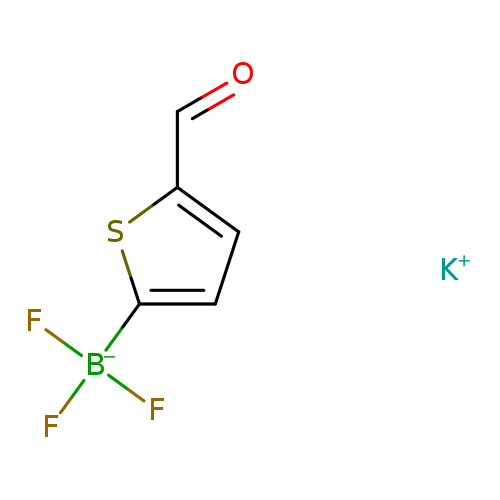
Potassium 5-formylthiophene-2-trifluoroborateCatalog No.:AA0007JR CAS No.:1025113-78-3 MDL No.:MFCD09800740 MF:C5H3BF3KOS MW:218.0462 |
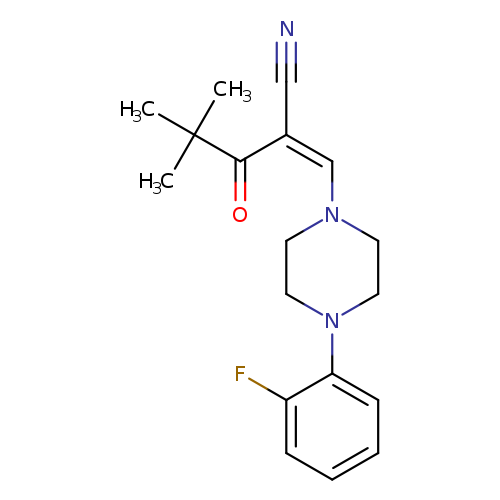
(2Z)-2-{[4-(2-fluorophenyl)piperazin-1-yl]methylidene}-4,4-dimethyl-3-oxopentanenitrileCatalog No.:AA00IZYH CAS No.:1025124-66-6 MDL No.:MFCD00245197 MF:C18H22FN3O MW:315.3852 |
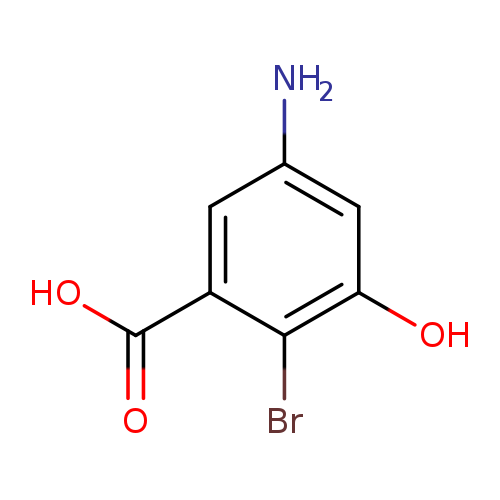
5-amino-2-bromo-3-hydroxybenzoic acidCatalog No.:AA01DSPB CAS No.:1025127-27-8 MDL No.:MFCD31559865 MF:C7H6BrNO3 MW:232.0314 |
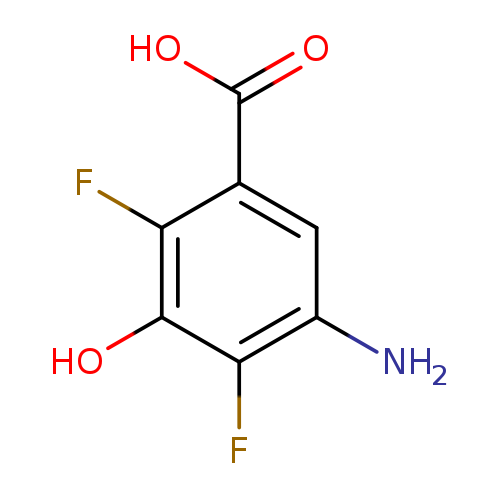
5-amino-2,4-difluoro-3-hydroxybenzoic acidCatalog No.:AA01DUUK CAS No.:1025127-35-8 MDL No.:MFCD31615801 MF:C7H5F2NO3 MW:189.1163 |
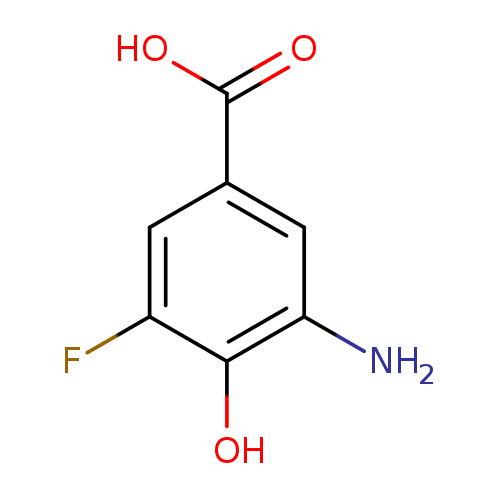
3-amino-5-fluoro-4-hydroxybenzoic acidCatalog No.:AA01C02Z CAS No.:1025127-44-9 MDL No.:MFCD28519246 MF:C7H6FNO3 MW:171.1258 |
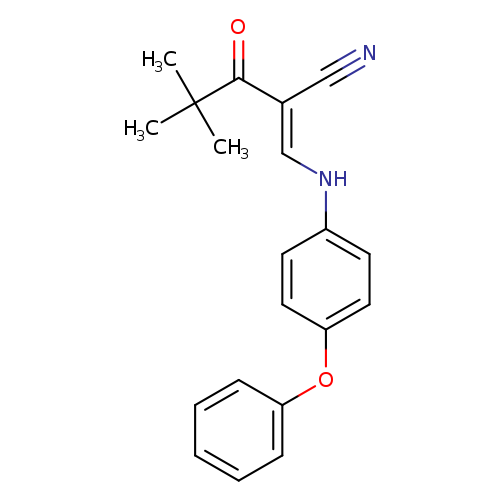
(2E)-4,4-dimethyl-3-oxo-2-{[(4-phenoxyphenyl)amino]methylidene}pentanenitrileCatalog No.:AA00IX6T CAS No.:1025129-86-5 MDL No.:MFCD03410415 MF:C20H20N2O2 MW:320.3850 |
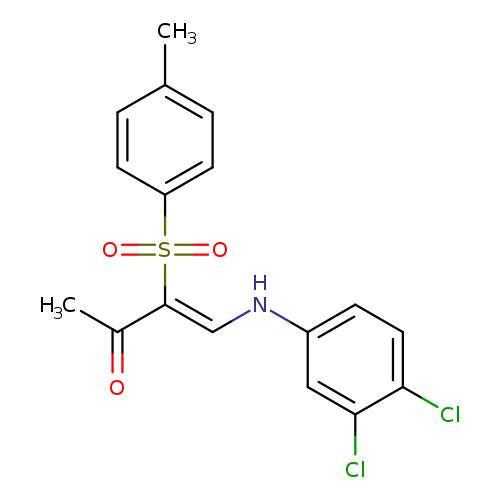
(3Z)-4-[(3,4-dichlorophenyl)amino]-3-(4-methylbenzenesulfonyl)but-3-en-2-oneCatalog No.:AA00IV60 CAS No.:1025138-31-1 MDL No.:MFCD00245552 MF:C17H15Cl2NO3S MW:384.2769 |
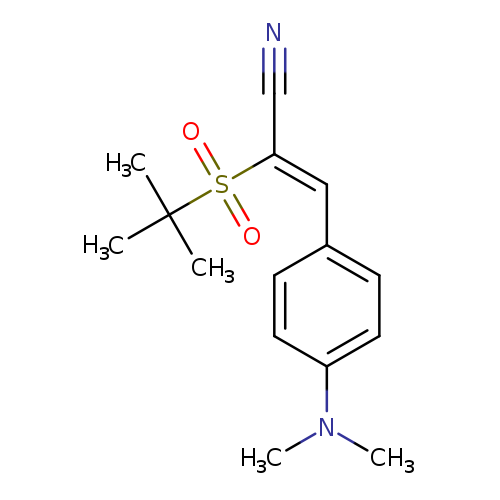
(2Z)-3-[4-(dimethylamino)phenyl]-2-(2-methylpropane-2-sulfonyl)prop-2-enenitrileCatalog No.:AA00IV7B CAS No.:1025143-31-0 MDL No.:MFCD00955177 MF:C15H20N2O2S MW:292.3965 |
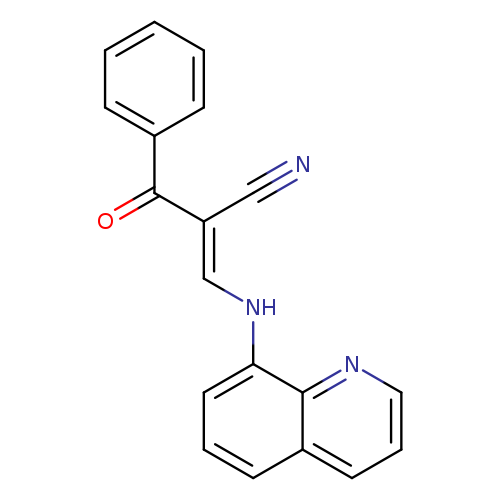
(2E)-2-[(E)-benzoyl]-3-[(quinolin-8-yl)amino]prop-2-enenitrileCatalog No.:AA00IX8F CAS No.:1025150-25-7 MDL No.:MFCD00245910 MF:C19H13N3O MW:299.3260 |
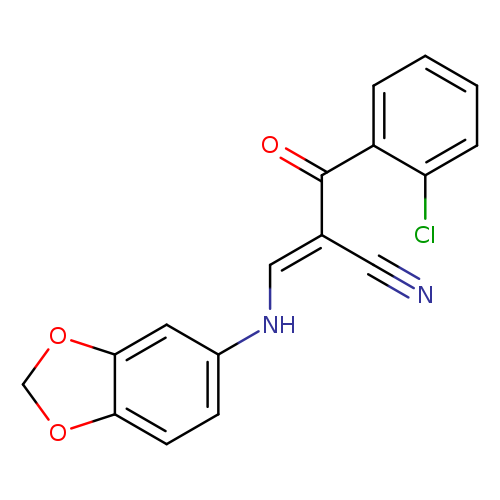
(2E)-3-[(2H-1,3-benzodioxol-5-yl)amino]-2-[(E)-2-chlorobenzoyl]prop-2-enenitrileCatalog No.:AA00ITEZ CAS No.:1025166-12-4 MDL No.:MFCD01567557 MF:C17H11ClN2O3 MW:326.7338 |
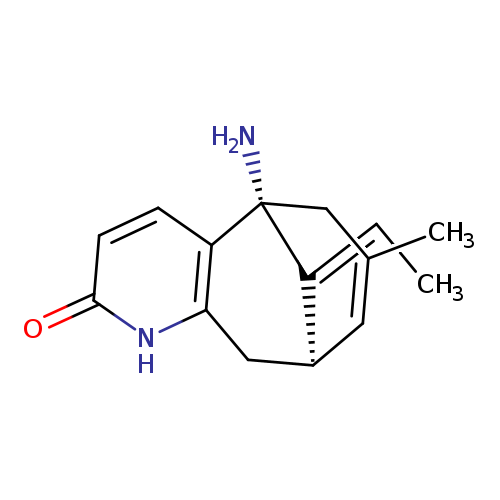
(1R,9R)-1-amino-13-ethylidene-11-methyl-6-azatricyclo[7.3.1.0^{2,7}]trideca-2(7),3,10-trien-5-oneCatalog No.:AA0007JW CAS No.:102518-79-6 MDL No.:MFCD01714949 MF:C15H18N2O MW:242.3162 |
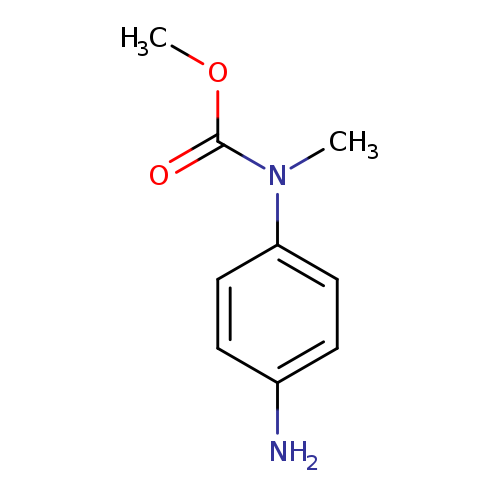
methyl N-(4-aminophenyl)-N-methylcarbamateCatalog No.:AA019X4Y CAS No.:10252-00-3 MDL No.:MFCD11184928 MF:C9H12N2O2 MW:180.2038 |
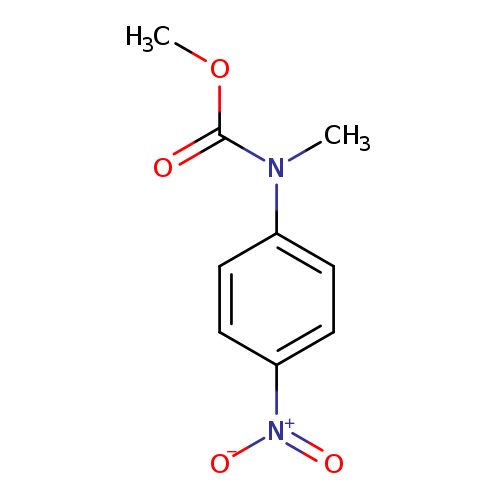
N-(4-Nitrophenyl)-N-methylcarbamic acid methyl esterCatalog No.:AA009R5M CAS No.:10252-27-4 MDL No.:MFCD12913151 MF:C9H10N2O4 MW:210.1867 |
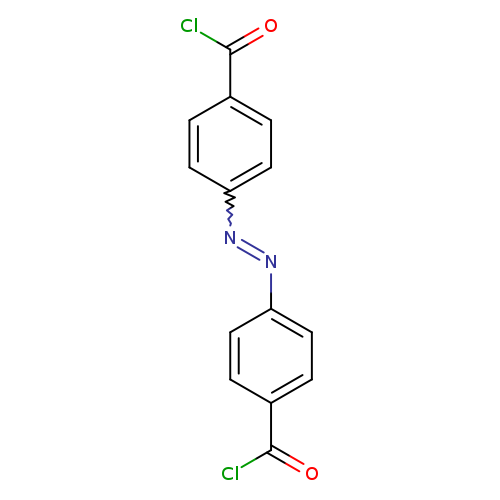
