2020-01-08 09:24:30
Lu Ding,[b] Run-Duo Gao,[a] and Shu-Li You
Fused indolenines are important structural cores of natural products and frequently appear in biologically active mole- cules (Figure 1).[1] Accordingly, extensive efforts have been de- voted to developing efficient methods for the construction of these scaffolds in a highly chemo- and enantioselective manner. In this regard, the recently emerging catalytic asymmetric dearomatization (CADA) reactions provide a large array of methods allowing for efficient access to fused indolenines from readily available indole derivatives.[2] Notably, transition- metal-catalyzed allylic dearomatization reactions of indoles have witnessed significant progresses in the past decade.[3,4,5] Recently, we realized the construction of fused indolenine skeletons in a cascade fashion.[6] In 2014, the Rawal group and we independently reported an intermolecular cascade dearo- matization reaction of indole-based bisnucleophiles with prop- argyl carbonate, leading to a series of spiroindolenines and spi- roindolines. Moderate enantioselectivity (, 77 % ee) was ach- ieved for limited substrates (Scheme 1, reaction 1).[6a,7,8] Rawal and co-workers found that the indole N@H moiety could par- ticipate in the cyclization process when the nucleophilic side chain was decorated at the C2 position of the C3 methyl sub- stituted indole (Scheme 1, reaction 2).[7,9] Herein, we report a chemo- and enantioselective synthesis of multisubstituted fused indolenines by Pd-catalyzed cascade dearomatization of indoles bearing a nucleophile at the C2 position with proparg- yl carbonate (Scheme 1, reaction 3).
We began our studies by examining the reaction of dimethyl 2-[(3-benzyl-1H-indol-2-yl)methyl]malonate (1 a) with methyl prop-2-yn-1-yl carbonate (2) in dimethylacetamide (DMA) in the presence of a Pd catalyst derived from Pd2dba3. Firstly, sev- eral commercially available chiral phosphorus ligands were tested (Scheme 2). The desired product 3a was obtained in moderate yields when Feringa ligand (S,S,Sa)-L1, BINAP (R)-L2, or Synphos (R)-L3 were used. However, the Pd complex de- rived from PHOX ligand (S)-L4 could hardly promote this reac- tion. It is worth noting that the reaction with L1 led to 3a in excellent enantioselectivity and good chemoselectivity (94 % ee, 10:1 3 a/4 a). Encouraged by these results, phosphora- midite ligands L5-L9 were investigated, however, no better re- sults were obtained. Next, further optimization of the reaction conditions was carried out using L1 (Table 1). Reactions in vari- ous solvents, such as DMF, CH2Cl2, and THF, afforded compara- ble yields and enantioselectivity albeit with lower 3 a/4a ratios (entries 2–4), whereas MeOH and PhMe led to almost no reac- tion (entries 5–6). Further evaluation of the concentration (entries 7–11) of 1a revealed that c = 0.2 mol L@1 was optimal in terms of yield (entry 8). Pd2(4-OMe-dba)3 was a better palladium source than Pd2dba3 and [Pd(allyl)Cl]2 (entries 12 and 13).[10] Furthermore, some additives were tested. To our delight, both yield and 3 a/4a ratio were improved remarkably when 4 a molecular sieves were used (82 % yield, 95 % ee, > 19:1 3 a/4 a, entry 15). Finally, the optimized reaction conditions were es- tablished as the following: Pd2(4-OMe-dba)3 (2.5 mol%), L1 (11 mol%), 4 a MS (100 mg) in DMA (1 mL) at 80 8C (entry 15). Notably, 4a could not be converted to 3a under the optimized
