2019-11-17 09:07:23
Yosuke Nakamura, Kyoji O-kawa, Satoshi Minami, Toshio Ogawa, Seiji Tobita, and Jun Nishimura*
Department of Chemistry, Gunma University, Tenjin-cho, Kiryu, Gunma 376-8515, Japan
[email protected]
Received September 20, 2001
Introduction
Among the functionalizations of [60]fullerene, DielsAlder reactions with o-quinodimethanes as dienes are very versatile because they provide thermally stable adducts that are usually not subjected to cycloreversion into their original components.1-6 The high stability is provided by the aromatic system generated in the adducts. o-Quinodimethane derivatives have been obtained as reactive intermediates, mainly from the thermolysis of benzocyclobutenes, the SO2 extrusion of sulfolenes or sultines, or the iodide-induced 1,4 elimination of 1,2-bis-(bromomethyl)benzene derivatives.7 These precursors, however, are not necessarily easily available. Furthermore, the generation of o-quinodimethane from benzocyclobutenes requires rather high temperatures, which can lead to undesirable side reactions. In the iodideinduced 1,4 eliminations of R,R′-dihalides, appropriate phase-transfer catalysts such as 18-crown-6 are necessary in addition to iodide sources because [60]fullerene is almost insoluble in many polar organic solvents such as DMF.
The photoirradiation of o-tolualdehyde (1a) and related carbonyl compounds is known to give an o-quinodimethane species carrying a hydroxy group via the biradical generated by the intramolecular hydrogen abstraction of the carbonyl group in the excited triplet state (n-π*) from the neighboring methyl group.8 o-Quinodimethane readily reacts with dienophiles, though it partially reverts to the
starting material. Hence, in principle, it seems possible to obtain [60]fullerene-o-quinodimethane adducts by using [60]fullerene as a dienophile. However, it is rather difficult to efficiently produce the photoexcited states of aromatic carbonyl compounds in the presence of [60]-fullerene because the latter has stronger absorption bands throughout most of the UV region. For this reason, many of the known photochemical reactions involving[60]fullerene have taken advantage of the excited triplet state of [60]fullerene formed by intersystem crossing from the singlet excited state.9

In this context, Tomioka et al. have examined the photoreaction of o-methylbenzophenone (1f) with [60]-fullerene.10 Unexpectedly, the desired adduct 2f was so unstable that they isolated only the monoalkyl-1,2-dihydrofullerene by the cleavage of the C-C bond connected to the fullerene core. In contrast, we have recently found that 1a and several related compounds afford sufficiently stable [60]fullerene adducts.11 This reaction is expected to be a useful method for preparing [60]-fullerene-o-quinodimethane adducts. Thus, we have further examined the reactions with various aromatic
carbonyl compounds carrying an alkyl group at the ortho position. Here we report the versatility of this reaction, the substituent effects on the reactivity and conformational behavior, and further functionalization of the resulting adducts in detail.
Results and Discussion
Photoreactions Between [60]Fullerene and 1an. The photoreactions of [60]fullerene with 1a-n, shown in Chart 1, were examined. The photoreaction with 1f and the isolation of the resulting adduct were also
reexamined. A 1:1 mixture of [60]fullerene and 1a-n in benzene (5× 10-4 M) was irradiated with a 400 W high-pressure mercury lamp in a Pyrex vessel with stirring at room temperature for 6 h. These irradiation conditions can produce the excited species of 1a-n as well as [60]-fullerene, though the latter is predominantly already in the excited state (e.g., the molar extinction coefficients() of 1a and [60]fullerene are 1 × 102 and 2 × 104 at 313nm, respectively). Among them, 1a, b, e, f, i, k, and m gave the desired monoadducts 2 along with the recovered[60]fullerene and a small amount of bisadducts (eq 1).

The results of the photoreactions are summarized in Table 1. o-Tolualdehyde (1a: R1 ) R2 ) H) and o-ethylbenzaldehyde (1b: R1 ) H, R2 ) Me) afforded the corresponding o-quinodimethane monoadducts 2a and b, respectively. 2a had been already prepared from benzocyclobutenol as a precursor of hydroxy-o-quinodimethane by Foote et al.3 It is noteworthy that 2a can be obtained from commercially available 1a in a single step. Although the isolated yield listed here (32%) is lower than that obtained by the reported method (59%),3 the yield based on the consumed [60]fullerene amounts to 87%. The
irradiation of [60]fullerene was also investigated in the presence of excess 1a. The yields of 2a and bisadducts are listed in Table 2, along with the recovered [60]-fullerene yield. With increasing amounts of 1a relative to the amount of [60]fullerene, the isolated yields of 2a and bisadducts gradually increased, and the recovery of [60]fullerene decreased. The highest yield of 2a was obtained for 1a/[60]fullerene ) 5:1. Using 9 equiv of 1a, the yield of 2a decreased while that of the bisadducts further increased. The decreased yield of 2a under this condition is ascribed to the formation of products with higher polarity, probably trisadducts, in addition to the formation of bisadducts. These results suggest that the reactivity of 2a with 1a is comparable to that of [60]-fullerene itself because the bisadditions should proceed
stepwise. The formation of bisadducts was confirmed by mass spectrometry. Their 1H NMR spectra, however, indicated the formation of complex mixtures of regioisomers and stereoisomers, which were not subjected to further purification.
In attempting to accomplish regioselective bisaddition, we have also examined the reactions using precursors 1o-r containing two o-tolualdehyde moieties tethered by oligomethylene or oligooxyethylene linkages of various lengths. Generally, covalent linkages between two reactive species effectively regulates their arrangement and distance and enhances regioselectivity in bisaddition, as demonstrated by several research groups.12 However, there has so far been no example of the application of this technique to photochemical reactions. The photoirradiation of 1o-r and [60]fullerene in benzene under
conditions similar to those mentioned above produced one o-quinodimethane species that reacted with [60]fullerene to yield monoaddition products 2o-r bearing an otolualdehyde moiety intact. Even by further irradiation of 2o-r, the remaining o-tolualdehyde moiety was unchanged, and thus no desired bisadducts were obtained. Because the intermolecular reaction between [60]fullerene and 1a undoubtedly gave bisadducts, 2o-r should have had sufficient reactivity for further photoreaction. The bridging linkage used here, especially that in 1q or r, seems sufficiently flexible so that it is unlikely to prevent the second addition because of steric hindrance. Thus, the failure of the second o-tolualdehyde moiety to react with [60]fullerene is interpreted as follows: The irradiation of the monoadduct can produce the excited states of the second o-tolualdehyde moiety, as in the case of the first addition step. However, these excited states appear to decay rapidly because of the intramolecular energy transfer to the [60]fullerene moiety because the otolualdehyde moiety is located relatively close to the fullerene surface, in contrast to the intermolecular reaction between 2a and 1a. Hence, no intramolecular
hydrogen abstraction that is necessary for the generation of o-quinodimethane takes place. The photoirradiation of 1o-r and [60]fullerene under the present conditions also failed to give compounds carrying two [60]fullerene moieties that reacted with both ends of 1o-r.

o-(Methoxymethyl)benzaldehyde 1c (R1 ) H, R2 ) OMe) yielded a product whose UV-vis spectrum indicated a peak around 430 nm, which is characteristic of [60]fullerene monoadducts. The 1H NMR spectra, however, were apparently inconsistent with the desired o-quinodimethane adducts; there were no suitable cyclohexene proton peaks. The identification of the products has been so far unsuccessful. The irradiation of 1d (R1 ) H, R2 ) Ph) resulted in the recovery of [60]fullerene. o-Methylacetophenone (1e) and o-methylbenzophenone (1f) also produced the desired monoadducts, though the
yield using 1e was rather low. In contrast, no desired adducts were formed from o-toluic acid (1g) and its methyl ester (1h).
The effects of the substituents directly attached to the benzene ring were also examined. 2,6-Dimethylbenzaldehyde (1i) gave adduct 2i, while 1j, bearing a hydroxy group at the ortho position of the formyl group, led to the recovery of [60]fullerene. In the excited state of 1j, the proton transfer from the hydroxyl group to the carbonyl group must be predominant, thus preventing the hydrogen abstraction from the methyl group.13 In contrast, 1k, carrying a hydroxy group at the para position of the formyl group, successfully afforded 2k, though the yield was extremely low.
The irradiation of aldehydes consisting of polycyclic aromatic or heteroaromatic rings was also examined. 2-Methyl-1-naphthaldehyde (1l) failed to give the desired monoadduct. In aromatic aldehydes containing larger π systems, the excited triplet state has π-π* character;14 this state cannot be involved in the hydrogen abstraction. Intriguingly, monoadduct 2m was obtained from 1m, which has a pyridine ring, whereas only [60]fullerene was recovered from 1n, which has a thiophene ring. The obtained monoadducts, except for 2f, were easily isolated as stable products by column chromatography
(silica gel), and no decomposition products were detected.
The purification of adducts is much easier than that in the reactions using benzocyclobutenes or 1,2-bis(bromomethyl)benzene derivatives as o-quinodimethane precursors. In contrast, the isolation of 2f has been unsuccessful, although 2f was a major product; a trace amount(ca. 1-2%) of the monoalkyl 1,2-dihydrofullerene reported in the literature10 was not completely removed, despite several attempts to isolate 2f, including recrystallization, HPLC, and GPC. Because this product was absent immediately after the photoirradiation, it was apparently formed during the purification process of 2f.
To clarify the reactive species involved in the photoreaction, a mixture of [60]fullerene and 1a was irradiated at 532 nm by using an Nd:YAG laser, in which [60]- fullerene can be exclusively photoexcited. Under these irradiation conditions, intact [60]fullerene was recovered without the formation of desired adducts. This observation apparently demonstrates that these photoreactions require the excited states of carbonyl compounds, which generate o-quinodimethane by intramolecular hydrogen abstraction; the excited triplet state of [60]fullerene does not react with the ground state 1a but decays back to
the ground state.
Considering the extremely small absorbance of carbonyl compounds such as 1a relative to that of [60]fullerene in the UV, it is noteworthy that the desired o-quinodimethane adducts were obtained by irradiation with a high-pressure mercury lamp in the presence of [60]-fullerene. The successful progress of this reaction indicates that the o-quinodimethane is quite efficiently formed from the excited triplet state of carbonyl compounds and, subsequently, undergoes the Diels-Alder reaction with [60]fullerene sufficiently fast within their lifetimes. The high efficiency of these processes should enable the seemingly unfavorable reaction, where the incident light is only slightly absorbed by the component(e.g., 1a) that needs to be photoexcited.
Structural Analysis of Monoadducts. The monoadducts obtained here were characterized by MS, UV, and NMR spectroscopy. 2a, b, e, f, i, k, and m had molecular ion peaks corresponding to the desired monoadducts in the FAB-MS spectra. Their UV-vis spectra were substantially identical to each other, despite the difference in substituents. All of these monoadducts displayed a sharp band around 430 nm and a weak band around 700 nm. These bands are characteristic of [60]fullerene monoadducts.1,2 1H NMR spectra provided the most decisive information on the structure and conformation of adducts. The chemical shifts of cyclohexene-ring protons are listed in Table 3.
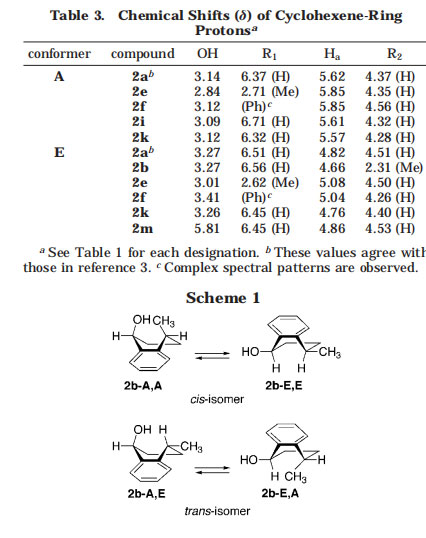
As reported by Foote et al.,3 2a is composed of two conformers 2a-A and 2a-E possessing a pseudoaxial and a pseudoequatorial hydroxy group, respectively, that slowly exchange on the NMR time scale at room temperature. The conformer ratio A/E was 6:4, which is approximately in agreement with that reported in the literature.3 Adduct 2b obtained from 1b carries another substituent on the cyclohexene ring. Therefore, two diastereoisomers, cis and trans isomers, are possible for 2b, each of which can be composed of two conformers, as shown in Scheme 1. Intriguingly, the 1H NMR spectrum of 2b indicated the existence of a single conformer of one diastereoisomer. Its stereochemistry was readily determined on the basis of NOE experiments. Because NOE interaction was observed between the two protons directly attached to the cyclohexene ring, the hydroxy and methyl groups were both assigned as pseudoequatorial (2b-E,E). This assignment is also supported by the similarity of chemical shifts of the cyclohexene and OH protons in 2b to those in 2a-E rather than to those in 2a-A (Table 3). The absence of the trans isomer (2b-E,A or 2b-A,E) and conformer 2b-A,A of the cis isomer is apparently related to the stereochemistry of reaction intermediates generated from 2b. As described above, the intramolecular hydrogen abstraction of 2b affords an o-quinodimethane species with a hydroxy group (i.e.,
dienol) via quite a short-lived biradical.6 The resulting dienol, for which four isomers I-IV are possible, undergoes a Diels-Alder reaction with [60]fullerene to give 2b (Scheme 2). Among them, (Z)-dienols II and IV are known to revert rapidly to the original aldehyde via a [1,5]-sigmatropic hydrogen shift.15 Of (E)-dienols I and III, I, which can lead to 2b-E,E, appears to be more stable than II because of less steric hindrance. The PM3 calculations suggest that I is more stable than III by 3.0 kcal/mol. Such relative stability results in the exclusive formation of 2b-E,E, which is unlikely to convert into 2bA,A because of the flagpole steric hindrance, thus resulting in the observation of the single conformer.

The 1H NMR spectra of both 2e and f indicated the existence of two conformers similar to 2a, although an inseparable decomposition product was also involved in 2f, as described above. The assignment of each conformer was established by comparison with the chemical shifts of 2a; in conformer E, the hydroxy proton resonates at a lower field than it does in conformer A, while the Ha proton resonates at a higher field. On the basis of these observations, the major conformers for 2e and f were assigned as A, which possesses a pseudoaxial hydroxy group, in contrast to that of 2a. The conformer ratio A/E
was 6:4 for both 2e and f. The decrease in the ratio of E in 2e and f relative to that of 2a seems to result from the destabilization of E caused by the steric hindrance between the Ha proton and the R1 (methyl or phenyl) group.
The interconversion between conformers E and A in 2e was investigated by the variable-temperature (VT) NMR technique. Foote et al. estimated the ∆Gq value for 2a to be 17.6 kcal/mol.3 This value is much larger than that for the [60]fullerene-o-quinodimethane adduct with no hydroxy group on the cyclohexene ring (15.2 kcal/mol).1b The higher barrier for 2a is attributable to the presence of the hydroxy group, which should destabilize the transition state and make the inversion difficult. In 2e bearing two geminal substituents, the methyl group coalesced at 120 °C in Cl2CDCDCl2, whereas two sets of
cyclohexene protons Ha and R2 did not coalesce even at this temperature, though there was slightly broadening with increasing temperature. On the basis of the coalescence temperature of the methyl group, the ∆Gq value was estimated to be 19.6 kcal/mol. It is apparent that the additional methyl group further prevents the ring inversion relative to 2a.
The 1H NMR spectrum of 2m produced from 1m showed a single conformer that was similar to 2b. This result is in remarkable contrast to 2a, which consists of two conformers. On the basis of NOE experiments, the hydroxy group was found to adopt a pseudoequatorial conformation similar to that of 2b; NOE interaction was observed between the R1 and Ha protons. The addition of trifluoroacetic acid to 2m induced considerable lowfield shifts in the 1H NMR spectrum, especially for the pyridine protons, indicating the protonation at the nitrogen atom, while only conformer E was still observed.
These results suggest that the hydrogen bonding between the pyridine nitrogen atom and the hydroxy group is not responsible for the exclusive presence of conformer E in 2m. The preference for E is probably ascribable to less steric interaction between the hydroxy group and the pyridine nitrogen atom than that found in 2a-E, which possesses a phenyl C-H proton. This steric interaction was clearly demonstrated by the structural analysis of 2i. Intriguingly, 2i was composed of a single conformer, which was assigned as A on the basis of the comparison with the 1H NMR spectral data of 2a; the chemical shifts of the OH, Ha, and R2 protons are comparable to those of 2a-A rather than to those of 2a-E (Table 3). The difference in the conformer ratios among 2a, 2m, and 2i is remarkable; 2m adopts only E, 2a, both A and E (4:6), and 2i, only A. These differences apparently correspond to the bulkiness of the X site of the aromatic ring; the value of A/E increases in the order X ) N < CH < CCH3.
In 2k, two conformers A and E were observed and were similar to those of 2a. However, A was slightly predominant over E (A/E ) 55:45), in contrast to the value of A/E for 2a. The relatively high stability of conformer A in 2k seems to be ascribed to some electronic effects of the hydroxy group that is attached to the benzene ring because this substituent is unlikely to affect sterically the relative stability of the two conformers.
Functionalization of Adducts Obtained. The hydroxy group of the monoadducts that were obtained is available for further functionalizations, which can alter the conformational behavior and also provide the adducts with additional properties. First, we have examined the oxidation of 2a, which should transform two conformers into a single entity. 2a was allowed to react with excess PCC in CH2Cl2/CS2 (1:1) at room temperature for 3 h to give the desired product 316 (eq 2). In the 1H NMR spectrum of 3, the two cyclohexene protons were observed at δ 4.82 as a sharp singlet, indicating that the inversion is sufficiently fast on the NMR time scale, in contrast with that of 2a. Such fast inversion was also demonstrated by the 13C NMR spectrum showing Cs symmetry.
The conversion of the hydroxy group of 2a into an ether or an ester group was investigated by Foote et al.3 We have examined the o-acylation of 2m bearing a pyridine residue and have prepared 4, 5, and 6 with a phenyl, a 1-naphthyl, and a 9-anthryl group, respectively (eq 3).
The o-acylation of 2m with benzoyl chloride and 1-naphthoyl chloride was carried out in the presence of 4-(dimethylamino)pyridine (DMAP) and pyridine in CH2-Cl2, in a manner similar to that reported by Foote et al.,3 to yield esters 4 and 5, respectively. For the preparation of 6, 2m was allowed to react with 9-anthroic acid in the presence of DCC and DMAP in toluene. 4, 5, and 6 are composed of two conformers each, in contrast to 2m. The introduction of acyl groups into the hydroxy group of 2m increases the steric interaction with the pyridine nitrogen atom, leading to the destabilization of conformer E relative to that of A. In each of 4-6, the conformer ratio A/E was estimated to be approximately 1:1. The dynamic behavior of A and E was examined by VT-NMR in Cl2-CDCDCl2 in a manner similar to that used with 2e.
Unfortunately, the coalescence of singlet peaks for the cyclohexene methine (R1) proton was not clearly observed because they overlapped with the signals of some of the aromatic protons. Instead, the pyridine R-protons were found to coalesce at 70 °C for both 4 and 5 and at 60 °C for 6. On the basis of these temperatures, the ∆Gq values for 4, 5, and 6 were estimated to be 17.2, 17.1, and 16.8 kcal/mol, respectively. These values are comparable to that for 2a (Table 4). The difference in the aromatic nuclei was not so significantly reflected in the ∆Gq values.

Summary
Stable o-quinodimethane adducts 2a, b, e, i, k, and m possessing a hydroxy group that is applicable to further transformations were obtained from photochemical reactions between the corresponding carbonyl compounds and [60]fullerene. A and E conformers existed for 2a, e, and k, while 2b, i, and m exclusively adopted E, A, and E conformations, respectively. The conformer ratios were mainly dependent on the bulkiness of the substituents attached to the aromatic or cyclohexene ring, as clearly demonstrated by 2a, i, and m. 2m, bearing a pyridine ring, was successfully transformed into esters 4-6, which adopted two conformations.