4,4'-Azodibenzoyl dichlorideCatalog No.:AA0007L2 CAS No.:10252-29-6 MDL No.:MFCD00053223 MF:C14H8Cl2N2O2 MW:307.1315 |
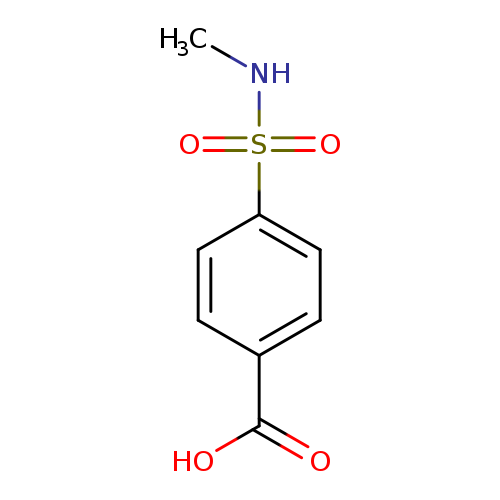
4-[(Methylamino)sulfonyl]benzoic acidCatalog No.:AA0007KY CAS No.:10252-63-8 MDL No.:MFCD05804382 MF:C8H9NO4S MW:215.2264 |
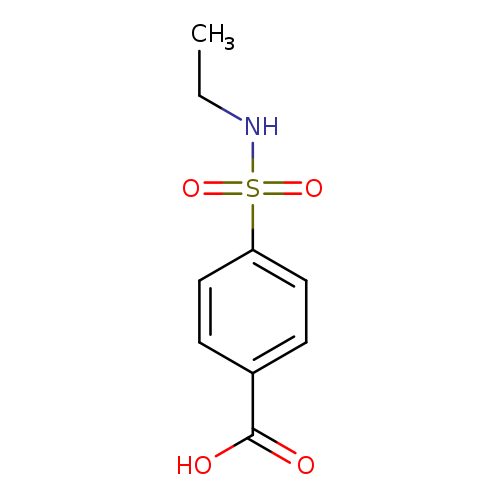
4-[(Ethylamino)sulfonyl]benzoic acidCatalog No.:AA0007KX CAS No.:10252-64-9 MDL No.:MFCD06357875 MF:C9H11NO4S MW:229.2529 |
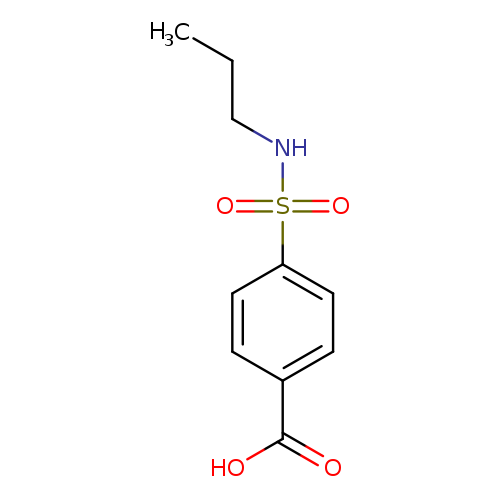
4-[(Propylamino)sulfonyl]benzoic acidCatalog No.:AA0007KW CAS No.:10252-65-0 MDL No.:MFCD05804383 MF:C10H13NO4S MW:243.2795 |
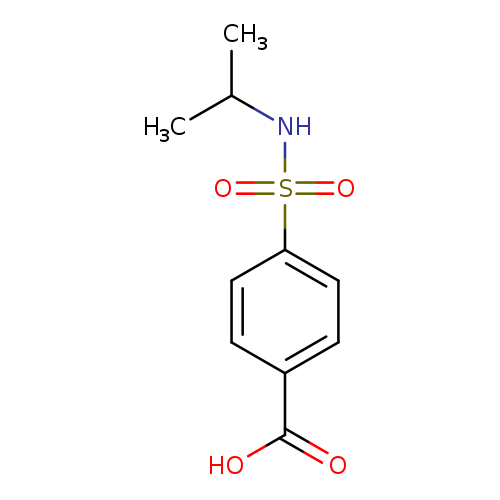
4-[(Isopropylamino)sulfonyl]benzoic acidCatalog No.:AA0007KV CAS No.:10252-66-1 MDL No.:MFCD06355943 MF:C10H13NO4S MW:243.2795 |
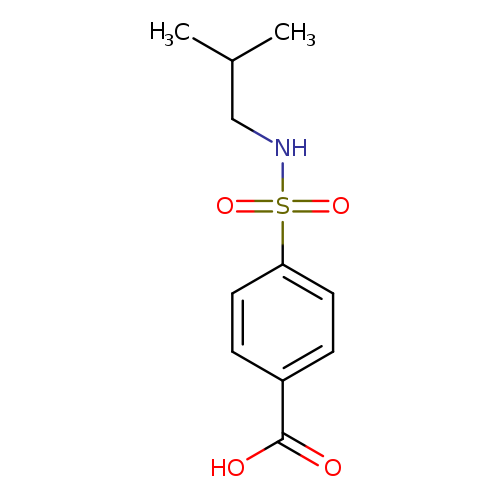
Benzoic acid, 4-[[(2-methylpropyl)amino]sulfonyl]-Catalog No.:AA0007KU CAS No.:10252-68-3 MDL No.:MFCD04035129 MF:C11H15NO4S MW:257.3061 |
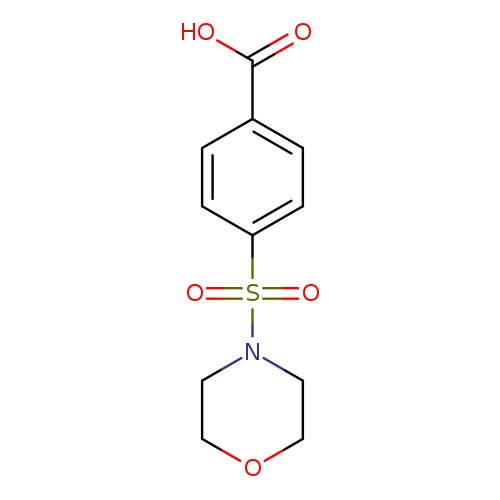
4-(Morpholine-4-sulfonyl)-benzoic acidCatalog No.:AA0007KS CAS No.:10252-82-1 MDL No.:MFCD00441966 MF:C11H13NO5S MW:271.2896 |
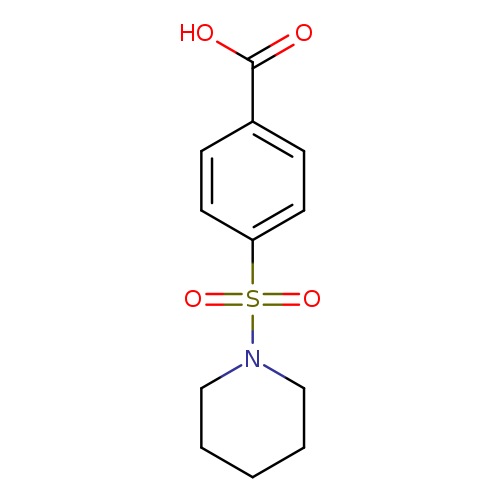
4-(Piperidine-1-sulfonyl)-benzoic acidCatalog No.:AA0007KR CAS No.:10252-83-2 MDL No.:MFCD01033230 MF:C12H15NO4S MW:269.3168 |
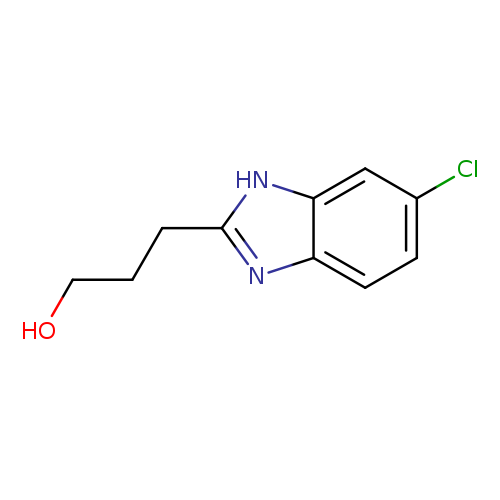
3-(5-Chloro-1H-benzo[d]imidazol-2-yl)propan-1-olCatalog No.:AA0007KP CAS No.:10252-89-8 MDL No.:MFCD00117218 MF:C10H11ClN2O MW:210.6601 |
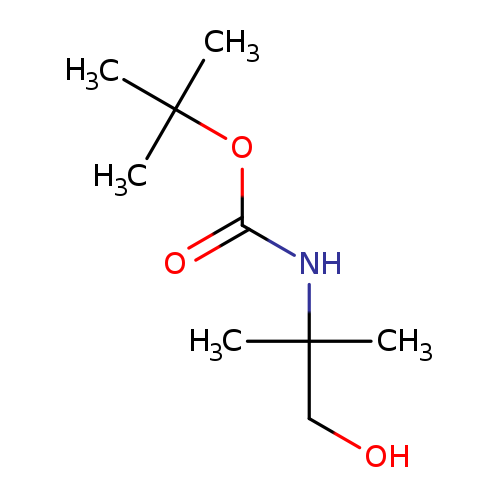
N-Boc-2-amino-2-methyl-1-propanolCatalog No.:AA0007KK CAS No.:102520-97-8 MDL No.:MFCD03788641 MF:C9H19NO3 MW:189.2521 |
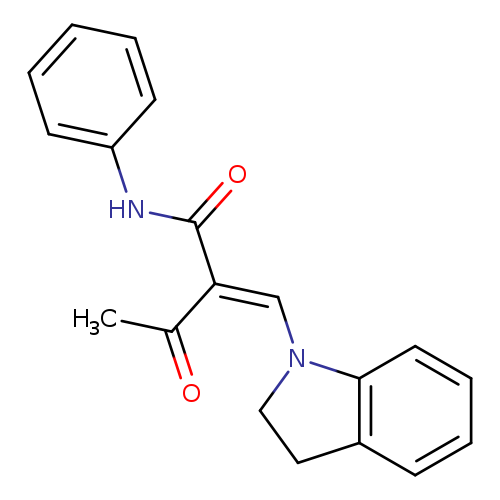
(2E)-2-[(2,3-dihydro-1H-indol-1-yl)methylidene]-3-oxo-N-phenylbutanamideCatalog No.:AA00IX8R CAS No.:1025209-24-8 MDL No.:MFCD00955263 MF:C19H18N2O2 MW:306.3584 |
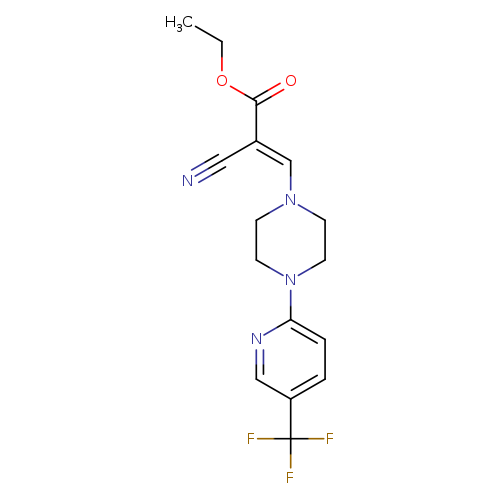
ethyl (2E)-2-cyano-3-{4-[5-(trifluoromethyl)pyridin-2-yl]piperazin-1-yl}prop-2-enoateCatalog No.:AA00ITFT CAS No.:1025218-95-4 MDL No.:MFCD01859633 MF:C16H17F3N4O2 MW:354.3270 |
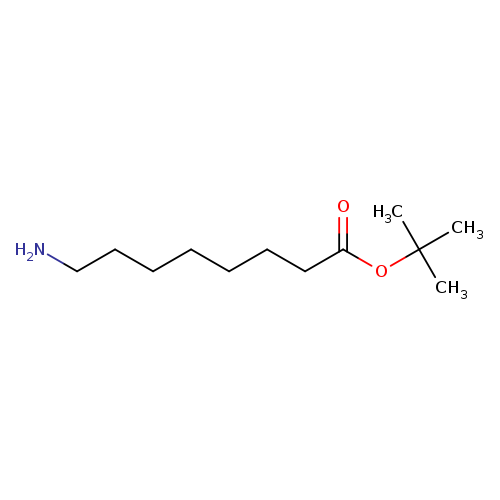
tert-Butyl 8-aminooctanoateCatalog No.:AA0007KI CAS No.:102522-32-7 MDL No.:MFCD24624132 MF:C12H25NO2 MW:215.3324 |
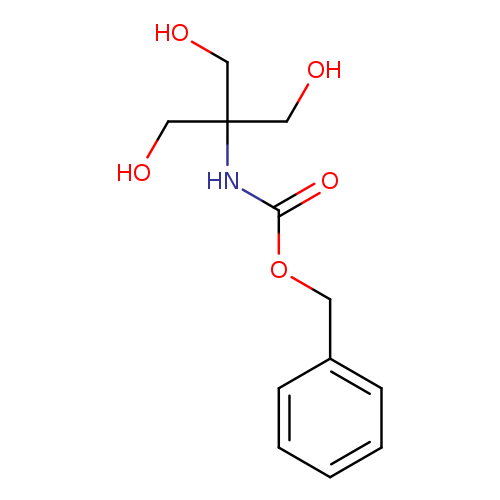
N-[2-Hydroxy-1,1-bis(hydroxyMethyl)ethyl]carbaMic Acid Benzyl EsterCatalog No.:AA008WYB CAS No.:102522-48-5 MDL No.:MFCD30186618 MF:C12H17NO5 MW:255.2671 |
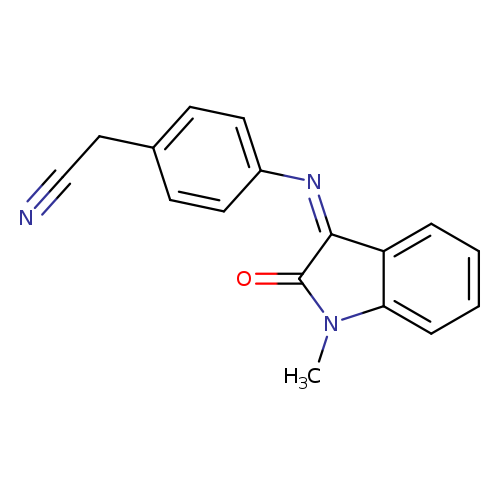
2-(4-{[(3Z)-1-methyl-2-oxo-2,3-dihydro-1H-indol-3-ylidene]amino}phenyl)acetonitrileCatalog No.:AA00ITDO CAS No.:1025223-99-7 MDL No.:MFCD03410317 MF:C17H13N3O MW:275.3046 |