conditions, suggesting the formation of C@N is likely an irrever- sible process here.
With the optimized conditions in hands, we then explored the substrate scope of this reaction (Scheme 3). Firstly, sub- strates with different substituents on the benzyl group were examined. Both electron-donating (3b: 4-OMe; 3c: 4-Me; 3d: 3-Me) and electron-withdrawing (3e: 4-F; 3 f: 4-Cl) groups were found to be well tolerated. The corresponding products were obtained in good to excellent yields, with excellent chemo- and enantioselectivities (3 b–3 f, 78–82 % yields, 16.2 :1 to > 19:1 3/4, 93–95 % ee). When the benzyl group was changed to 2-thienyl or 2-naphthyl, the desired products could also be obtained with gratifying re- sults (3g: 79% yield, 15.6:1 3 g/4 g, 96% ee; 3h: 85% yield, > 19:1 3 h/4 h, 96% ee). Next, substrates with different substituents on the indole ring were investi- gated. The dearomatized products were afforded in excellent yields, chemo- and enantioselectivities when an electron-donating group (3i: 5-OMe, 3j: 5- Me) was installed on the indole ring (3i: 84% yield,> 19:1 3 i/4 i, 96% ee ; 3j: 86% yield, > 19:1 3 j/4 j, 92 % ee).
Remarkably, when an electron-withdrawing group was introduced on the indole ring (3k: 5-F; 3l: 5-Cl; 3m: 5-Br; 3n: 6-F; 3o: 6-Cl), good to excel- lent yields, C/N ratios and excellent enantioselectivity were obtained (3 k–3o: 73–77 % yields, 4.2:1 to 11.7:1 3/4, 92–94 % ee). Notably, the formation of al- lylic amination products was more favorable when stronger electron-withdrawing group was introduced (from 5-F to 5-Br, 6-F to 6-Cl). These are probably due to the increased acidity of the indole N@H and more easily formed N nucleophiles caused by the electron-withdrawing substituent. When the elec- tron-withdrawing group on substrate 1 was changed from methyl carbonate to ethyl carbonate, the desired product could also be obtained in good results (3p: 71% yield, 18.3 :1 3 p/4 p, 91% ee). Furthermore, when an aliphatic group was in- stalled at the C3 position, all substrates were well tolerated (3 q–3v: 71–85 % yields, > 19:1 3/4 in all cases, 92–94 % ee). The structure and absolute configuration of product 3r were confirmed by an X-ray crystallographic analysis of an enantio- pure example (see the Supporting Information for details). Moreover, substrate 1w bearing a nucleophilic chain at the C3 position underwent the reaction smoothly, affording the bridged indoline 3w in 71 % yield and 96 % ee.
To demonstrate the utility of the current method, a gram-scale reaction and several transformations of the product were carried out (Scheme 4). The asymmetric cascade dearomatization reaction of 1a with 2 on a 3.0 mmol scale provided product 3a in 84 % yield and 95 % ee. Under Pd/C hydrogena- tion conditions, the exocyclic double bond of product 3a was reduced and the benzyl group was also removed at the same time likely due to the driving force of aromatiza- tion, giving 5 in 92 % yield. The imine moiety in 3a could be reduced by NaBH3CN in acetic acid to afford 6 in 91 % yield. The relative configuration of 6 was determined by NOE analysis (for details, see the Sup- porting Information). Treatment of 6 with acetyl chloride and NaH produced 7 in quantitative yield. Finally, the double bond in 7 was oxidized with ozone to give ketone 8 in 64 % yield.