2-((3-Methylisoxazol-5-yl)methoxy)acetic acidCatalog No.:AA019Y6S CAS No.:1018051-09-6 MDL No.:MFCD09859453 MF:C7H9NO4 MW:171.1507 |
4-(3,4-dihydro-2H-1,5-benzodioxepin-7-yl)butanoic acidCatalog No.:AA01BXF8 CAS No.:1018054-02-8 MDL No.:MFCD09881140 MF:C13H16O4 MW:236.2637 |
Ethyl 3-cyclopropyl-1,2,4-oxadiazole-5-carboxylateCatalog No.:AA00JFJ9 CAS No.:1018125-29-5 MDL No.:MFCD09701787 MF:C8H10N2O3 MW:182.1766 |
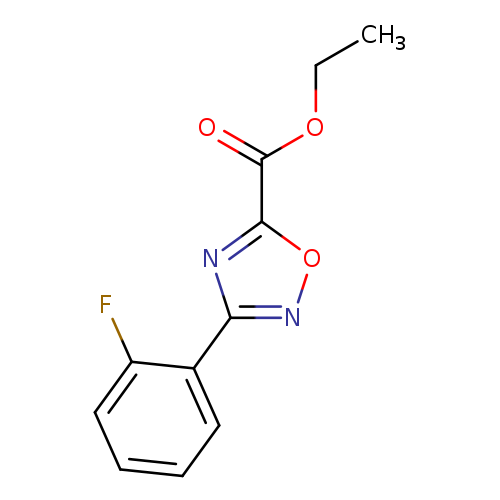
Ethyl 3-(2-Fluorophenyl)-1,2,4-oxadiazole-5-carboxylateCatalog No.:AA00J0NW CAS No.:1018125-37-5 MDL No.:MFCD09701808 MF:C11H9FN2O3 MW:236.1992 |
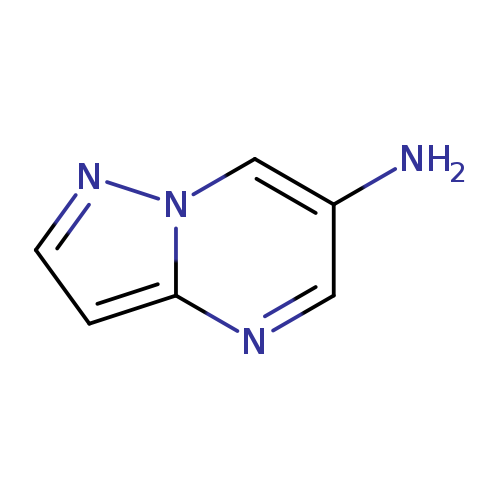
Pyrazolo[1,5-a]pyrimidin-6-amineCatalog No.:AA0005J5 CAS No.:1018125-39-7 MDL No.:MFCD09701813 MF:C6H6N4 MW:134.1386 |
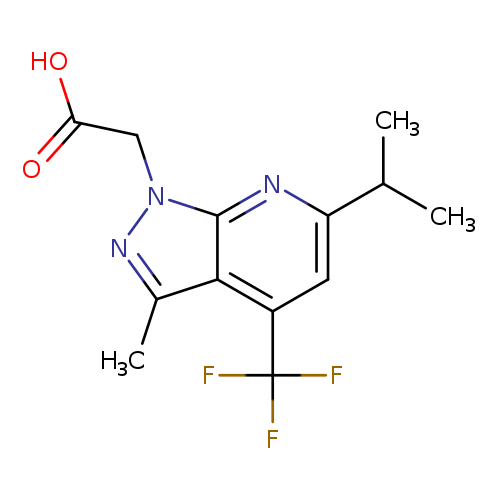
2-(6-Isopropyl-3-methyl-4-(trifluoromethyl)-1H-pyrazolo[3,4-b]pyridin-1-yl)acetic acidCatalog No.:AA00JFKI CAS No.:1018125-53-5 MDL No.:MFCD09701842 MF:C13H14F3N3O2 MW:301.2644 |
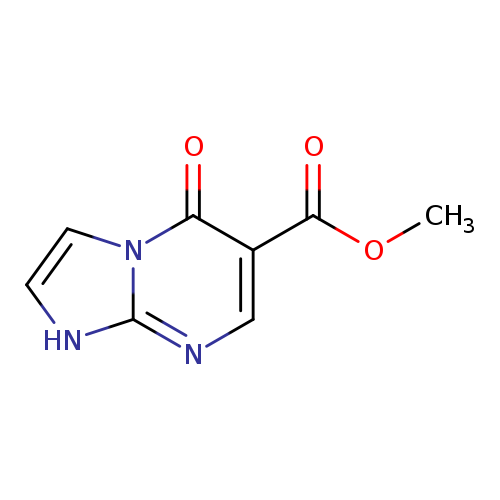
Methyl 5-oxo-1,5-dihydroimidazo[1,2-a]pyrimidine-6-carboxylateCatalog No.:AA01B77L CAS No.:1018125-57-9 MDL No.:MFCD09701857 MF:C8H7N3O3 MW:193.1595 |
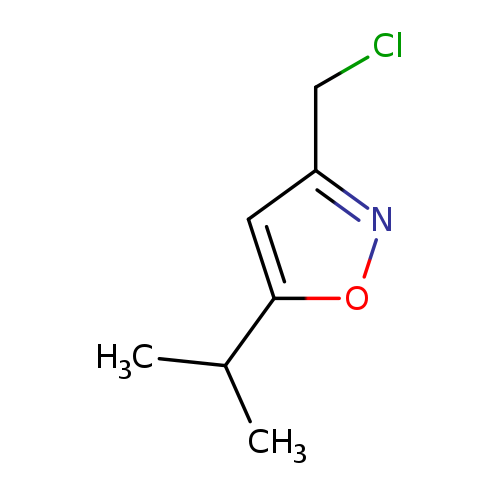
3-(Chloromethyl)-5-isopropylisoxazoleCatalog No.:AA0005J3 CAS No.:1018128-18-1 MDL No.:MFCD09859196 MF:C7H10ClNO MW:159.6134 |
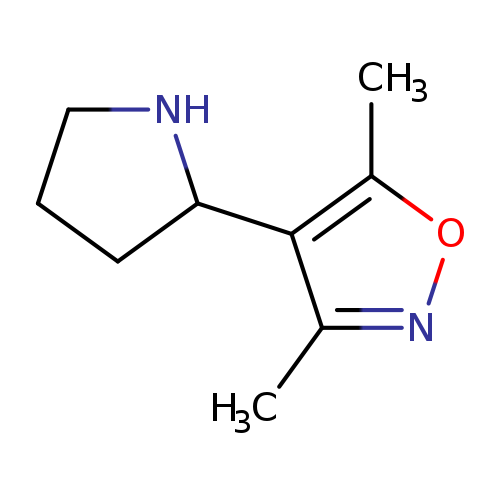
3,5-dimethyl-4-pyrrolidin-2-ylisoxazoleCatalog No.:AA008VKF CAS No.:1018128-26-1 MDL No.:MFCD09859204 MF:C9H14N2O MW:166.2203 |
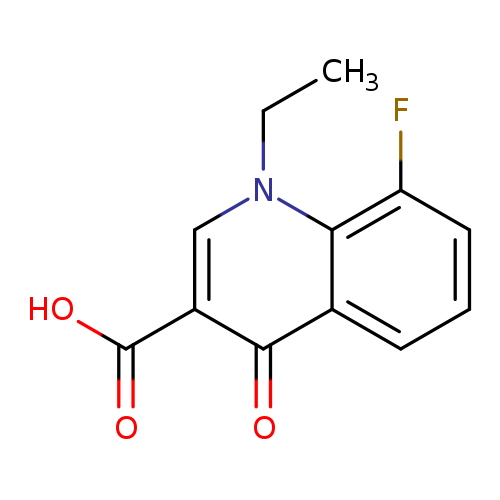
1-Ethyl-8-fluoro-4-oxo-1,4-dihydroquinoline-3-carboxylic acidCatalog No.:AA01ARF7 CAS No.:1018135-10-8 MDL No.:MFCD11058036 MF:C12H10FNO3 MW:235.2111 |
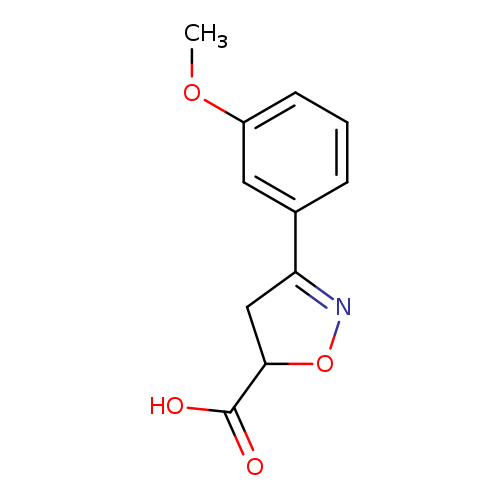
3-(3-Methoxyphenyl)-4,5-dihydroisoxazole-5-carboxylic acidCatalog No.:AA00JFI9 CAS No.:1018143-20-8 MDL No.:MFCD09701747 MF:C11H11NO4 MW:221.2093 |
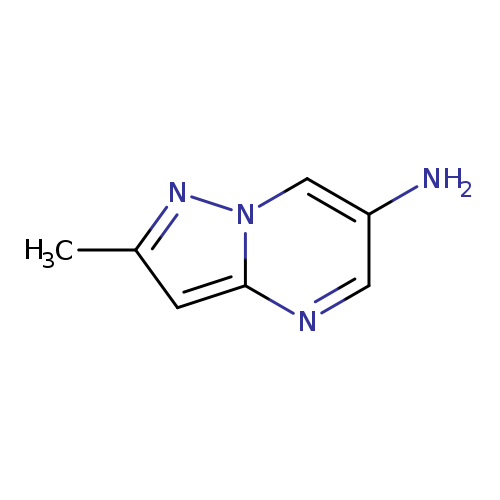
2-Methylpyrazolo[1,5-a]pyrimidin-6-amineCatalog No.:AA01BW78 CAS No.:1018143-51-5 MDL No.:MFCD09701780 MF:C7H8N4 MW:148.1652 |
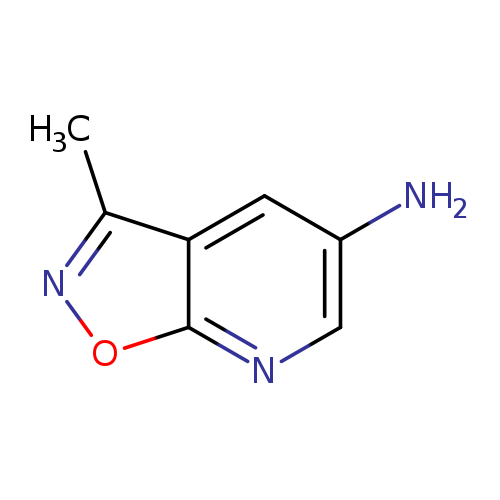
3-Methyl-[1,2]oxazolo[5,4-b]pyridin-5-amineCatalog No.:AA01A94R CAS No.:1018144-08-5 MDL No.:MFCD09701862 MF:C7H7N3O MW:149.1500 |
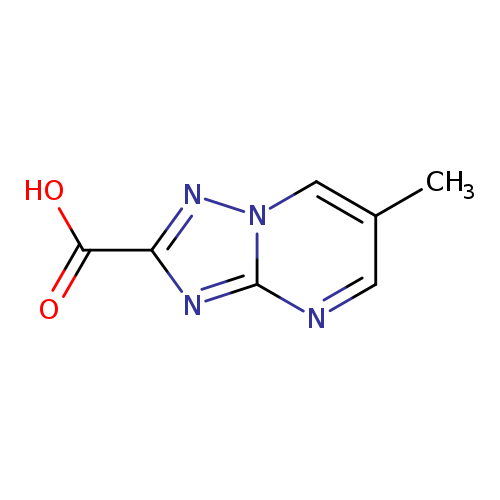
6-Methyl-[1,2,4]triazolo[1,5-a]pyrimidine-2-carboxylic acidCatalog No.:AA019D95 CAS No.:1018144-12-1 MDL No.:MFCD09701872 MF:C7H6N4O2 MW:178.1481 |
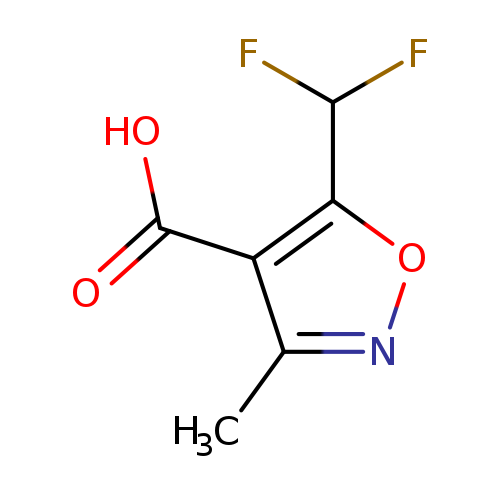
5-(Difluoromethyl)-3-methyl-1,2-oxazole-4-carboxylic acidCatalog No.:AA019RUA CAS No.:1018166-17-0 MDL No.:MFCD09859239 MF:C6H5F2NO3 MW:177.1056 |
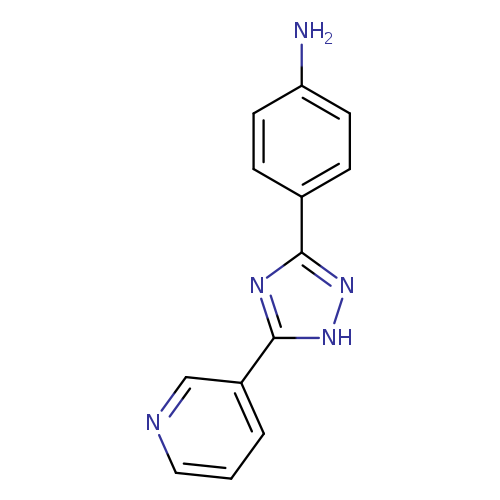
[4-(5-Pyridin-3-yl-1h-1,2,4-triazol-3-yl)phenyl]amineCatalog No.:AA00JAVM CAS No.:1018168-01-8 MDL No.:MFCD11058002 MF:C13H11N5 MW:237.2599 |
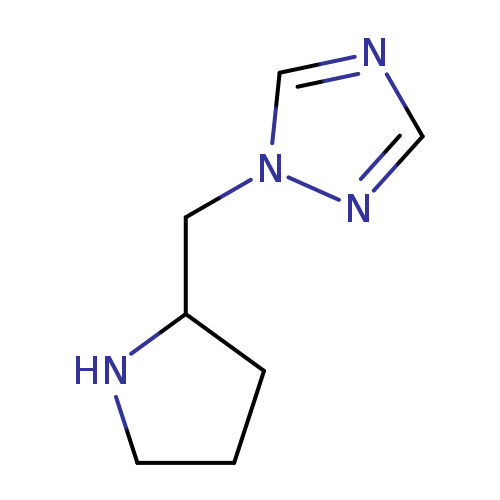
1-(Pyrrolidin-2-ylmethyl)-1H-1,2,4-triazoleCatalog No.:AA00JFQ9 CAS No.:1018170-67-6 MDL No.:MFCD10001573 MF:C7H12N4 MW:152.1970 |
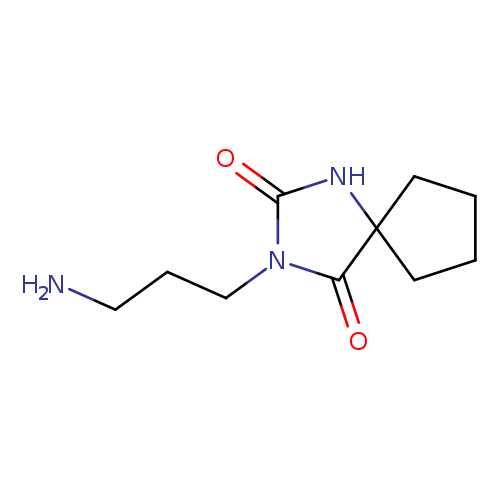
3-(3-aminopropyl)-1,3-diazaspiro[4.4]nonane-2,4-dioneCatalog No.:AA019P4W CAS No.:1018185-30-2 MDL No.:MFCD09880453 MF:C10H17N3O2 MW:211.2609 |
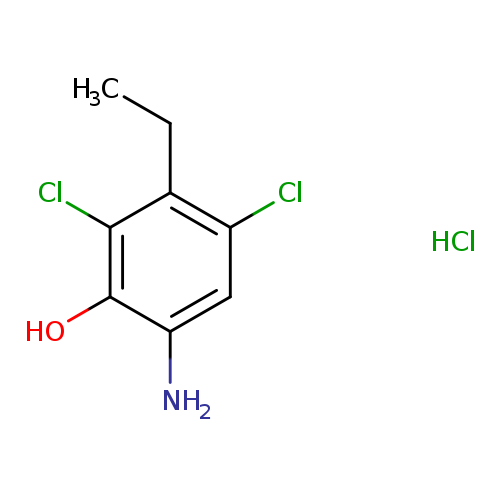
2,4-Dichloro-3-ethyl-6-aminophenol HClCatalog No.:AA0005K3 CAS No.:101819-99-2 MDL No.:MFCD00270793 MF:C8H10Cl3NO MW:242.5301 |
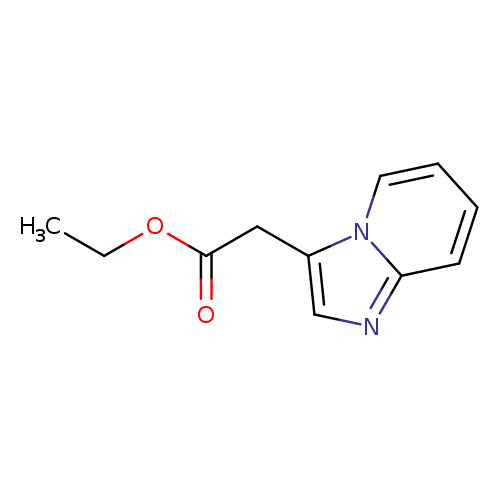
Ethyl 2-(imidazo[1,2-a]pyridin-3-yl)acetateCatalog No.:AA0005K0 CAS No.:101820-69-3 MDL No.:MFCD13183145 MF:C11H12N2O2 MW:204.2252 |
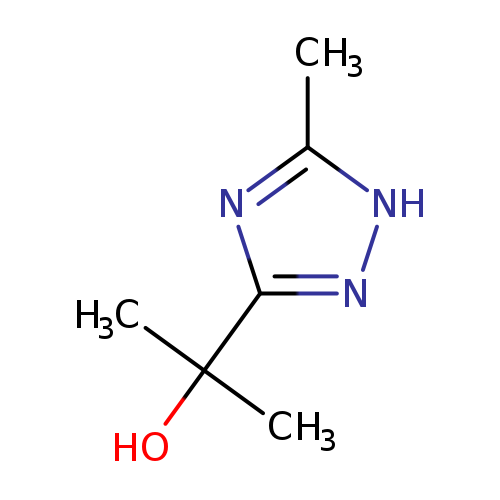
2-(5-methyl-1H-1,2,4-triazol-3-yl)propan-2-olCatalog No.:AA01AJI0 CAS No.:101822-15-5 MDL No.:MFCD19686271 MF:C6H11N3O MW:141.1710 |
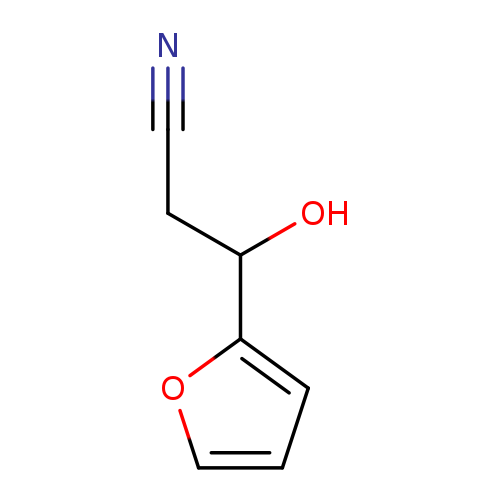
3-(furan-2-yl)-3-hydroxypropanenitrileCatalog No.:AA01BNAP CAS No.:101824-11-7 MDL No.:MFCD16300739 MF:C7H7NO2 MW:137.1360 |
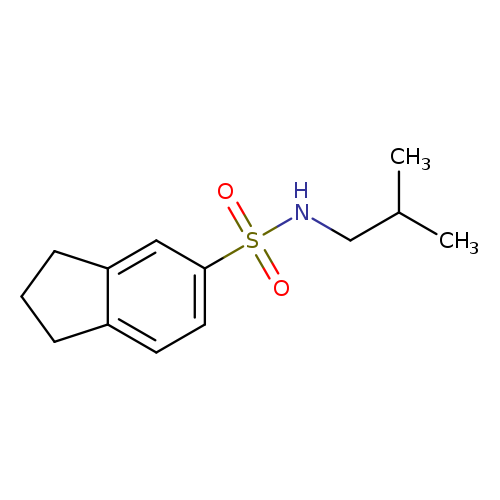
N-(2-Methylpropyl)-2,3-dihydro-1h-indene-5-sulfonamideCatalog No.:AA01B270 CAS No.:1018240-42-0 MDL No.:MFCD13520397 MF:C13H19NO2S MW:253.3605 |
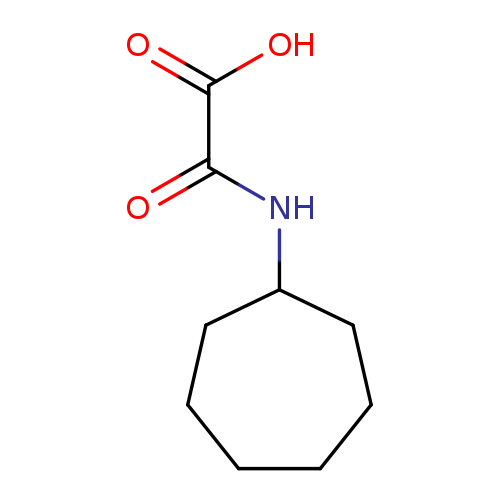
(Cycloheptylamino)(oxo)acetic acidCatalog No.:AA0005KO CAS No.:1018243-04-3 MDL No.:MFCD10018440 MF:C9H15NO3 MW:185.2203 |
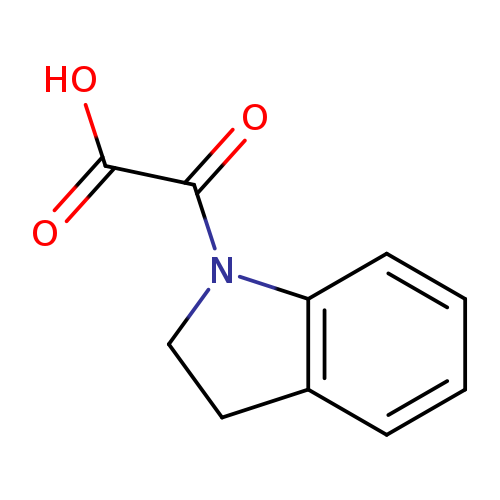
2,3-Dihydro-1h-indol-1-yl(oxo)acetic acidCatalog No.:AA0005KN CAS No.:1018243-08-7 MDL No.:MFCD10018450 MF:C10H9NO3 MW:191.1834 |
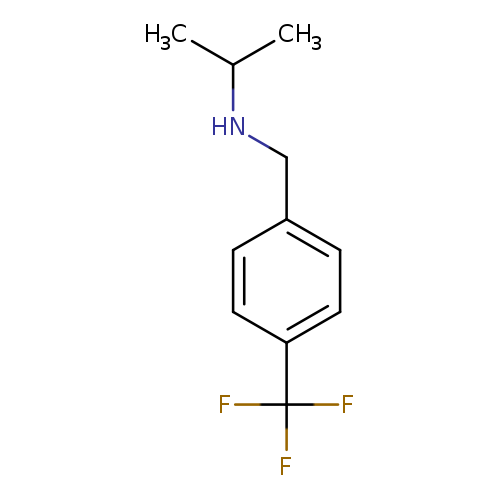
(propan-2-yl)({[4-(trifluoromethyl)phenyl]methyl})amineCatalog No.:AA00JT99 CAS No.:101825-12-1 MDL No.:MFCD08060648 MF:C11H14F3N MW:217.2308 |
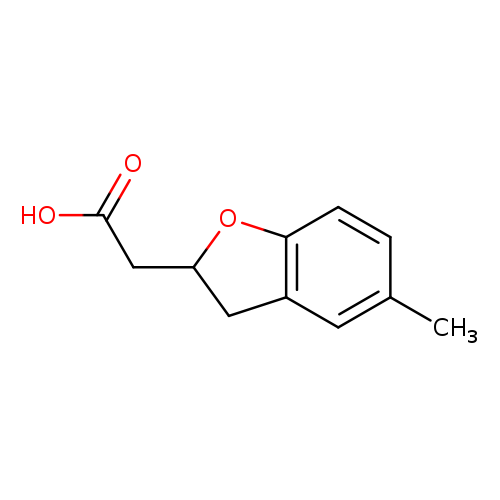
2-(5-methyl-2,3-dihydro-1-benzofuran-2-yl)acetic acidCatalog No.:AA01BV60 CAS No.:1018251-36-9 MDL No.:MFCD10008548 MF:C11H12O3 MW:192.2112 |
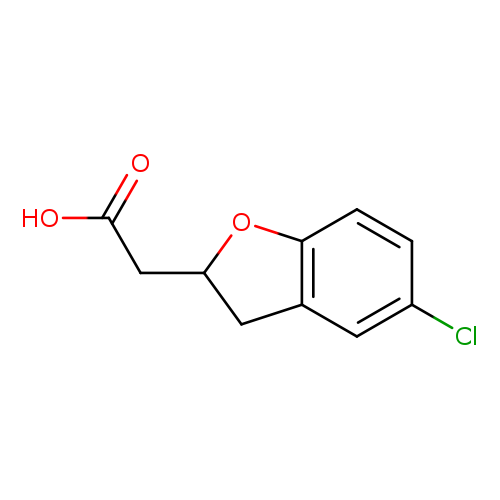
2-(5-chloro-2,3-dihydro-1-benzofuran-2-yl)acetic acidCatalog No.:AA01BUZN CAS No.:1018251-39-2 MDL No.:MFCD10008552 MF:C10H9ClO3 MW:212.6297 |
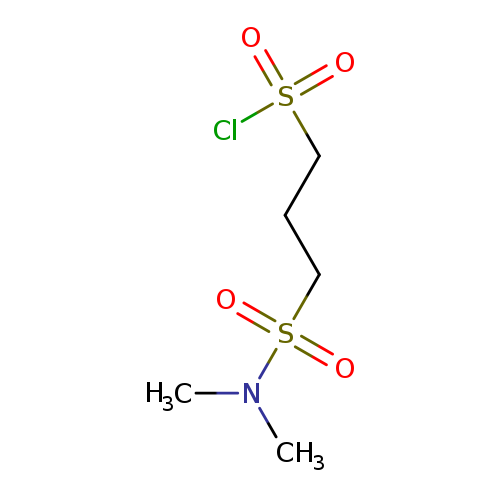
3-(dimethylsulfamoyl)propane-1-sulfonyl chlorideCatalog No.:AA01ACKU CAS No.:1018251-96-1 MDL No.:MFCD10015475 MF:C5H12ClNO4S2 MW:249.7361 |
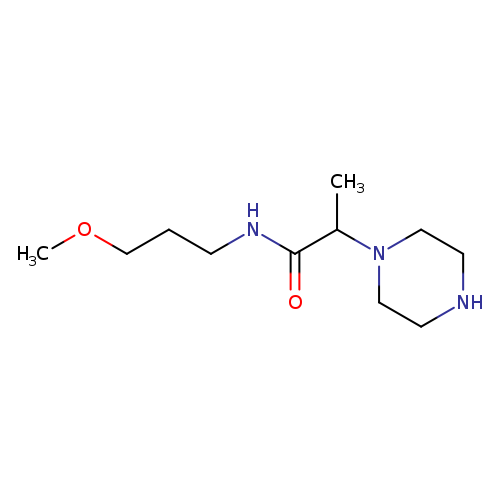
N-(3-methoxypropyl)-2-(piperazin-1-yl)propanamideCatalog No.:AA01AAOT CAS No.:1018252-09-9 MDL No.:MFCD10011194 MF:C11H23N3O2 MW:229.3192 |