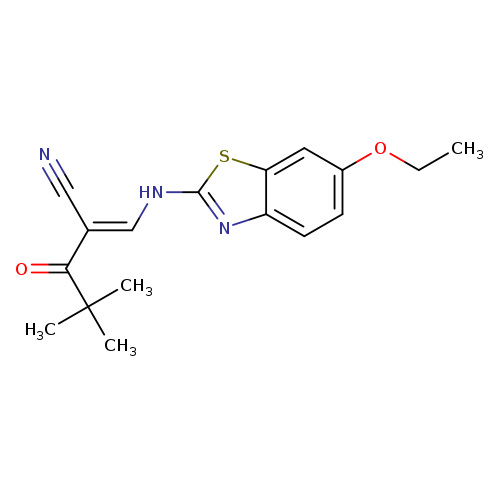
(2E)-2-{[(6-ethoxy-1,3-benzothiazol-2-yl)amino]methylidene}-4,4-dimethyl-3-oxopentanenitrileCatalog No.:AA00IZZV CAS No.:1025224-09-2 MDL No.:MFCD01567351 MF:C17H19N3O2S MW:329.4167 |
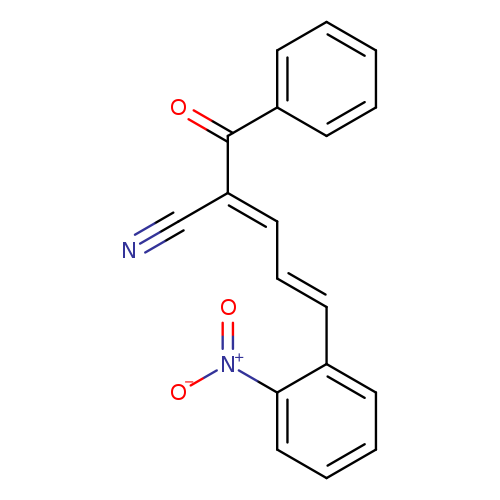
(2E,4E)-2-[(E,E)-benzoyl]-5-(2-nitrophenyl)penta-2,4-dienenitrileCatalog No.:AA00IX9K CAS No.:1025224-57-0 MDL No.:MFCD01567422 MF:C18H12N2O3 MW:304.2995 |
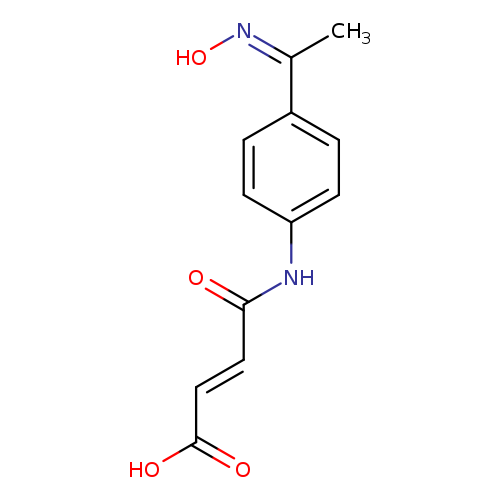
(2E)-3-({4-[(1Z)-1-(hydroxyimino)ethyl]phenyl}carbamoyl)prop-2-enoic acidCatalog No.:AA00ITJQ CAS No.:1025227-47-7 MDL No.:MFCD00245596 MF:C12H12N2O4 MW:248.2347 |
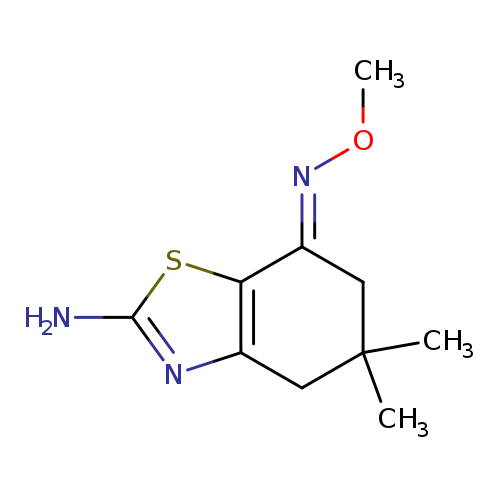
(7E)-7-(methoxyimino)-5,5-dimethyl-4,5,6,7-tetrahydro-1,3-benzothiazol-2-amineCatalog No.:AA00IUWV CAS No.:1025228-72-1 MDL No.:MFCD03410394 MF:C10H15N3OS MW:225.3106 |
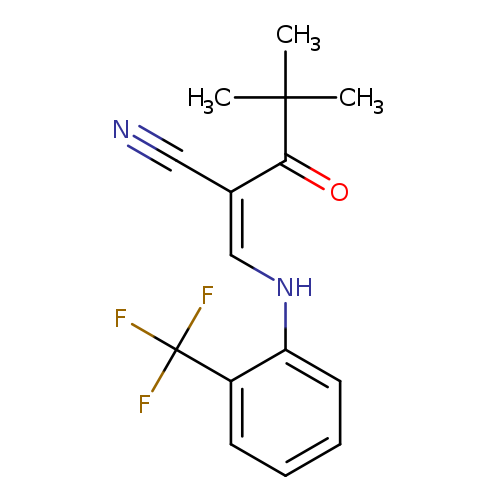
(2Z)-4,4-dimethyl-3-oxo-2-({[2-(trifluoromethyl)phenyl]amino}methylidene)pentanenitrileCatalog No.:AA00IV4X CAS No.:1025230-57-2 MDL No.:MFCD00245117 MF:C15H15F3N2O MW:296.2876 |
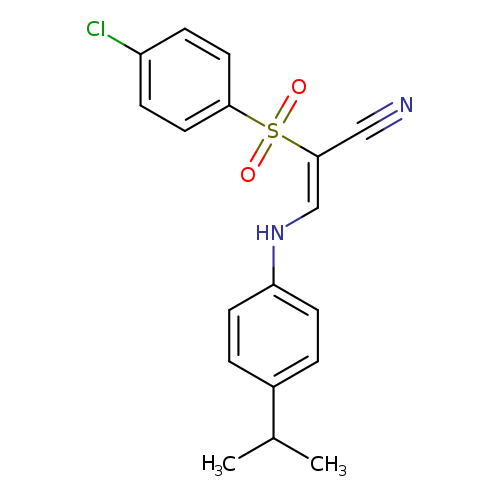
(2Z)-2-(4-chlorobenzenesulfonyl)-3-{[4-(propan-2-yl)phenyl]amino}prop-2-enenitrileCatalog No.:AA00J01U CAS No.:1025242-00-5 MDL No.:MFCD01567602 MF:C18H17ClN2O2S MW:360.8578 |
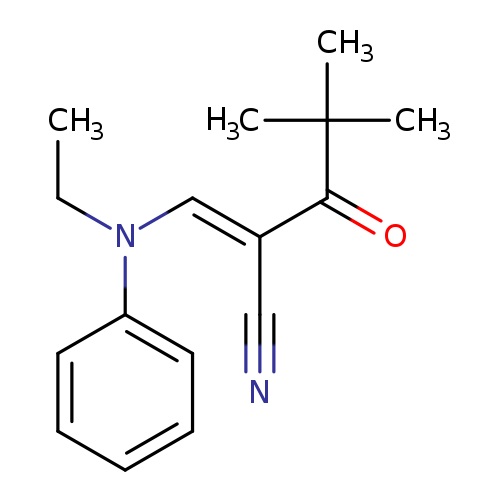
(2E)-2-{[ethyl(phenyl)amino]methylidene}-4,4-dimethyl-3-oxopentanenitrileCatalog No.:AA00IX8I CAS No.:1025242-61-8 MDL No.:MFCD00245963 MF:C16H20N2O MW:256.3428 |
(1,5-Cyclooctadiene)-η5-indenyl)iridium(I)Catalog No.:AA0007KG CAS No.:102525-11-1 MDL No.:MFCD07782020 MF:C17H19Ir MW:415.5498 |
(2Z)-3-oxo-N-phenyl-2-({[3-(trifluoromethyl)phenyl]amino}methylidene)butanamideCatalog No.:AA00J01E CAS No.:1025257-06-0 MDL No.:MFCD01567450 MF:C18H15F3N2O2 MW:348.3191 |

(E)-2-[(4-bromophenyl)sulfonyl]-3-[5-nitro-2-(2-pyridinylsulfanyl)phenyl]-2-propenenitrileCatalog No.:AA01FOL4 CAS No.:1025259-60-2 MDL No.: MF: MW: |
1,1,1,3,3,5,5,5-OctafluoropentaneCatalog No.:AA01EQJ8 CAS No.:102526-10-3 MDL No.:MFCD30530152 MF:C5H4F8 MW:216.0725 |
(2Z)-2-amino-3-[(E)-[(2E)-3-(2-nitrophenyl)prop-2-en-1-ylidene]amino]but-2-enedinitrileCatalog No.:AA00ITBH CAS No.:1025261-91-9 MDL No.:MFCD00245250 MF:C13H9N5O2 MW:267.2429 |
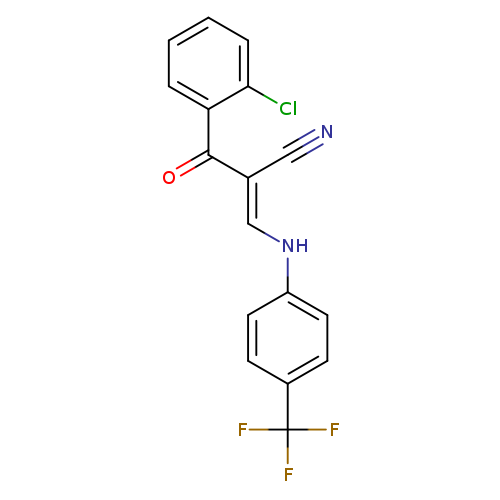
(2E)-2-[(E)-2-chlorobenzoyl]-3-{[4-(trifluoromethyl)phenyl]amino}prop-2-enenitrileCatalog No.:AA00IXA0 CAS No.:1025263-05-1 MDL No.:MFCD01567558 MF:C17H10ClF3N2O MW:350.7223 |
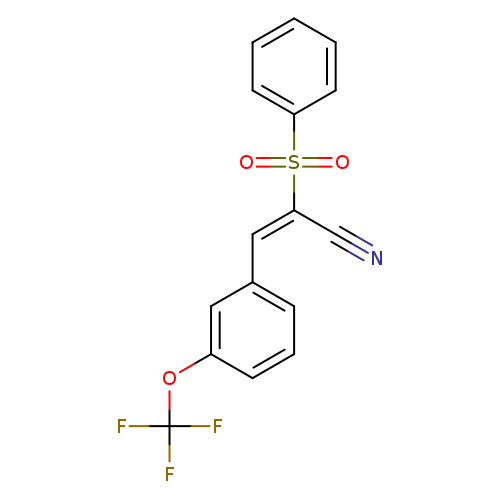