A proposed catalytic cycle is depicted in Scheme 5. The propargyl carbonate is acti- vated by Pd0 to give intermediate. Then, the nucleophilic side chain attacks intermediate to generate p-allyl palladium species. Upon the deprotonation of indole N@H, the C3 position of indole in III attacks p-allyl palla- dium moiety to deliver IV. Finally, product 3 is provided upon the liberation of palladium catalyst.
In conclusion, we have achieved an effi- cient palladium(0)-catalyzed intermolecular asymmetric cascade dearomatization reac- tion of indole derivatives with propargyl car- bonate. A series of multiply substituted fused indolenines were obtained in good to excellent yields, excellent chemo- and enan- tioselectivities. This method features mild reaction conditions and broad substrate scope. Further studies on the reaction mechanism are currently underway in our laboratory.
Acknowledgements
We thank the National Key R&D Program of China (2016YFA0202900), the National Basic Research Program of China (2015CB856600), NSFC (21572252, 21821002), Program of Shanghai Subject Chief Scientist (16XD1404300), and the CAS (XDB20000000, QYZDY-SSW-SLH012) for generous financial support.
Mac13243Catalog No.:AA008TEV CAS No.:1071638-38-4 MDL No.:MFCD18782745 MF:C20H25Cl2N3O2S MW:442.4024 |
3-amino-1-(3-ethynylphenyl)thioureaCatalog No.:AA01A9WT CAS No.:1071638-49-7 MDL No.:MFCD16817510 MF:C9H9N3S MW:191.2529 |
2-(dimethyltriaz-1-en-1-yl)benzoic acidCatalog No.:AA019OWL CAS No.:1071638-58-8 MDL No.:MFCD00013975 MF:C9H11N3O2 MW:193.2025 |
1,3-bis(bromomethyl)-5-iodobenzeneCatalog No.:AA01BTIB CAS No.:107164-93-2 MDL No.:MFCD22492791 MF:C8H7Br2I MW:389.8537 |
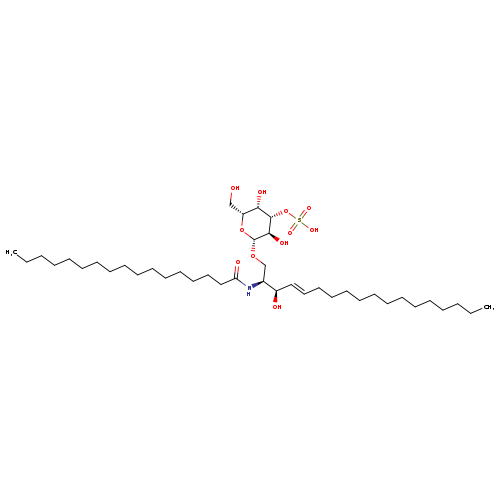
N-[(1S,2R,3E)-2-hydroxy-1-[[(3-O-sulfo-β-D-galactopyranosyl)oxy]methyl]-3-heptadecen-1-yl]-heptadecanamideCatalog No.:AA01EPUS CAS No.:1071654-84-6 MDL No.: MF:C41H79NO11S MW:794.1311 |
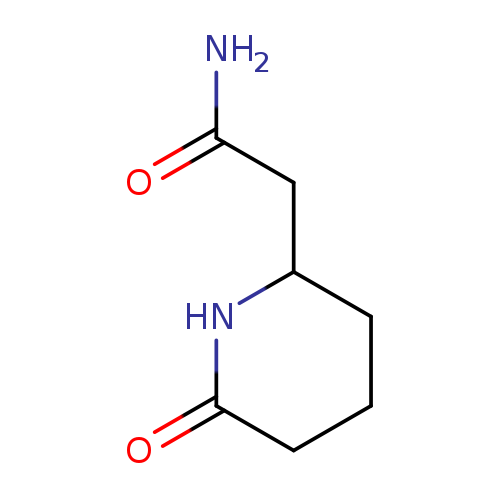
2-(6-Oxopiperidin-2-yl)acetamideCatalog No.:AA01AJ5Y CAS No.:1071661-20-5 MDL No.:MFCD19382158 MF:C7H12N2O2 MW:156.1824 |
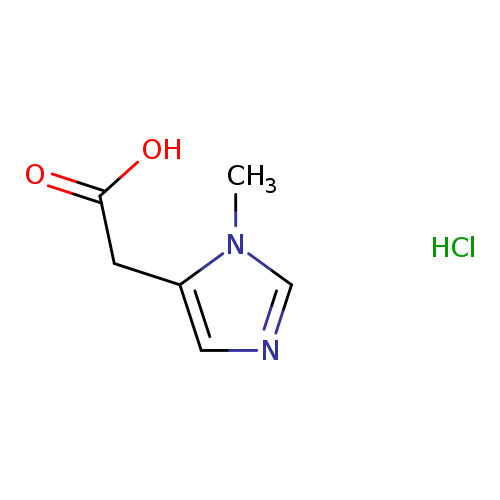
2-(1-Methyl-1h-imidazol-5-yl)acetic acid hydrochlorideCatalog No.:AA01AF3X CAS No.:1071661-55-6 MDL No.:MFCD27500744 MF:C6H9ClN2O2 MW:176.6009 |
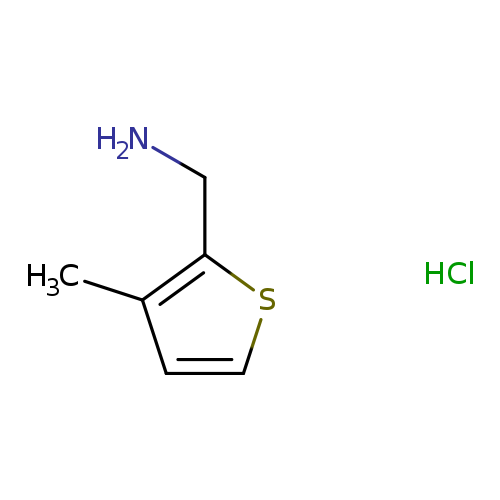
[(3-Methyl-2-thienyl)methyl]amine hydrochlorideCatalog No.:AA00HAV0 CAS No.:1071674-80-0 MDL No.:MFCD09971550 MF:C6H10ClNS MW:163.6683 |
3,4-diethylaniline hydrochlorideCatalog No.:AA01A4II CAS No.:1071678-12-0 MDL No.:MFCD18827535 MF:C10H16ClN MW:185.6937 |
N-[1-[2-(2-methylphenyl)ethyl]-4-piperidinyl]-N-phenyl-acetamide,monohydrochlorideCatalog No.:AA01EQ9T CAS No.:1071703-90-6 MDL No.: MF:C22H29ClN2O MW:372.9315 |
N-(1-(4-Methylphenethyl)piperidin-4-yl)-N-phenylacetamideHydrochlorideCatalog No.:AA01DZFI CAS No.:1071703-95-1 MDL No.:MFCD30186655 MF:C22H29ClN2O MW:372.9315 |
N-[1-[2-(4-methylphenyl)ethyl]-4-piperidinyl]-N-phenyl-propanamide,monohydrochlorideCatalog No.:AA01EPP7 CAS No.:1071703-97-3 MDL No.: MF:C23H31ClN2O MW:386.9580 |
N-[1-[2-(3-methylphenyl)ethyl]-4-piperidinyl]-N-phenyl-acetamide,monohydrochlorideCatalog No.:AA01EQ9P CAS No.:1071704-02-3 MDL No.: MF:C22H29ClN2O MW:372.9315 |
methyl 3-(2,4-dichlorophenoxy)propanoateCatalog No.:AA019Z2T CAS No.:1071715-08-6 MDL No.:MFCD00152418 MF:C10H10Cl2O3 MW:249.0906 |
(Z)-5H-Cyclopenta[b]pyridin-7(6h)-one o-methyl oximeCatalog No.:AA0096KM CAS No.:1071727-78-0 MDL No.:MFCD28975314 MF:C9H10N2O MW:162.1885 |