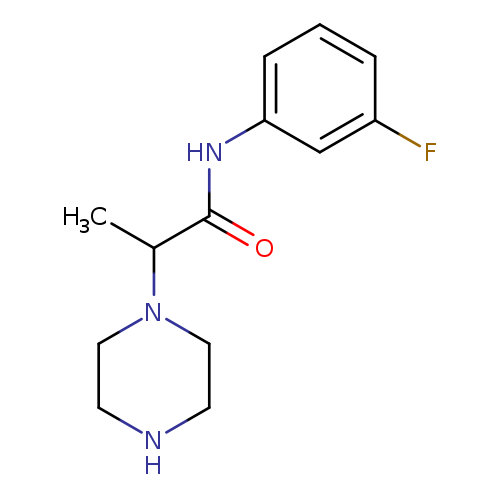
N-(3-fluorophenyl)-2-(piperazin-1-yl)propanamideCatalog No.:AA019UNS CAS No.:1018252-85-1 MDL No.:MFCD10011294 MF:C13H18FN3O MW:251.2999 |
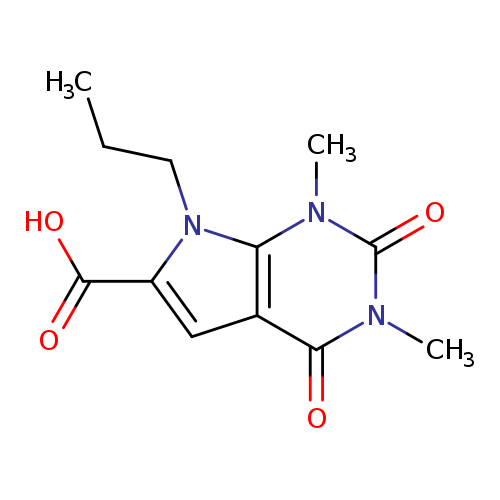
1,3-Dimethyl-2,4-dioxo-7-propyl-2,3,4,7-tetrahydro-1H-pyrrolo[2,3-d]pyrimidine-6-carboxylic acidCatalog No.:AA00H9K6 CAS No.:1018256-11-5 MDL No.:MFCD10007810 MF:C12H15N3O4 MW:265.2652 |
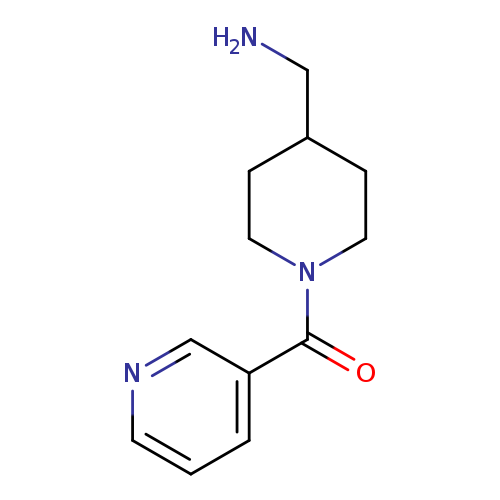
{[1-(pyridin-3-ylcarbonyl)piperidin-4-yl]methyl}amineCatalog No.:AA0192XC CAS No.:1018258-85-9 MDL No.:MFCD10016318 MF:C12H17N3O MW:219.2829 |
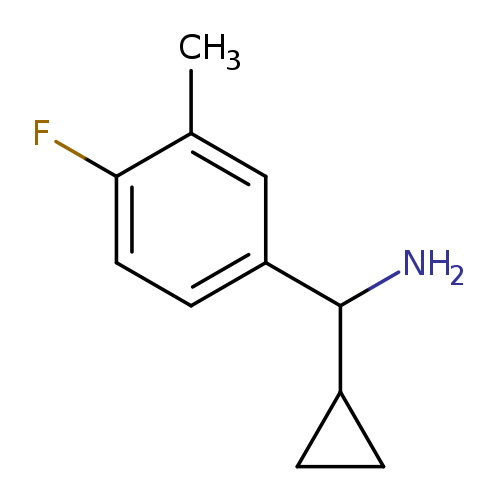
cyclopropyl(4-fluoro-3-methylphenyl)methanamineCatalog No.:AA01A326 CAS No.:1018265-03-6 MDL No.:MFCD10018234 MF:C11H14FN MW:179.2340 |
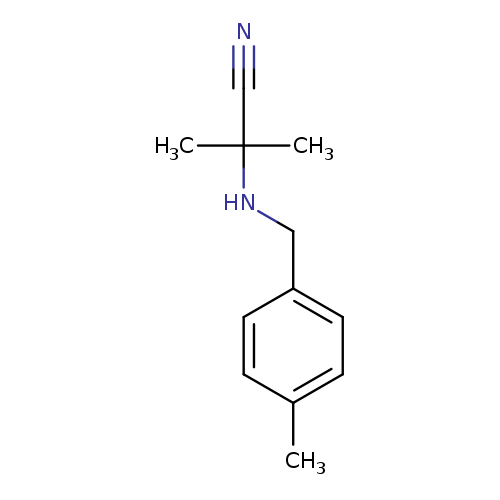
2-Methyl-2-([(4-methylphenyl)methyl]amino)propanenitrileCatalog No.:AA00IZ98 CAS No.:1018265-76-3 MDL No.:MFCD10014475 MF:C12H16N2 MW:188.2688 |
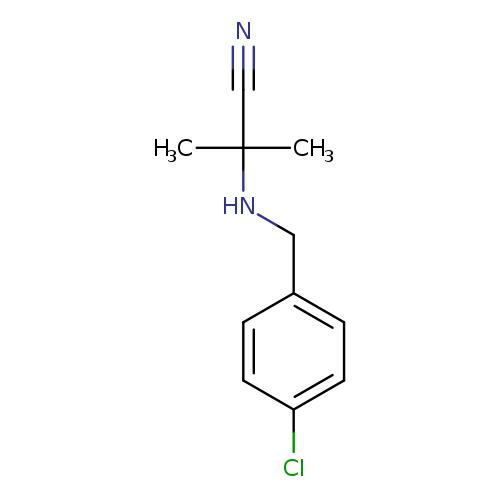
2-((4-Chlorobenzyl)amino)-2-methylpropanenitrileCatalog No.:AA00ITYV CAS No.:1018265-80-9 MDL No.:MFCD10014479 MF:C11H13ClN2 MW:208.6873 |
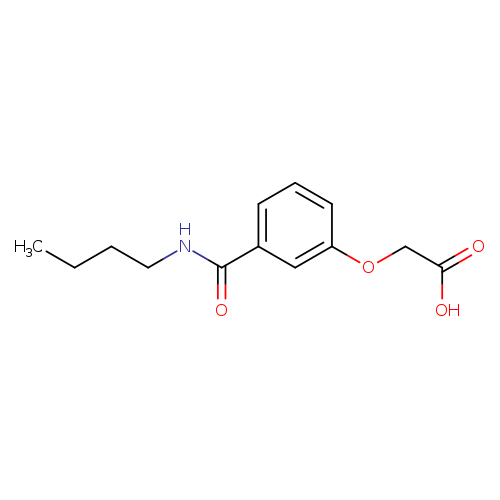
3-(Butylcarbamoyl)phenoxyacetic acidCatalog No.:AA00H9K7 CAS No.:1018266-14-2 MDL No.:MFCD10027954 MF:C13H17NO4 MW:251.2784 |
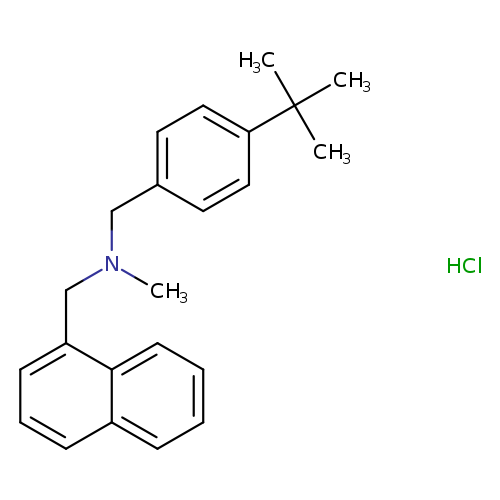
Butenafine HClCatalog No.:AA0005KW CAS No.:101827-46-7 MDL No.:MFCD00917064 MF:C23H28ClN MW:353.9281 |
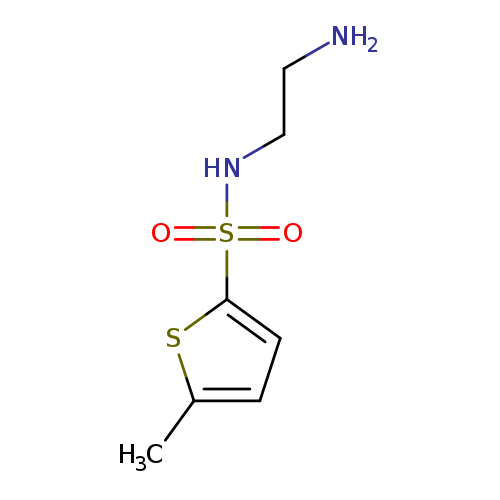
N-(2-aminoethyl)-5-methylthiophene-2-sulfonamideCatalog No.:AA01A1F0 CAS No.:1018273-59-0 MDL No.:MFCD10024764 MF:C7H12N2O2S2 MW:220.3124 |
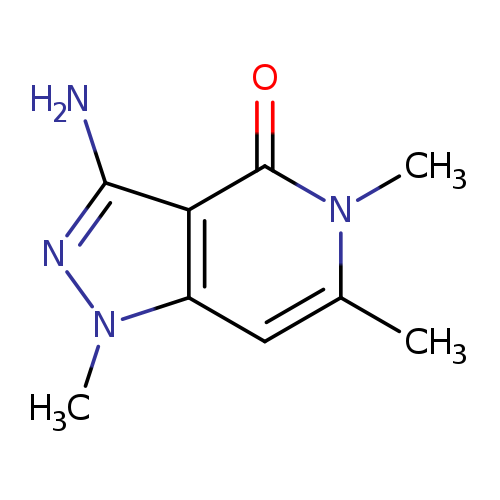
3-amino-1,5,6-trimethyl-1H,4H,5H-pyrazolo[4,3-c]pyridin-4-oneCatalog No.:AA01BE5K CAS No.:1018274-29-7 MDL No.:MFCD10008264 MF:C9H12N4O MW:192.2178 |
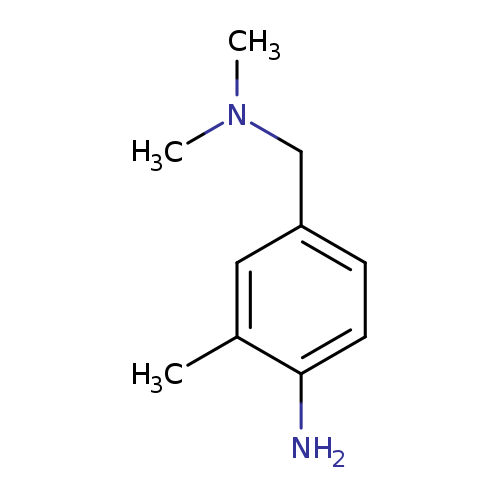
4-[(dimethylamino)methyl]-2-methylanilineCatalog No.:AA01C2EZ CAS No.:1018276-97-5 MDL No.:MFCD30499535 MF:C10H16N2 MW:164.2474 |
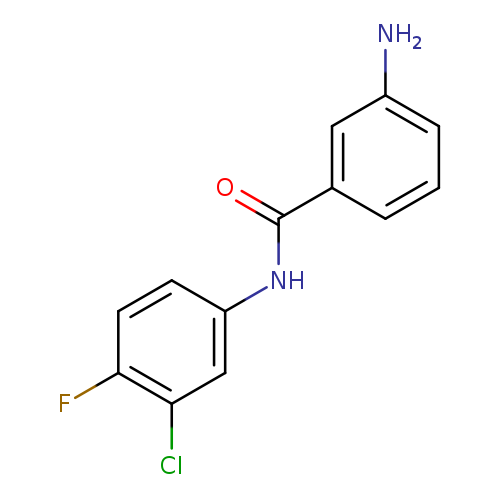
3-Amino-N-(3-chloro-4-fluorophenyl)benzamideCatalog No.:AA01FPK3 CAS No.:1018277-98-9 MDL No.: MF:C13H10ClFN2O MW:264.6827 |
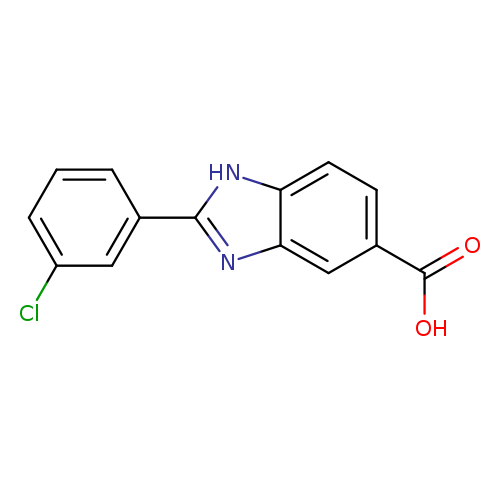
2-(3-Chlorophenyl)-1h-benzimidazole-5-carboxylic acidCatalog No.:AA01EKFJ CAS No.:1018278-53-9 MDL No.:MFCD10007770 MF:C14H9ClN2O2 MW:272.6865 |
2-(4-ethylphenyl)-2-(methylamino)acetonitrileCatalog No.:AA01BB8W CAS No.:1018281-34-9 MDL No.:MFCD10013261 MF:C11H14N2 MW:174.2423 |
4-(Pyrrolidine-1-sulfonyl)butanoic acidCatalog No.:AA019VYY CAS No.:1018283-85-6 MDL No.:MFCD10012310 MF:C8H15NO4S MW:221.2740 |
3-(4-Aminophenyl)-N,N-dimethylpropanamideCatalog No.:AA0005KK CAS No.:1018284-46-2 MDL No.:MFCD10022146 MF:C11H16N2O MW:192.2575 |
1-(6-aminoindolin-1-yl)propan-1-oneCatalog No.:AA01C818 CAS No.:1018286-45-7 MDL No.:MFCD10016469 MF:C11H14N2O MW:190.2417 |
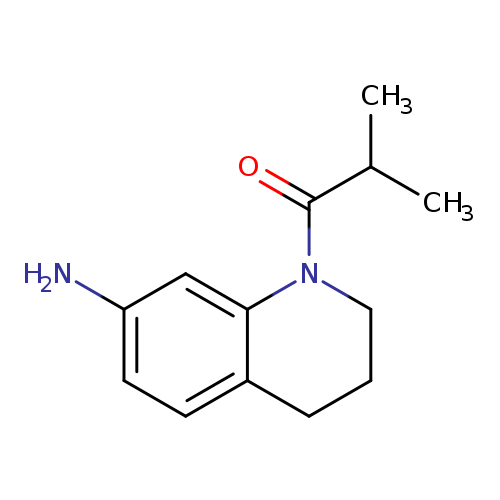
1-isobutyryl-1,2,3,4-tetrahydroquinolin-7-amineCatalog No.:AA0192UO CAS No.:1018287-36-9 MDL No.:MFCD10016587 MF:C13H18N2O MW:218.2948 |
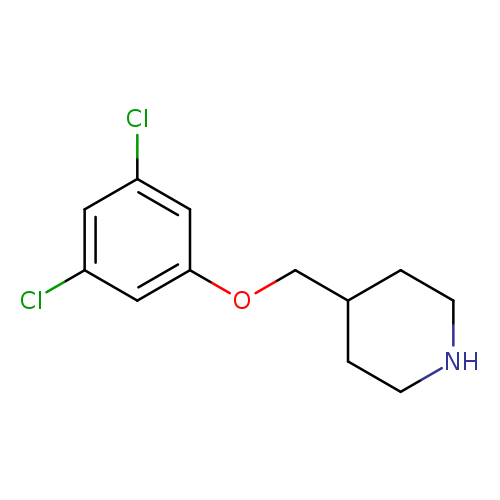
4-(3,5-dichlorophenoxymethyl)piperidineCatalog No.:AA01AKTQ CAS No.:1018288-02-2 MDL No.:MFCD10022918 MF:C12H15Cl2NO MW:260.1596 |
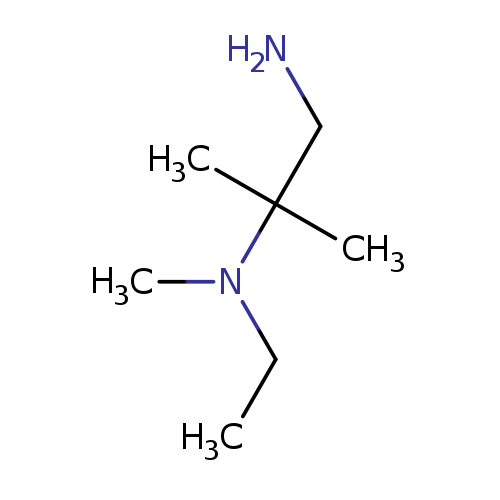
N2-Ethyl-N2,2-dimethylpropane-1,2-diamineCatalog No.:AA0005KI CAS No.:1018289-07-0 MDL No.:MFCD10026603 MF:C7H18N2 MW:130.2312 |
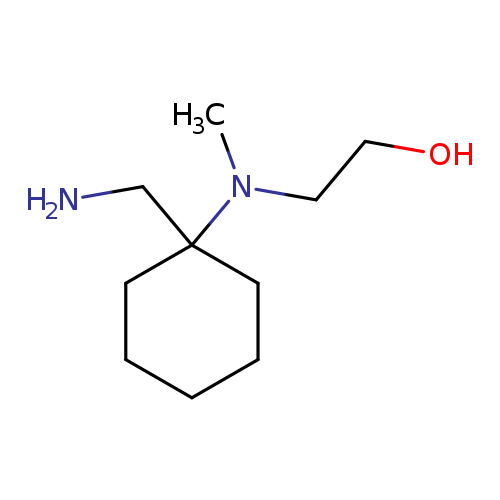
2-{[1-(aminomethyl)cyclohexyl](methyl)amino}ethan-1-olCatalog No.:AA01AI9I CAS No.:1018289-25-2 MDL No.:MFCD10026623 MF:C10H22N2O MW:186.2945 |
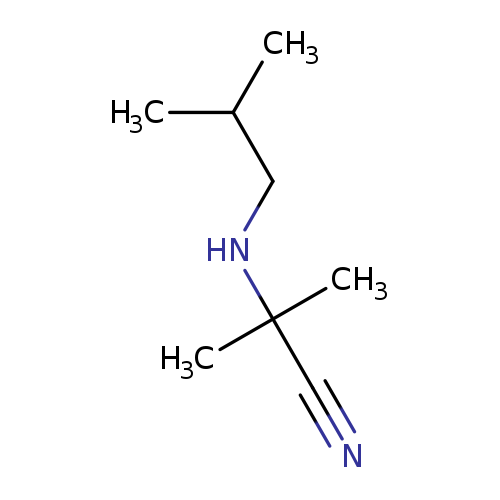
2-methyl-2-[(2-methylpropyl)amino]propanenitrileCatalog No.:AA00ITRT CAS No.:1018291-88-7 MDL No.:MFCD10014464 MF:C8H16N2 MW:140.2260 |
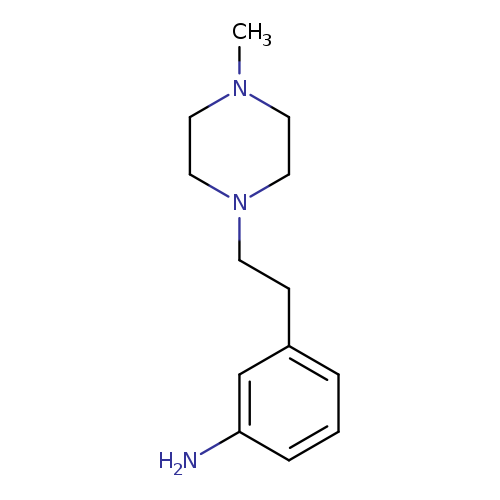
3-[2-(4-methylpiperazin-1-yl)ethyl]anilineCatalog No.:AA01A43S CAS No.:1018294-23-9 MDL No.:MFCD10021281 MF:C13H21N3 MW:219.3259 |
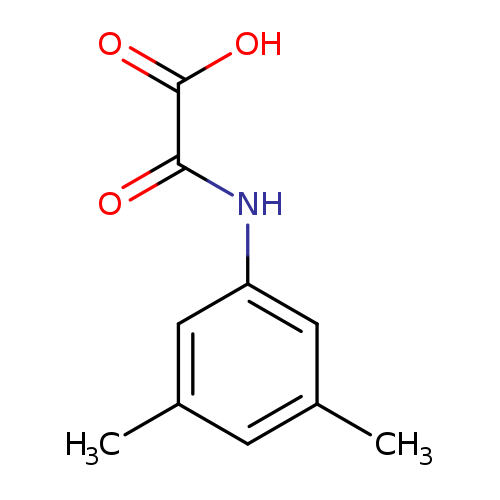
[(3,5-Dimethylphenyl)amino](oxo)acetic acidCatalog No.:AA008VI2 CAS No.:1018295-15-2 MDL No.:MFCD10018452 MF:C10H11NO3 MW:193.1992 |
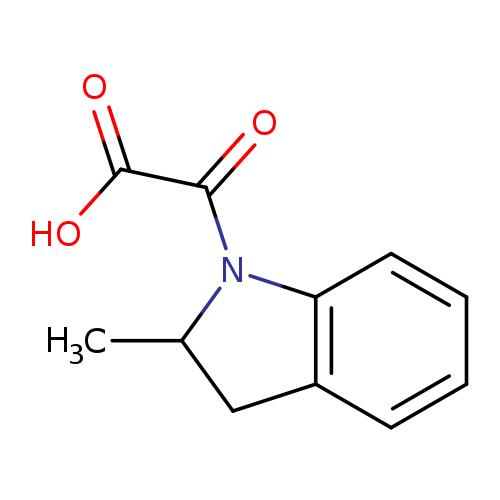
(2-Methyl-2,3-dihydro-1h-indol-1-yl)(oxo)acetic acidCatalog No.:AA0005KG CAS No.:1018295-36-7 MDL No.:MFCD10018504 MF:C11H11NO3 MW:205.2099 |
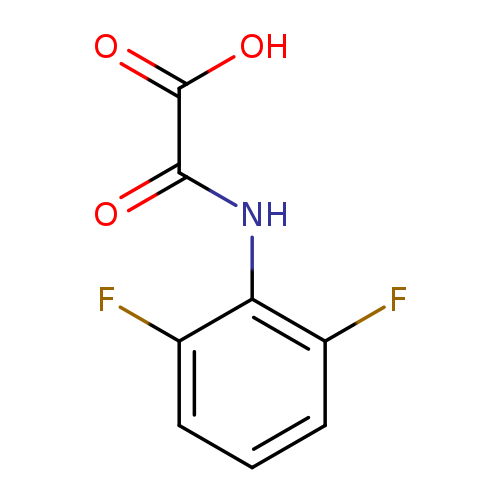
[(2,6-Difluorophenyl)carbamoyl]formic acidCatalog No.:AA0096D0 CAS No.:1018295-42-5 MDL No.:MFCD10018517 MF:C8H5F2NO3 MW:201.1270 |
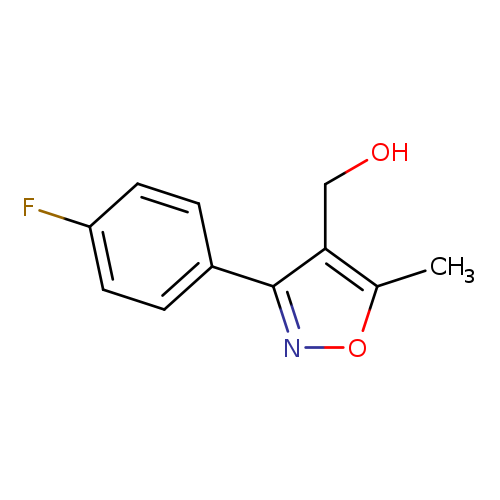
(3-(4-Fluorophenyl)-5-methylisoxazol-4-yl)methanolCatalog No.:AA0005KF CAS No.:1018297-63-6 MDL No.:MFCD10008830 MF:C11H10FNO2 MW:207.2010 |
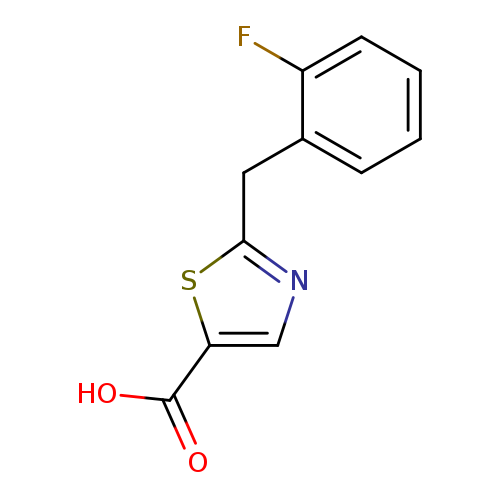
2-[(2-fluorophenyl)methyl]-1,3-thiazole-5-carboxylic acidCatalog No.:AA01AFBB CAS No.:1018297-66-9 MDL No.:MFCD10008833 MF:C11H8FNO2S MW:237.2501 |
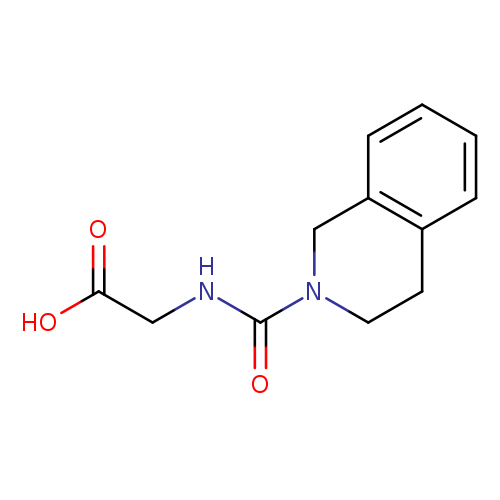
2-[(1,2,3,4-tetrahydroisoquinoline-2-carbonyl)amino]acetic acidCatalog No.:AA01AHA4 CAS No.:1018299-34-7 MDL No.:MFCD10011461 MF:C12H14N2O3 MW:234.2512 |
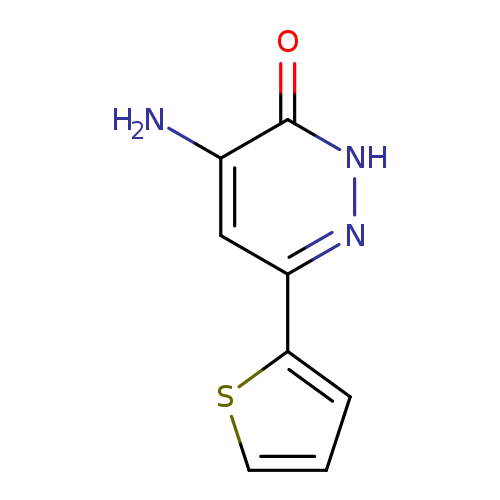
4-Amino-6-(2-thienyl)pyridazin-3(2(h))-oneCatalog No.:AA01ARCR CAS No.:1018299-56-3 MDL No.:MFCD10009040 MF:C8H7N3OS MW:193.2257 |