(2E)-2-(benzenesulfonyl)-3-[3-(trifluoromethoxy)phenyl]prop-2-enenitrileCatalog No.:AA00IX81 CAS No.:1025264-69-0 MDL No.:MFCD00245708 MF:C16H10F3NO3S MW:353.3157 |

(2Z)-2-(benzenesulfonyl)-3-[(3-chlorophenyl)amino]-3-(methylsulfanyl)prop-2-enenitrileCatalog No.:AA00IZY3 CAS No.:1025268-52-3 MDL No.:MFCD00245004 MF:C16H13ClN2O2S2 MW:364.8696 |
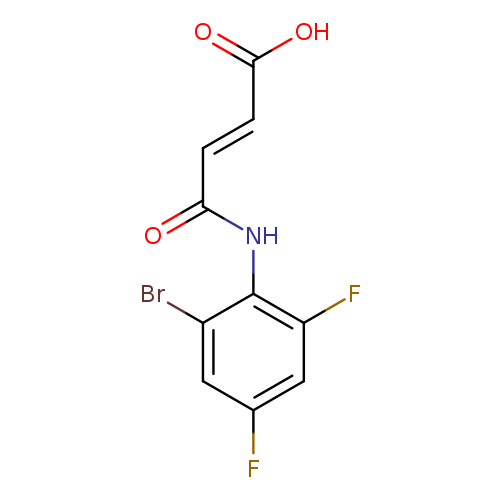
(2E)-3-[(2-bromo-4,6-difluorophenyl)carbamoyl]prop-2-enoic acidCatalog No.:AA00IVAK CAS No.:1025273-83-9 MDL No.:MFCD03001993 MF:C10H6BrF2NO3 MW:306.0603 |
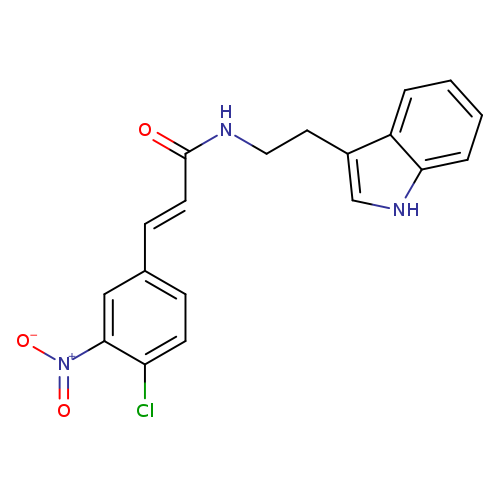
(2E)-3-(4-chloro-3-nitrophenyl)-N-[2-(1H-indol-3-yl)ethyl]prop-2-enamideCatalog No.:AA00ITAN CAS No.:1025276-57-6 MDL No.:MFCD00170951 MF:C19H16ClN3O3 MW:369.8016 |
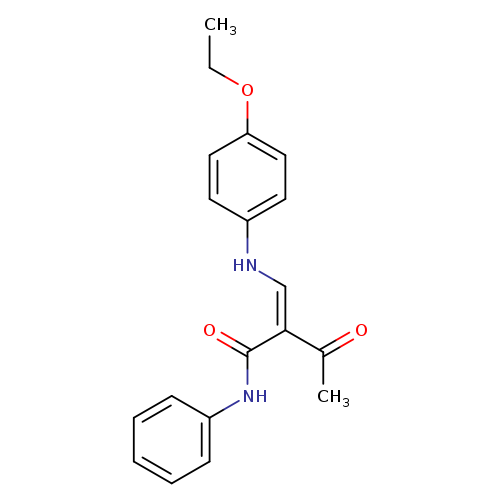
(2Z)-2-{[(4-ethoxyphenyl)amino]methylidene}-3-oxo-N-phenylbutanamideCatalog No.:AA00IV7D CAS No.:1025278-05-0 MDL No.:MFCD00955257 MF:C19H20N2O3 MW:324.3737 |
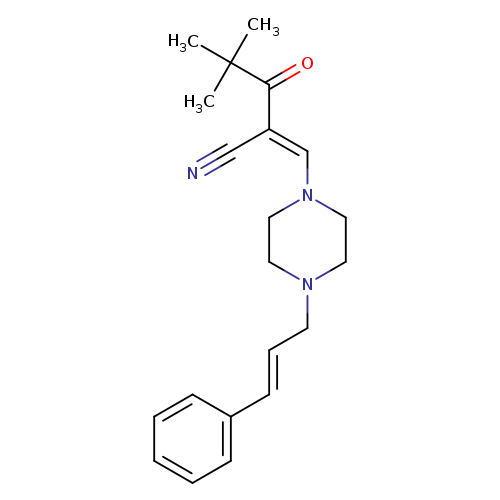
(2E)-4,4-dimethyl-3-oxo-2-({4-[(2E)-3-phenylprop-2-en-1-yl]piperazin-1-yl}methylidene)pentanenitrileCatalog No.:AA00J01W CAS No.:1025295-23-1 MDL No.:MFCD01567635 MF:C21H27N3O MW:337.4586 |
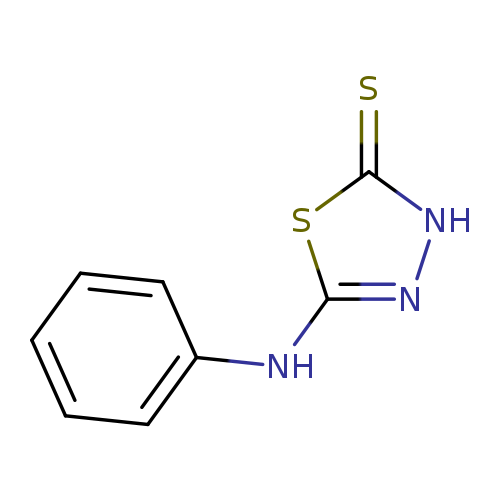
2-Mercapto-5-anilino-1,3,4-thiadiazoleCatalog No.:AA0007KO CAS No.:10253-83-5 MDL No.:MFCD00125876 MF:C8H7N3S2 MW:209.2913 |
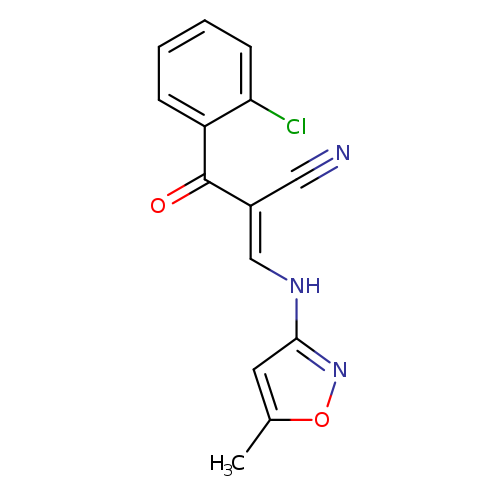
(2E)-2-[(E)-2-chlorobenzoyl]-3-[(5-methyl-1,2-oxazol-3-yl)amino]prop-2-enenitrileCatalog No.:AA00IV8I CAS No.:1025301-03-4 MDL No.:MFCD08272778 MF:C14H10ClN3O2 MW:287.7011 |
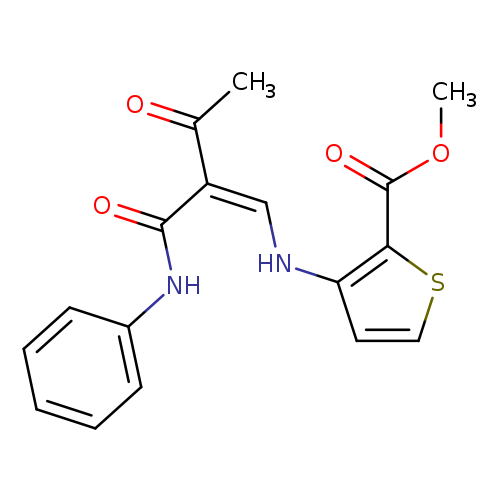
methyl 3-{[(1Z)-2-acetyl-2-(phenylcarbamoyl)eth-1-en-1-yl]amino}thiophene-2-carboxylateCatalog No.:AA00J01H CAS No.:1025302-40-2 MDL No.:MFCD01567470 MF:C17H16N2O4S MW:344.3849 |
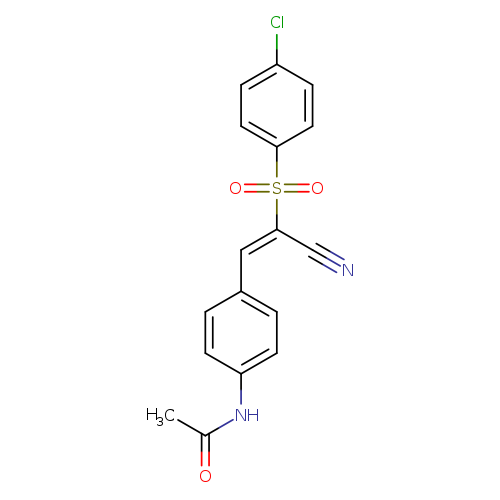
N-{4-[(1E)-2-(4-chlorobenzenesulfonyl)-2-cyanoeth-1-en-1-yl]phenyl}acetamideCatalog No.:AA00J00E CAS No.:1025304-57-7 MDL No.:MFCD00245992 MF:C17H13ClN2O3S MW:360.8147 |
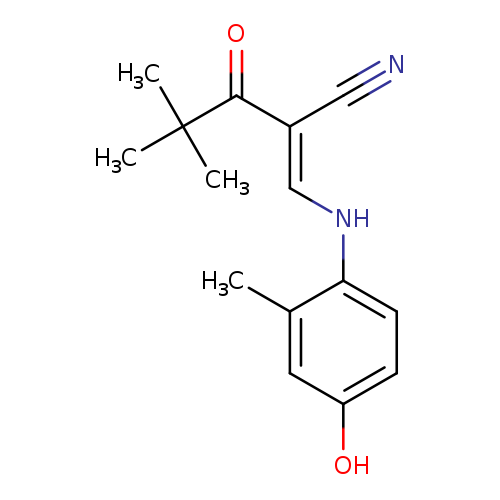
(2E)-2-{[(4-hydroxy-2-methylphenyl)amino]methylidene}-4,4-dimethyl-3-oxopentanenitrileCatalog No.:AA00J003 CAS No.:1025306-51-7 MDL No.:MFCD11656745 MF:C15H18N2O2 MW:258.3156 |
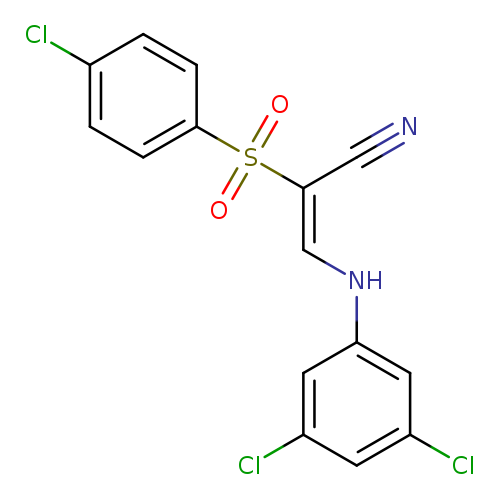
(2E)-2-(4-chlorobenzenesulfonyl)-3-[(3,5-dichlorophenyl)amino]prop-2-enenitrileCatalog No.:AA00ITB9 CAS No.:1025311-95-8 MDL No.:MFCD00178158 MF:C15H9Cl3N2O2S MW:387.6682 |
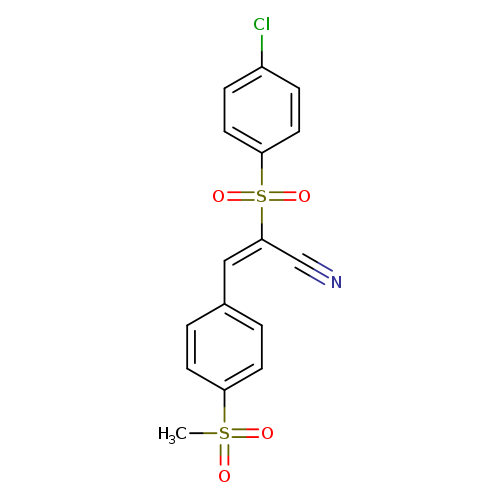
(2E)-2-(4-chlorobenzenesulfonyl)-3-(4-methanesulfonylphenyl)prop-2-enenitrileCatalog No.:AA00IV68 CAS No.:1025312-02-0 MDL No.:MFCD03410330 MF:C16H12ClNO4S2 MW:381.8538 |