7-iodohept-1-eneCatalog No.:AA00KG7E CAS No.:107175-49-5 MDL No.:MFCD10570159 MF:C7H13I MW:224.0826 |
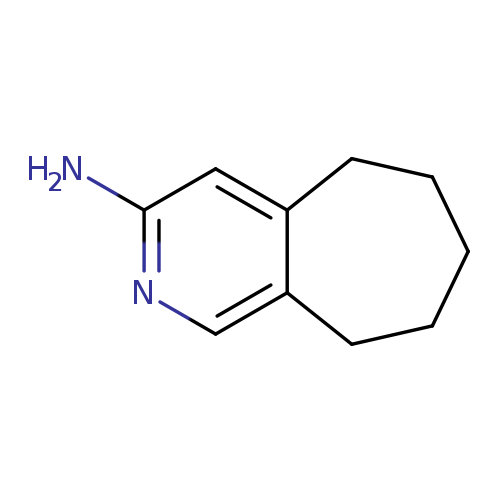
5H,6H,7H,8H,9H-cyclohepta[c]pyridin-3-amineCatalog No.:AA01DQQ9 CAS No.:1071763-80-8 MDL No.:MFCD22550643 MF:C10H14N2 MW:162.2316 |
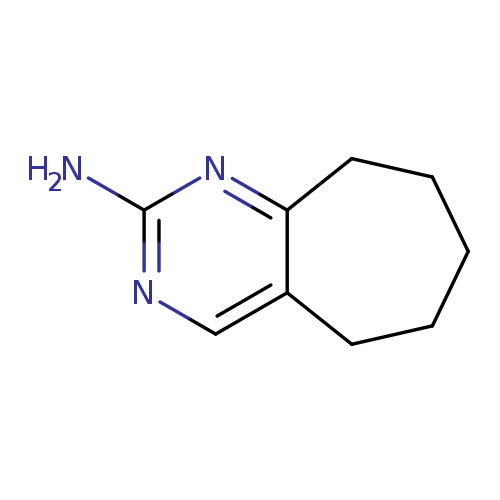
6,7,8,9-Tetrahydro-5h-cyclohepta[d]pyrimidin-2-amineCatalog No.:AA00HAV3 CAS No.:1071763-85-3 MDL No.:MFCD18361690 MF:C9H13N3 MW:163.2196 |
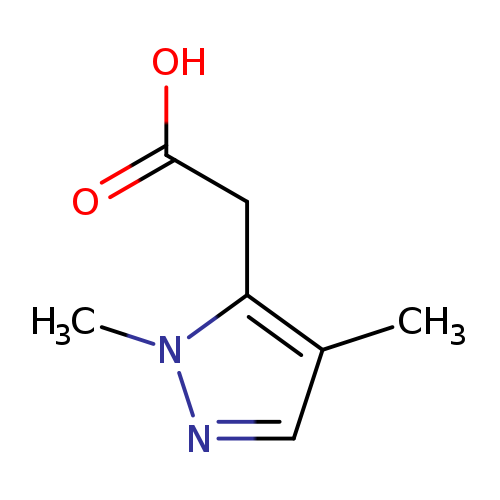
2-(1,4-dimethyl-1H-pyrazol-5-yl)acetic acidCatalog No.:AA01EK33 CAS No.:1071814-38-4 MDL No.:MFCD20660111 MF:C7H10N2O2 MW:154.1665 |
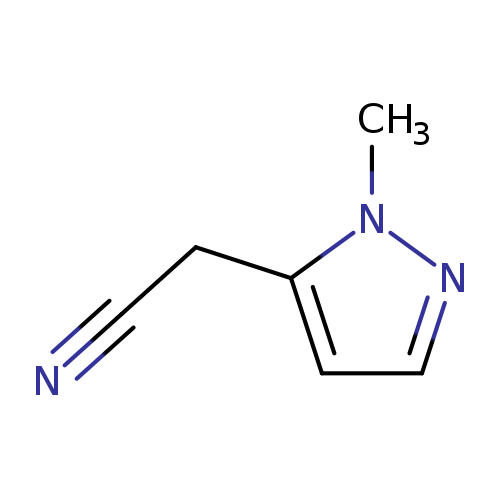
(1-Methyl-1h-pyrazol-5-yl)acetonitrileCatalog No.:AA00397V CAS No.:1071814-43-1 MDL No.:MFCD09859321 MF:C6H7N3 MW:121.1399 |
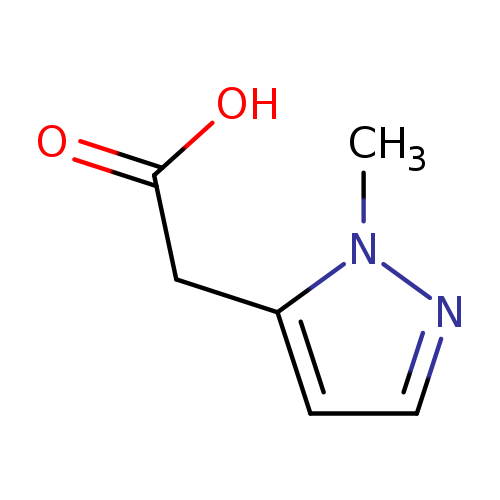
2-(1-Methyl-1h-pyrazol-5-yl)acetic acidCatalog No.:AA0096FO CAS No.:1071814-44-2 MDL No.:MFCD18262290 MF:C6H8N2O2 MW:140.1399 |
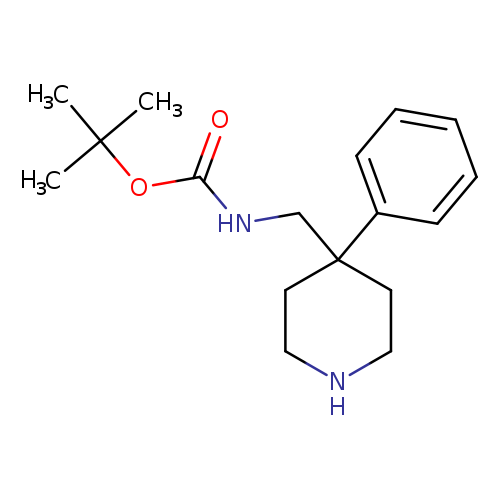
(4-Phenyl-piperidin-4-ylmethyl)-carbamic acid tert-butyl esterCatalog No.:AA00HAV4 CAS No.:1071866-01-7 MDL No.:MFCD09753905 MF:C17H26N2O2 MW:290.4005 |
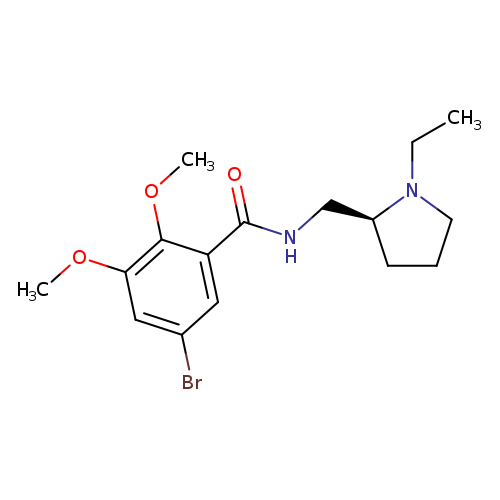
(S)-5-Bromo-N-((1-ethylpyrrolidin-2-yl)methyl)-2,3-dimethoxybenzamideCatalog No.:AA007T64 CAS No.:107188-74-9 MDL No.:MFCD00894744 MF:C16H23BrN2O3 MW:371.2694 |
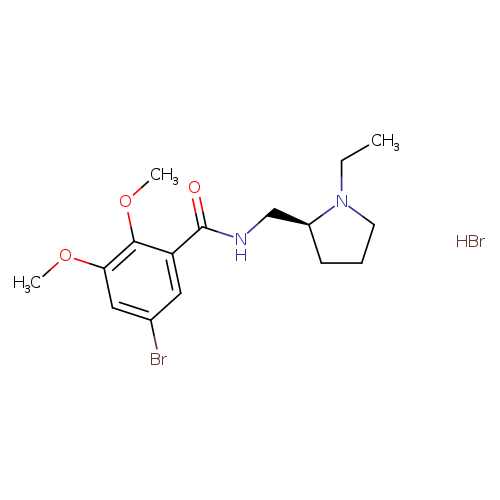
FLB 457 HydrobromideCatalog No.:AA007ARR CAS No.:107188-92-1 MDL No.:MFCD18252334 MF:C16H24Br2N2O3 MW:452.1814 |
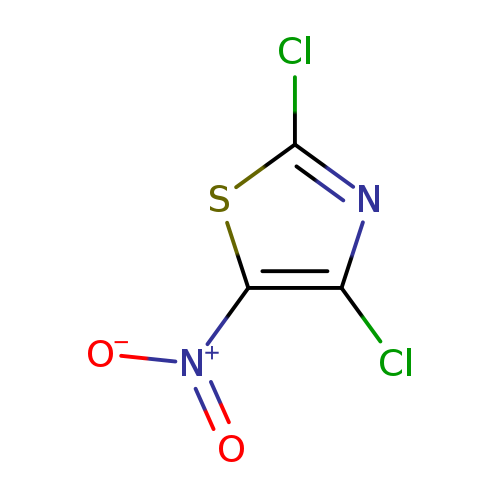
2,4-Dichloro-5-nitrothiazoleCatalog No.:AA009TAV CAS No.:107190-42-1 MDL No.:MFCD11131237 MF:C3Cl2N2O2S MW:199.0153 |
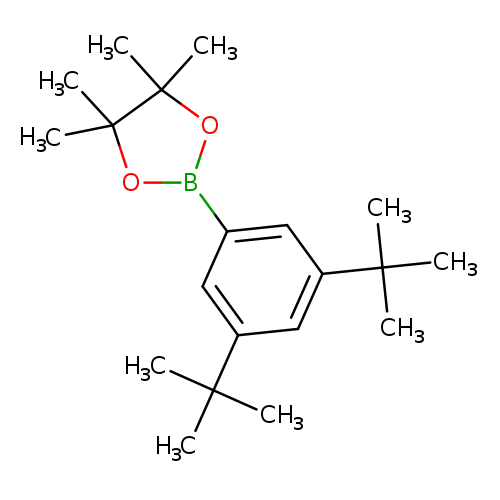
2-(3,5-Di-tert-butylphenyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolaneCatalog No.:AA0094NL CAS No.:1071924-13-4 MDL No.:MFCD24039797 MF:C20H33BO2 MW:316.2858 |
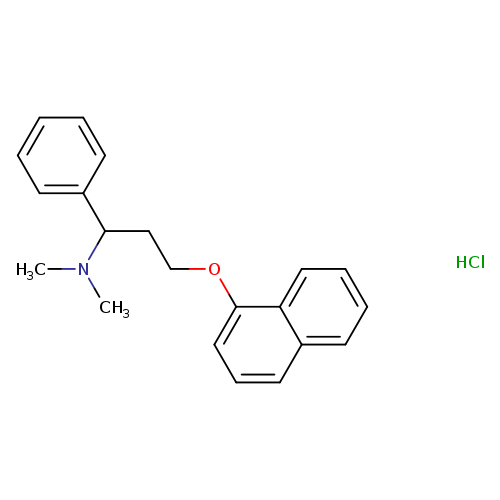
Dl-dapoxetine hclCatalog No.:AA008XLC CAS No.:1071929-03-7 MDL No.:MFCD20922882 MF:C21H24ClNO MW:341.8744 |
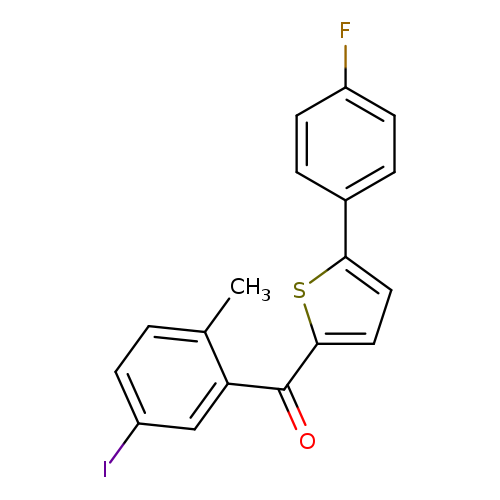
(5-(4-Fluorophenyl)thiophen-2-yl)(5-iodo-2-methylphenyl)methanoneCatalog No.:AA00HAV5 CAS No.:1071929-08-2 MDL No.:MFCD12405594 MF:C18H12FIOS MW:422.2552 |
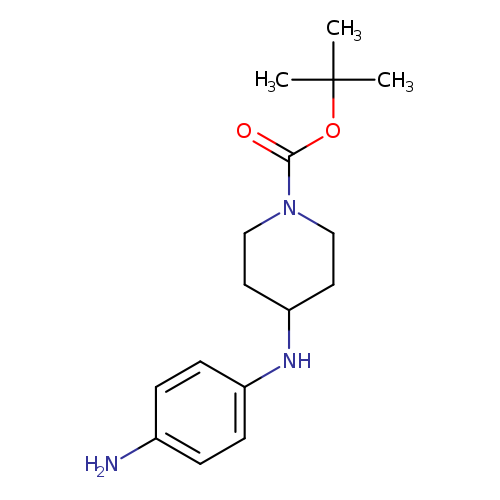
4-(4-Amino-phenylamino)-piperidine-1-carboxylic acid tert-butyl esterCatalog No.:AA007T63 CAS No.:1071932-29-0 MDL No.:MFCD12026437 MF:C16H25N3O2 MW:291.3886 |
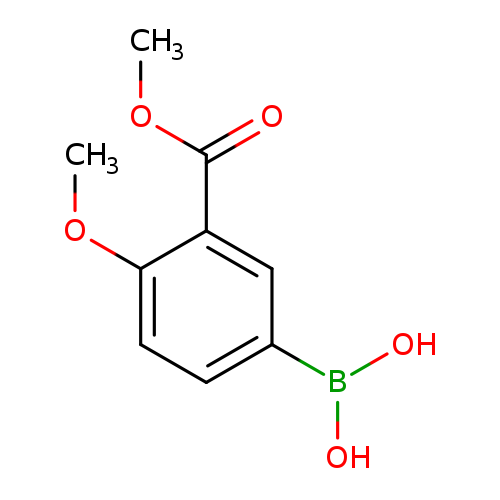
4-methoxy-3-(methoxycarbonyl)phenylboronic acidCatalog No.:AA00HAV6 CAS No.:1071958-96-7 MDL No.:MFCD23380361 MF:C9H11BO5 MW:209.9916 |
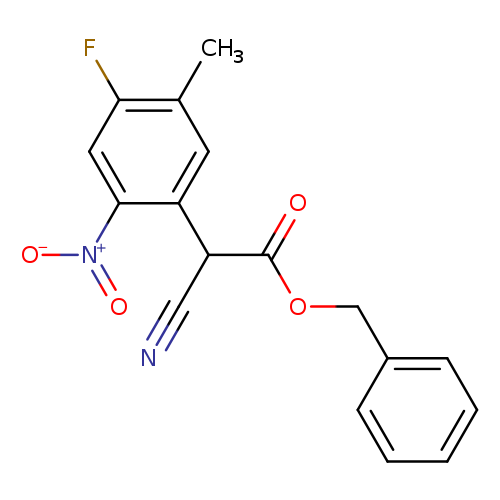
Benzyl 2-cyano-2-(4-fluoro-5-methyl-2-nitrophenyl)acetateCatalog No.:AA0096L1 CAS No.:1071974-01-0 MDL No.:MFCD28369447 MF:C17H13FN2O4 MW:328.2945 |
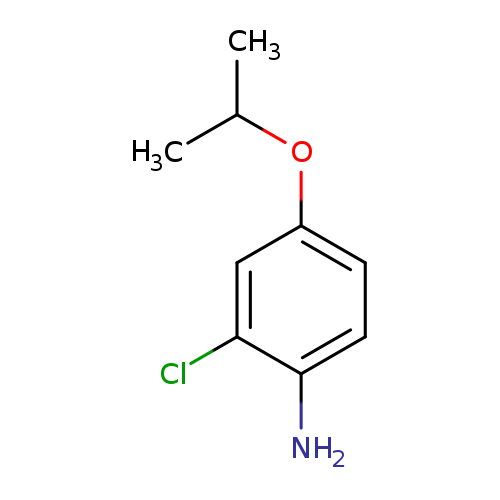
2-chloro-4-(propan-2-yloxy)anilineCatalog No.:AA01AG32 CAS No.:1071989-24-6 MDL No.:MFCD16680491 MF:C9H12ClNO MW:185.6507 |
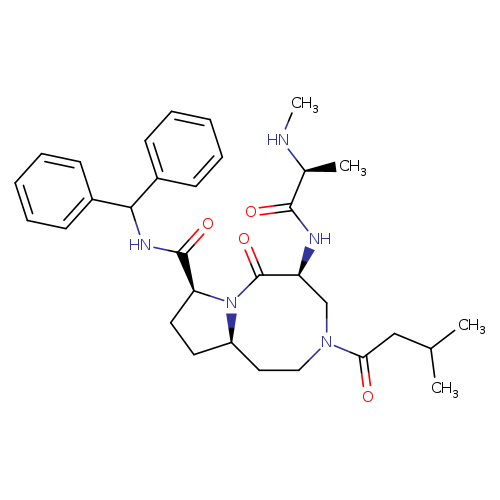
AT406Catalog No.:AA008WG6 CAS No.:1071992-99-8 MDL No.:MFCD22124467 MF:C32H43N5O4 MW:561.7149 |
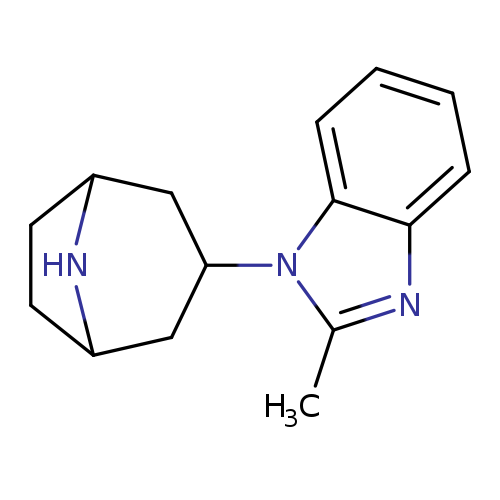
1-(8-Azabicyclo[3.2.1]oct-3-yl)-2-methyl-1h-benzimidazoleCatalog No.:AA00906Q CAS No.:1071993-26-4 MDL No.:MFCD08064258 MF:C15H19N3 MW:241.3315 |
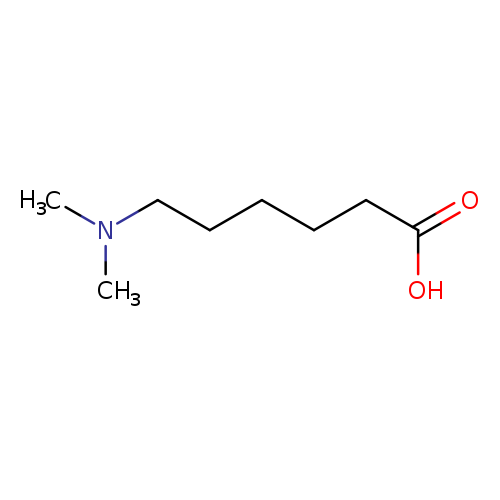
6-(dimethylamino)hexanoic acidCatalog No.:AA01EK2X CAS No.:1072-09-9 MDL No.:MFCD20318750 MF:C8H17NO2 MW:159.2261 |