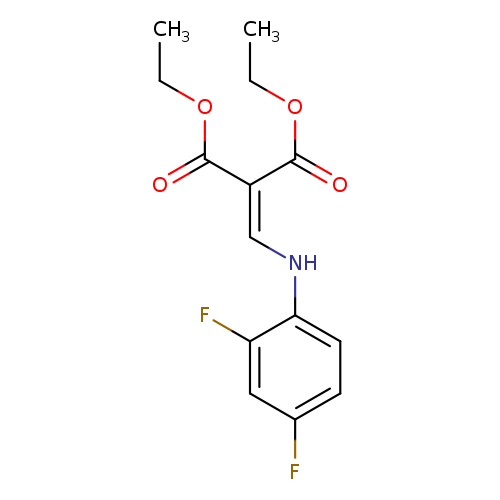
1,3-diethyl 2-{[(2,4-difluorophenyl)amino]methylidene}propanedioateCatalog No.:AA00IU9O CAS No.:101830-90-4 MDL No.:MFCD00173344 MF:C14H15F2NO4 MW:299.2700 |
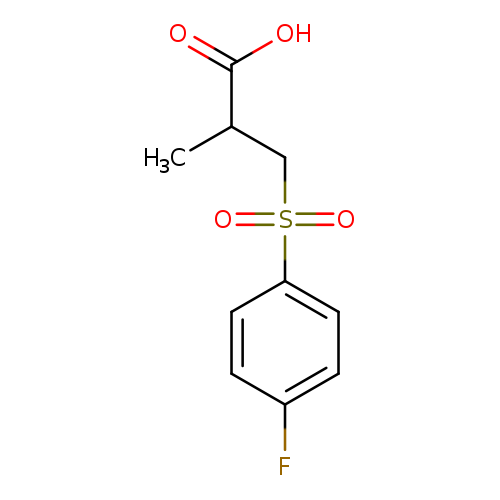
3-(4-fluorobenzenesulfonyl)-2-methylpropanoic acidCatalog No.:AA019ZAF CAS No.:1018300-28-1 MDL No.:MFCD10024397 MF:C10H11FO4S MW:246.2553 |
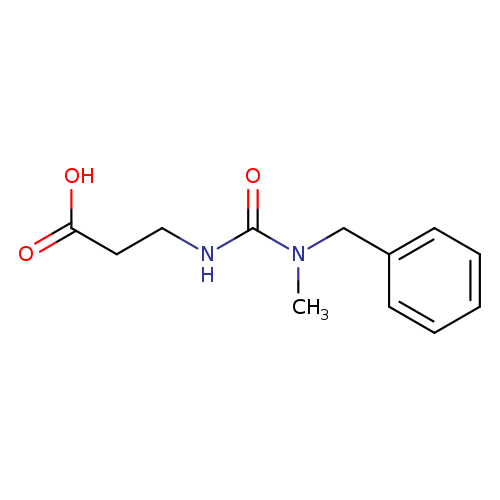
3-([Benzyl(methyl)carbamoyl]amino)propanoic acidCatalog No.:AA01A9UX CAS No.:1018300-35-0 MDL No.:MFCD10011914 MF:C12H16N2O3 MW:236.2670 |
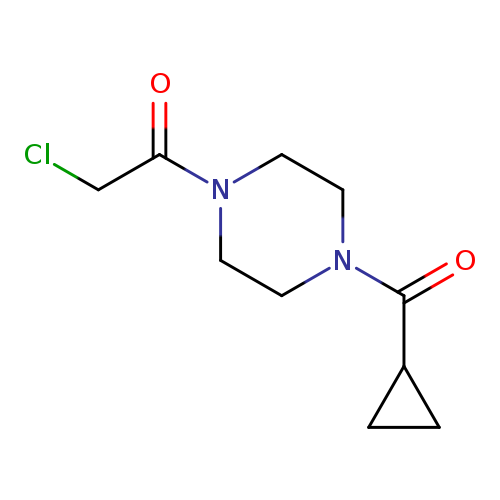
2-chloro-1-(4-cyclopropanecarbonylpiperazin-1-yl)ethan-1-oneCatalog No.:AA019VK3 CAS No.:1018304-01-2 MDL No.:MFCD10015802 MF:C10H15ClN2O2 MW:230.6913 |
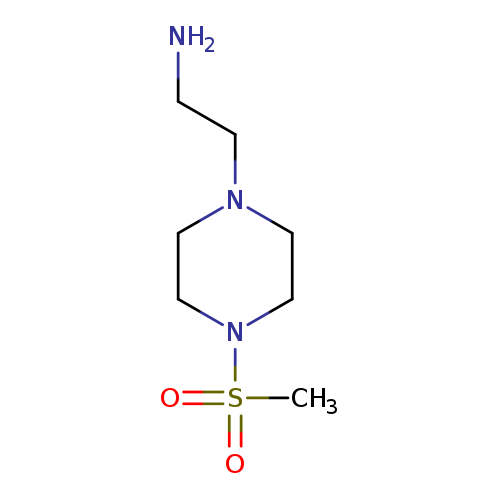
2-(4-(Methylsulfonyl)piperazin-1-yl)ethanamineCatalog No.:AA0098XX CAS No.:1018305-83-3 MDL No.:MFCD10015996 MF:C7H17N3O2S MW:207.2938 |
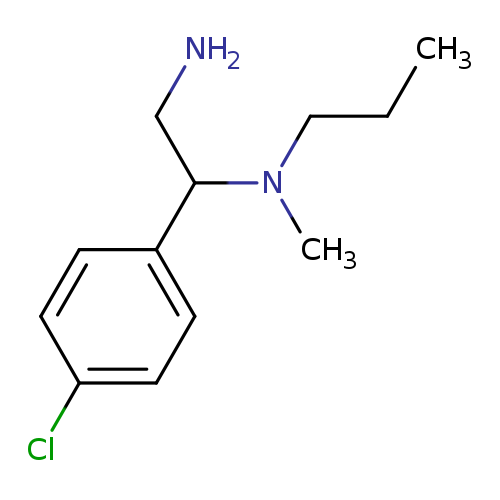
[2-amino-1-(4-chlorophenyl)ethyl](methyl)propylamineCatalog No.:AA01B9Y2 CAS No.:1018308-78-5 MDL No.:MFCD10026192 MF:C12H19ClN2 MW:226.7457 |
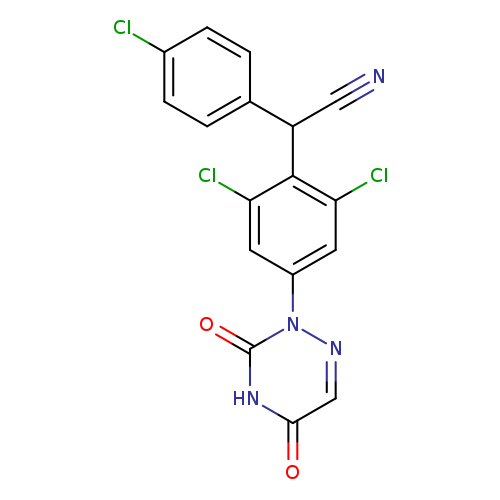
Benzeneacetonitrile, 2,6-dichloro-α-(4-chlorophenyl)-4-(4,5-dihydro-3,5-dioxo-1,2,4-triazin-2(3H)-yl)-Catalog No.:AA0005LQ CAS No.:101831-37-2 MDL No.:MFCD00867203 MF:C17H9Cl3N4O2 MW:407.6380 |
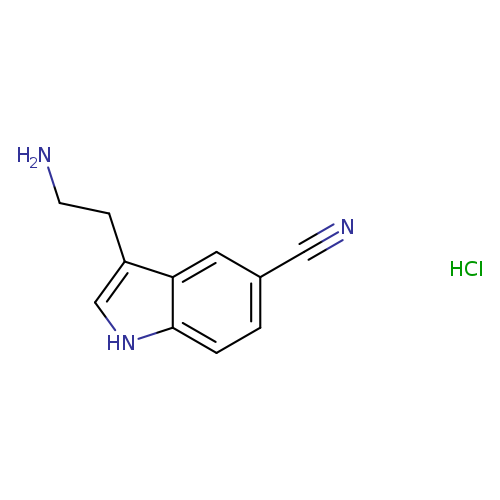
3-(2-Aminoethyl)-1h-indole-5-carbonitrile hydrochlorideCatalog No.:AA0005LM CAS No.:101831-71-4 MDL No.:MFCD01719136 MF:C11H12ClN3 MW:221.6861 |
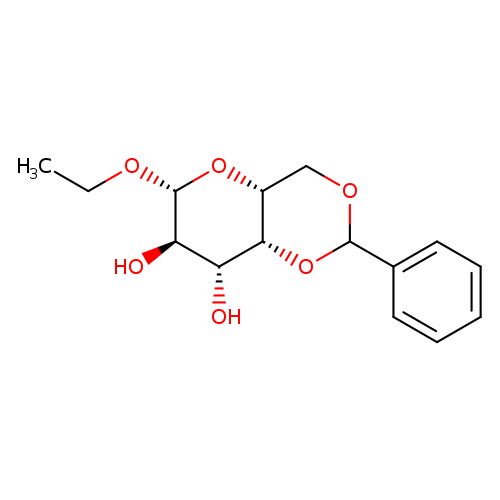
Ethyl4,6-o-benzylidene-beta-d-galactopyranosideCatalog No.:AA0005L7 CAS No.:101833-22-1 MDL No.:MFCD07367505 MF:C15H20O6 MW:296.3157 |
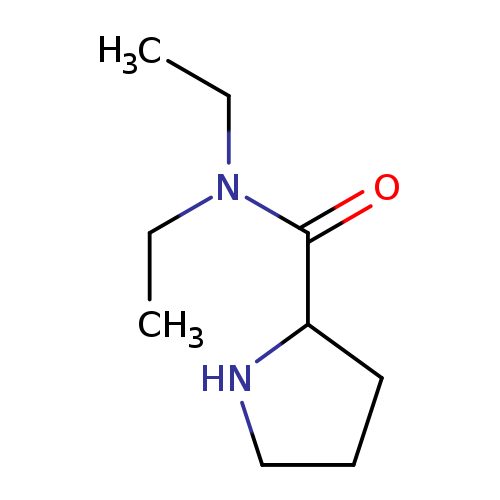
N,N-diethylpyrrolidine-2-carboxamideCatalog No.:AA01A7ER CAS No.:1018331-52-6 MDL No.:MFCD06357924 MF:C9H18N2O MW:170.2520 |
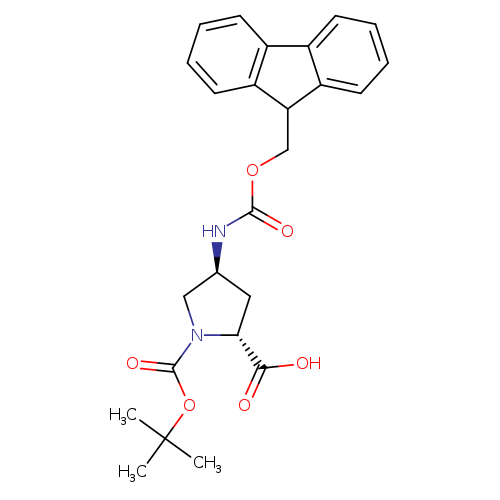
N-Boc-trans-4-n-fmoc-amino-d-prolineCatalog No.:AA00H9KC CAS No.:1018332-23-4 MDL No.:MFCD11519438 MF:C25H28N2O6 MW:452.4996 |
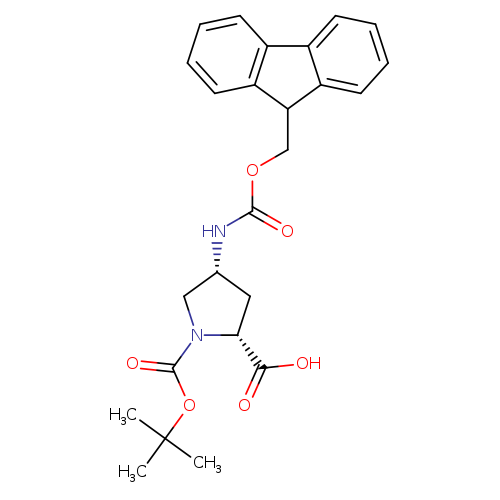
(2R,4R)-4-(((9H-Fluoren-9-yl)methoxy)carbonylamino)-1-(tert-butoxycarbonyl)pyrrolidine-2-carboxylic acidCatalog No.:AA0098XZ CAS No.:1018332-24-5 MDL No.:MFCD11519437 MF:C25H28N2O6 MW:452.4996 |
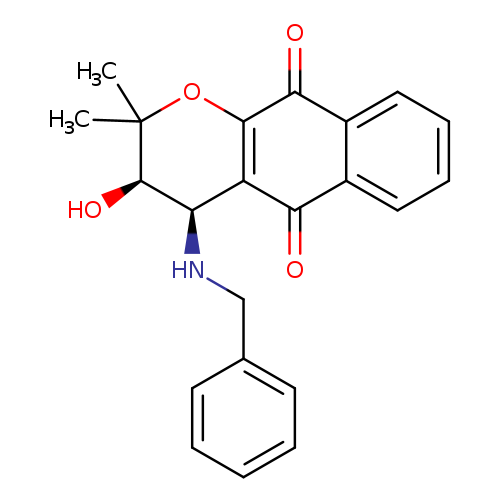
2H-Naphtho[2,3-b]pyran-5,10-dione, 3,4-dihydro-3-hydroxy-2,2-dimethyl-4-[(phenylmethyl)amino]-, (3R,4R)-rel-Catalog No.:AA0005LU CAS No.:1018340-07-2 MDL No.:MFCD18428034 MF:C22H21NO4 MW:363.4064 |
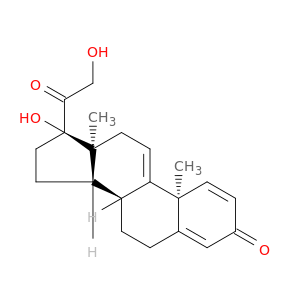
17,21-Dihydroxypregna-1,4,9(11)-triene-3,20-dioneCatalog No.:AA01DZ9N CAS No.:10184-69-7 MDL No.: MF: MW: |
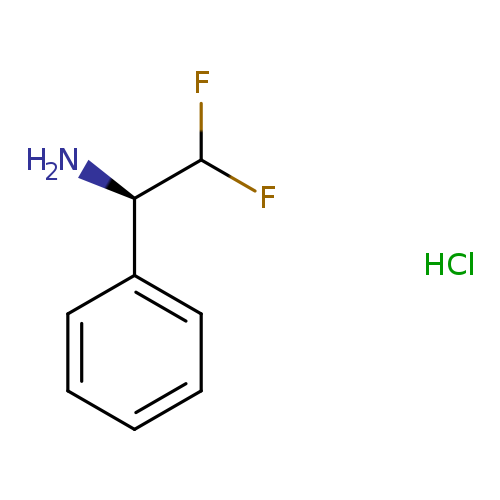
(αR)-α-(Difluoromethyl)-benzenemethanamineHydrochlorideCatalog No.:AA01DZ9O CAS No.:1018434-64-4 MDL No.:MFCD26572458 MF:C8H10ClF2N MW:193.6215 |
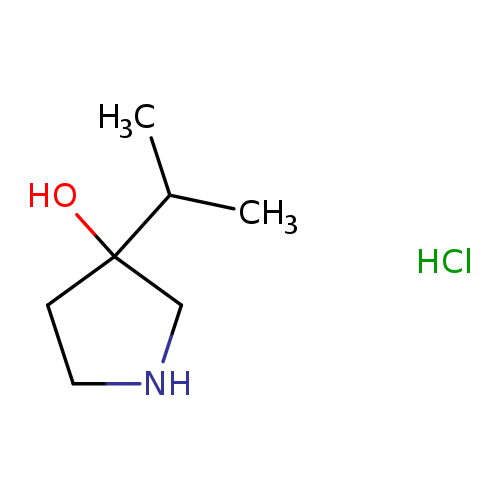
3-(propan-2-yl)pyrrolidin-3-ol hydrochlorideCatalog No.:AA01EM8L CAS No.:1018442-97-1 MDL No.:MFCD31616973 MF:C7H16ClNO MW:165.6610 |
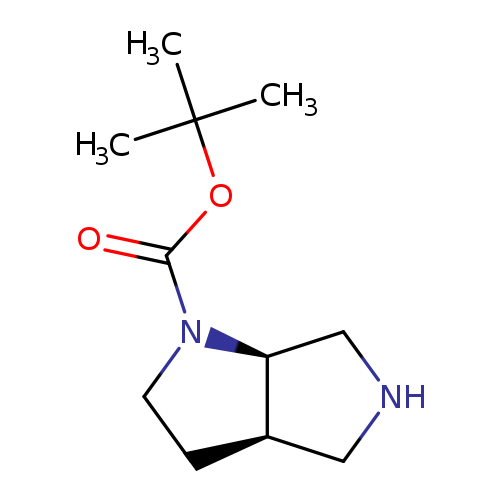
(3As,6as)-rel-tert-butyl hexahydropyrrolo[3,4-b]pyrrole-1(2h)-carboxylateCatalog No.:AA0005MP CAS No.:1018443-32-7 MDL No.:MFCD12198661 MF:C11H20N2O2 MW:212.2887 |
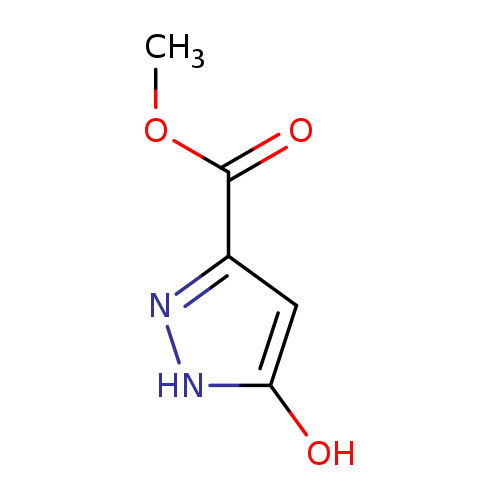
Methyl 5-hydroxy-pyrazole-3-carboxylateCatalog No.:AA0005MO CAS No.:1018446-60-0 MDL No.:MFCD09878841 MF:C5H6N2O3 MW:142.1127 |
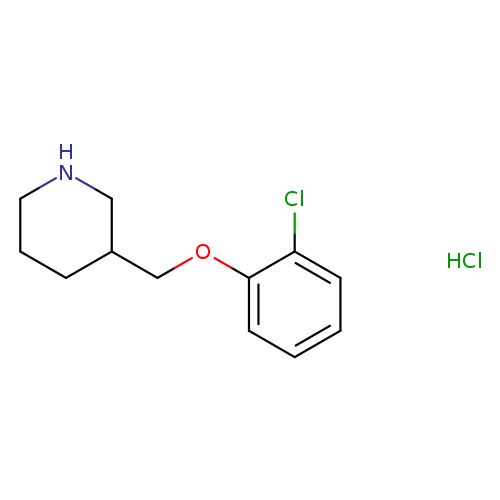
3-[(2-Chlorophenoxy)methyl]piperidine hydrochlorideCatalog No.:AA01A2CB CAS No.:1018446-64-4 MDL No.:MFCD08447187 MF:C12H17Cl2NO MW:262.1755 |
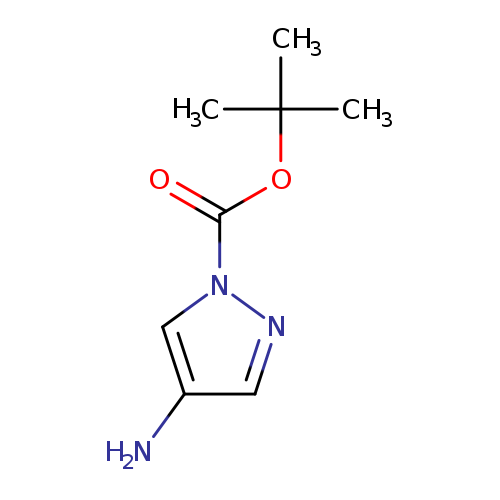
tert-Butyl 4-amino-1h-pyrazole-1-carboxylateCatalog No.:AA0005MN CAS No.:1018446-95-1 MDL No.:MFCD20697038 MF:C8H13N3O2 MW:183.2077 |
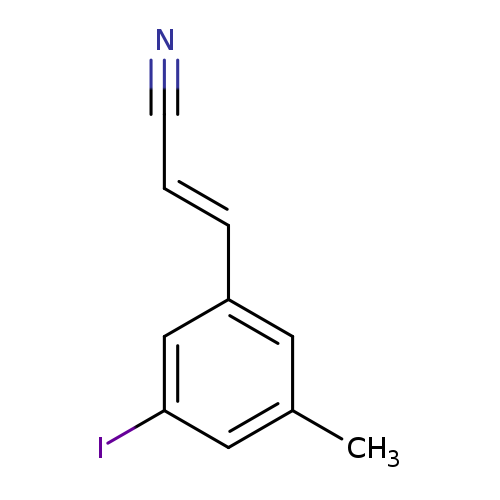
3-Iodo-5-methylcinnamonitrileCatalog No.:AA00H9KF CAS No.:1018450-07-1 MDL No.:MFCD21496416 MF:C10H8IN MW:269.0817 |
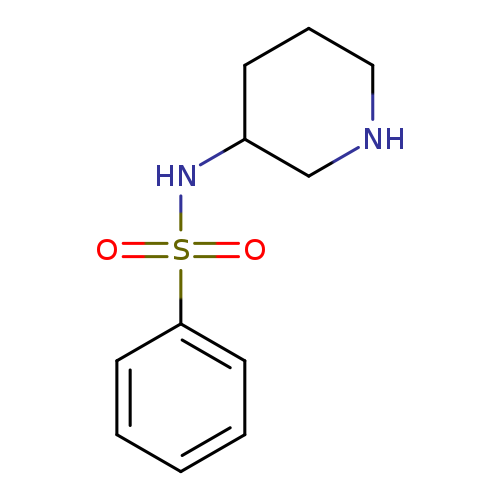
N-(piperidin-3-yl)benzenesulfonamideCatalog No.:AA01AB07 CAS No.:1018458-26-8 MDL No.:MFCD09810197 MF:C11H16N2O2S MW:240.3219 |
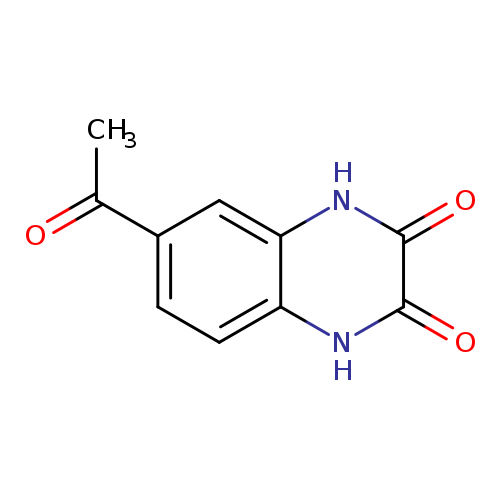
6-Acetyl-1,2,3,4-tetrahydroquinoxaline-2,3-dioneCatalog No.:AA01EB9C CAS No.:1018458-42-8 MDL No.:MFCD09813951 MF:C10H8N2O3 MW:204.1821 |
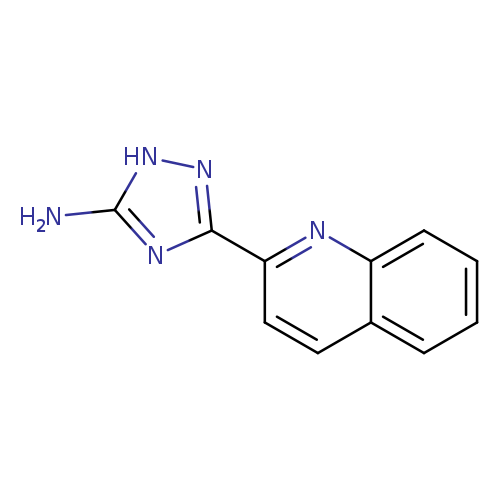
3-(quinolin-2-yl)-1H-1,2,4-triazol-5-amineCatalog No.:AA01AAA7 CAS No.:1018458-44-0 MDL No.:MFCD16990688 MF:C11H9N5 MW:211.2227 |
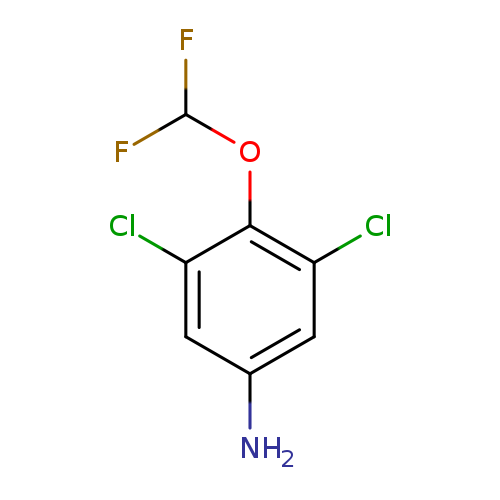
3,5-dichloro-4-(difluoromethoxy)anilineCatalog No.:AA019VEY CAS No.:101847-52-3 MDL No.:MFCD11647706 MF:C7H5Cl2F2NO MW:228.0235 |
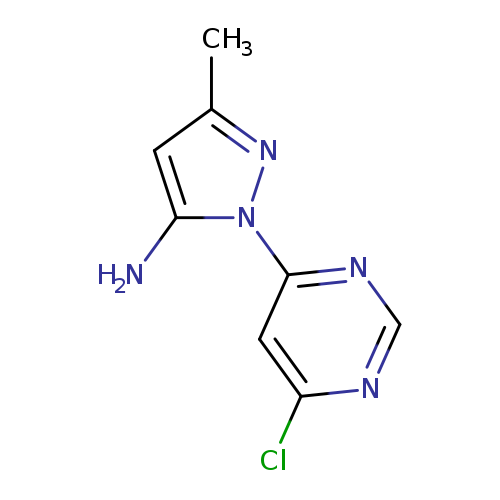
1-(6-Chloropyrimidin-4-yl)-3-methyl-1H-pyrazol-5-amineCatalog No.:AA0005MK CAS No.:1018473-22-7 MDL No.:MFCD19440630 MF:C8H8ClN5 MW:209.6356 |
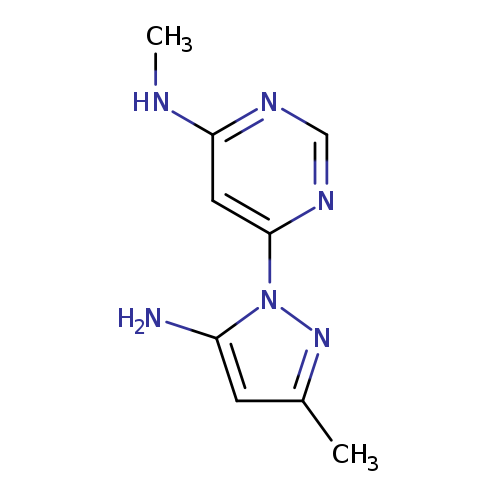
6-(5-Amino-3-methyl-1h-pyrazol-1-yl)-n-methylpyrimidin-4-amineCatalog No.:AA01BG4B CAS No.:1018473-23-8 MDL No.:MFCD24613077 MF:C9H12N6 MW:204.2318 |
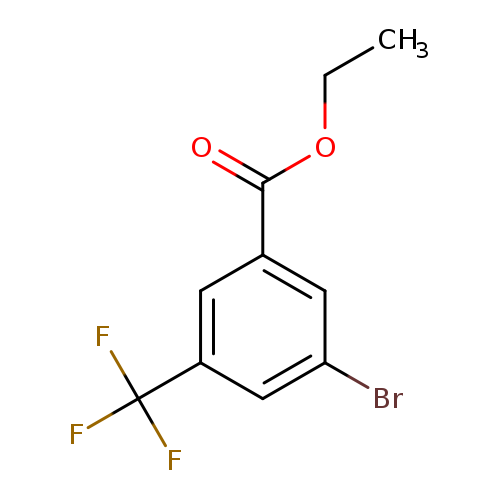
Ethyl 3-bromo-5-(trifluoromethyl)benzoateCatalog No.:AA01FBDC CAS No.:1018473-24-9 MDL No.:MFCD14698180 MF:C10H8BrF3O2 MW:297.0685 |
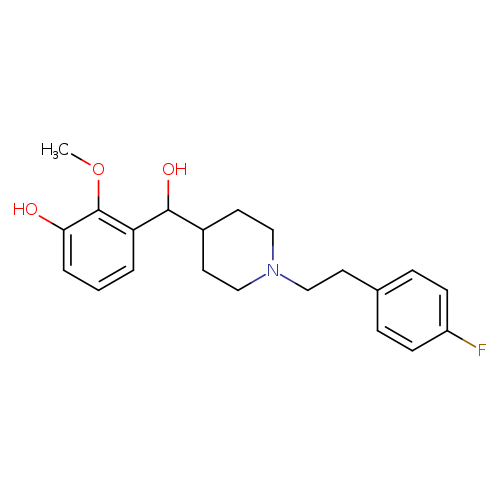