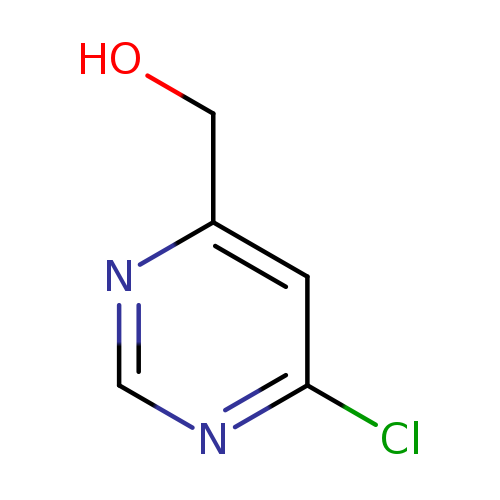
6-Chloro-4-hydroxymethylpyrimidineCatalog No.:AA0007L4 CAS No.:1025351-41-0 MDL No.:MFCD20482236 MF:C5H5ClN2O MW:144.5590 |
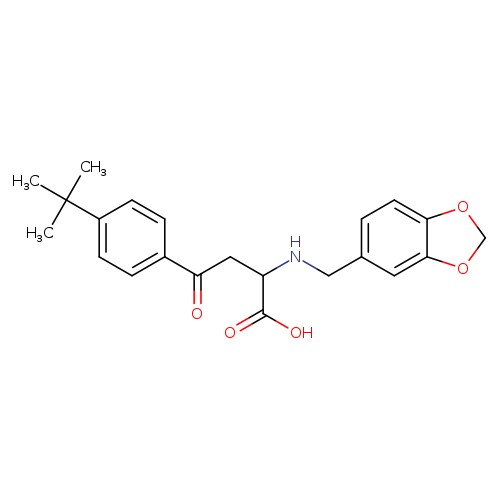
2-{[(2H-1,3-benzodioxol-5-yl)methyl]amino}-4-(4-tert-butylphenyl)-4-oxobutanoic acidCatalog No.:AA00ISSQ CAS No.:1025355-17-2 MDL No.:MFCD00955084 MF:C22H25NO5 MW:383.4376 |
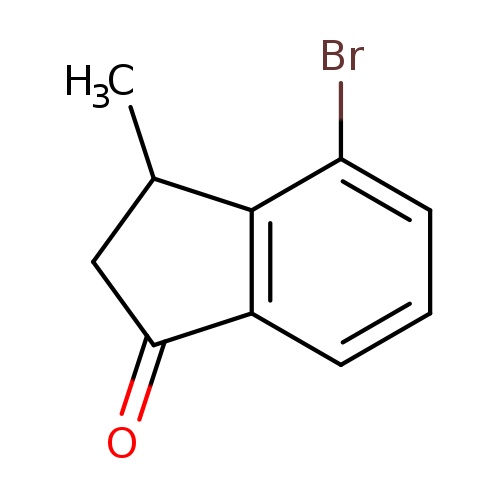
1H-Inden-1-one, 4-bromo-2,3-dihydro-3-methyl-Catalog No.:AA0007LC CAS No.:102539-53-7 MDL No.:MFCD11149281 MF:C10H9BrO MW:225.0819 |
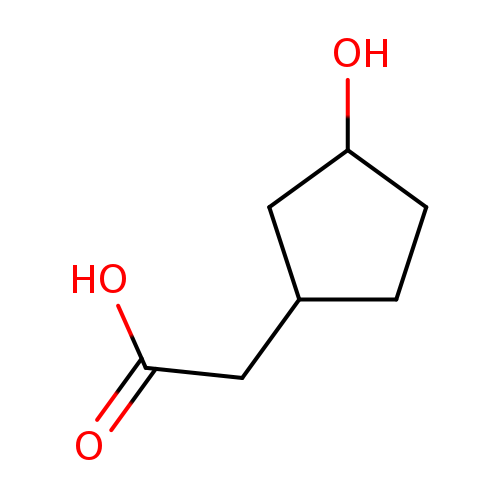
2-(3-Hydroxycyclopentyl)acetic acidCatalog No.:AA01DMLV CAS No.:102539-66-2 MDL No.:MFCD19230530 MF:C7H12O3 MW:144.1684 |
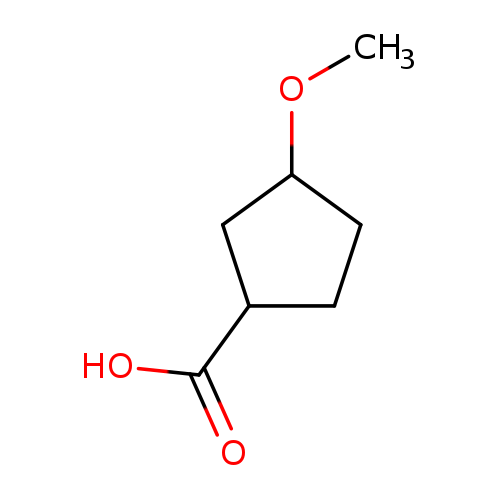
3-Methoxycyclopentane-1-carboxylic acidCatalog No.:AA01B5AH CAS No.:102539-67-3 MDL No.:MFCD24499323 MF:C7H12O3 MW:144.1684 |
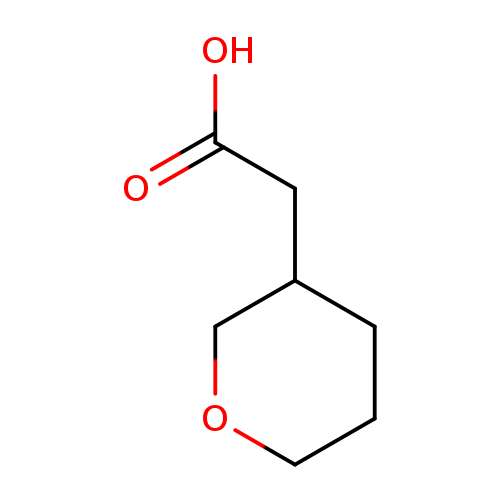
Tetrahydro-2h-pyran-3-ylacetic acidCatalog No.:AA0007LA CAS No.:102539-71-9 MDL No.:MFCD10700199 MF:C7H12O3 MW:144.1684 |
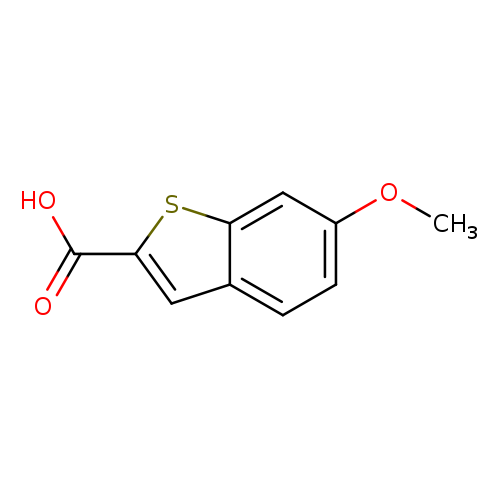
6-Methoxybenzo[b]thiophene-2-carboxylic acidCatalog No.:AA0007L9 CAS No.:102539-79-7 MDL No.:MFCD07371542 MF:C10H8O3S MW:208.2337 |
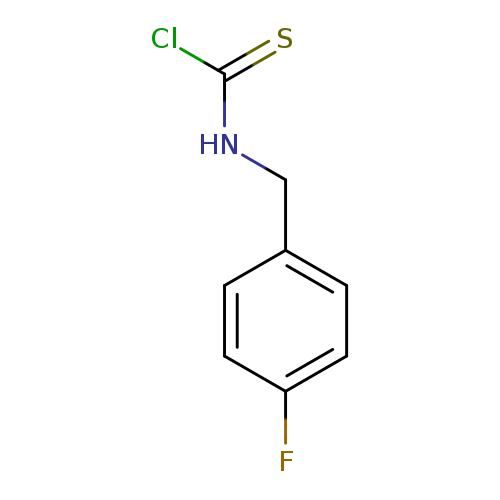
Carbamothioic chloride, (4-fluorophenyl)methyl- (9CI)Catalog No.:AA0007LN CAS No.:10254-60-1 MDL No.:MFCD03093779 MF:C8H7ClFNS MW:203.6643 |
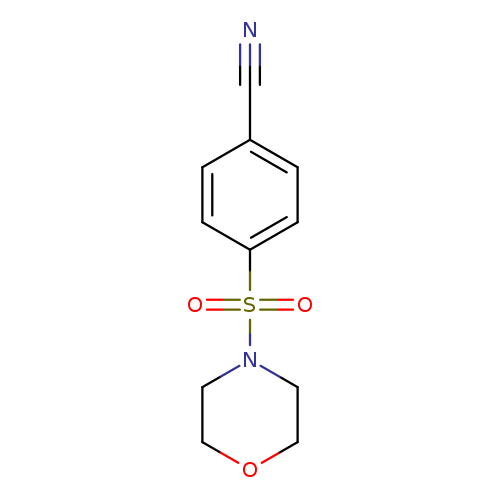
4-(Morpholin-4-ylsulfonyl)benzonitrileCatalog No.:AA0007LM CAS No.:10254-89-4 MDL No.:MFCD00111187 MF:C11H12N2O3S MW:252.2896 |
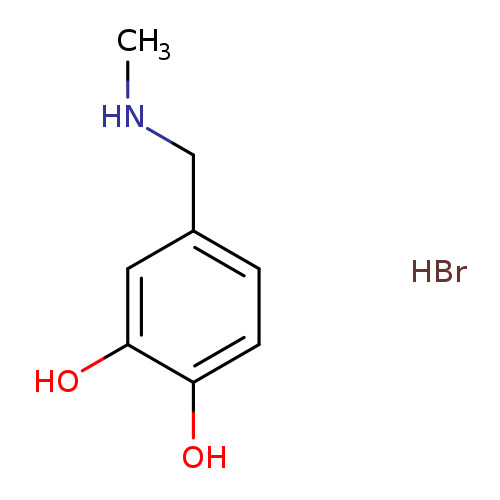
4-[(Methylamino)methyl]pyrocatechol hydrobromideCatalog No.:AA0007M6 CAS No.:1025423-95-3 MDL No.:MFCD26045226 MF:C8H12BrNO2 MW:234.0904 |
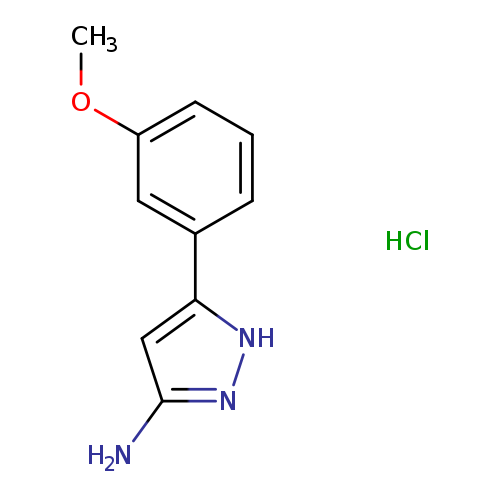
5-(3-Methoxy-phenyl)-2h-pyrazol-3-ylamine, HClCatalog No.:AA0007M3 CAS No.:1025447-42-0 MDL No.:MFCD05256460 MF:C10H12ClN3O MW:225.6748 |
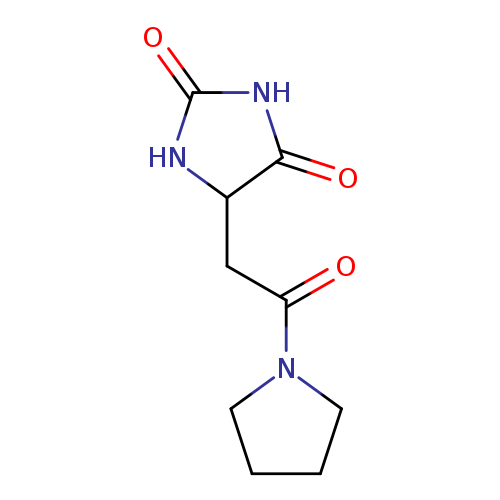
5-[2-oxo-2-(pyrrolidin-1-yl)ethyl]imidazolidine-2,4-dioneCatalog No.:AA019K7B CAS No.:1025448-38-7 MDL No.:MFCD08691224 MF:C9H13N3O3 MW:211.2178 |
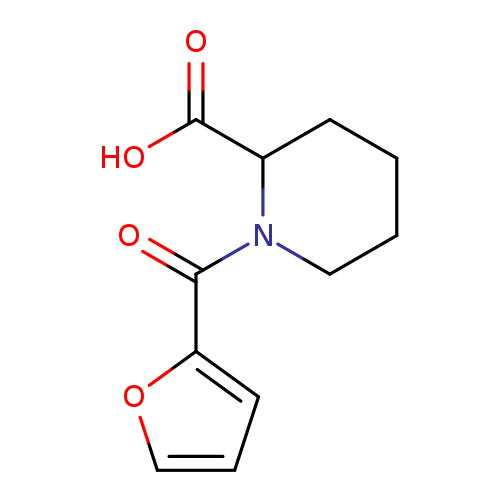
1-(Furan-2-carbonyl)piperidine-2-carboxylic acidCatalog No.:AA008TN7 CAS No.:1025450-05-8 MDL No.:MFCD08444976 MF:C11H13NO4 MW:223.2252 |
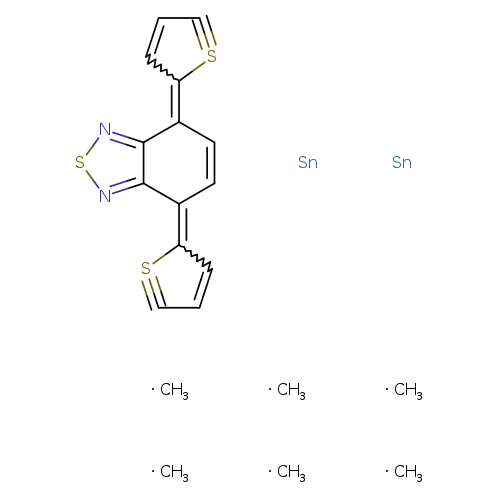
4,7-Bis(5-(trimethylstannyl)thiophen-2-yl)benzo[c][1,2,5]thiadiazoleCatalog No.:AA0091WK CAS No.:1025451-57-3 MDL No.:MFCD28049209 MF:C20H26N2S3Sn2 MW:628.0308 |