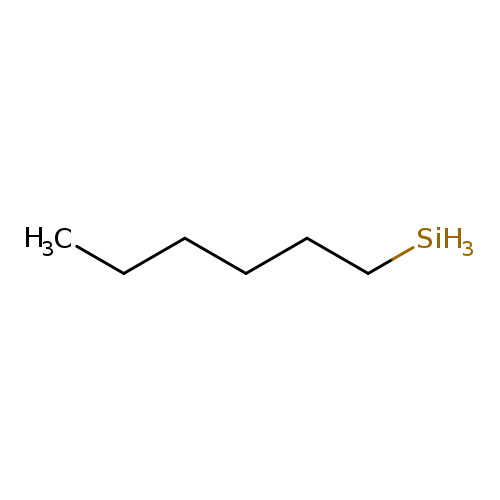
Silane, hexyl-Catalog No.:AA007T5T CAS No.:1072-14-6 MDL No.:MFCD00027291 MF:C6H16Si MW:116.2767 |
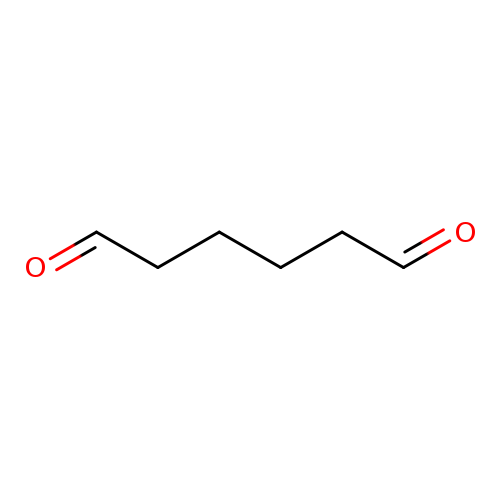
AdipaldehydeCatalog No.:AA008XJU CAS No.:1072-21-5 MDL No.:MFCD13186001 MF:C6H10O2 MW:114.1424 |
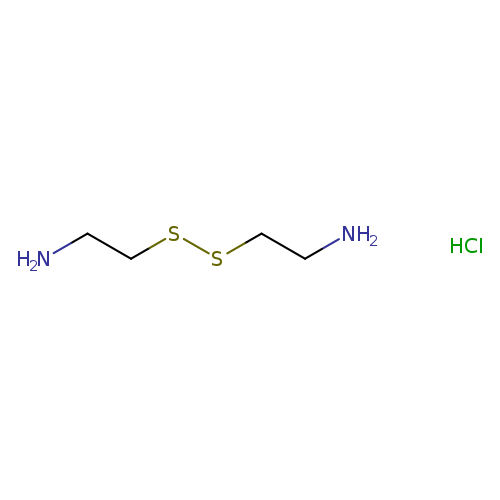
2-[(2-aminoethyl)disulfanyl]ethan-1-amine hydrochlorideCatalog No.:AA01A7VY CAS No.:1072-22-6 MDL No.:MFCD06804621 MF:C4H13ClN2S2 MW:188.7424 |
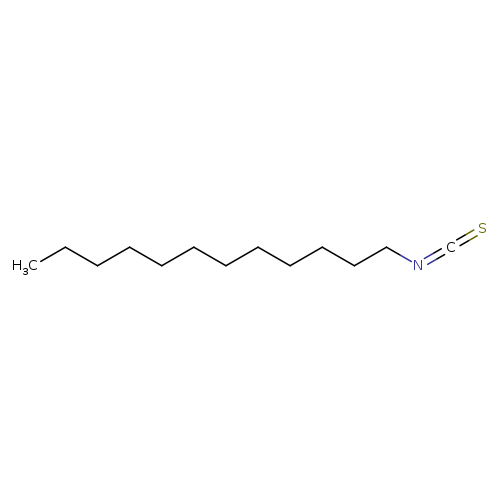
Dodecyl isothiocyanateCatalog No.:AA003PSY CAS No.:1072-32-8 MDL No.:MFCD00041121 MF:C13H25NS MW:227.4093 |
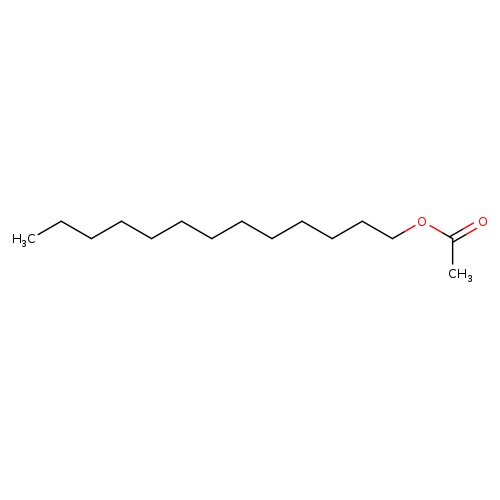
Acetic acid tridecyl esterCatalog No.:AA003UYF CAS No.:1072-33-9 MDL No.:MFCD00056197 MF:C15H30O2 MW:242.3975 |
2,5,8,11,14,17,20-HeptaoxaheneicosaneCatalog No.:AA0082C2 CAS No.:1072-40-8 MDL No.:MFCD00038357 MF:C14H30O7 MW:310.3838 |
4-Methyl-1,3-dioxolaneCatalog No.:AA003LMM CAS No.:1072-47-5 MDL No.:MFCD00014109 MF:C4H8O2 MW:88.1051 |
1-AziridineethanolCatalog No.:AA0038W6 CAS No.:1072-52-2 MDL No.:MFCD00005119 MF:C4H9NO MW:87.1204 |
EthylenesulfateCatalog No.:AA0037VT CAS No.:1072-53-3 MDL No.:MFCD00221769 MF:C2H4O4S MW:124.1158 |
CyclotetramethylenedimethylsilaneCatalog No.:AA007T5B CAS No.:1072-54-4 MDL No.:MFCD00040240 MF:C6H14Si MW:114.2609 |
1-Methoxy-1-cyclopenteneCatalog No.:AA009413 CAS No.:1072-59-9 MDL No.:MFCD30474836 MF:C6H10O MW:98.1430 |
1,3-Dioxolane,4-(chloromethyl)-Catalog No.:AA0082BV CAS No.:1072-61-3 MDL No.:MFCD02171168 MF:C4H7ClO2 MW:122.5502 |
2-EthylimidazoleCatalog No.:AA003B54 CAS No.:1072-62-4 MDL No.:MFCD00005192 MF:C5H8N2 MW:96.1304 |
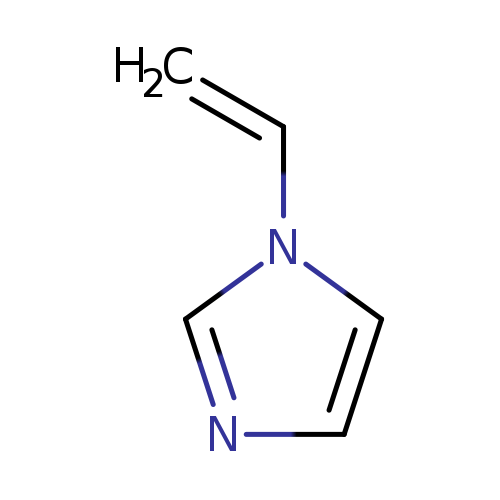
1-VinylimidazoleCatalog No.:AA00HAV8 CAS No.:1072-63-5 MDL No.:MFCD00005297 MF:C5H6N2 MW:94.1145 |
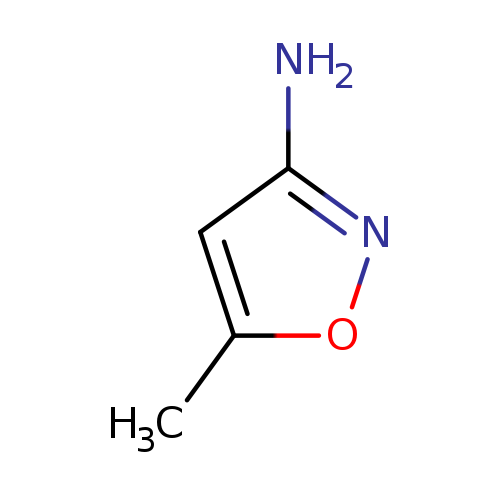
3-Amino-5-methylisoxazoleCatalog No.:AA003ITW CAS No.:1072-67-9 MDL No.:MFCD00003155 MF:C4H6N2O MW:98.1032 |
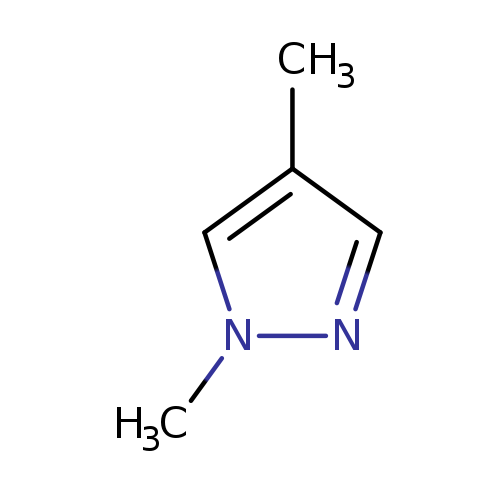
1,4-DimethylpyrazoleCatalog No.:AA003DM3 CAS No.:1072-68-0 MDL No.:MFCD01075158 MF:C5H8N2 MW:96.1304 |
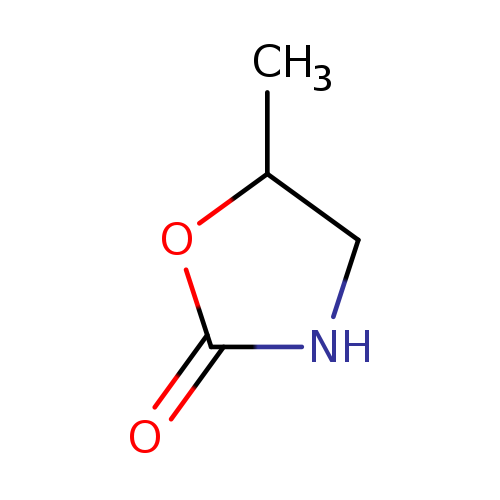
5-Methyl-1,3-oxazolidin-2-oneCatalog No.:AA007AQV CAS No.:1072-70-4 MDL No.:MFCD00068871 MF:C4H7NO2 MW:101.1039 |
1,3,4-Thiadiazolidine-2,5-dithioneCatalog No.:AA00HAV9 CAS No.:1072-71-5 MDL No.:MFCD00003103 MF:C2H2N2S3 MW:150.2457 |
Tetrahydro-4H-thiopyran-4-oneCatalog No.:AA003UMH CAS No.:1072-72-6 MDL No.:MFCD00006660 MF:C5H8OS MW:116.1814 |
3-AcetylpyrroleCatalog No.:AA0038W4 CAS No.:1072-82-8 MDL No.:MFCD00067759 MF:C6H7NO MW:109.1259 |

2-AcetylpyrroleCatalog No.:AA003G7Y CAS No.:1072-83-9 MDL No.:MFCD00005220 MF:C6H7NO MW:109.1259 |
1H-Imidazole-5-carboxylic acidCatalog No.:AA0032P2 CAS No.:1072-84-0 MDL No.:MFCD01365907 MF:C4H4N2O2 MW:112.0868 |
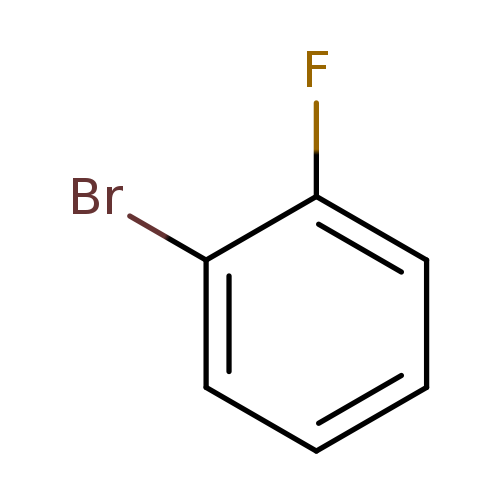
1-Bromo-2-fluorobenzeneCatalog No.:AA00333Q CAS No.:1072-85-1 MDL No.:MFCD00000282 MF:C6H4BrF MW:174.9984 |
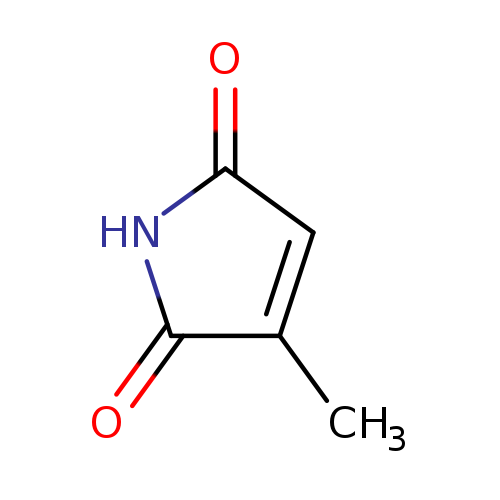
3-methylpyrrole-2,5-dioneCatalog No.:AA009QTF CAS No.:1072-87-3 MDL No.:MFCD10699598 MF:C5H5NO2 MW:111.0987 |
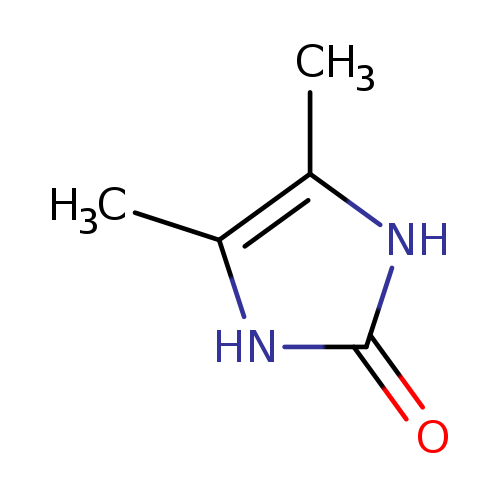
4,5-Dimethyl-1,3-dihydro-2h-imidazol-2-oneCatalog No.:AA007T4Y CAS No.:1072-89-5 MDL No.:MFCD00459895 MF:C5H8N2O MW:112.1298 |
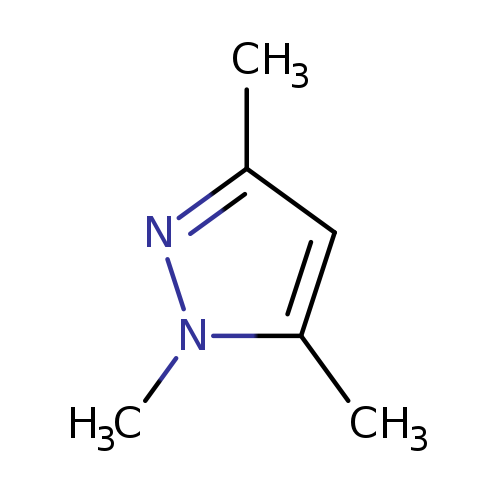
1,3,5-Trimethyl-1H-pyrazoleCatalog No.:AA003DDW CAS No.:1072-91-9 MDL No.:MFCD00015536 MF:C6H10N2 MW:110.1570 |
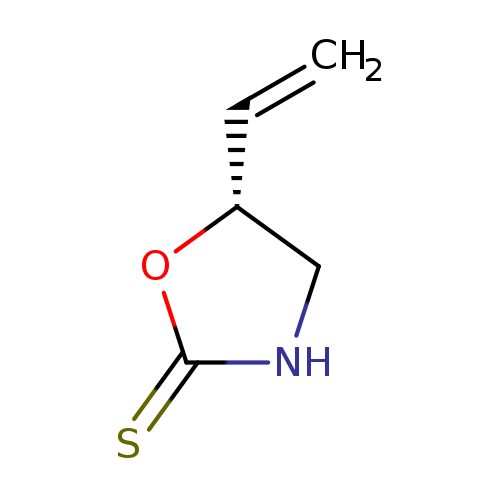