(±)-MDL 105725Catalog No.:AA0097C4 CAS No.:1018473-89-6 MDL No.:MFCD10565917 MF:C21H26FNO3 MW:359.4344 |
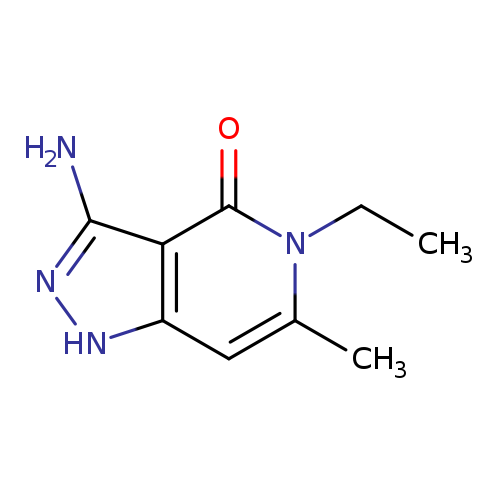
3-amino-5-ethyl-6-methyl-1H,4H,5H-pyrazolo[4,3-c]pyridin-4-oneCatalog No.:AA01C2Y2 CAS No.:1018497-10-3 MDL No.:MFCD10008266 MF:C9H12N4O MW:192.2178 |
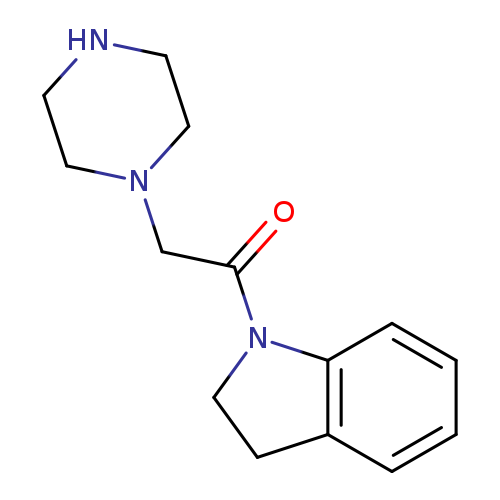
1-(2,3-dihydro-1H-indol-1-yl)-2-(piperazin-1-yl)ethan-1-oneCatalog No.:AA019UOI CAS No.:1018497-78-3 MDL No.:MFCD10011126 MF:C14H19N3O MW:245.3202 |
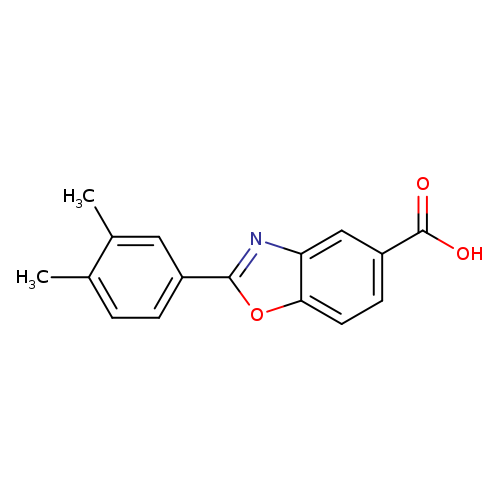
2-(3,4-Dimethylphenyl)-1,3-benzoxazole-5-carboxylic acidCatalog No.:AA01EKHF CAS No.:1018498-33-3 MDL No.:MFCD10024020 MF:C16H13NO3 MW:267.2793 |
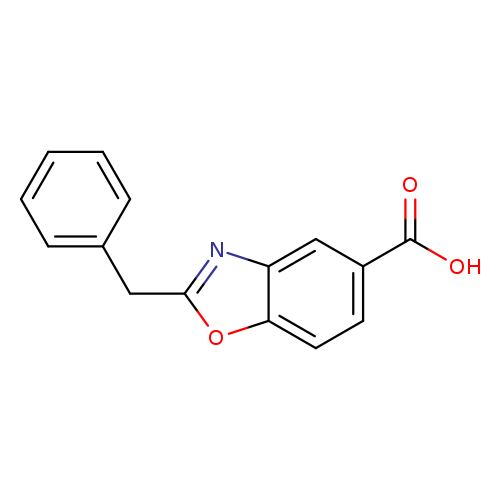
2-benzyl-1,3-benzoxazole-5-carboxylic acidCatalog No.:AA01DX43 CAS No.:1018498-36-6 MDL No.:MFCD10024025 MF:C15H11NO3 MW:253.2527 |
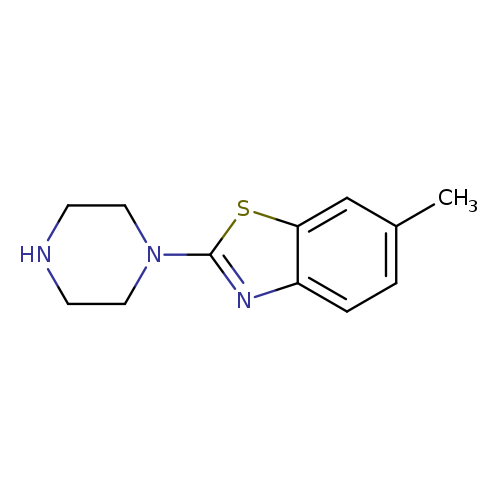
6-Methyl-2-piperazin-1-yl-1,3-benzothiazoleCatalog No.:AA01ARCL CAS No.:1018498-61-7 MDL No.:MFCD10008524 MF:C12H15N3S MW:233.3326 |
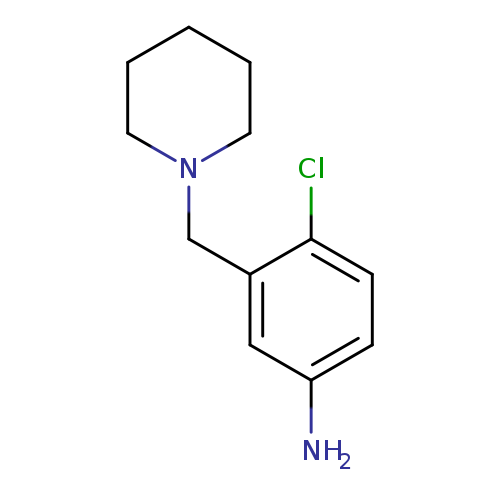
4-chloro-3-(piperidin-1-ylmethyl)anilineCatalog No.:AA019TD3 CAS No.:1018498-91-3 MDL No.:MFCD10021402 MF:C12H17ClN2 MW:224.7298 |
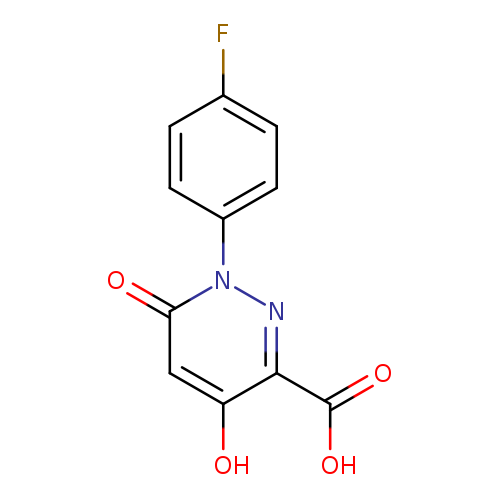
1-(4-Fluorophenyl)-4-hydroxy-6-oxo-1,6-dihydropyridazine-3-carboxylic acidCatalog No.:AA01FMZK CAS No.:1018498-97-9 MDL No.:MFCD10008579 MF:C11H7FN2O4 MW:250.1827 |

1,2,3,6-Tetrahydro-α,4-bis(p-methoxyphenyl)-1-methyl-3-pyridinemethanolCatalog No.:AA01CCIE CAS No.:10185-04-3 MDL No.:MFCD31568303 MF: MW: |
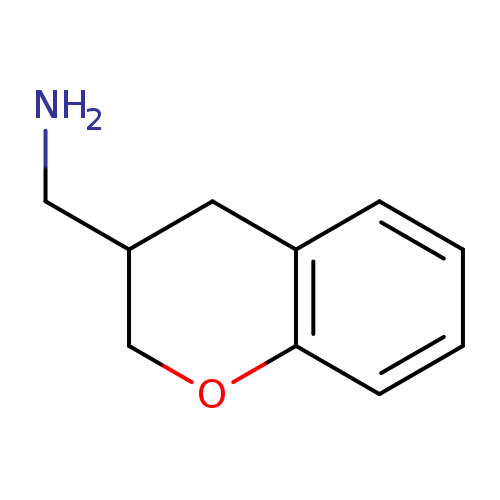
Chroman-3-yl-methylamineCatalog No.:AA0005N3 CAS No.:10185-46-3 MDL No.:MFCD04114627 MF:C10H13NO MW:163.2163 |
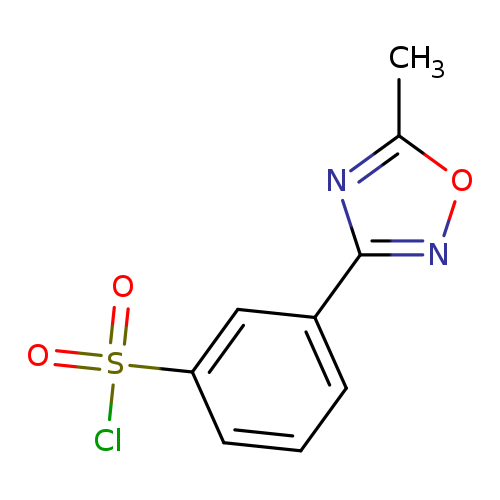
3-(5-Methyl-1,2,4-oxadiazol-3-yl)benzenesulfonyl chlorideCatalog No.:AA0005N2 CAS No.:10185-62-3 MDL No.:MFCD06797486 MF:C9H7ClN2O3S MW:258.6815 |
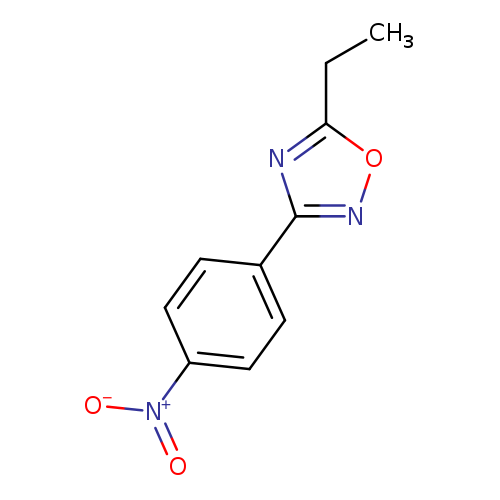
5-Ethyl-3-(4-nitrophenyl)-1,2,4-oxadiazoleCatalog No.:AA0005NV CAS No.:10185-65-6 MDL No.:MFCD09972166 MF:C10H9N3O3 MW:219.1968 |
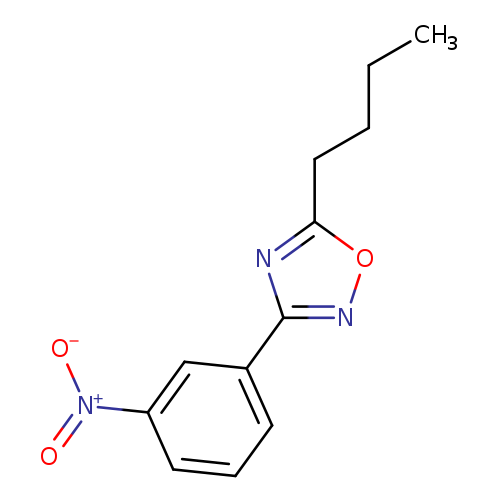
5-Butyl-3-(3-nitrophenyl)-1,2,4-oxadiazoleCatalog No.:AA0005NU CAS No.:10185-66-7 MDL No.:MFCD10699664 MF:C12H13N3O3 MW:247.2499 |
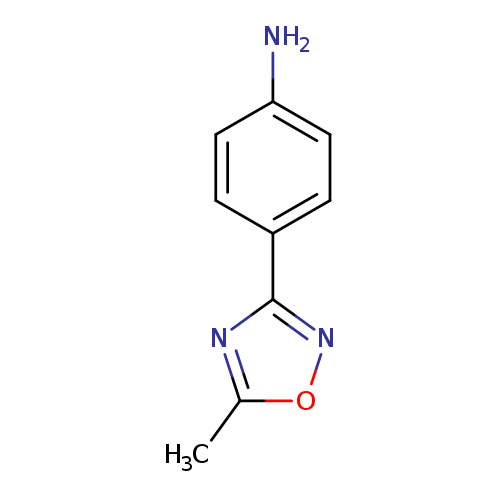
4-(5-methyl-1,2,4-oxadiazol-3-yl)anilineCatalog No.:AA0005NT CAS No.:10185-68-9 MDL No.:MFCD08435856 MF:C9H9N3O MW:175.1873 |
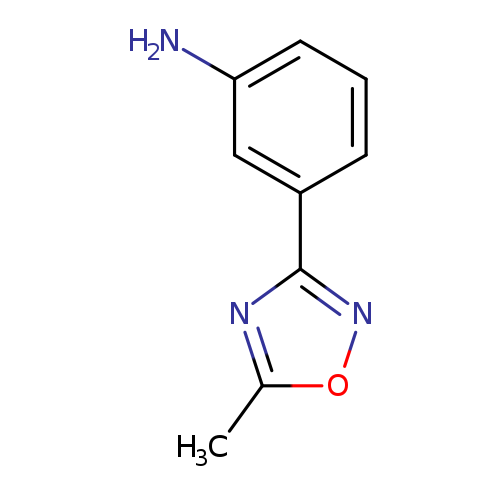
3-(5-Methyl-1,2,4-oxadiazol-3-yl)anilineCatalog No.:AA0005NS CAS No.:10185-69-0 MDL No.:MFCD08443707 MF:C9H9N3O MW:175.1873 |
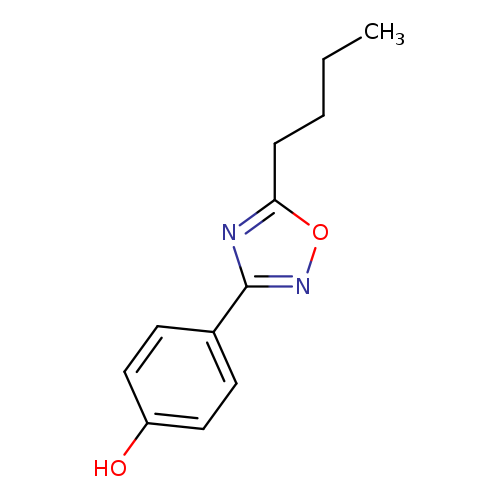
4-(5-butyl-1,2,4-oxadiazol-3-yl)phenolCatalog No.:AA019X09 CAS No.:10185-79-2 MDL No.:MFCD10691947 MF:C12H14N2O2 MW:218.2518 |
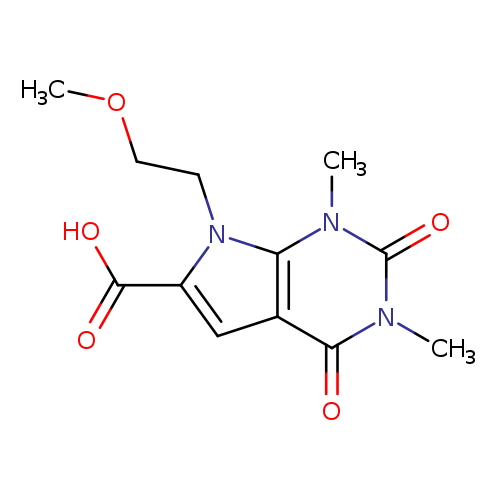
7-(2-Methoxyethyl)-1,3-dimethyl-2,4-dioxo-2,3,4,7-tetrahydro-1H-pyrrolo[2,3-d]pyrimidine-6-carboxylic acidCatalog No.:AA00900C CAS No.:1018501-32-0 MDL No.:MFCD10007813 MF:C12H15N3O5 MW:281.2646 |
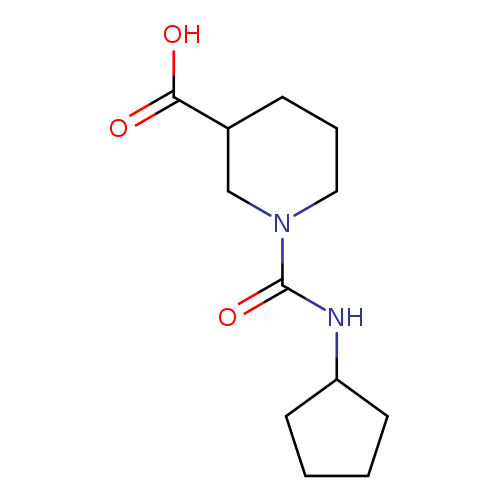
1-(cyclopentylcarbamoyl)piperidine-3-carboxylic acidCatalog No.:AA019T4C CAS No.:1018503-75-7 MDL No.:MFCD10012142 MF:C12H20N2O3 MW:240.2988 |
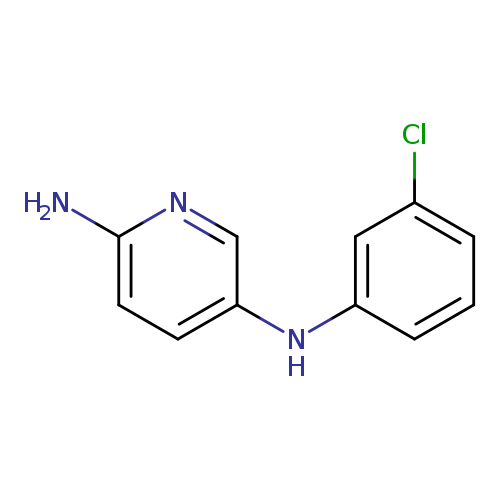
N5-(3-chlorophenyl)pyridine-2,5-diamineCatalog No.:AA01B4Y9 CAS No.:1018503-96-2 MDL No.:MFCD10009414 MF:C11H10ClN3 MW:219.6702 |
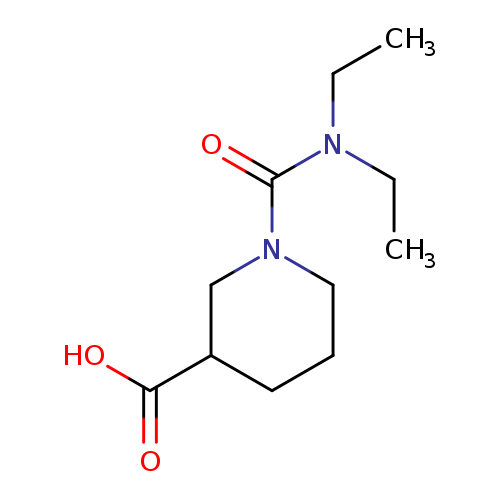
1-(diethylcarbamoyl)piperidine-3-carboxylic acidCatalog No.:AA019MQW CAS No.:1018504-07-8 MDL No.:MFCD10012173 MF:C11H20N2O3 MW:228.2881 |
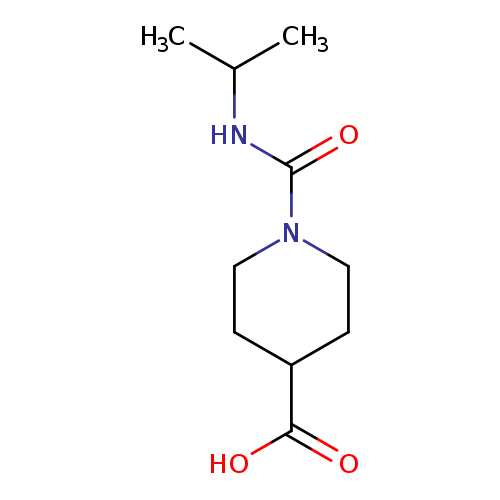
1-[(propan-2-yl)carbamoyl]piperidine-4-carboxylic acidCatalog No.:AA019MPP CAS No.:1018504-63-6 MDL No.:MFCD10012220 MF:C10H18N2O3 MW:214.2615 |
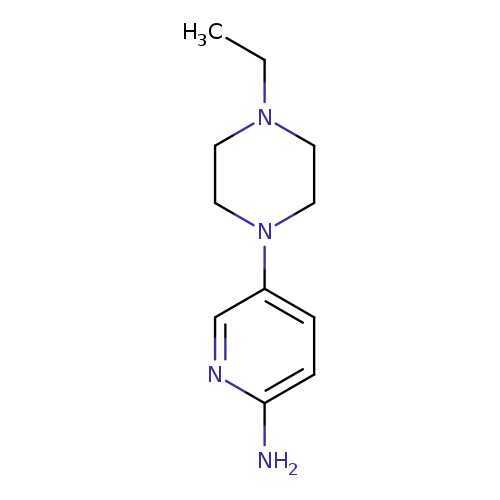
5-(4-Ethylpiperazin-1-yl)pyridin-2-amineCatalog No.:AA0005NB CAS No.:1018505-59-3 MDL No.:MFCD10009575 MF:C11H18N4 MW:206.2874 |
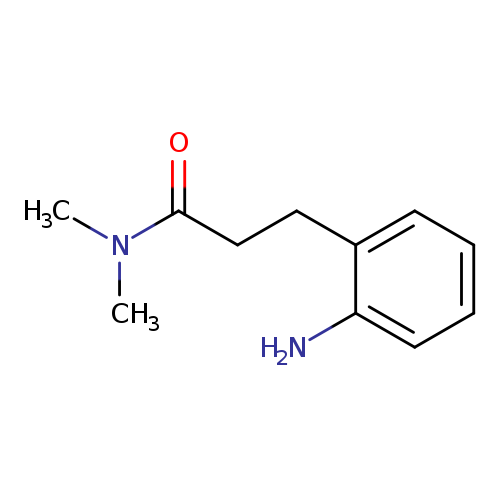
Benzenepropanamide, 2-amino-N,N-dimethyl-Catalog No.:AA0005NA CAS No.:1018506-33-6 MDL No.:MFCD10022148 MF:C11H16N2O MW:192.2575 |
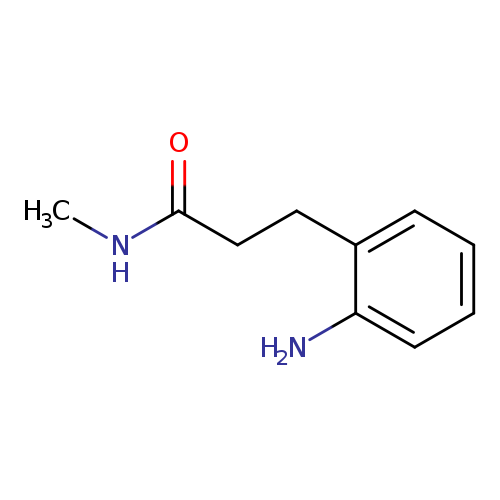
3-(2-aminophenyl)-N-methylpropanamideCatalog No.:AA008V4S CAS No.:1018506-37-0 MDL No.:MFCD10022153 MF:C10H14N2O MW:178.2310 |
([1-(PHENOXYACETYL)PIPERIDIN-4-YL]METHYL)AMINECatalog No.:AA01APEY CAS No.:1018507-00-0 MDL No.:MFCD10016311 MF:C14H20N2O2 MW:248.3208 |
2-[(3-hydroxypropyl)amino]propanenitrileCatalog No.:AA01BABA CAS No.:1018512-65-6 MDL No.:MFCD10014403 MF:C6H12N2O MW:128.1723 |
3-Isopropylimidazo[1,5-a]pyridine-1-carbaldehydeCatalog No.:AA01AIV6 CAS No.:1018516-49-8 MDL No.:MFCD07381139 MF:C11H12N2O MW:188.2258 |
2-([BENZYL(ETHYL)AMINO]METHYL)ANILINECatalog No.:AA01C667 CAS No.:1018517-28-6 MDL No.:MFCD10021235 MF:C16H20N2 MW:240.3434 |
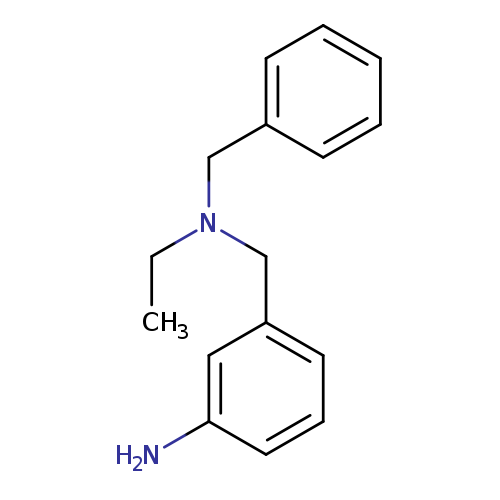
3-((Benzyl(ethyl)amino)methyl)anilineCatalog No.:AA01C668 CAS No.:1018517-42-4 MDL No.:MFCD10021240 MF:C16H20N2 MW:240.3434 |
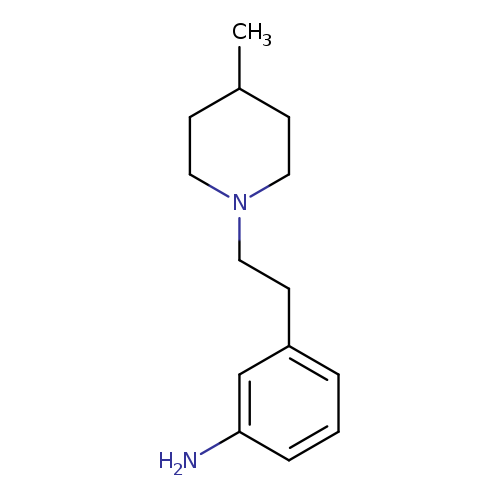
3-[2-(4-methylpiperidin-1-yl)ethyl]anilineCatalog No.:AA01A40O CAS No.:1018517-70-8 MDL No.:MFCD10021265 MF:C14H22N2 MW:218.3379 |
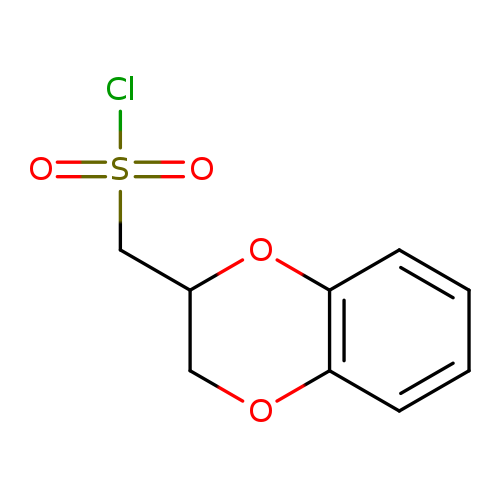
(2,3-dihydro-1,4-benzodioxin-2-yl)methanesulfonyl chlorideCatalog No.:AA01B316 CAS No.:1018520-20-1 MDL No.:MFCD10015221 MF:C9H9ClO4S MW:248.6834 |