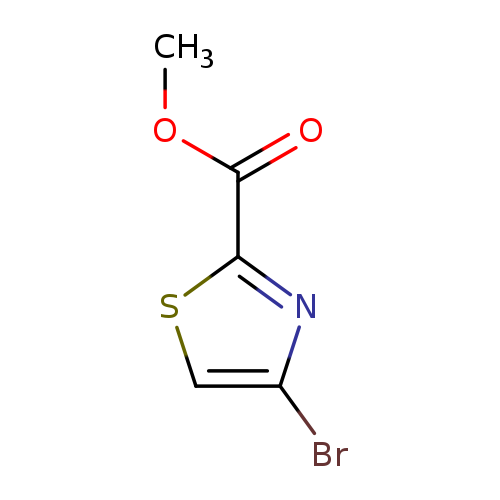
Methyl 4-bromothiazole-2-carboxylateCatalog No.:AA0007LZ CAS No.:1025468-06-7 MDL No.:MFCD11114545 MF:C5H4BrNO2S MW:222.0598 |
9-Methyl-3,9-diazabicyclo[4.2.1]nonaneCatalog No.:AA0007MA CAS No.:102547-84-2 MDL No.:MFCD13192515 MF:C8H16N2 MW:140.2260 |
Diacetato{(S)-(+)-5,5'-bis[di(3,5-di-t-butyl-4-methoxyphenyl)phosphino]-4,4'-bi-1,3-benzodioxole}ruthenium(II)Catalog No.:AA008V2X CAS No.:1025476-84-9 MDL No.:MFCD17018804 MF:C78H106O12P2Ru MW:1398.6866 |
Diacetato{(R)-(-)-5,5'-bis[di(3,5-di-t-butyl-4-methoxyphenyl)phosphino]-4,4'-bi-1,3-benzodioxole}ruthenium(II)Catalog No.:AA008VI3 CAS No.:1025477-38-6 MDL No.:MFCD17018804 MF:C78H106O12P2Ru MW:1398.6866 |
4-(3,5-Dimethyl-1H-pyrazol-1-yl)phenylboronic acidCatalog No.:AA0007LV CAS No.:1025495-85-5 MDL No.:MFCD08572133 MF:C11H13BN2O2 MW:216.0441 |
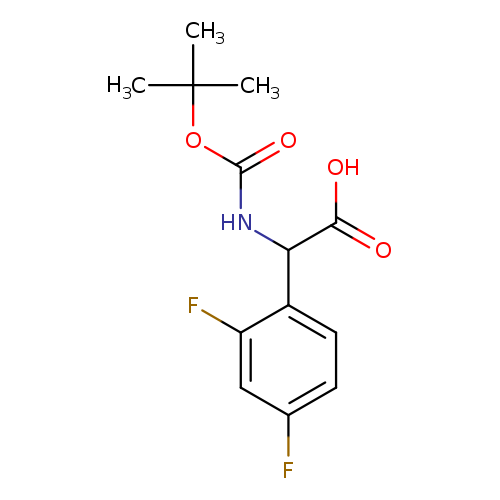
Benzeneacetic acid, α-[[(1,1-dimethylethoxy)carbonyl]amino]-2,4-difluoro-Catalog No.:AA0007LU CAS No.:1025496-16-5 MDL No.:MFCD07388901 MF:C13H15F2NO4 MW:287.2593 |
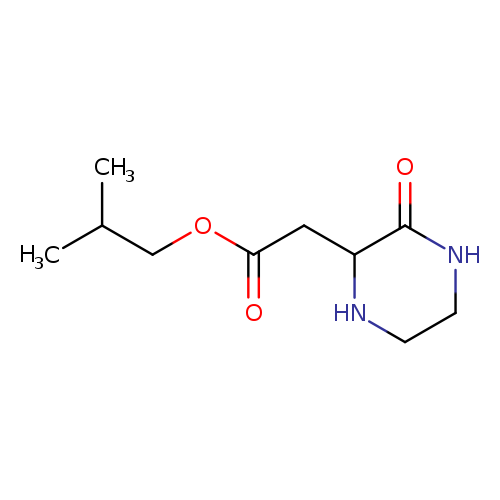
Isobutyl 2-(3-oxo-2-piperazinyl)acetateCatalog No.:AA01FO7G CAS No.:1025496-29-0 MDL No.: MF:C10H18N2O3 MW:214.2615 |
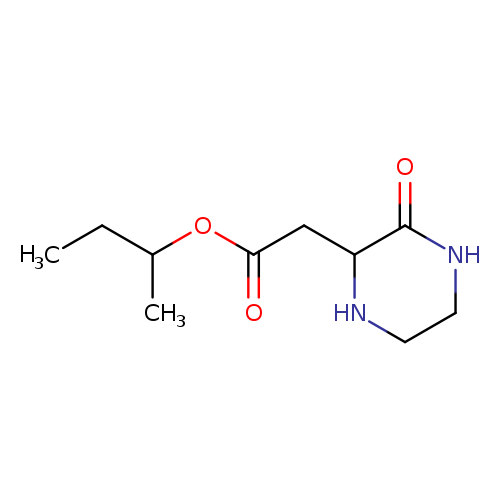
sec-Butyl 2-(3-oxo-2-piperazinyl)acetateCatalog No.:AA01FO8I CAS No.:1025496-30-3 MDL No.: MF:C10H18N2O3 MW:214.2615 |
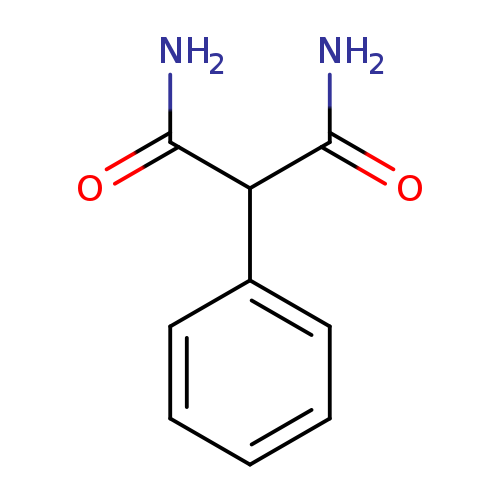
Propanediamide, 2-phenyl-Catalog No.:AA0007N4 CAS No.:10255-95-5 MDL No.:MFCD00051776 MF:C9H10N2O2 MW:178.1879 |
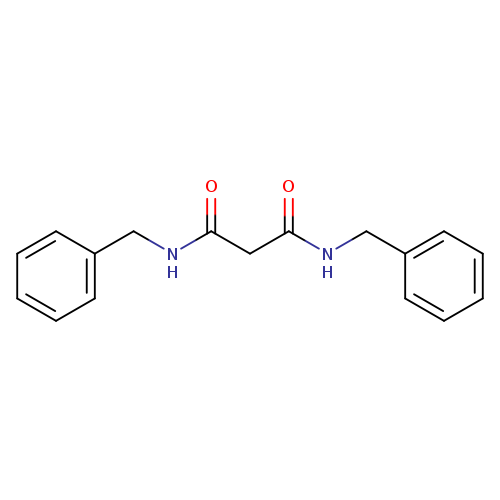
Propanediamide, N1,N3-bis(phenylmethyl)-Catalog No.:AA0007N3 CAS No.:10255-99-9 MDL No.:MFCD00420018 MF:C17H18N2O2 MW:282.3370 |
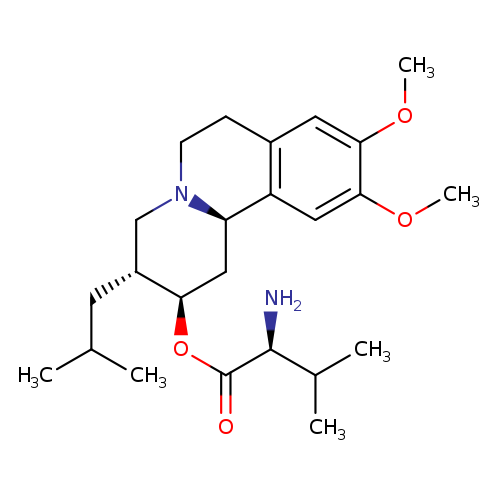
ValbenazineCatalog No.:AA0007MM CAS No.:1025504-45-3 MDL No.:MFCD28963976 MF:C24H38N2O4 MW:418.5695 |
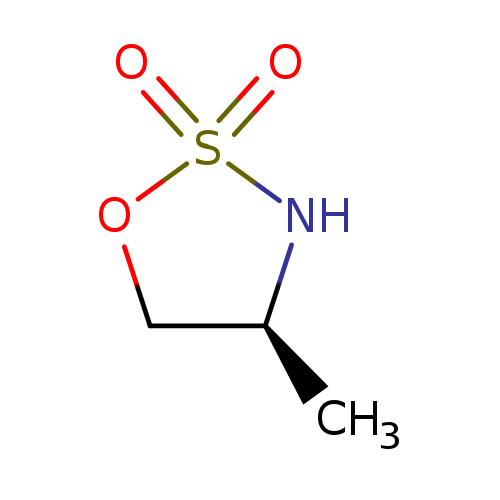
(4S)-4-methyl-1,2,3-oxathiazolidine-2,2-dioneCatalog No.:AA01EIMT CAS No.:1025505-82-1 MDL No.:MFCD31690208 MF:C3H7NO3S MW:137.1576 |
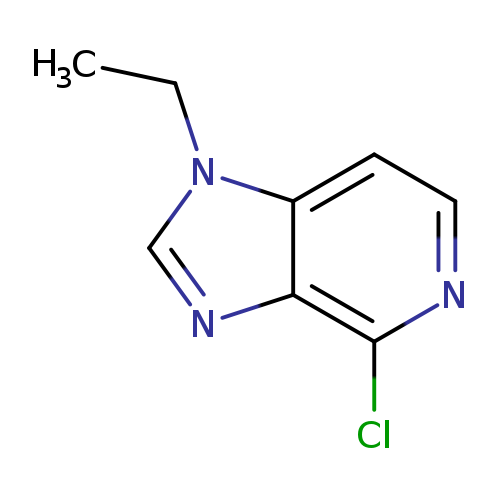
4-chloro-1-ethyl-1H-imidazo[4,5-c]pyridineCatalog No.:AA01BI4R CAS No.:1025509-11-8 MDL No.:MFCD29068189 MF:C8H8ClN3 MW:181.6222 |
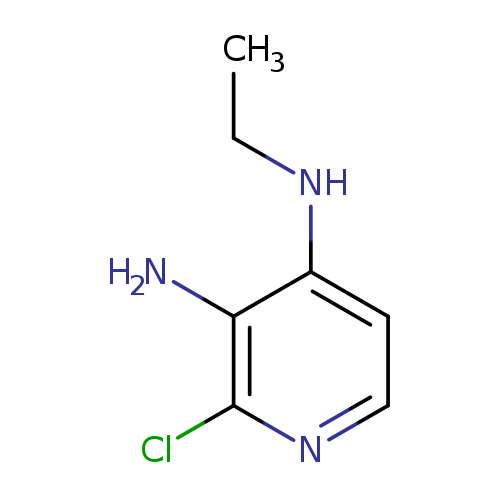
2-Chloro-n4-ethylpyridine-3,4-diamineCatalog No.:AA0007MK CAS No.:1025509-12-9 MDL No.:MFCD02670925 MF:C7H10ClN3 MW:171.6274 |
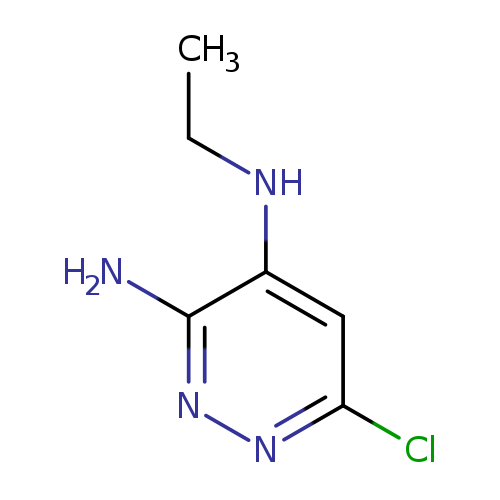
6-chloro-4-N-ethylpyridazine-3,4-diamineCatalog No.:AA01A16M CAS No.:1025509-63-0 MDL No.:MFCD20686403 MF:C6H9ClN4 MW:172.6155 |
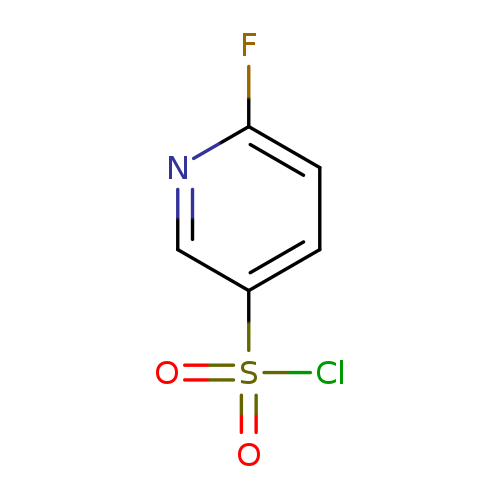