Ethyl-p-MethoxycinnamateCatalog No.:AA008RKP CAS No.:1072-93-1 MDL No.:MFCD01709801 MF:C5H7NOS MW:129.1802 |
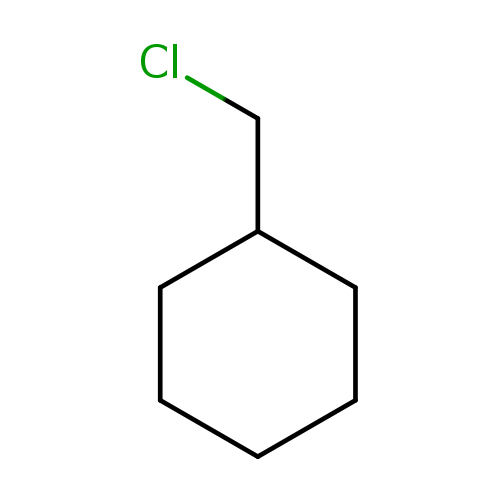
Cyclohexane, (chloromethyl)-Catalog No.:AA007WFC CAS No.:1072-95-3 MDL No.:MFCD09908175 MF:C7H13Cl MW:132.6311 |
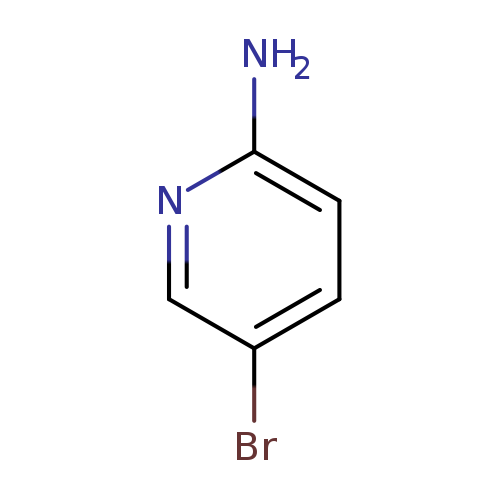
2-Amino-5-bromopyridineCatalog No.:AA00330T CAS No.:1072-97-5 MDL No.:MFCD00006323 MF:C5H5BrN2 MW:173.0106 |
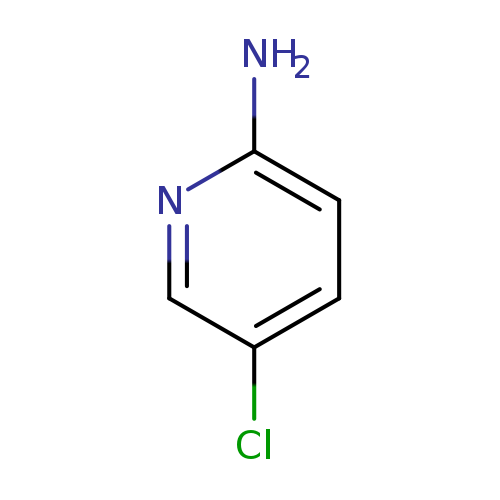
2-Amino-5-chloropyridineCatalog No.:AA003310 CAS No.:1072-98-6 MDL No.:MFCD00006324 MF:C5H5ClN2 MW:128.5596 |
5-Fluoro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1h-indoleCatalog No.:AA0094QF CAS No.:1072009-08-5 MDL No.:MFCD16987816 MF:C14H17BFNO2 MW:261.0997 |
Benzyl n-methyl-n-(2-oxoethyl)carbamateCatalog No.:AA007T60 CAS No.:107201-33-2 MDL No.:MFCD14635609 MF:C11H13NO3 MW:207.2258 |
(R)-3-Aminotetrahydrofuran, HClCatalog No.:AA0039BC CAS No.:1072015-52-1 MDL No.:MFCD08461720 MF:C4H10ClNO MW:123.5813 |
N-Boc-l-cyclohexylglycinolCatalog No.:AA003SR8 CAS No.:107202-39-1 MDL No.:MFCD04112591 MF:C13H25NO3 MW:243.3425 |
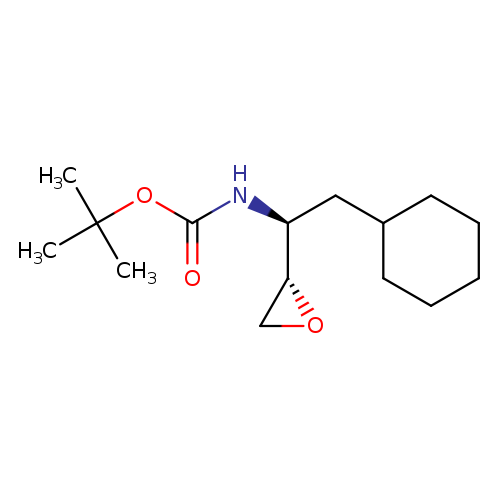
Erythro-n-boc-l-cyclohexylalanine epoxideCatalog No.:AA008R2J CAS No.:107202-62-0 MDL No.:MFCD08061628 MF:C15H27NO3 MW:269.3798 |
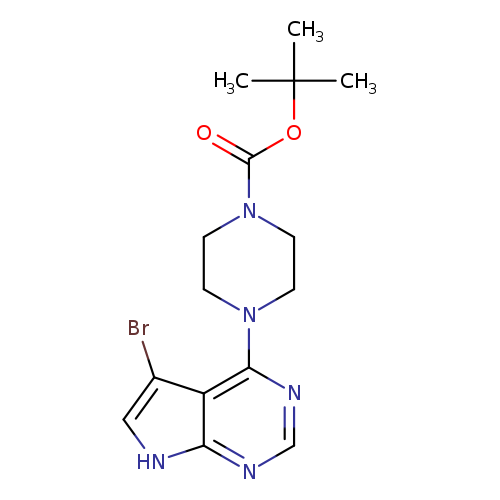
4-(4-Boc-1-piperazinyl)-5-bromo-7h-pyrrolo[2,3-d]pyrimidineCatalog No.:AA003JZG CAS No.:1072027-36-1 MDL No.:MFCD18642459 MF:C15H20BrN5O2 MW:382.2556 |
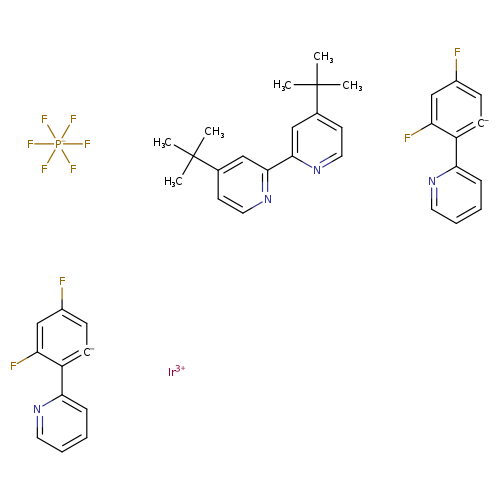
iridium(3+) ion bis(3,5-difluoro-2-(pyridin-2-yl)benzen-1-ide) 4-tert-butyl-2-(4-tert-butylpyridin-2-yl)pyridine hexafluoro-lambda5-phosphanuideCatalog No.:AA01EIFJ CAS No.:1072067-44-7 MDL No.:MFCD31657504 MF:C40H36F10IrN4P MW:985.9154 |
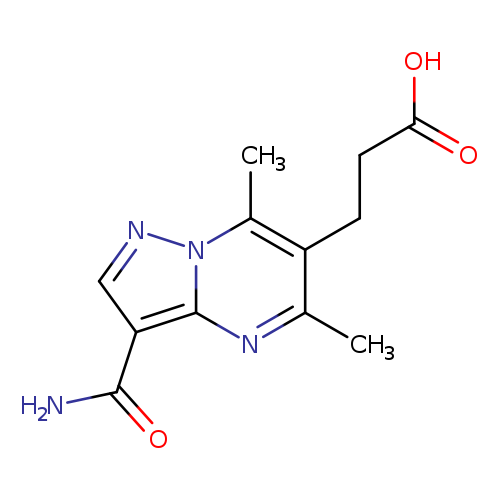
3-{3-carbamoyl-5,7-dimethylpyrazolo[1,5-a]pyrimidin-6-yl}propanoic acidCatalog No.:AA019UQC CAS No.:1072069-75-0 MDL No.:MFCD11857944 MF:C12H14N4O3 MW:262.2646 |
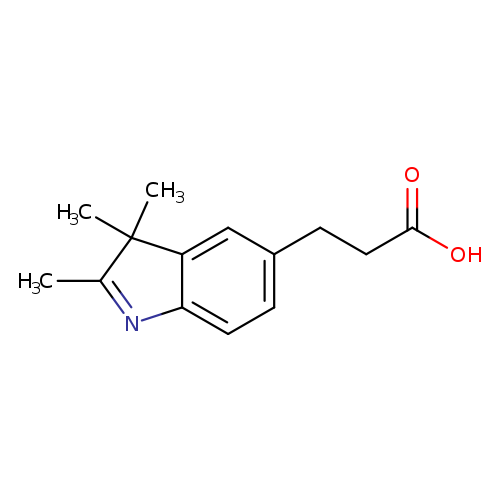
3-(2,3,3-trimethyl-3H-indol-5-yl)propanoic acidCatalog No.:AA00906R CAS No.:1072069-91-0 MDL No.:MFCD12922972 MF:C14H17NO2 MW:231.2903 |
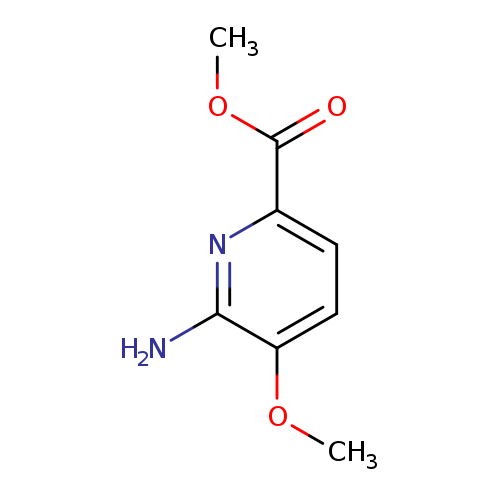
6-Amino-5-methoxy-pyridine-2-carboxylic acid methyl esterCatalog No.:AA0093KM CAS No.:1072077-54-3 MDL No.:MFCD18261109 MF:C8H10N2O3 MW:182.1766 |
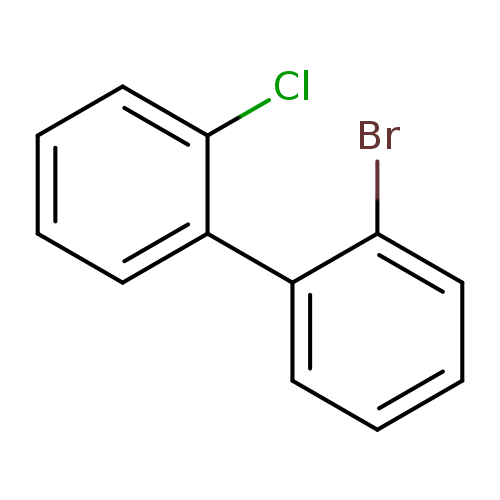
2-bromo-2-chloro-1,1-biphenylCatalog No.:AA019EQZ CAS No.:107208-70-8 MDL No.:MFCD00466291 MF:C12H8BrCl MW:267.5489 |
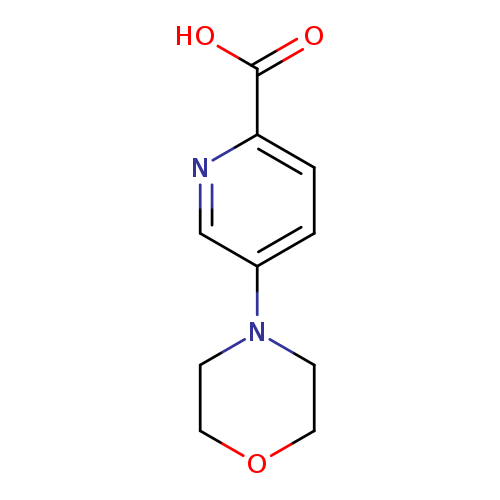
5-Morpholinopicolinic acidCatalog No.:AA00995G CAS No.:1072103-29-7 MDL No.:MFCD18251373 MF:C10H12N2O3 MW:208.2139 |
(1R)-2-chloro-1-[4-(trifluoromethyl)phenyl]ethan-1-olCatalog No.:AA01BCL2 CAS No.:1072105-58-8 MDL No.:MFCD24346567 MF:C9H8ClF3O MW:224.6074 |
3-(5-AMINO-3-METHYL-1H-1,2,4-TRIAZOL-1-YL)PROPANENITRILECatalog No.:AA01EJML CAS No.:1072106-44-5 MDL No.:MFCD16845192 MF:C6H9N5 MW:151.1692 |
3-(5-amino-3-phenyl-1H-1,2,4-triazol-1-yl)propanenitrileCatalog No.:AA01EJQL CAS No.:1072106-46-7 MDL No.:MFCD24641918 MF:C11H11N5 MW:213.2385 |
2-METHYL-6H,7H-[1,2,4]TRIAZOLO[1,5-A]PYRIMIDIN-5-AMINECatalog No.:AA01ELLE CAS No.:1072106-47-8 MDL No.:MFCD24641915 MF:C6H9N5 MW:151.1692 |
3-Iodo-5-nitropyridin-4-olCatalog No.:AA008SAD CAS No.:1072140-97-6 MDL No.:MFCD11052833 MF:C5H3IN2O3 MW:265.9934 |
4-Chloro-1h-pyrrolo[2,3-b]pyridine-5-boronic acid pinacol esterCatalog No.:AA008U3S CAS No.:1072145-24-4 MDL No.:MFCD09965897 MF:C13H16BClN2O2 MW:278.5423 |