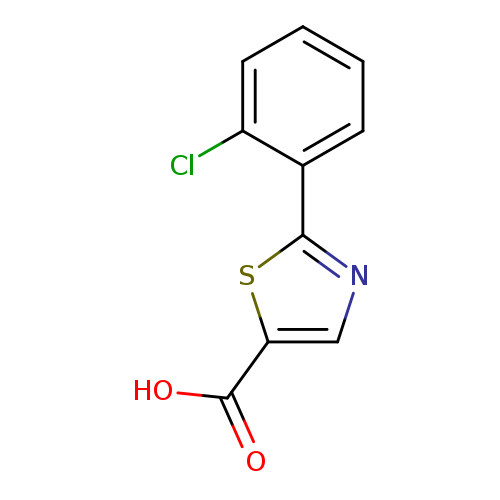
2-(2-Chlorophenyl)thiazole-5-carboxylic acidCatalog No.:AA01A829 CAS No.:1018520-26-7 MDL No.:MFCD07376793 MF:C10H6ClNO2S MW:239.6781 |
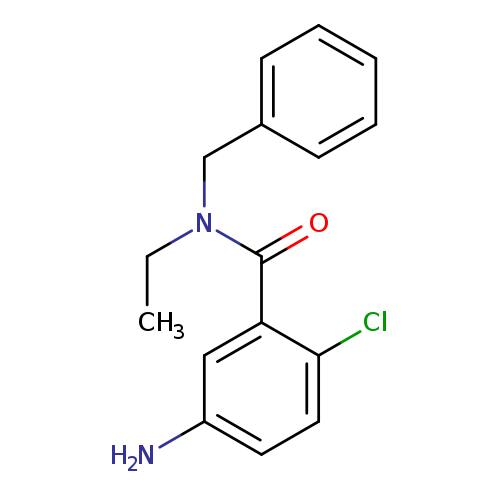
5-amino-N-benzyl-2-chloro-N-ethylbenzamideCatalog No.:AA019VPX CAS No.:1018522-83-2 MDL No.:MFCD10021773 MF:C16H17ClN2O MW:288.7720 |
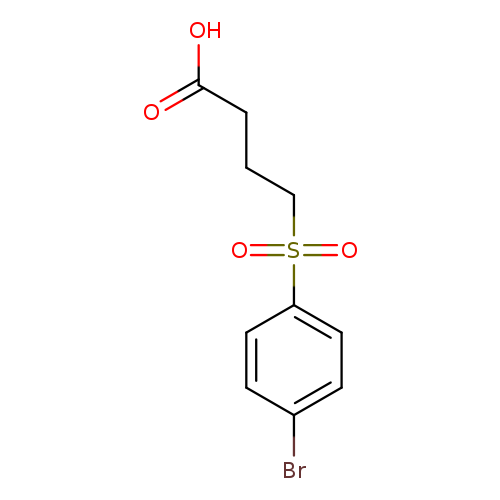
4-(4-bromobenzenesulfonyl)butanoic acidCatalog No.:AA01AGLB CAS No.:1018522-96-7 MDL No.:MFCD10024476 MF:C10H11BrO4S MW:307.1609 |
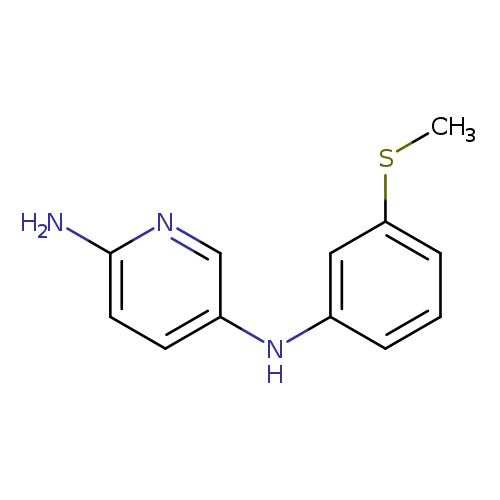
N5-[3-(methylsulfanyl)phenyl]pyridine-2,5-diamineCatalog No.:AA01B4W2 CAS No.:1018523-30-2 MDL No.:MFCD10009226 MF:C12H13N3S MW:231.3167 |
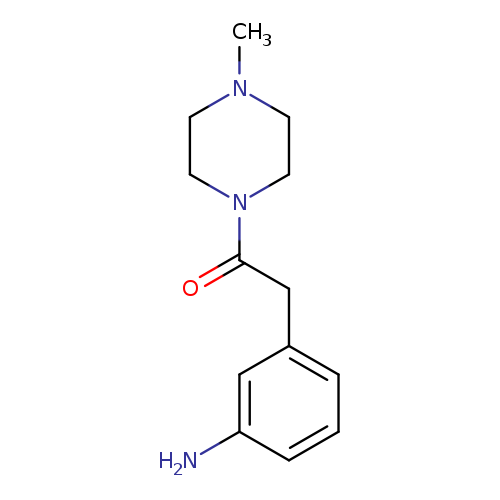
2-(3-aminophenyl)-1-(4-methylpiperazin-1-yl)ethan-1-oneCatalog No.:AA019TGV CAS No.:1018525-88-6 MDL No.:MFCD10019024 MF:C13H19N3O MW:233.3095 |
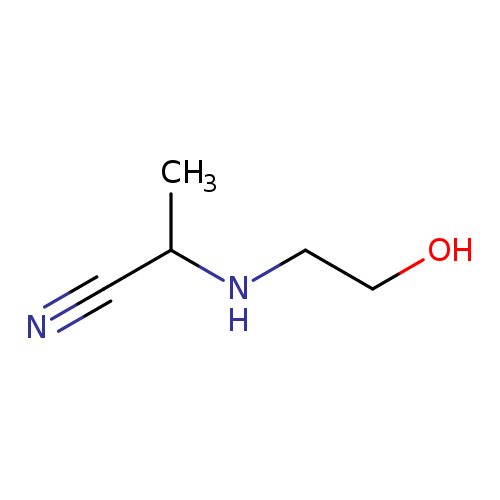
2-[(2-hydroxyethyl)amino]propanenitrileCatalog No.:AA01BAR8 CAS No.:1018539-22-4 MDL No.:MFCD10014401 MF:C5H10N2O MW:114.1457 |
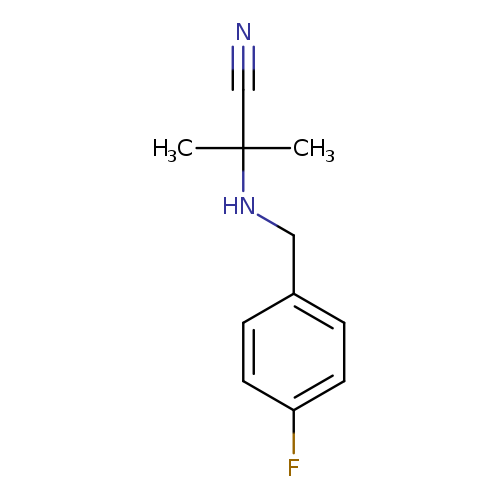
2-((4-Fluorobenzyl)amino)-2-methylpropanenitrileCatalog No.:AA00IZ97 CAS No.:1018539-78-0 MDL No.:MFCD10014477 MF:C11H13FN2 MW:192.2327 |
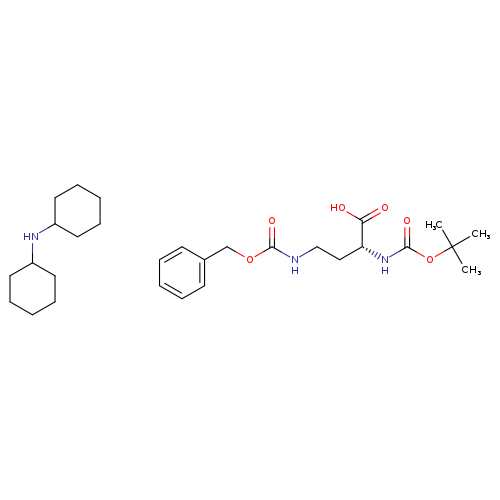
Boc-d-dab(z)-oh dchaCatalog No.:AA0005OI CAS No.:101854-42-6 MDL No.:MFCD00798628 MF:C29H47N3O6 MW:533.7000 |
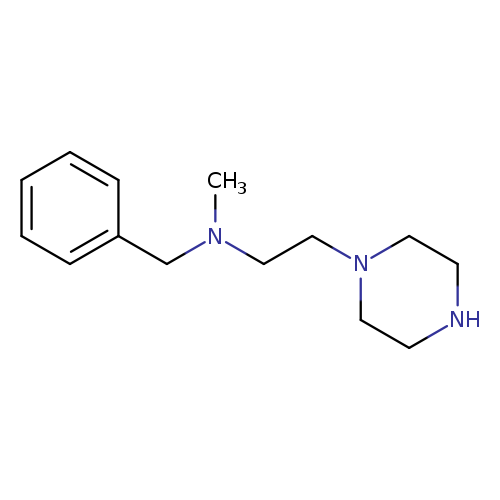
benzyl(methyl)[2-(piperazin-1-yl)ethyl]amineCatalog No.:AA01AARF CAS No.:1018545-42-0 MDL No.:MFCD10015229 MF:C14H23N3 MW:233.3525 |
2-(4-Fluorophenoxy)ethanesulfonyl chlorideCatalog No.:AA008V58 CAS No.:1018548-27-0 MDL No.:MFCD10022931 MF:C8H8ClFO3S MW:238.6637 |
2-(4-cyclohexanecarbonylpiperazin-1-yl)ethan-1-amineCatalog No.:AA01AHTR CAS No.:1018552-50-5 MDL No.:MFCD10015952 MF:C13H25N3O MW:239.3571 |
1-{6-methylthieno[2,3-d]pyrimidin-4-yl}piperazineCatalog No.:AA00VSRY CAS No.:1018554-94-3 MDL No.:MFCD10009395 MF:C11H14N4S MW:234.3207 |
1-(6-Amino-2,3-dihydro-1h-indol-1-yl)-2-phenylethan-1-oneCatalog No.:AA01AMRA CAS No.:1018559-05-1 MDL No.:MFCD10016495 MF:C16H16N2O MW:252.3110 |
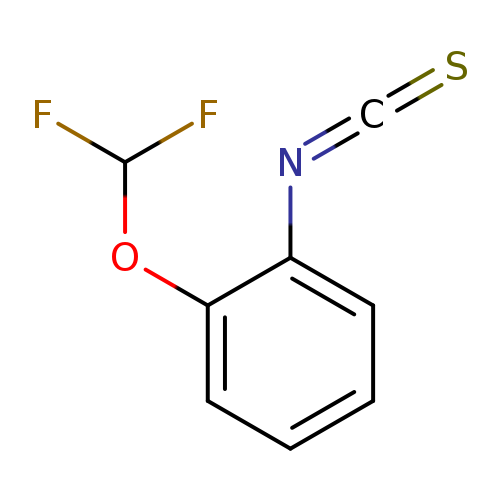
2-DIFLUOROMETHOXYPHENYL ISOTHIOCYANATECatalog No.:AA008YKB CAS No.:101856-90-0 MDL No.:MFCD02683534 MF:C8H5F2NOS MW:201.1932 |
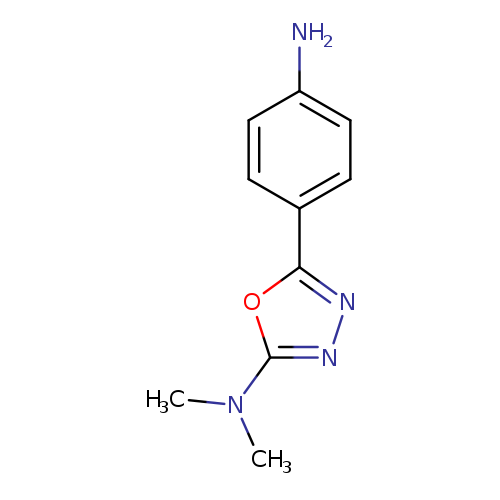
5-(4-aminophenyl)-N,N-dimethyl-1,3,4-oxadiazol-2-amineCatalog No.:AA01A0XZ CAS No.:1018563-25-1 MDL No.:MFCD10021144 MF:C10H12N4O MW:204.2285 |
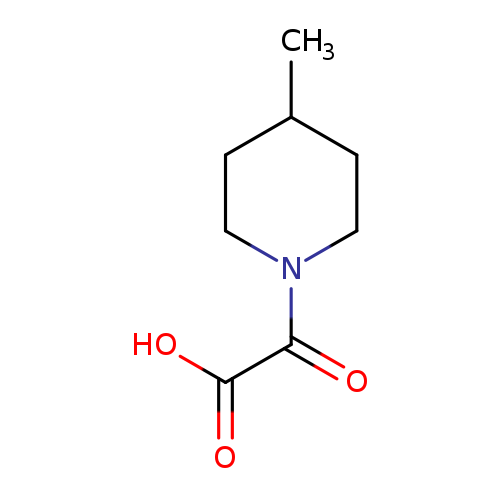
2-(4-Methylpiperidin-1-yl)-2-oxoacetic acidCatalog No.:AA01AC3F CAS No.:1018565-49-5 MDL No.:MFCD10018380 MF:C8H13NO3 MW:171.1937 |
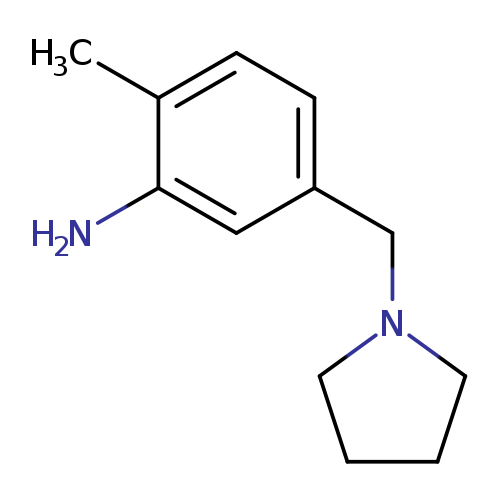
2-methyl-5-(pyrrolidin-1-ylmethyl)anilineCatalog No.:AA01A14C CAS No.:1018565-55-3 MDL No.:MFCD10021454 MF:C12H18N2 MW:190.2847 |
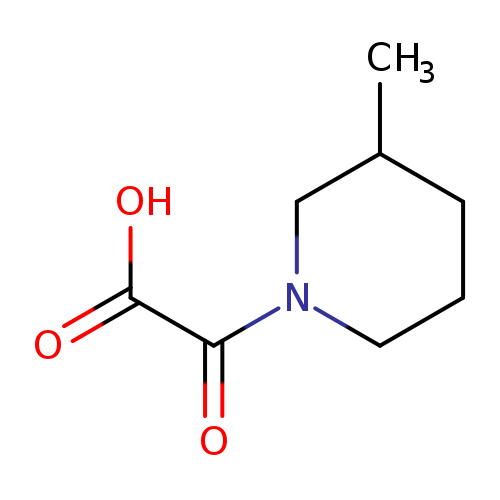
2-(3-methylpiperidin-1-yl)-2-oxoacetic acidCatalog No.:AA01AC3G CAS No.:1018565-95-1 MDL No.:MFCD10018446 MF:C8H13NO3 MW:171.1937 |
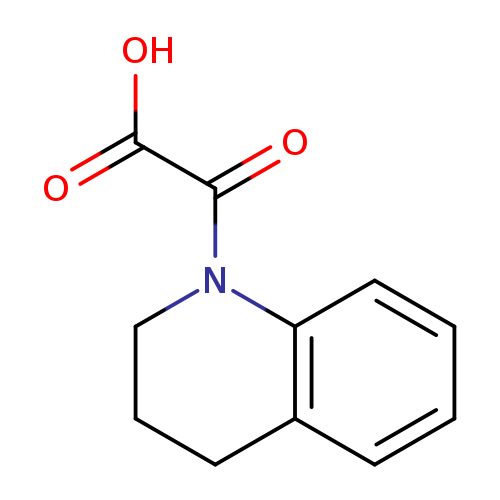
3,4-Dihydroquinolin-1(2h)-yl(oxo)acetic acidCatalog No.:AA0005NW CAS No.:1018565-99-5 MDL No.:MFCD10018451 MF:C11H11NO3 MW:205.2099 |
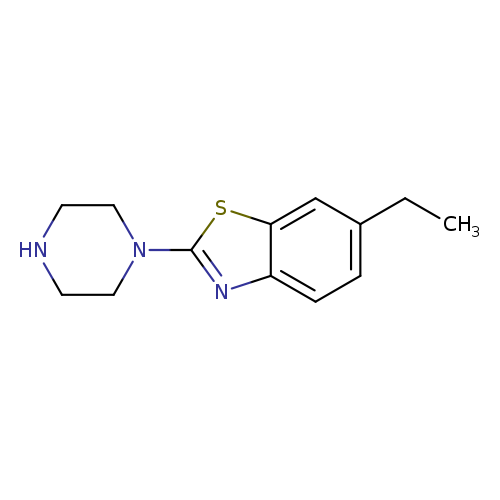
6-Ethyl-2-piperazin-1-yl-1,3-benzothiazoleCatalog No.:AA01ARCM CAS No.:1018566-85-2 MDL No.:MFCD10008525 MF:C13H17N3S MW:247.3592 |
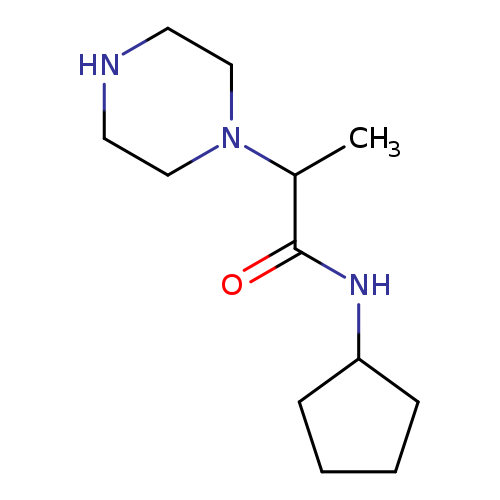
N-cyclopentyl-2-(piperazin-1-yl)propanamideCatalog No.:AA01AANM CAS No.:1018568-87-0 MDL No.:MFCD10011188 MF:C12H23N3O MW:225.3305 |
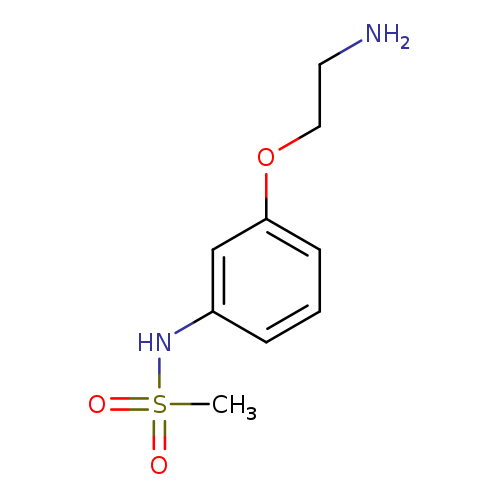
N-[3-(2-Aminoethoxy)phenyl]methanesulfonamideCatalog No.:AA01A3RC CAS No.:1018569-96-4 MDL No.:MFCD10025262 MF:C9H14N2O3S MW:230.2841 |
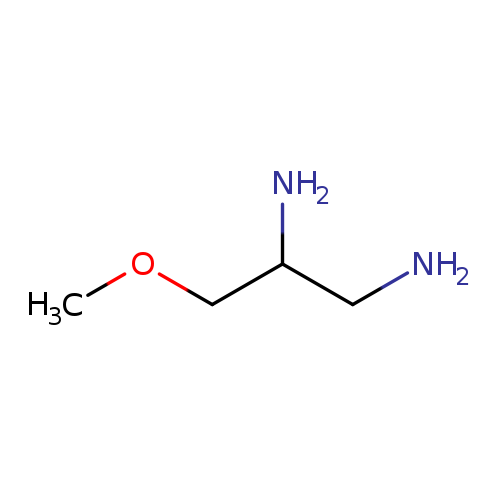
1,2-Propanediamine, 3-methoxy-Catalog No.:AA0005OB CAS No.:101857-37-8 MDL No.:MFCD19204176 MF:C4H12N2O MW:104.1509 |
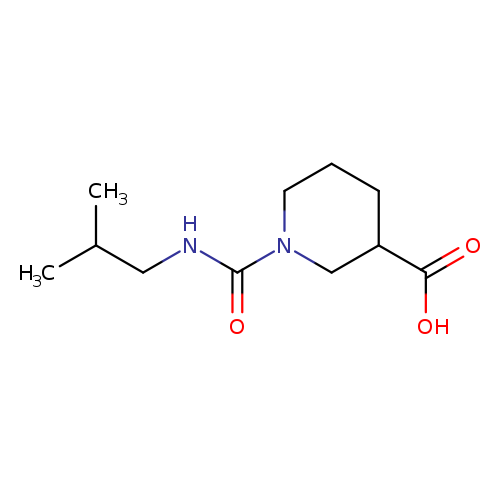
1-[(2-methylpropyl)carbamoyl]piperidine-3-carboxylic acidCatalog No.:AA01AKGM CAS No.:1018573-51-7 MDL No.:MFCD10012139 MF:C11H20N2O3 MW:228.2881 |
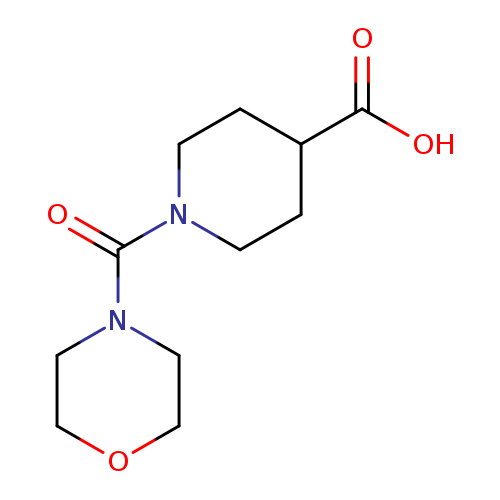
1-(morpholine-4-carbonyl)piperidine-4-carboxylic acidCatalog No.:AA019X17 CAS No.:1018574-31-6 MDL No.:MFCD10012232 MF:C11H18N2O4 MW:242.2716 |
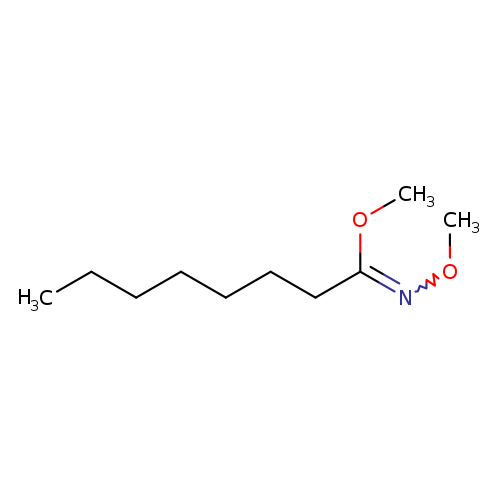
N-Methoxy-N-methyloctanamideCatalog No.:AA0005P1 CAS No.:101858-33-7 MDL No.:MFCD01711119 MF:C10H21NO2 MW:187.2792 |
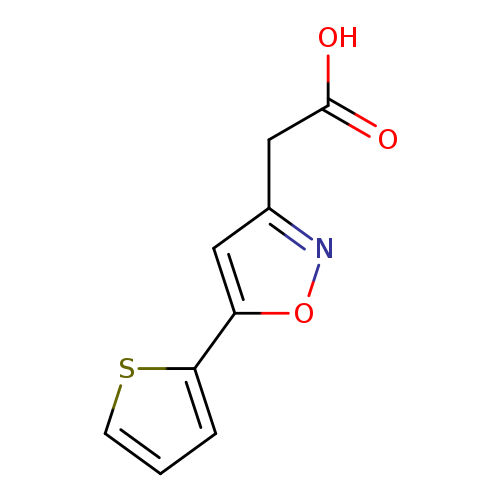
2-(5-(Thiophen-2-yl)isoxazol-3-yl)acetic acidCatalog No.:AA01ARE7 CAS No.:1018584-21-8 MDL No.:MFCD10036140 MF:C9H7NO3S MW:209.2218 |
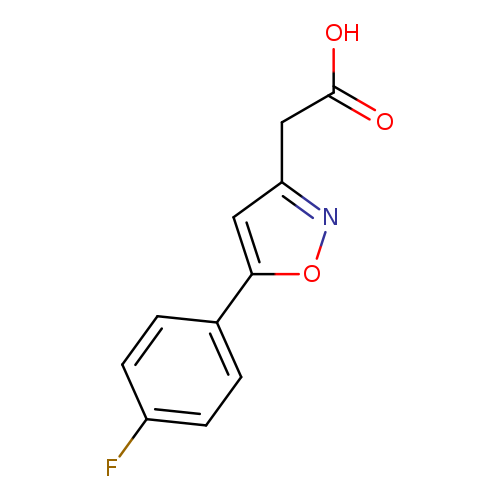
2-(5-(4-Fluorophenyl)isoxazol-3-yl)acetic acidCatalog No.:AA01BBYB CAS No.:1018584-37-6 MDL No.:MFCD10036149 MF:C11H8FNO3 MW:221.1845 |
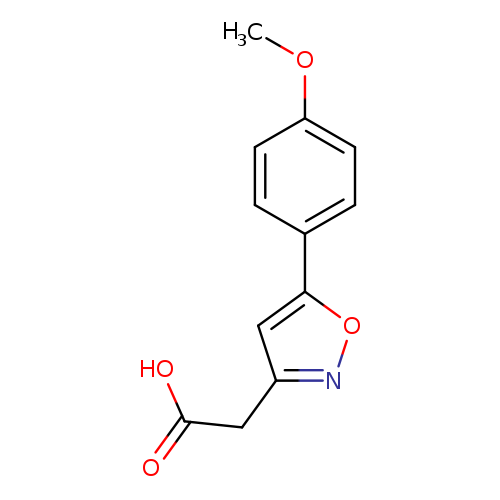
2-(5-(4-Methoxyphenyl)isoxazol-3-yl)acetic acidCatalog No.:AA01APHF CAS No.:1018584-41-2 MDL No.:MFCD10036152 MF:C12H11NO4 MW:233.2200 |
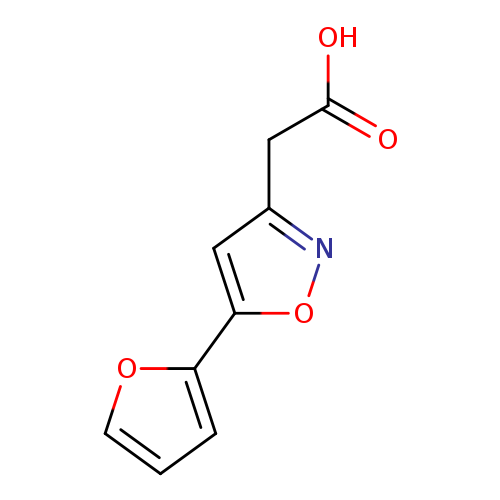
2-[5-(furan-2-yl)-1,2-oxazol-3-yl]acetic acidCatalog No.:AA01APHG CAS No.:1018584-57-0 MDL No.:MFCD10036160 MF:C9H7NO4 MW:193.1562 |
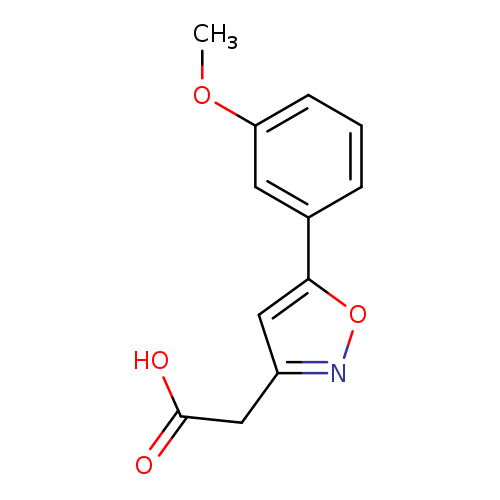
[5-(3-Methoxyphenyl)isoxazol-3-yl]acetic acidCatalog No.:AA00VSXR CAS No.:1018584-61-6 MDL No.:MFCD10036162 MF:C12H11NO4 MW:233.2200 |