6-fluoropyridine-3-sulfonyl chlorideCatalog No.:AA008Z3D CAS No.:1025509-80-1 MDL No.:MFCD20275532 MF:C5H3ClFNO2S MW:195.5992 |
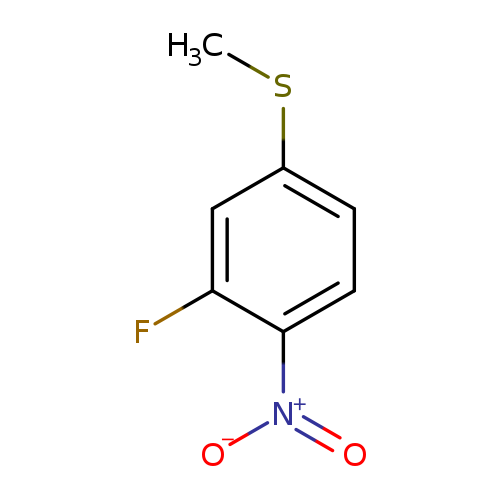
2-Fluoro-4-methylthio-1-nitrobenzeneCatalog No.:AA0007MI CAS No.:1025509-81-2 MDL No.:MFCD18070982 MF:C7H6FNO2S MW:187.1914 |
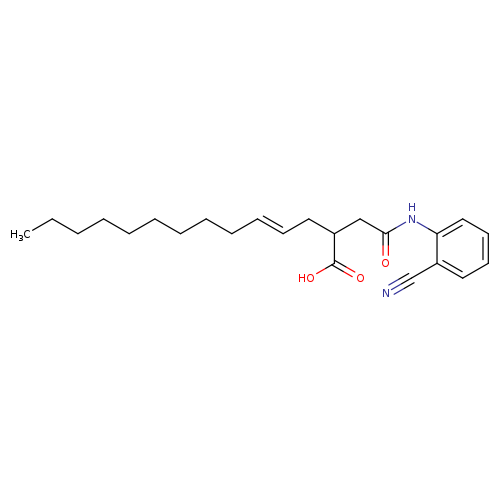
(4E)-2-{[(2-cyanophenyl)carbamoyl]methyl}tetradec-4-enoic acidCatalog No.:AA00ITD3 CAS No.:1025526-59-3 MDL No.:MFCD00245902 MF:C23H32N2O3 MW:384.5118 |
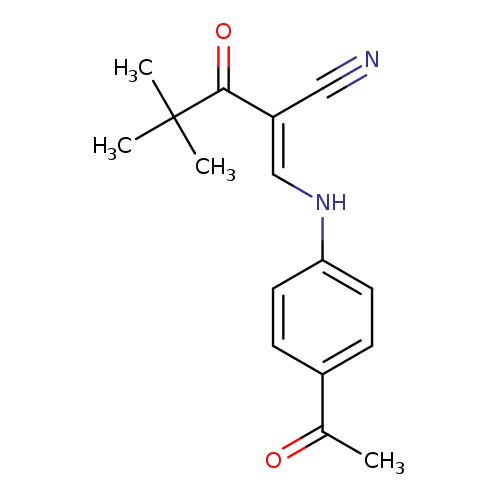
(2E)-2-{[(4-acetylphenyl)amino]methylidene}-4,4-dimethyl-3-oxopentanenitrileCatalog No.:AA00IX6R CAS No.:1025529-30-9 MDL No.:MFCD00245105 MF:C16H18N2O2 MW:270.3263 |
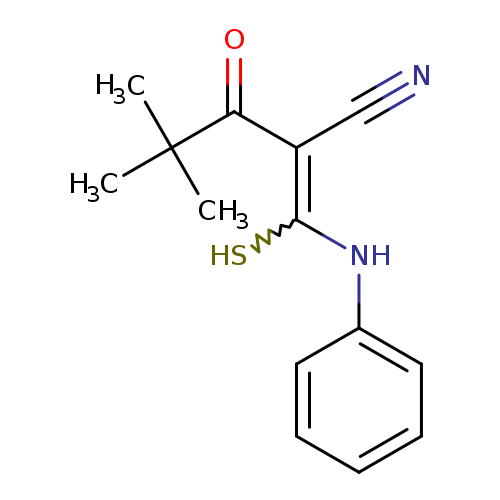
4,4-dimethyl-3-oxo-2-[(phenylamino)(sulfanyl)methylidene]pentanenitrileCatalog No.:AA00IX4R CAS No.:1025530-23-7 MDL No.:MFCD00955501 MF:C14H16N2OS MW:260.3546 |
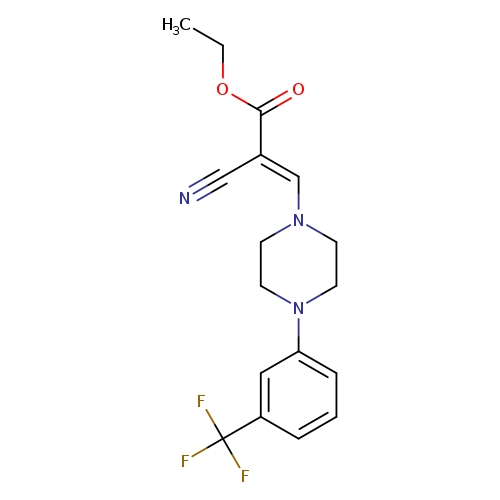
ethyl (2E)-2-cyano-3-{4-[3-(trifluoromethyl)phenyl]piperazin-1-yl}prop-2-enoateCatalog No.:AA00ITFD CAS No.:1025533-14-5 MDL No.:MFCD01567640 MF:C17H18F3N3O2 MW:353.3389 |
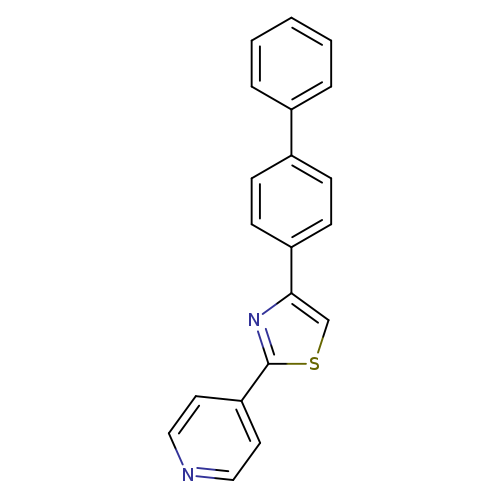
4-(4-Biphenylyl)-2-(4-pyridyl)thiazoleCatalog No.:AA00H9QK CAS No.:102554-97-2 MDL No.:MFCD12937797 MF:C20H14N2S MW:314.4036 |
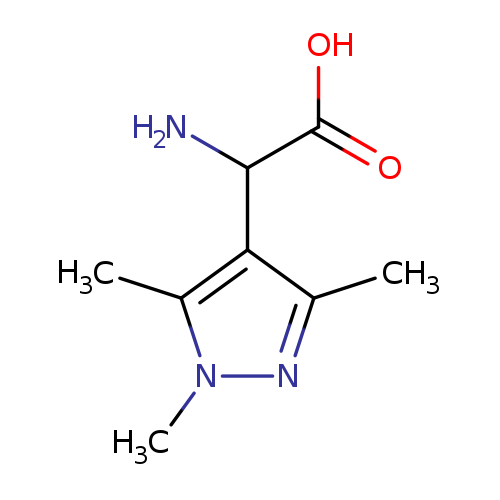
amino(1,3,5-trimethyl-1H-pyrazol-4-yl)acetic acidCatalog No.:AA008VJX CAS No.:1025555-34-3 MDL No.:MFCD07366340 MF:C8H13N3O2 MW:183.2077 |
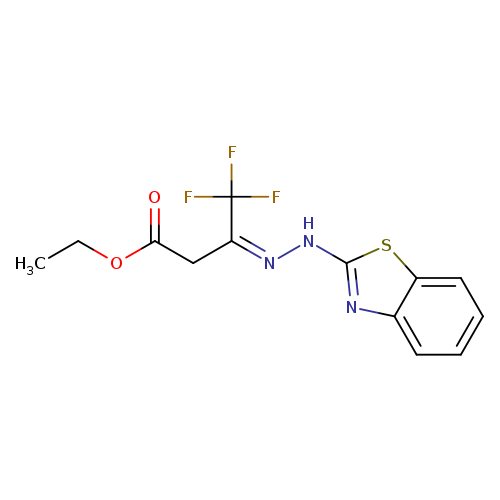
ethyl (3Z)-3-[2-(1,3-benzothiazol-2-yl)hydrazin-1-ylidene]-4,4,4-trifluorobutanoateCatalog No.:AA00IX8Y CAS No.:1025562-02-0 MDL No.:MFCD00955274 MF:C13H12F3N3O2S MW:331.3135 |
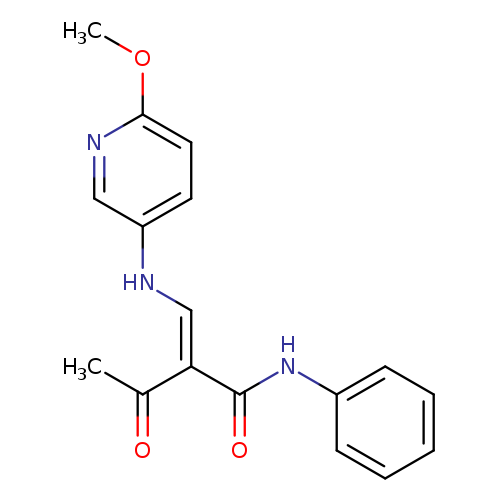
(2E)-2-{[(6-methoxypyridin-3-yl)amino]methylidene}-3-oxo-N-phenylbutanamideCatalog No.:AA00IX8Q CAS No.:1025562-35-9 MDL No.:MFCD00955260 MF:C17H17N3O3 MW:311.3352 |
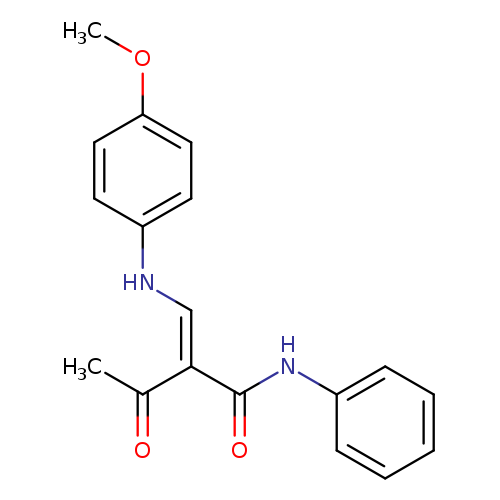
(2E)-2-{[(4-methoxyphenyl)amino]methylidene}-3-oxo-N-phenylbutanamideCatalog No.:AA00IV8C CAS No.:1025564-26-4 MDL No.:MFCD01567500 MF:C18H18N2O3 MW:310.3471 |
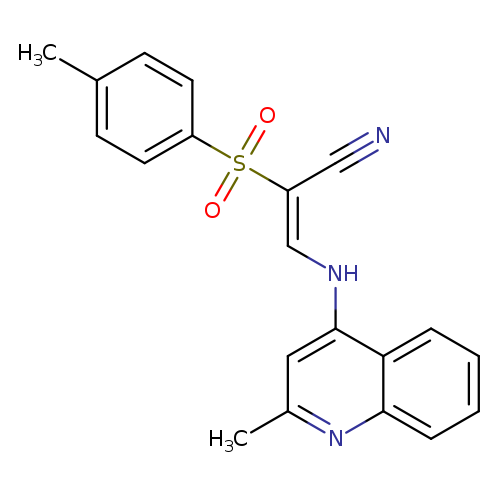
(2E)-2-(4-methylbenzenesulfonyl)-3-[(2-methylquinolin-4-yl)amino]prop-2-enenitrileCatalog No.:AA00ITB2 CAS No.:1025568-34-6 MDL No.:MFCD00245012 MF:C20H17N3O2S MW:363.4329 |
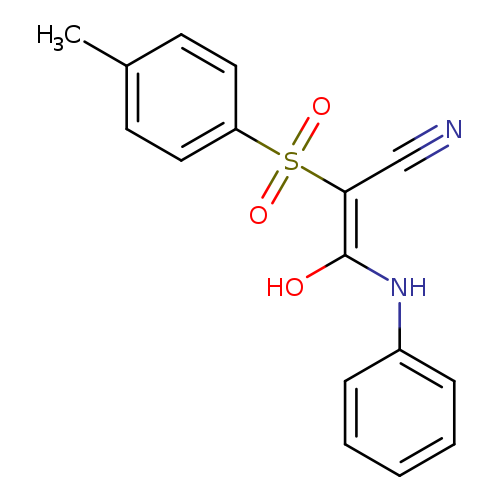
(2Z)-3-hydroxy-2-(4-methylbenzenesulfonyl)-3-(phenylamino)prop-2-enenitrileCatalog No.:AA00IX6I CAS No.:1025568-98-2 MDL No.:MFCD00245017 MF:C16H14N2O3S MW:314.3590 |
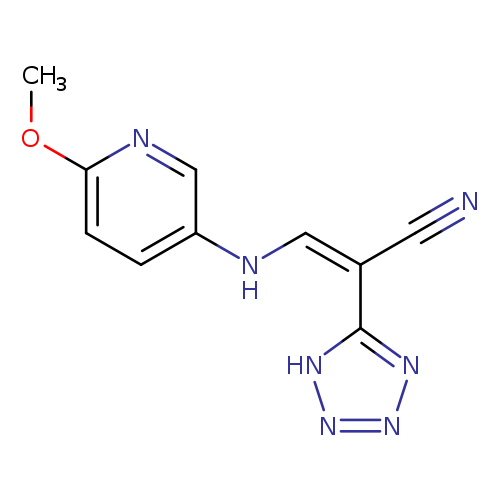