1,2-Difluoro-4-[2-[(trans,trans)-4'-propyl[1,1'-bicyclohexyl]-4-yl]ethyl]benzeneCatalog No.:AA0094QB CAS No.:107215-66-7 MDL No.:MFCD28963786 MF:C23H34F2 MW:348.5129 |
Ethyl 2-(3-amino-2-hydroxyphenyl)acetateCatalog No.:AA01B8DM CAS No.:1072150-23-2 MDL No.:MFCD24642393 MF:C10H13NO3 MW:195.2151 |
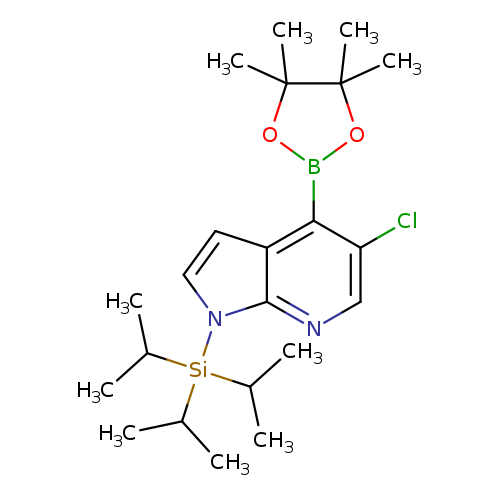
5-Chloro-1-(triisopropylsilyl)-1h-pyrrolo[2,3-b]pyridine-4-boronic acid pinacol esterCatalog No.:AA007ARF CAS No.:1072152-34-1 MDL No.:MFCD09965900 MF:C22H36BClN2O2Si MW:434.8829 |
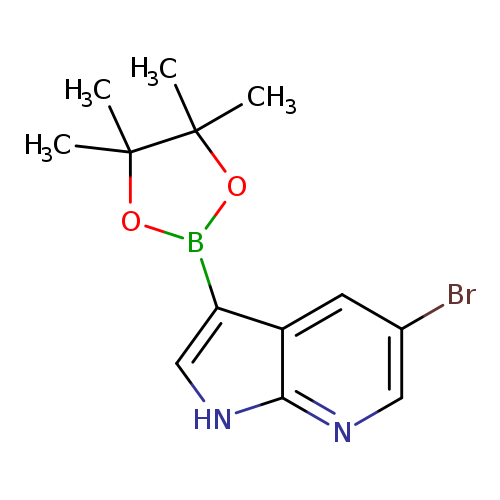
5-Bromo-3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1h-pyrrolo[2,3-b]pyridineCatalog No.:AA0094GZ CAS No.:1072152-50-1 MDL No.:MFCD10574985 MF:C13H16BBrN2O2 MW:322.9933 |
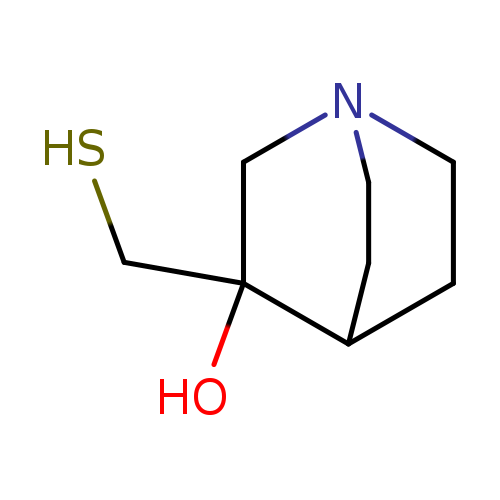
rac3-Hydroxy-3-mercaptomethylquinuclidineCatalog No.:AA01DZFL CAS No.:107220-26-8 MDL No.:MFCD11042346 MF:C8H15NOS MW:173.2758 |
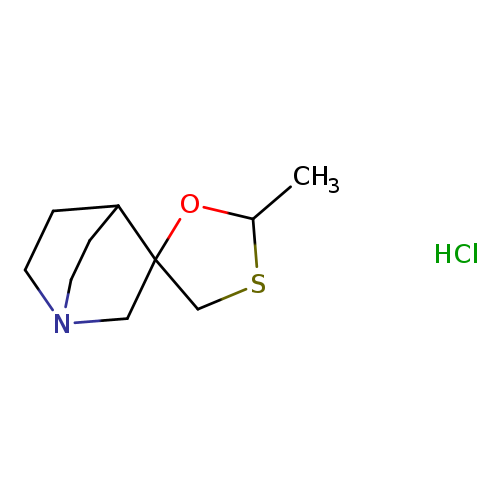
Cevimeline hydrochlorideCatalog No.:AA007ARD CAS No.:107220-28-0 MDL No.:MFCD01961045 MF:C10H18ClNOS MW:235.7740 |
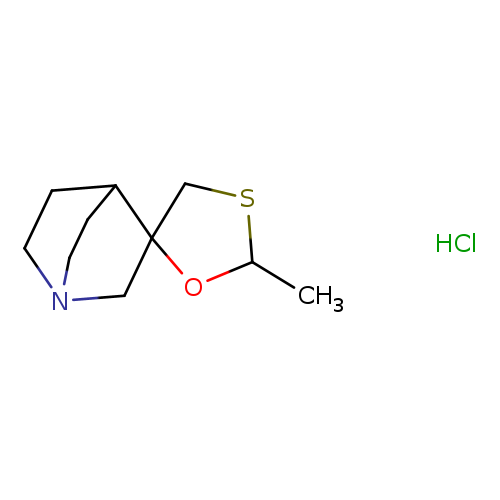
trans-CevimelineHydrochlorideCatalog No.:AA01DZFM CAS No.:107220-29-1 MDL No.:MFCD01961046 MF:C10H18ClNOS MW:235.7740 |
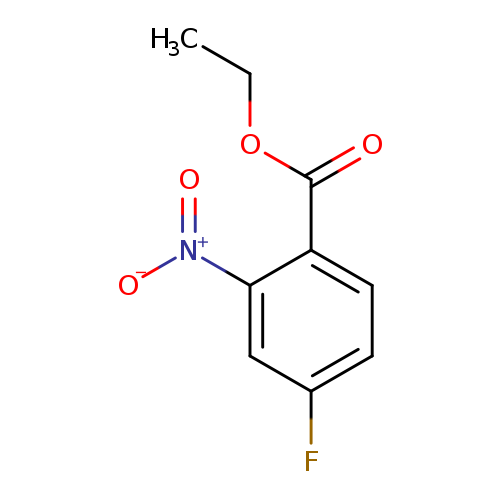
Ethyl 4-fluoro-2-nitrobenzoateCatalog No.:AA009NXM CAS No.:1072207-10-3 MDL No.:MFCD10566267 MF:C9H8FNO4 MW:213.1625 |
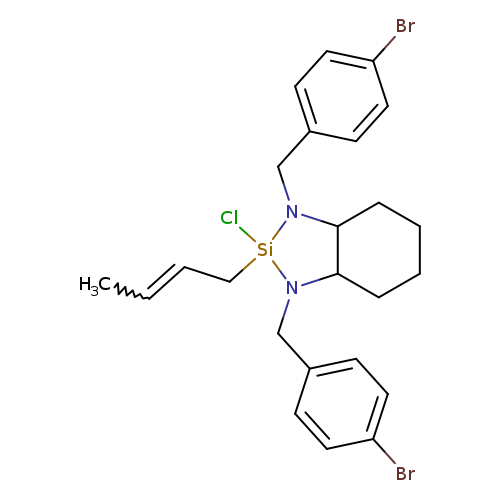
(S,S)-1,3-Bis(4-bromobenzyl)-2-chlorooctahydro-2-(2E)-crotyl-1H-1,3,2-benzodiazasiloleCatalog No.:AA003CM4 CAS No.:1072220-37-1 MDL No.:MFCD18632567 MF:C24H29Br2ClN2Si MW:568.8470 |
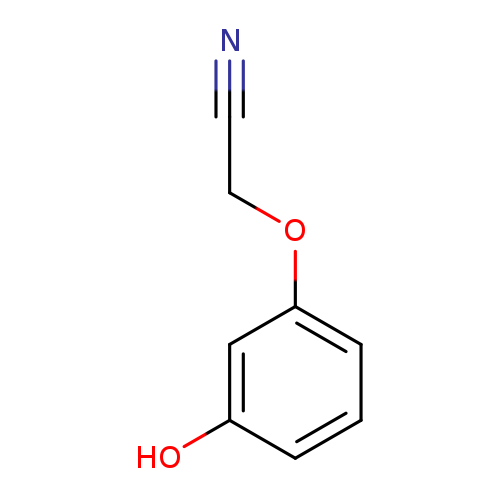
2-(3-Hydroxyphenoxy)acetonitrileCatalog No.:AA00HAVG CAS No.:107223-69-8 MDL No.:MFCD11181964 MF:C8H7NO2 MW:149.1467 |
3-Methyl-1H-pyrazolo[3,4-c]pyridineCatalog No.:AA0082CB CAS No.:1072249-89-8 MDL No.:MFCD13191779 MF:C7H7N3 MW:133.1506 |
3,6-Dichloro-4,5-diethylpyridazineCatalog No.:AA007AR7 CAS No.:107228-53-5 MDL No.:MFCD17014878 MF:C8H10Cl2N2 MW:205.0844 |
3,6-Dichloro-4-ethylpyridazineCatalog No.:AA0037VU CAS No.:107228-54-6 MDL No.:MFCD17014877 MF:C6H6Cl2N2 MW:177.0312 |
3,6-Dichloro-4-cyclobutylpyridazineCatalog No.:AA00HAVH CAS No.:107228-57-9 MDL No.:MFCD25955161 MF:C8H8Cl2N2 MW:203.0685 |
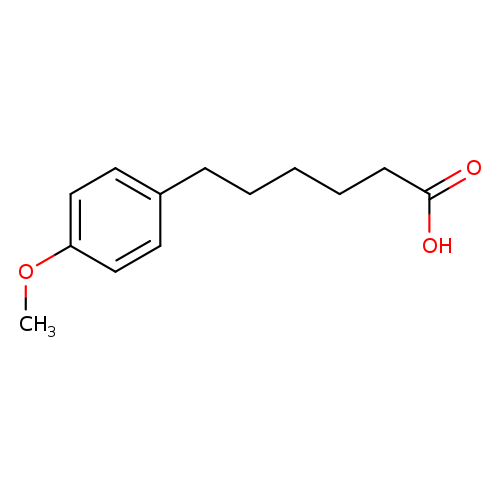
6-(4-Methoxyphenyl)hexanoic acidCatalog No.:AA01AA0M CAS No.:107228-87-5 MDL No.:MFCD12172613 MF:C13H18O3 MW:222.2802 |
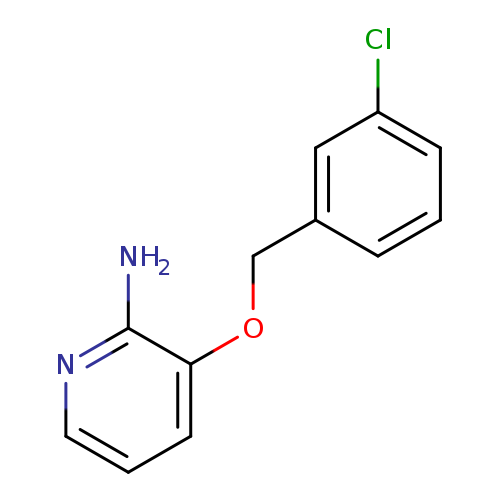
2-Pyridinamine, 3-[(3-chlorophenyl)methoxy]-Catalog No.:AA0082C5 CAS No.:107229-62-9 MDL No.:MFCD11195108 MF:C12H11ClN2O MW:234.6815 |
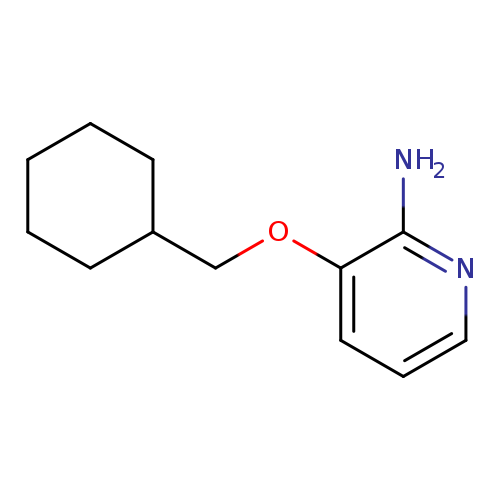
3-(cyclohexylmethoxy)pyridin-2-amineCatalog No.:AA01A8DY CAS No.:107229-69-6 MDL No.:MFCD10689932 MF:C12H18N2O MW:206.2841 |
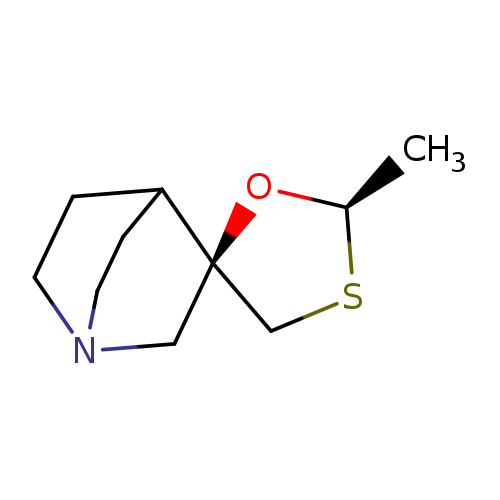
CevimelineCatalog No.:AA0082C4 CAS No.:107233-08-9 MDL No.:MFCD22577792 MF:C10H17NOS MW:199.3131 |
1-(2-Chloropropanoyl)indolineCatalog No.:AA009PNU CAS No.:107236-27-1 MDL No.:MFCD05263162 MF:C11H12ClNO MW:209.6721 |
Zirconium 1,4-dicarboxybenzene MOF (UiO-66)Catalog No.:AA01FOVW CAS No.:1072413-89-8 MDL No.:MFCD29037163 MF:C48H24O32Zr6 MW:1660.0290 |
5-Iodo-1-methyl-1h-indazoleCatalog No.:AA007AR0 CAS No.:1072433-59-0 MDL No.:MFCD11977535 MF:C8H7IN2 MW:258.0590 |
methyl 4-(aminomethyl)-3-fluorobenzoate hydrochlorideCatalog No.:AA01B6DP CAS No.:1072438-51-7 MDL No.:MFCD17171306 MF:C9H11ClFNO2 MW:219.6405 |