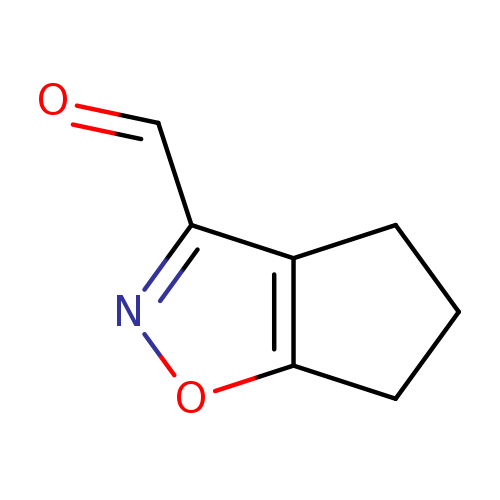
4H,5H,6H-cyclopenta[d][1,2]oxazole-3-carbaldehydeCatalog No.:AA01ELZU CAS No.:1018584-65-0 MDL No.:MFCD10036251 MF:C7H7NO2 MW:137.1360 |
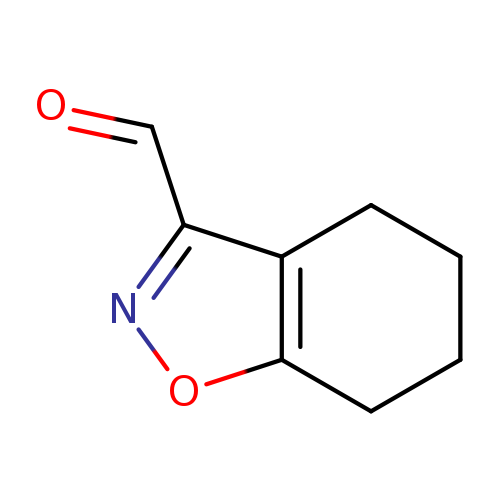
4,5,6,7-Tetrahydro-1,2-benzoxazole-3-carbaldehydeCatalog No.:AA01DUU5 CAS No.:1018584-77-4 MDL No.:MFCD10036257 MF:C8H9NO2 MW:151.1626 |
1-(2-Aminopropoxy)-3-bromobenzeneCatalog No.:AA01EMU9 CAS No.:1018586-11-2 MDL No.:MFCD10034446 MF:C9H12BrNO MW:230.1017 |
1-(2,5-dimethylphenyl)propan-1-amineCatalog No.:AA01BXDO CAS No.:1018586-68-9 MDL No.:MFCD10032946 MF:C11H17N MW:163.2594 |
Propanoic acid, 2,3,3,3-tetrafluoro-2-methoxy-, methyl esterCatalog No.:AA0005P7 CAS No.:10186-63-7 MDL No.:MFCD00155951 MF:C5H6F4O3 MW:190.0930 |
Ethyl 2-bromo-2,3,3,3-tetrafluoropropionateCatalog No.:AA0005P3 CAS No.:10186-73-9 MDL No.:MFCD00153721 MF:C5H5BrF4O2 MW:252.9896 |
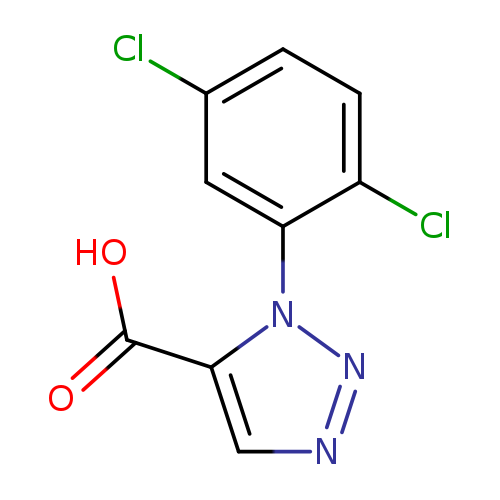
1-(2,5-dichlorophenyl)-1H-1,2,3-triazole-5-carboxylic acidCatalog No.:AA01E7FS CAS No.:101860-73-5 MDL No.:MFCD11559211 MF:C9H5Cl2N3O2 MW:258.0609 |
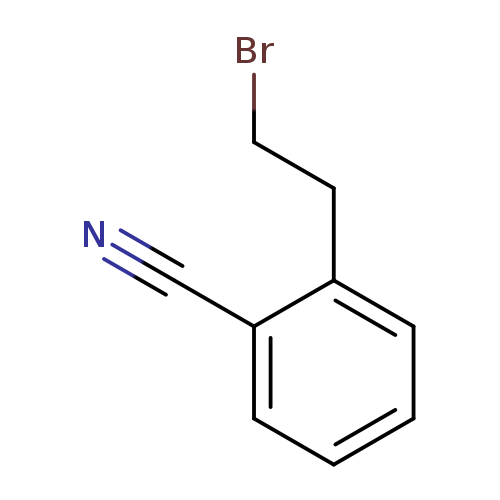
2-(2-Bromoethyl)benzonitrileCatalog No.:AA0096QC CAS No.:101860-81-5 MDL No.:MFCD24462563 MF:C9H8BrN MW:210.0705 |
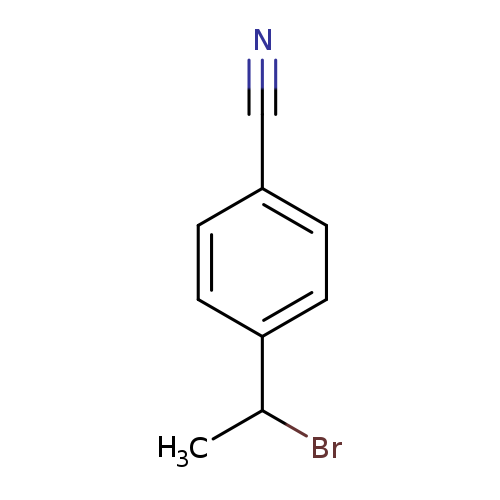
4-(1-Bromoethyl)benzonitrileCatalog No.:AA01AJEY CAS No.:101860-82-6 MDL No.:MFCD11180285 MF:C9H8BrN MW:210.0705 |
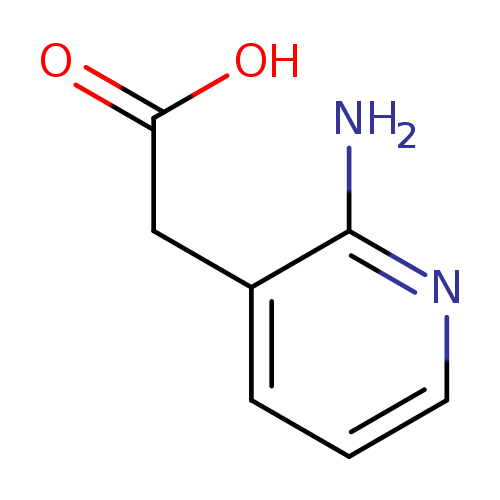
2-(2-Aminopyridin-3-yl)acetic acidCatalog No.:AA0005OQ CAS No.:101860-97-3 MDL No.:MFCD11848318 MF:C7H8N2O2 MW:152.1506 |
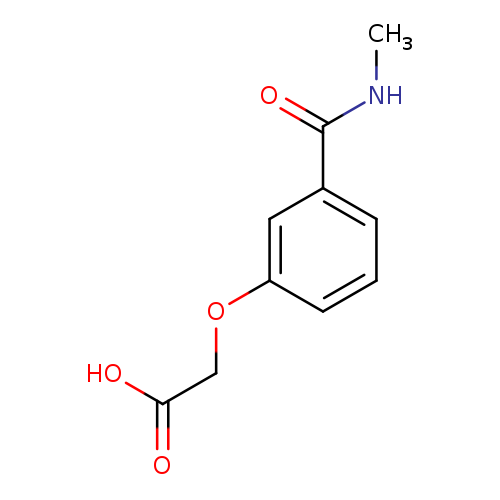
2-[3-(methylcarbamoyl)phenoxy]acetic acidCatalog No.:AA01A00P CAS No.:1018600-26-4 MDL No.:MFCD10037128 MF:C10H11NO4 MW:209.1986 |
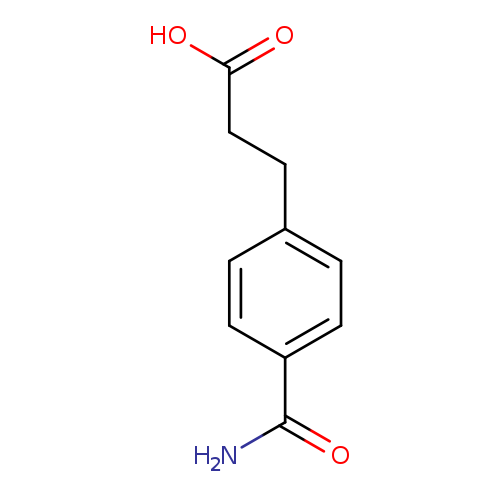
3-(4-Carbamoylphenyl)propanoic acidCatalog No.:AA01AM7D CAS No.:1018600-38-8 MDL No.:MFCD10037135 MF:C10H11NO3 MW:193.1992 |
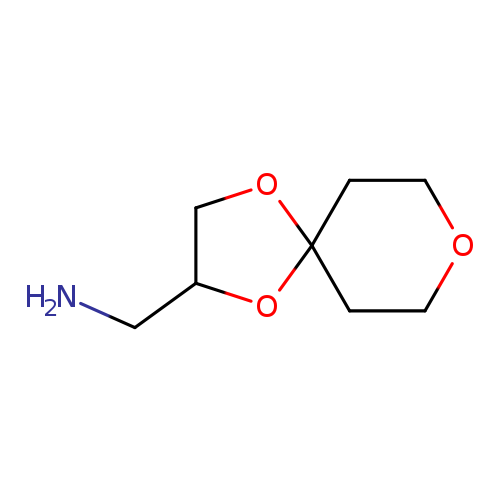
1,4,8-trioxaspiro[4.5]decan-2-ylmethanamineCatalog No.:AA01A4WO CAS No.:1018603-76-3 MDL No.:MFCD10032751 MF:C8H15NO3 MW:173.2096 |
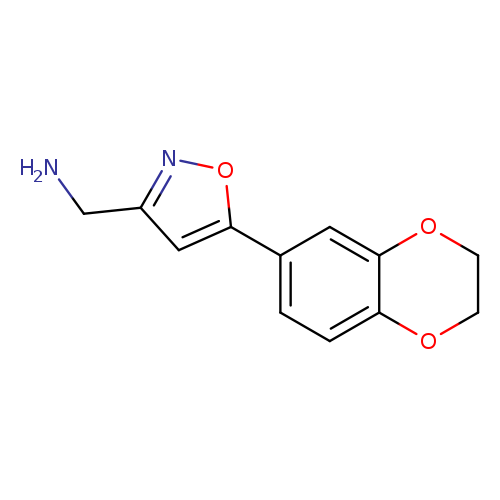
[5-(2,3-dihydro-1,4-benzodioxin-6-yl)-1,2-oxazol-3-yl]methanamineCatalog No.:AA01EJEB CAS No.:1018605-08-7 MDL No.:MFCD10036103 MF:C12H12N2O3 MW:232.2353 |
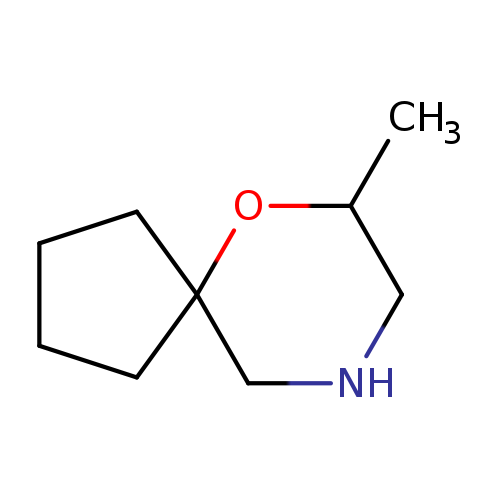
7-methyl-6-oxa-9-azaspiro[4.5]decaneCatalog No.:AA01C53O CAS No.:1018608-06-4 MDL No.:MFCD10032394 MF:C9H17NO MW:155.2374 |
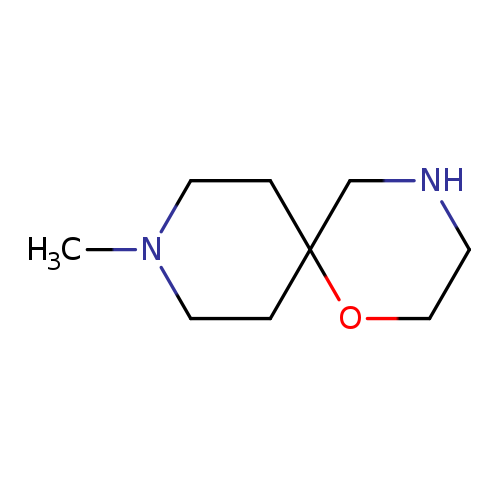
9-methyl-1-oxa-4,9-diazaspiro[5.5]undecaneCatalog No.:AA01C538 CAS No.:1018608-10-0 MDL No.:MFCD10032396 MF:C9H18N2O MW:170.2520 |
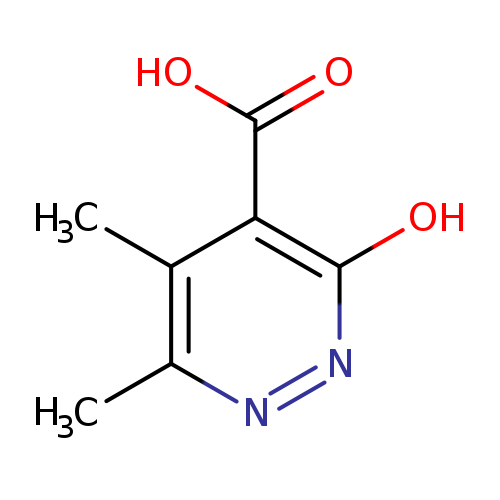
3-hydroxy-5,6-dimethylpyridazine-4-carboxylic acidCatalog No.:AA018GVD CAS No.:101861-29-4 MDL No.:MFCD10686669 MF:C7H8N2O3 MW:168.1500 |
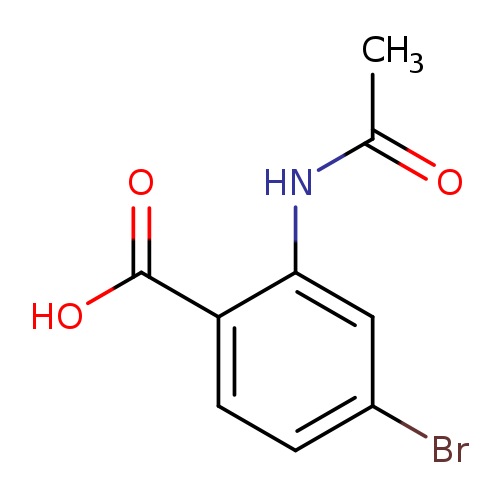
4-Bromo-2-acetamidobenzoic acidCatalog No.:AA0005PW CAS No.:101861-53-4 MDL No.:MFCD12805958 MF:C9H8BrNO3 MW:258.0687 |
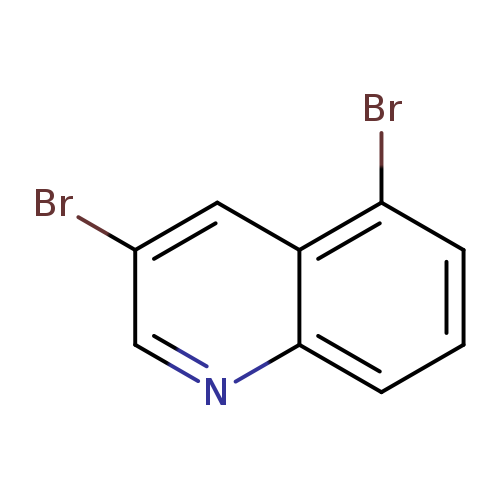
3,5-DibromoquinolineCatalog No.:AA0005PV CAS No.:101861-59-0 MDL No.:MFCD12024477 MF:C9H5Br2N MW:286.9507 |
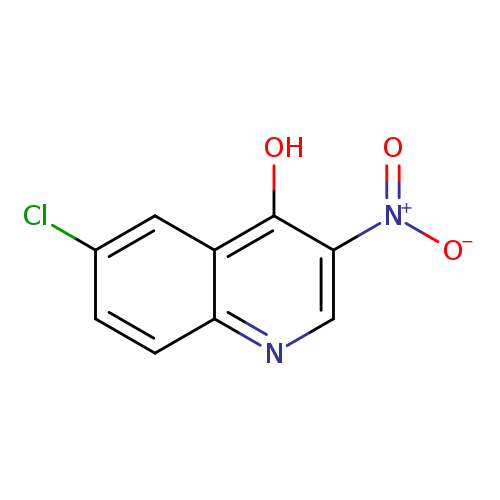
6-Chloro-3-nitroquinolin-4-olCatalog No.:AA0005PU CAS No.:101861-61-4 MDL No.:MFCD09800425 MF:C9H5ClN2O3 MW:224.6006 |
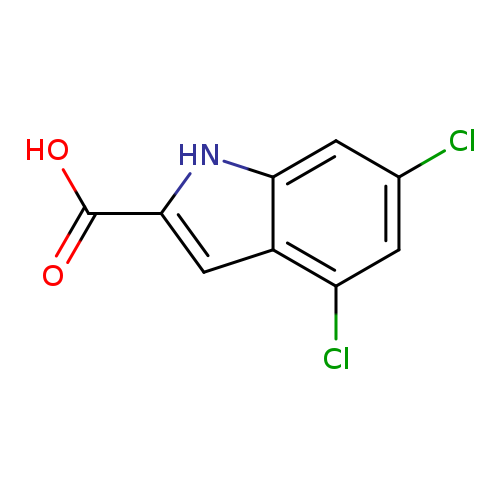
4,6-Dichloro-1h-indole-2-carboxylic acidCatalog No.:AA0005PT CAS No.:101861-63-6 MDL No.:MFCD00209870 MF:C9H5Cl2NO2 MW:230.0475 |
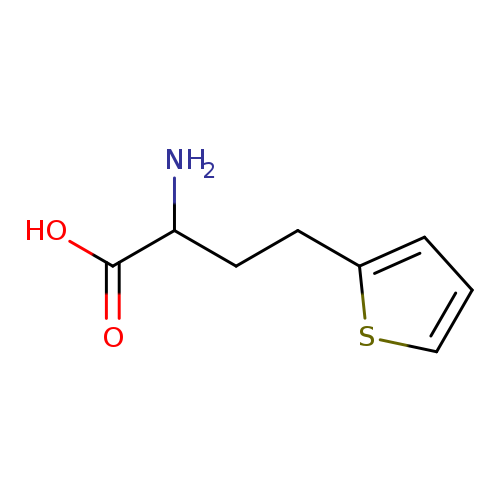
2-amino-4-(thiophen-2-yl)butanoic acidCatalog No.:AA01B984 CAS No.:101861-70-5 MDL No.:MFCD18884585 MF:C8H11NO2S MW:185.2434 |
(1,1-dioxo-1lambda6-thiolan-3-yl)methanesulfonyl chlorideCatalog No.:AA01BG4H CAS No.:1018611-90-9 MDL No.:MFCD10035251 MF:C5H9ClO4S2 MW:232.7056 |
2-(3-Bromophenyl)morpholineCatalog No.:AA01A8AO CAS No.:1018612-02-6 MDL No.:MFCD10035261 MF:C10H12BrNO MW:242.1124 |
{4-[4-(cyclopropylmethyl)piperazin-1-yl]phenyl}methanamineCatalog No.:AA01A3Y0 CAS No.:1018613-28-9 MDL No.:MFCD10033297 MF:C15H23N3 MW:245.3632 |
2-Methyl-1,4-oxazepaneCatalog No.:AA008V7E CAS No.:1018614-41-9 MDL No.:MFCD10035564 MF:C6H13NO MW:115.1735 |
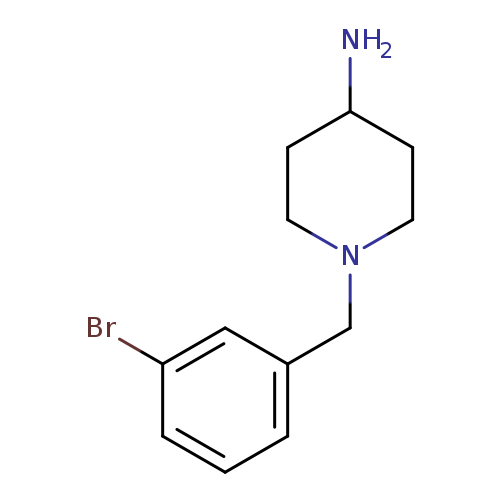
1-[(3-bromophenyl)methyl]piperidin-4-amineCatalog No.:AA01C32C CAS No.:1018617-44-1 MDL No.:MFCD10033710 MF:C12H17BrN2 MW:269.1808 |
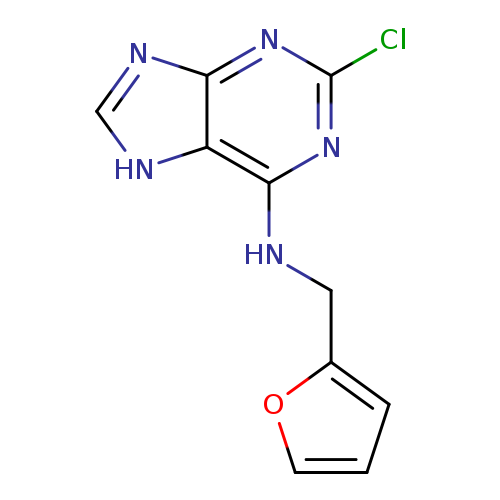
(2-Chloro-7H-purin-6-yl)-furan-2-ylmethyl-amineCatalog No.:AA0092LU CAS No.:101862-47-9 MDL No.:MFCD09792860 MF:C10H8ClN5O MW:249.6564 |
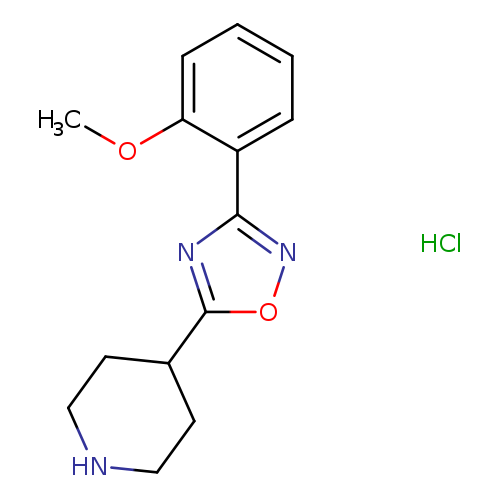
4-[3-(2-Methoxyphenyl)-1,2,4-oxadiazol-5-yl]piperidine hydrochlorideCatalog No.:AA00H9KM CAS No.:1018626-66-8 MDL No.:MFCD27960949 MF:C14H18ClN3O2 MW:295.7646 |
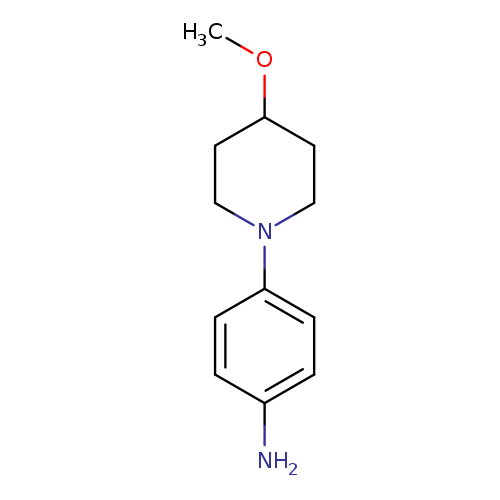
4-(4-methoxypiperidin-1-yl)anilineCatalog No.:AA01931U CAS No.:1018635-74-9 MDL No.:MFCD10034078 MF:C12H18N2O MW:206.2841 |
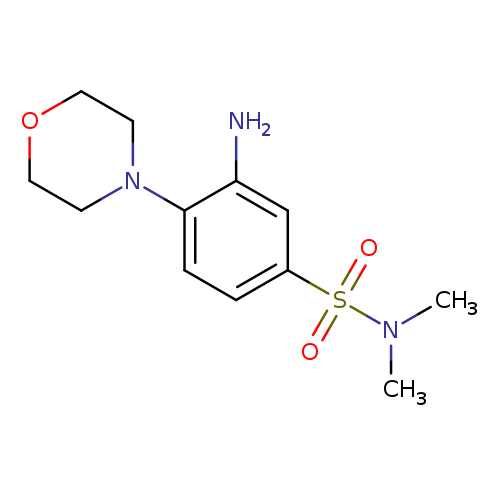
3-Amino-n,n-dimethyl-4-(morpholin-4-yl)benzene-1-sulfonamideCatalog No.:AA01A2HO CAS No.:1018638-08-8 MDL No.:MFCD10032291 MF:C12H19N3O3S MW:285.3626 |
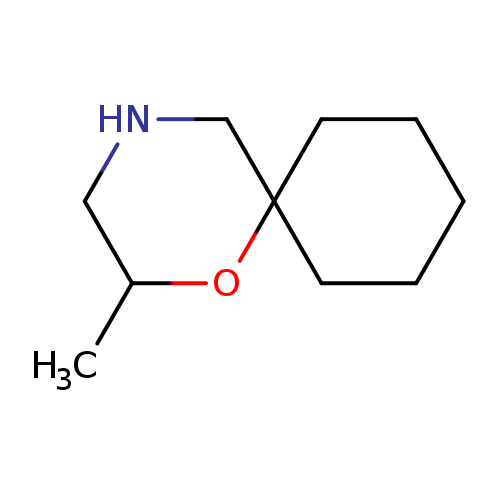
2-methyl-1-oxa-4-azaspiro[5.5]undecaneCatalog No.:AA01B28R CAS No.:1018639-87-6 MDL No.:MFCD10032395 MF:C10H19NO MW:169.2640 |