(2Z)-3-[(6-methoxypyridin-3-yl)amino]-2-(1H-1,2,3,4-tetrazol-5-yl)prop-2-enenitrileCatalog No.:AA00IV41 CAS No.:1025570-25-5 MDL No.:MFCD00178162 MF:C10H9N7O MW:243.2248 |
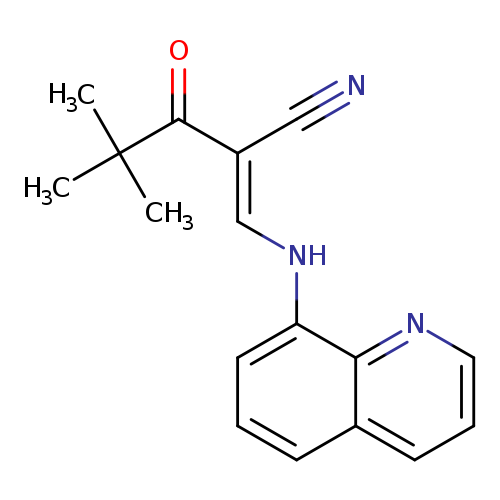
(2E)-4,4-dimethyl-3-oxo-2-{[(quinolin-8-yl)amino]methylidene}pentanenitrileCatalog No.:AA00IV4Y CAS No.:1025578-01-1 MDL No.:MFCD00245124 MF:C17H17N3O MW:279.3364 |
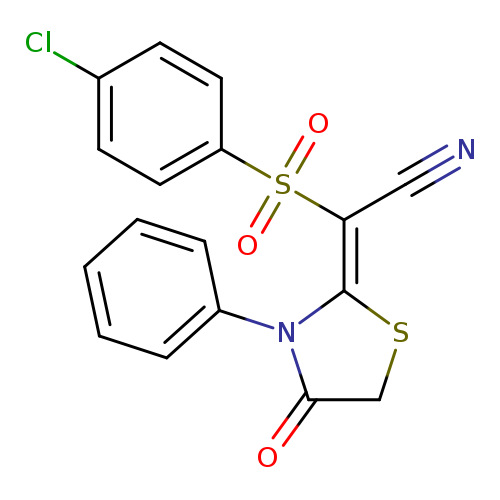
2-(4-chlorobenzenesulfonyl)-2-[(2E)-4-oxo-3-phenyl-1,3-thiazolidin-2-ylidene]acetonitrileCatalog No.:AA00IV4R CAS No.:1025578-26-0 MDL No.:MFCD00245028 MF:C17H11ClN2O3S2 MW:390.8638 |
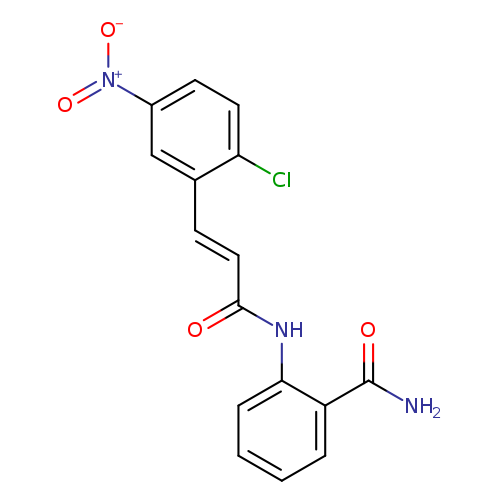
2-[(2E)-3-(2-chloro-5-nitrophenyl)prop-2-enamido]benzamideCatalog No.:AA00IZZ1 CAS No.:1025578-44-2 MDL No.:MFCD00245466 MF:C16H12ClN3O4 MW:345.7372 |
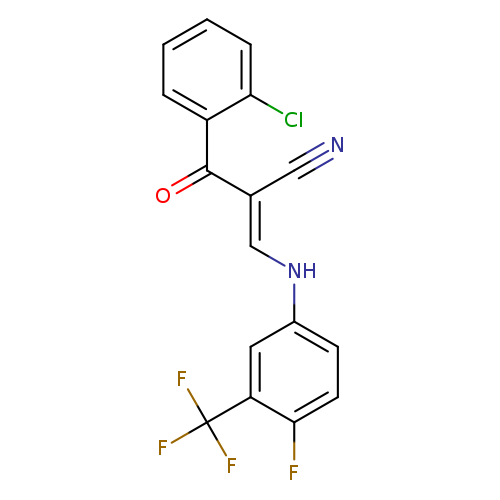
(2E)-2-[(E)-2-chlorobenzoyl]-3-{[4-fluoro-3-(trifluoromethyl)phenyl]amino}prop-2-enenitrileCatalog No.:AA00J01O CAS No.:1025596-46-6 MDL No.:MFCD01567560 MF:C17H9ClF4N2O MW:368.7128 |
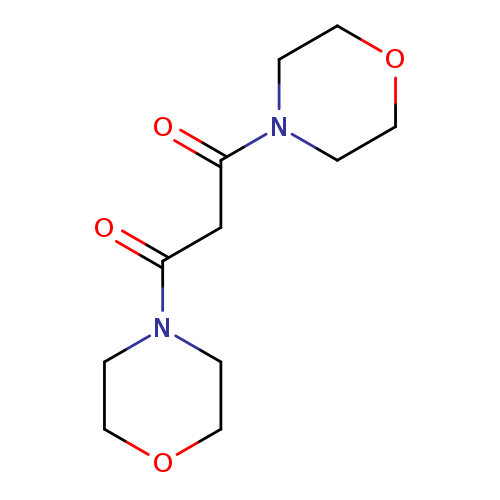
1,3-Di-morpholin-4-yl-propane-1,3-dioneCatalog No.:AA0007N1 CAS No.:10256-01-6 MDL No.:MFCD00023355 MF:C11H18N2O4 MW:242.2716 |
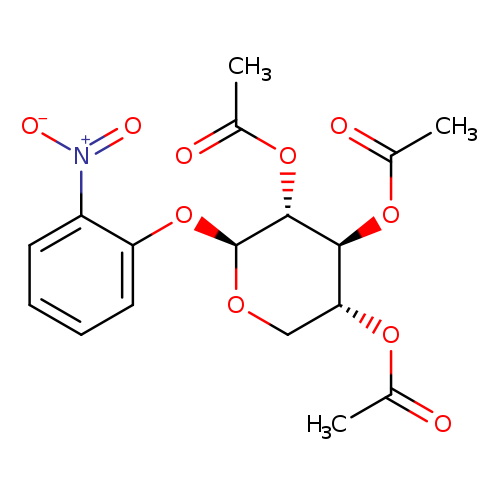
β-D-Xylopyranoside, 2-nitrophenyl, 2,3,4-triacetateCatalog No.:AA0007MZ CAS No.:10256-24-3 MDL No.:MFCD07369611 MF:C17H19NO10 MW:397.3335 |
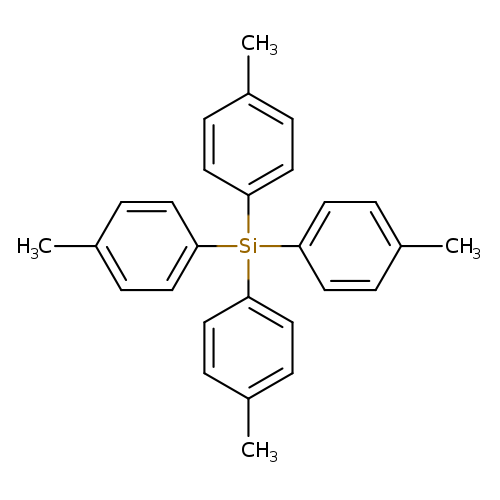
Benzene, 1,1',1'',1'''-silanetetrayltetrakis[4-methyl-Catalog No.:AA0007MV CAS No.:10256-83-4 MDL No.:MFCD00092688 MF:C28H28Si MW:392.6074 |
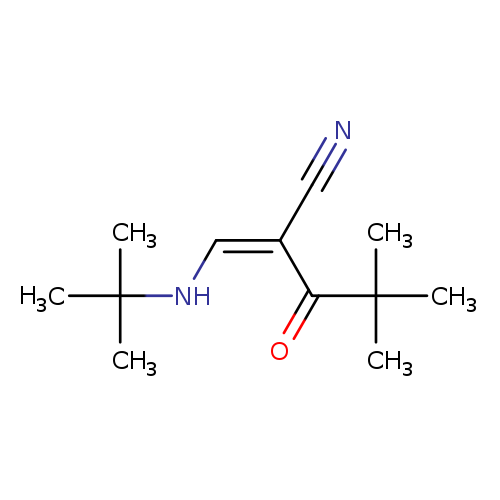
(2Z)-2-[(tert-butylamino)methylidene]-4,4-dimethyl-3-oxopentanenitrileCatalog No.:AA00IX69 CAS No.:1025600-70-7 MDL No.:MFCD00171053 MF:C12H20N2O MW:208.3000 |
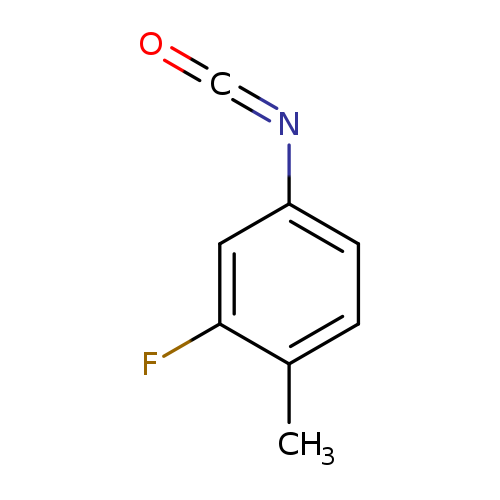
2-Fluoro-4-isocyanato-1-methylbenzeneCatalog No.:AA0007MO CAS No.:102561-42-2 MDL No.:MFCD00037071 MF:C8H6FNO MW:151.1377 |
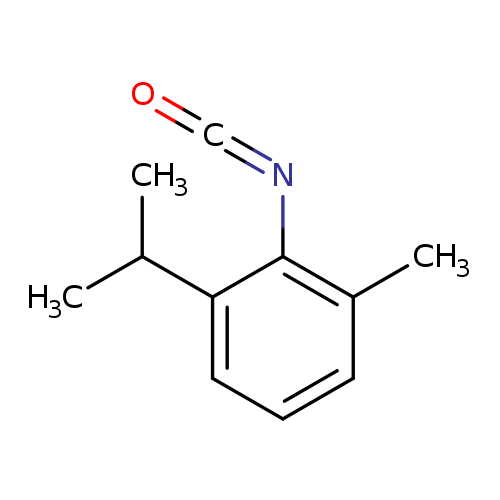
Benzene, 2-isocyanato-1-methyl-3-(1-methylethyl)-Catalog No.:AA0007MN CAS No.:102561-43-3 MDL No.:MFCD00019911 MF:C11H13NO MW:175.2270 |
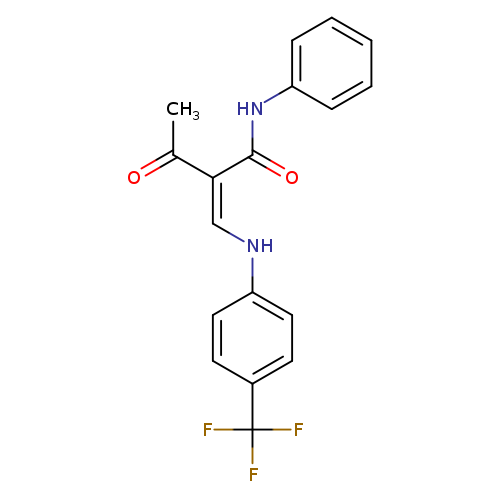
(2Z)-3-oxo-N-phenyl-2-({[4-(trifluoromethyl)phenyl]amino}methylidene)butanamideCatalog No.:AA00J01J CAS No.:1025617-09-7 MDL No.:MFCD01567474 MF:C18H15F3N2O2 MW:348.3191 |
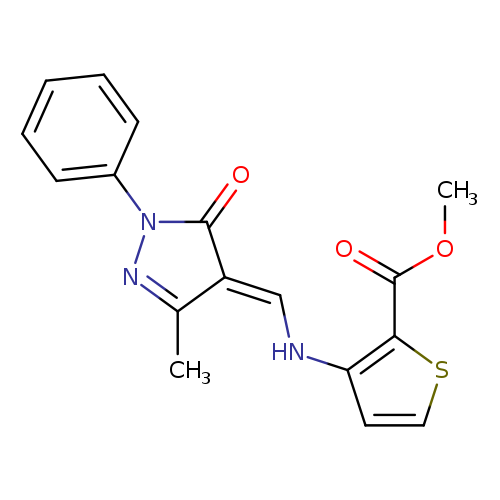
methyl 3-({[(4E)-3-methyl-5-oxo-1-phenyl-4,5-dihydro-1H-pyrazol-4-ylidene]methyl}amino)thiophene-2-carboxylateCatalog No.:AA00IV8D CAS No.:1025619-73-1 MDL No.:MFCD08272775 MF:C17H15N3O3S MW:341.3843 |