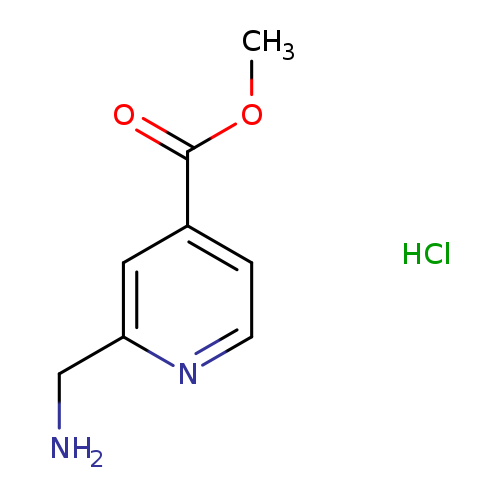
Methyl 2-(aminomethyl)pyridine-4-carboxylate, HClCatalog No.:AA007T5E CAS No.:1072438-54-0 MDL No.:MFCD09878665 MF:C8H11ClN2O2 MW:202.6381 |
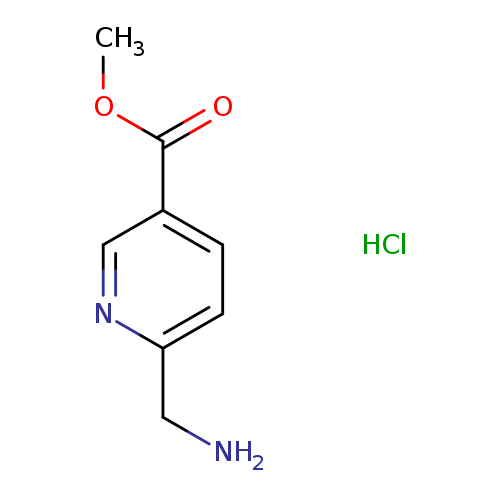
Methyl 6-(aminomethyl)nicotinate hydrochlorideCatalog No.:AA008ZD7 CAS No.:1072438-56-2 MDL No.:MFCD11045450 MF:C8H11ClN2O2 MW:202.6381 |
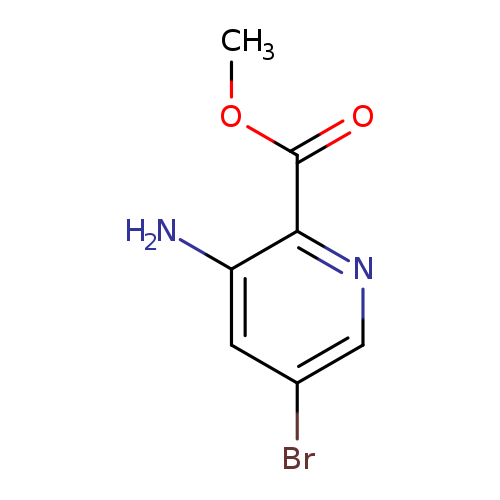
Methyl 3-amino-5-bromopicolinateCatalog No.:AA008U25 CAS No.:1072448-08-8 MDL No.:MFCD17015929 MF:C7H7BrN2O2 MW:231.0467 |
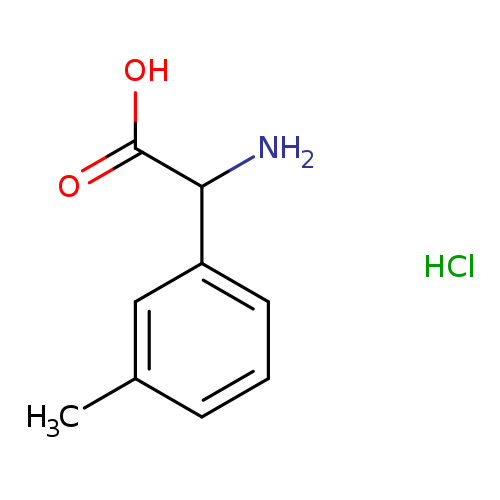
2-Amino-2-(3-methylphenyl)acetic acid HClCatalog No.:AA0082C0 CAS No.:1072449-62-7 MDL No.:MFCD06410554 MF:C9H12ClNO2 MW:201.6501 |
1-(Dimethylamino)-6-methylheptan-3-oneCatalog No.:AA00HAVI CAS No.:107245-26-1 MDL No.:MFCD22683883 MF:C10H21NO MW:171.2798 |

monoMETHACRYLOXYPROPYL FUNCTIONAL POLYTRIFLUOROPROPYLMETHYLSILOXANE, symmetric, 50-70 cStCatalog No.:AA01EIBU CAS No.:1072456-00-8 MDL No.: MF: MW: |
1-Fmoc-4-(hydroxymethyl)piperidineCatalog No.:AA00995K CAS No.:1072502-03-4 MDL No.:MFCD02094489 MF:C21H23NO3 MW:337.4122 |
5-Nitro-2-((3-phenylpropyl)amino)benzoic acidCatalog No.:AA00385A CAS No.:107254-86-4 MDL No.:MFCD00153851 MF:C16H16N2O4 MW:300.3092 |
4-[[3-[2-(3-Chlorophenyl)ethyl]-2-pyridinyl]carbonyl]-1-piperidinecarboxylic AcidCatalog No.:AA008W2U CAS No.:107256-32-6 MDL No.:MFCD16251304 MF:C22H25ClN2O3 MW:400.8985 |
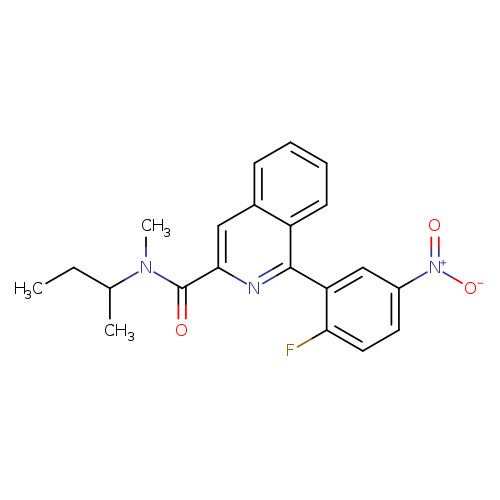
Pk14105Catalog No.:AA003TQE CAS No.:107257-28-3 MDL No.:MFCD28969041 MF:C21H20FN3O3 MW:381.4002 |
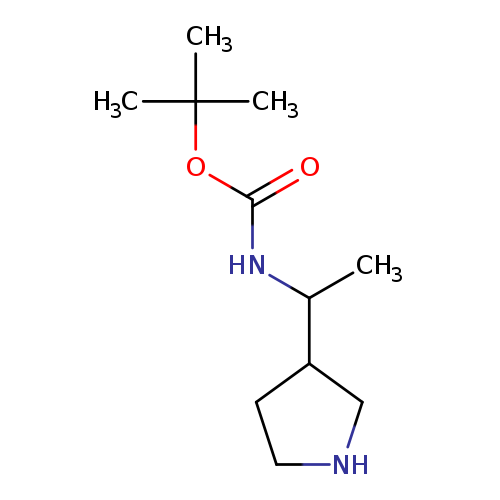
3-(1-(Boc-amino)ethyl)-pyrrolidineCatalog No.:AA00HAVK CAS No.:107258-90-2 MDL No.:MFCD18073167 MF:C11H22N2O2 MW:214.3046 |
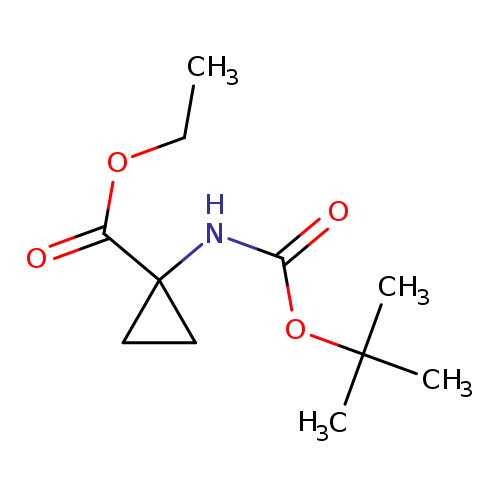
Ethyl 1-(boc-amino)cyclopropanecarboxylateCatalog No.:AA003PXD CAS No.:107259-05-2 MDL No.:MFCD11845623 MF:C11H19NO4 MW:229.2729 |
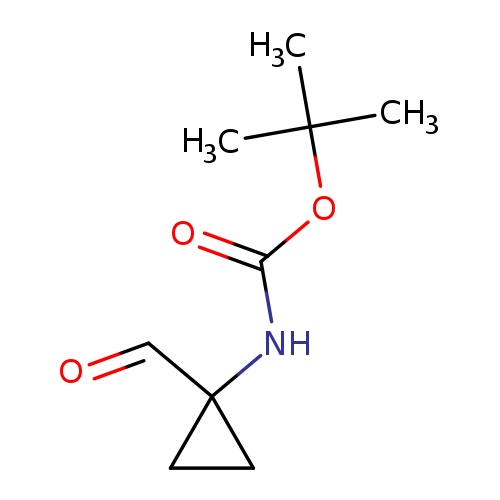
(1-Formyl-cyclopropyl)-carbamic acid tert-butyl esterCatalog No.:AA0037VS CAS No.:107259-06-3 MDL No.:MFCD10697899 MF:C9H15NO3 MW:185.2203 |
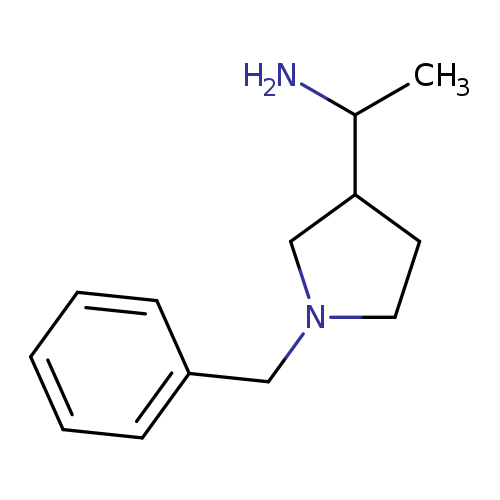
1-(1-Benzylpyrrolidin-3-yl)ethanamineCatalog No.:AA01A52R CAS No.:107259-16-5 MDL No.:MFCD22057269 MF:C13H20N2 MW:204.3113 |
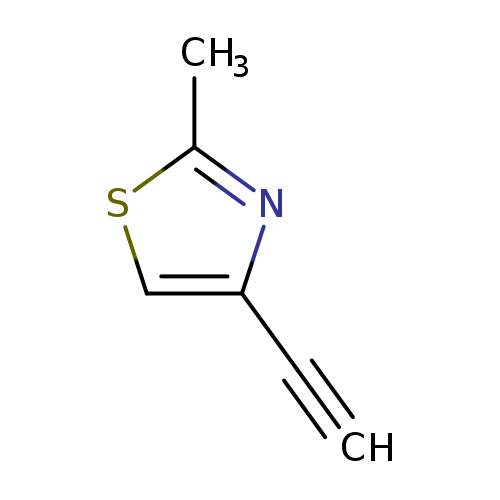
4-Ethynyl-2-methylthiazoleCatalog No.:AA008U6X CAS No.:107263-89-8 MDL No.:MFCD18207414 MF:C6H5NS MW:123.1756 |
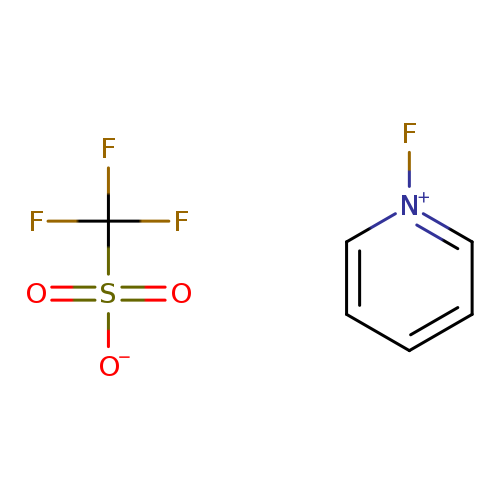
1-Fluoropyridinium TriflateCatalog No.:AA003AZ0 CAS No.:107263-95-6 MDL No.:MFCD00013458 MF:C6H5F4NO3S MW:247.1674 |
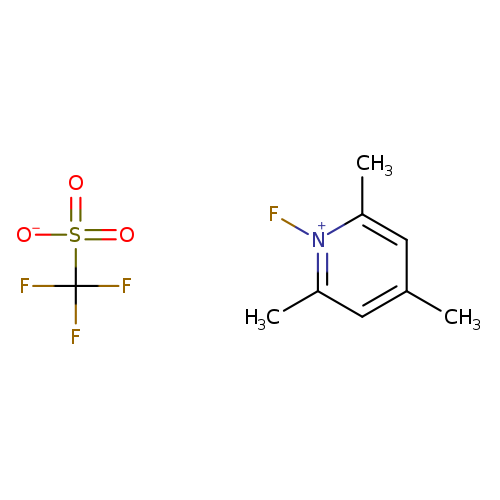
1-Fluoro-2,4,6-trimethylpyridin-1-ium trifluoromethanesulfonateCatalog No.:AA003E92 CAS No.:107264-00-6 MDL No.:MFCD00067525 MF:C9H11F4NO3S MW:289.2472 |
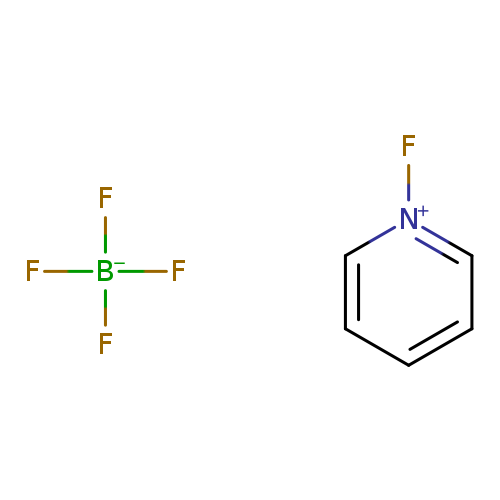
1-Fluoropyridinium tetrafluoroborateCatalog No.:AA003E9M CAS No.:107264-09-5 MDL No.:MFCD00153176 MF:C5H5BF5N MW:184.9029 |
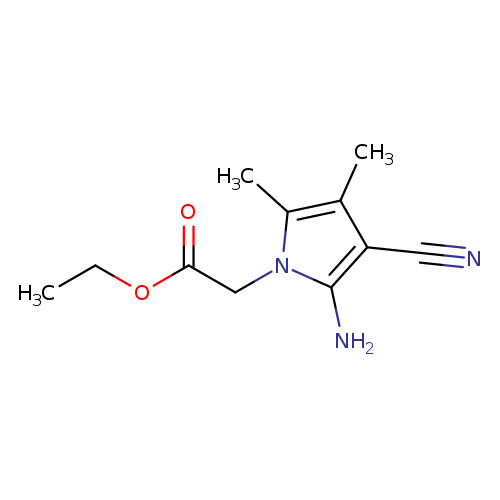
ethyl 2-(2-amino-3-cyano-4,5-dimethyl-1H-pyrrol-1-yl)acetateCatalog No.:AA00IYYI CAS No.:107267-88-9 MDL No.:MFCD04126142 MF:C11H15N3O2 MW:221.2557 |
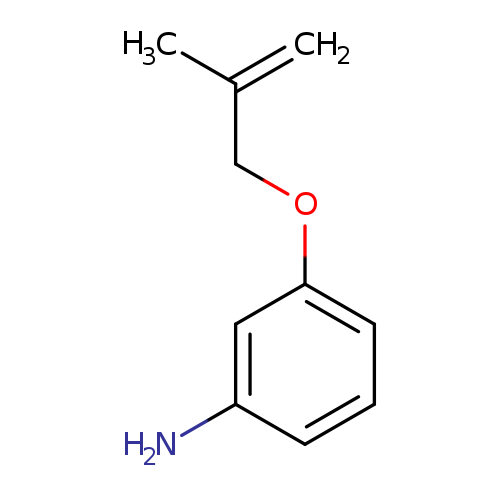
3-((2-Methylallyl)oxy)anilineCatalog No.:AA0082BS CAS No.:107268-37-1 MDL No.:MFCD06801092 MF:C10H13NO MW:163.2163 |
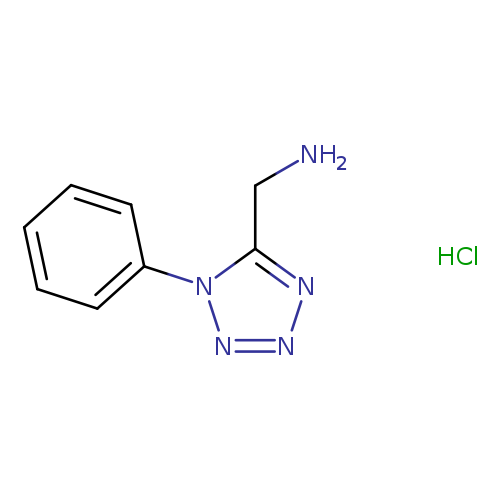
IFLAB-BB F1703-0109Catalog No.:AA008X8V CAS No.:107269-65-8 MDL No.:MFCD06623794 MF:C8H10ClN5 MW:211.6515 |