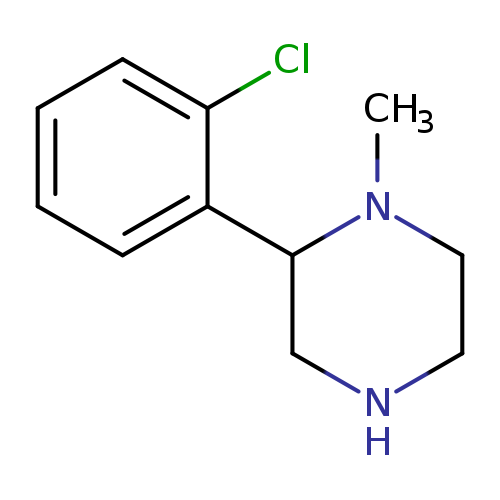
2-(2-Chlorophenyl)-1-methylpiperazineCatalog No.:AA01BBA7 CAS No.:1018645-91-4 MDL No.:MFCD10034704 MF:C11H15ClN2 MW:210.7032 |
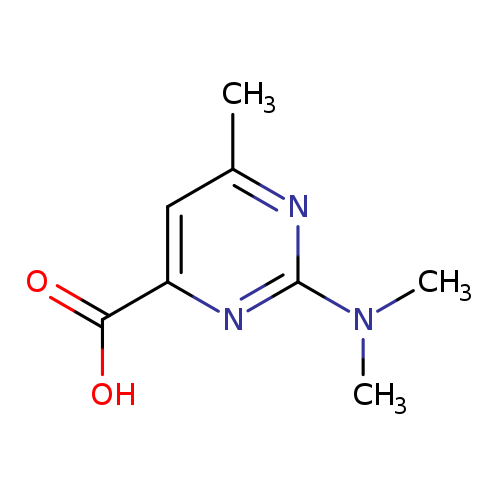
2-(Dimethylamino)-6-methylpyrimidine-4-carboxylic acidCatalog No.:AA0091GA CAS No.:1018648-19-5 MDL No.:MFCD10032942 MF:C8H11N3O2 MW:181.1918 |
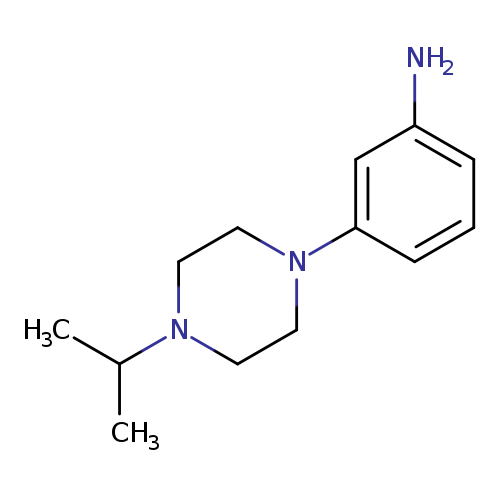
3-[4-(propan-2-yl)piperazin-1-yl]anilineCatalog No.:AA01A2BJ CAS No.:1018648-79-7 MDL No.:MFCD10032984 MF:C13H21N3 MW:219.3259 |
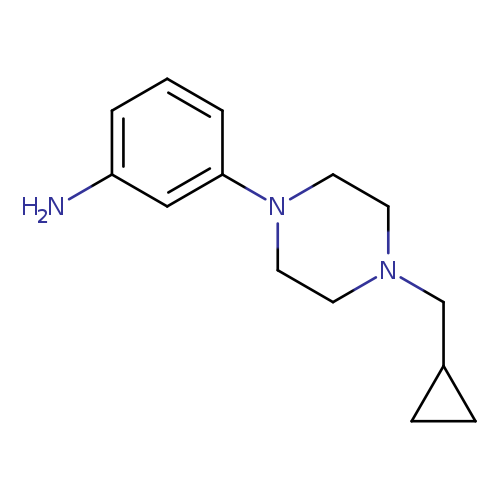
3-[4-(Cyclopropylmethyl)piperazin-1-yl]anilineCatalog No.:AA019X57 CAS No.:1018649-76-7 MDL No.:MFCD10033036 MF:C14H21N3 MW:231.3366 |
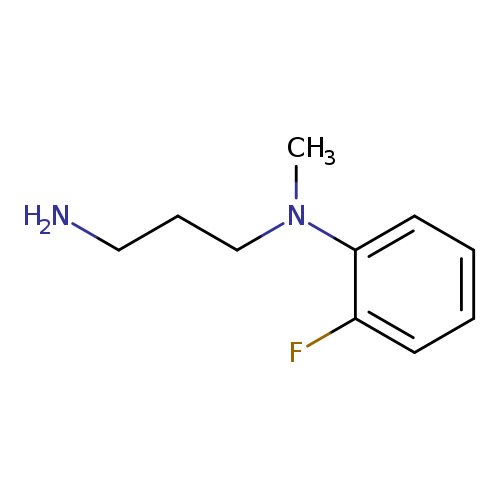
N-(3-aminopropyl)-2-fluoro-N-methylanilineCatalog No.:AA019WBH CAS No.:1018650-31-1 MDL No.:MFCD10031317 MF:C10H15FN2 MW:182.2379 |
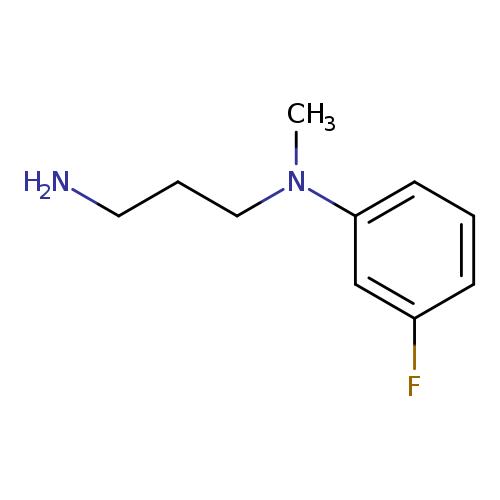
N-(3-Aminopropyl)-3-fluoro-n-methylanilineCatalog No.:AA019WJZ CAS No.:1018650-35-5 MDL No.:MFCD10031318 MF:C10H15FN2 MW:182.2379 |
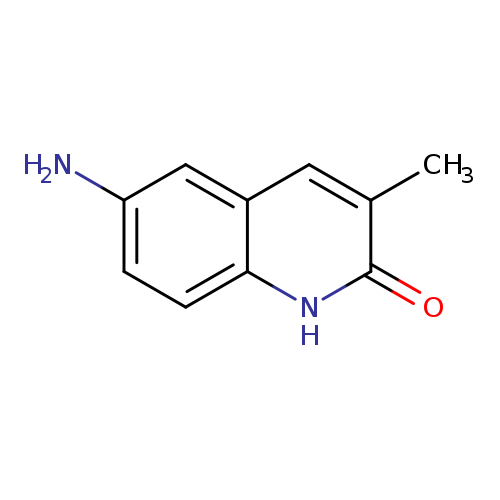
6-amino-3-methyl-1,2-dihydroquinolin-2-oneCatalog No.:AA01E95V CAS No.:1018652-50-0 MDL No.:MFCD10036880 MF:C10H10N2O MW:174.1992 |
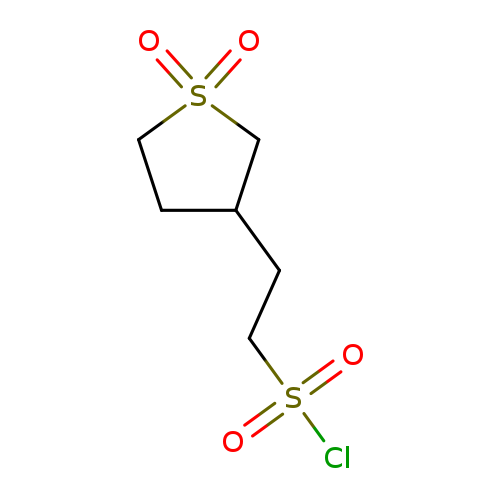
2-(1,1-dioxidotetrahydro-3-thienyl)ethanesulfonyl chlorideCatalog No.:AA00J2JA CAS No.:1018656-49-9 MDL No.:MFCD10035252 MF:C6H11ClO4S2 MW:246.7321 |
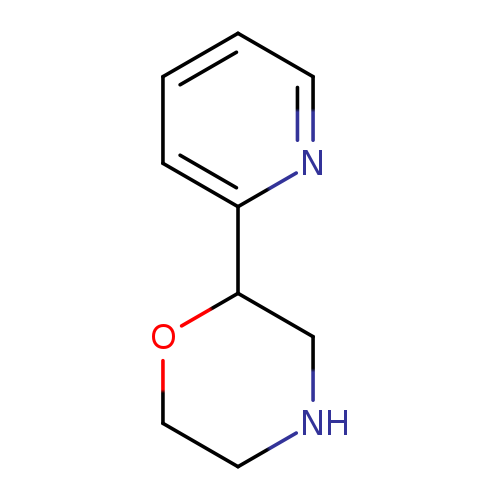
2-(Pyridin-2-yl)morpholineCatalog No.:AA0005PM CAS No.:1018656-53-5 MDL No.:MFCD10035254 MF:C9H12N2O MW:164.2044 |
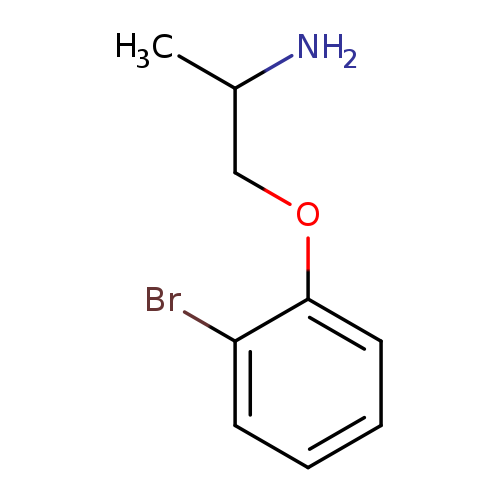
1-(2-Aminopropoxy)-2-bromobenzeneCatalog No.:AA01EMUI CAS No.:1018662-79-7 MDL No.:MFCD10034444 MF:C9H12BrNO MW:230.1017 |
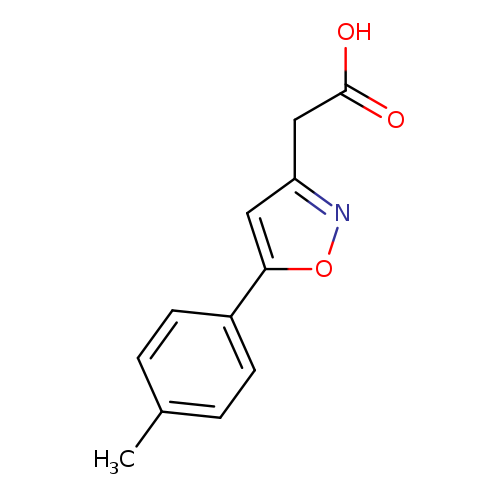
[5-(4-Methylphenyl)isoxazol-3-yl]acetic acidCatalog No.:AA01ARE8 CAS No.:1018663-09-6 MDL No.:MFCD10036142 MF:C12H11NO3 MW:217.2206 |
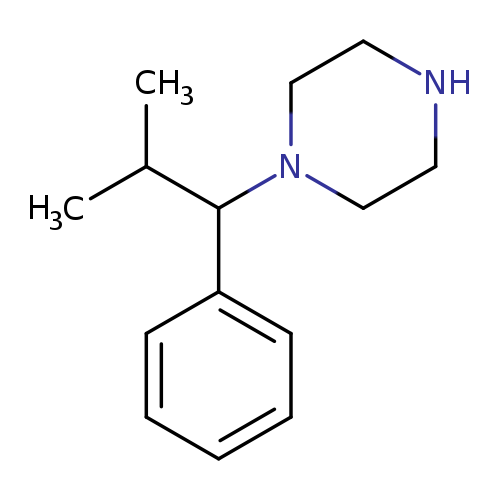
1-(2-methyl-1-phenylpropyl)piperazineCatalog No.:AA01FQ9Q CAS No.:1018666-38-0 MDL No.:MFCD10032779 MF:C14H22N2 MW:218.3379 |
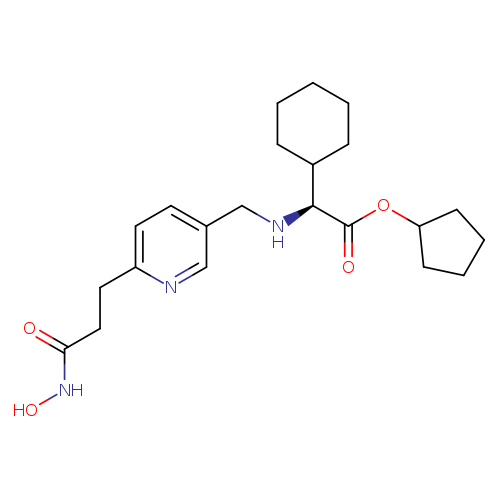
cyclopentyl 2-cyclohexyl-2-((6-(3-(hydroxyamino)-3-oxopropyl)pyridin-3-yl)methylamino)acetateCatalog No.:AA008TM8 CAS No.:1018673-42-1 MDL No.:MFCD29924725 MF:C22H33N3O4 MW:403.5151 |
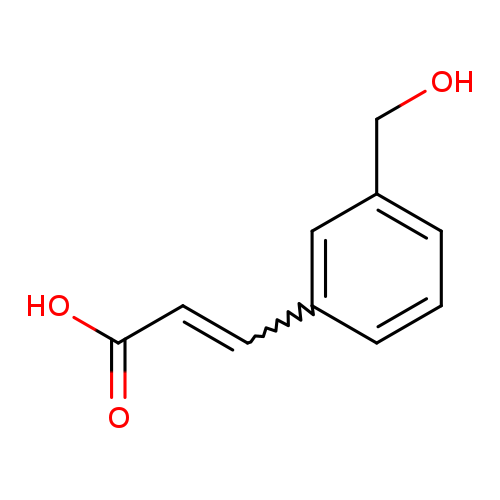
3-[3-(hydroxymethyl)phenyl]prop-2-enoic acidCatalog No.:AA019ZWP CAS No.:1018674-37-7 MDL No.:MFCD14687996 MF:C10H10O3 MW:178.1846 |
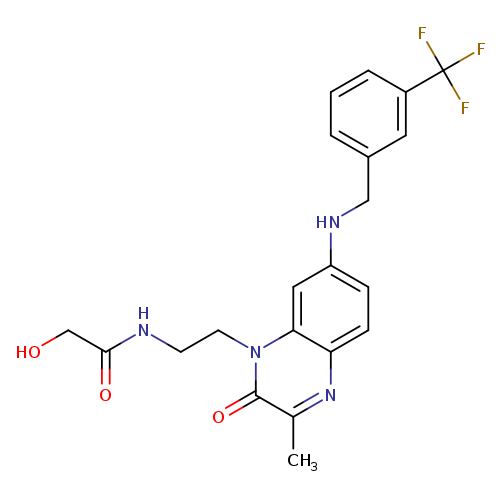
CVT-12012Catalog No.:AA01EO9U CAS No.:1018675-35-8 MDL No.:MFCD18251587 MF:C21H21F3N4O3 MW:434.4116 |
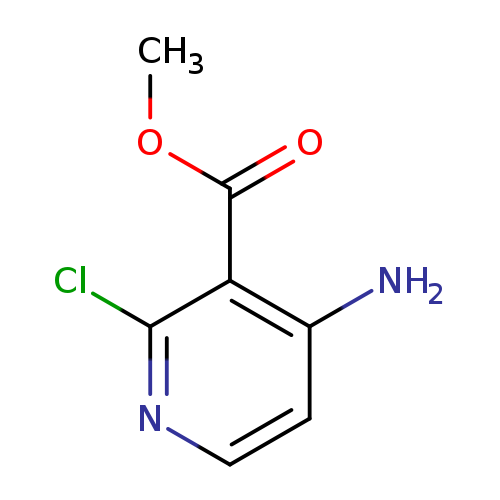
Methyl 4-amino-2-chloronicotinateCatalog No.:AA0005PE CAS No.:1018678-37-9 MDL No.:MFCD20726014 MF:C7H7ClN2O2 MW:186.5957 |
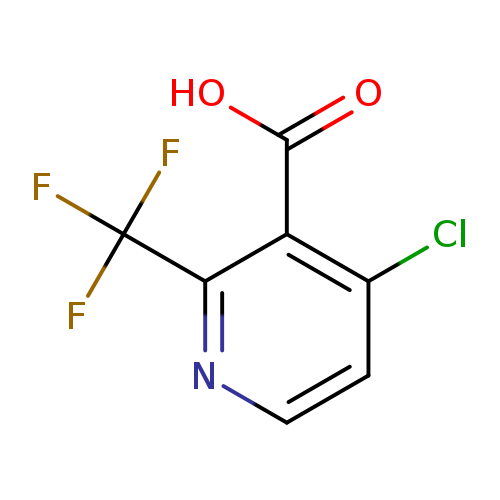
4-Chloro-2-(trifluoromethyl)nicotinic acidCatalog No.:AA0005PC CAS No.:1018678-39-1 MDL No.:MFCD22546902 MF:C7H3ClF3NO2 MW:225.5524 |
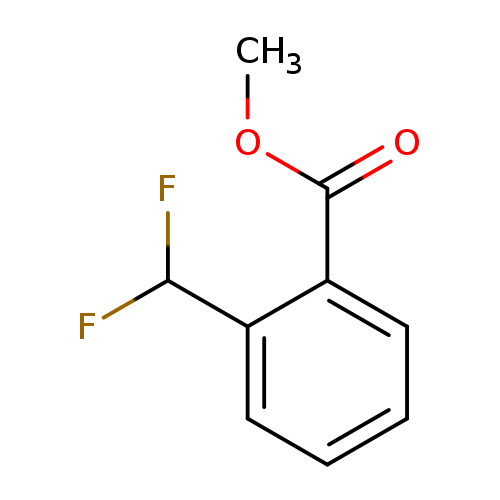
2-Difluoromethyl-benzoic acid methyl esterCatalog No.:AA0005PB CAS No.:1018678-48-2 MDL No.:MFCD16140192 MF:C9H8F2O2 MW:186.1554 |
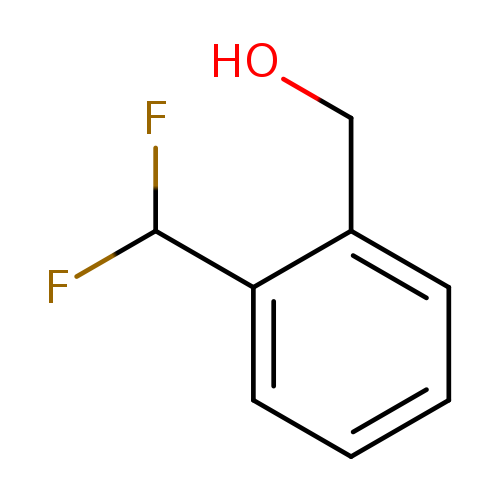
[2-(Difluoromethyl)phenyl]methanolCatalog No.:AA00H9KS CAS No.:1018678-49-3 MDL No.:MFCD26728124 MF:C8H8F2O MW:158.1453 |
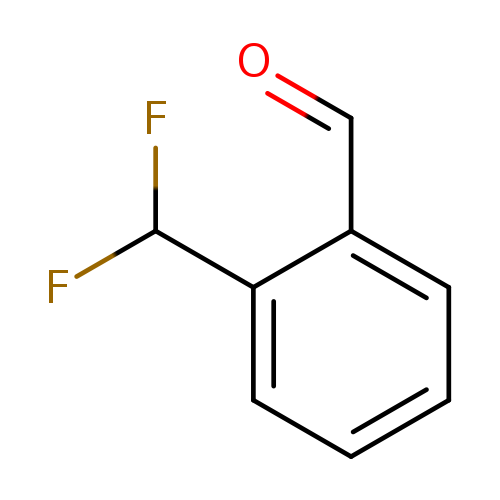
2-(Difluoromethyl)benzaldehydeCatalog No.:AA0005PA CAS No.:1018678-50-6 MDL No.:MFCD16875639 MF:C8H6F2O MW:156.1294 |
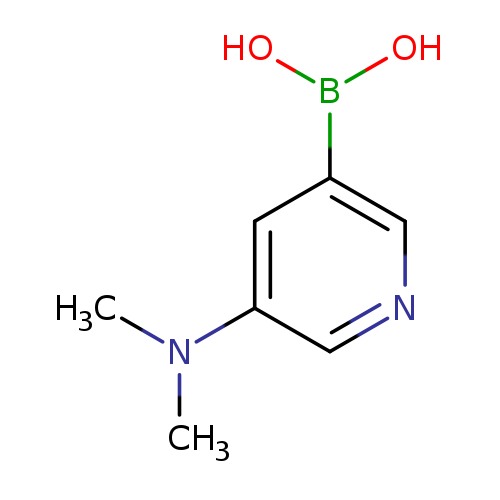
3-Dimethylaminopyridine-5-boronic acidCatalog No.:AA0005Q2 CAS No.:1018680-09-5 MDL No.:MFCD12964565 MF:C7H11BN2O2 MW:165.9854 |
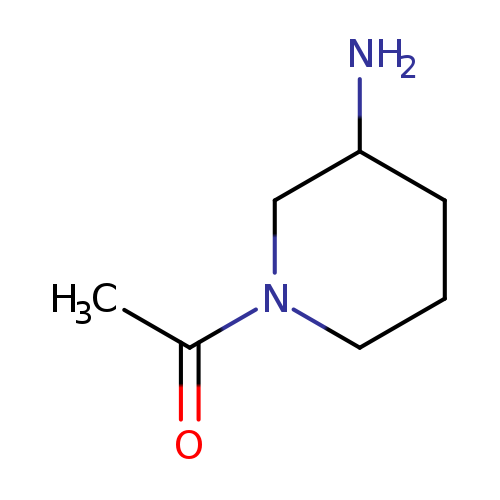
1-Acetyl-3-aminopiperidineCatalog No.:AA0005Q1 CAS No.:1018680-22-2 MDL No.:MFCD09264572 MF:C7H14N2O MW:142.1989 |
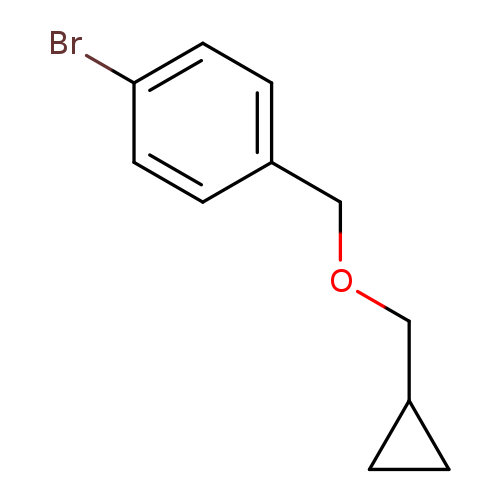
1-Bromo-4-[(cyclopropylmethoxy)methyl]benzeneCatalog No.:AA0005Q0 CAS No.:1018681-31-6 MDL No.:MFCD14687047 MF:C11H13BrO MW:241.1243 |
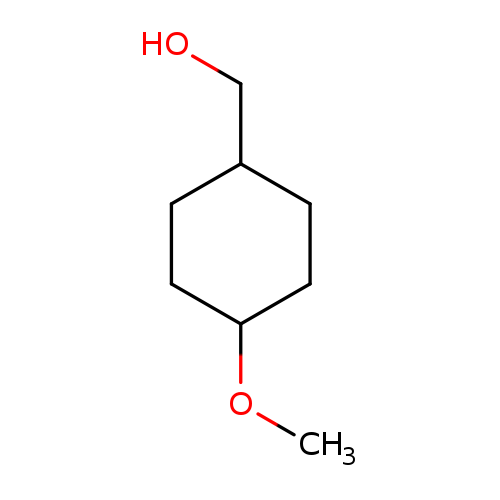
(4-Methoxycyclohexyl)methanolCatalog No.:AA0005QD CAS No.:101869-74-3 MDL No.:MFCD26885663 MF:C8H16O2 MW:144.2114 |
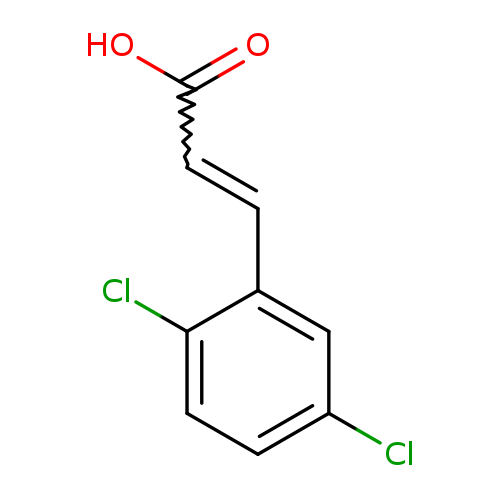
2-Propenoic acid, 3-(2,5-dichlorophenyl)-Catalog No.:AA0005QC CAS No.:101869-82-3 MDL No.:MFCD03002786 MF:C9H6Cl2O2 MW:217.0487 |
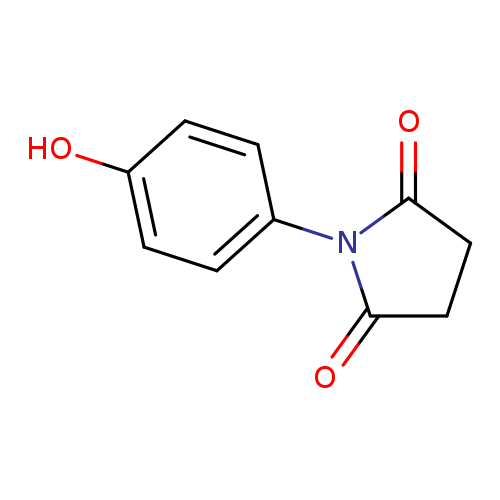
1-(4-Hydroxyphenyl)pyrrolidine-2,5-dioneCatalog No.:AA0005QN CAS No.:10187-21-0 MDL No.:MFCD00466649 MF:C10H9NO3 MW:191.1834 |
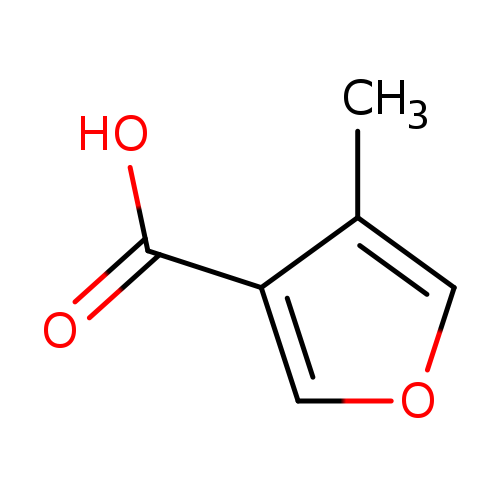
4-Methyl-3-furancarboxylic acidCatalog No.:AA00H9KU CAS No.:101870-15-9 MDL No.:MFCD15143778 MF:C6H6O3 MW:126.1100 |
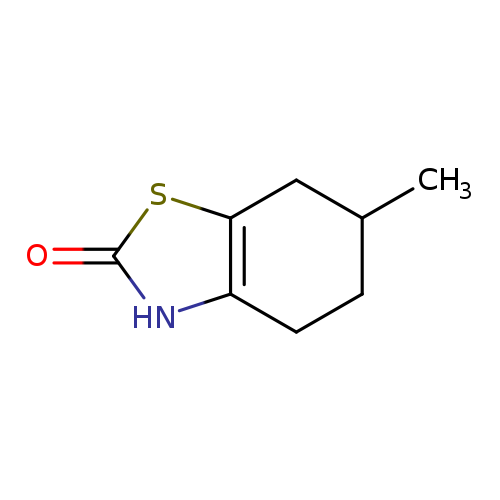
6-methyl-2,3,4,5,6,7-hexahydro-1,3-benzothiazol-2-oneCatalog No.:AA01A8OZ CAS No.:101870-28-4 MDL No.:MFCD11632139 MF:C8H11NOS MW:169.2440 |
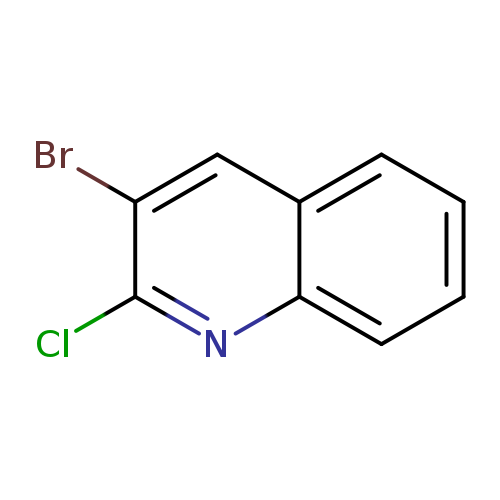
3-Bromo-2-chloroquinolineCatalog No.:AA0005Q7 CAS No.:101870-60-4 MDL No.:MFCD08457743 MF:C9H5BrClN MW:242.4997 |
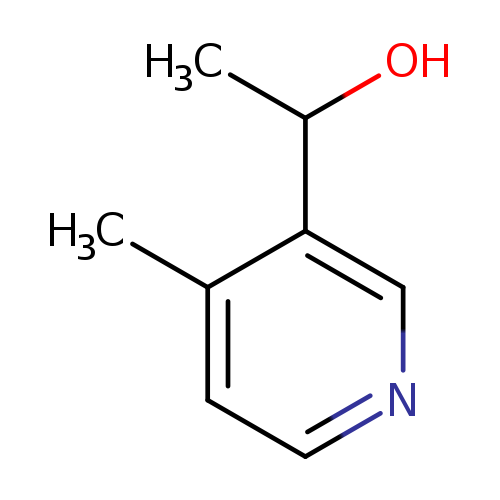
3-Pyridinemethanol,alpha,4-dimethyl-(6CI,9CI)Catalog No.:AA009S5E CAS No.:101870-76-2 MDL No.:MFCD21740226 MF:C8H11NO MW:137.1790 |
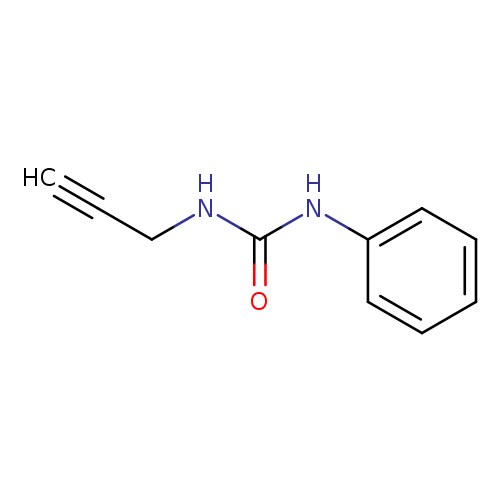
Urea, N-phenyl-N'-2-propyn-1-yl-Catalog No.:AA0005Q4 CAS No.:101871-81-2 MDL No.:MFCD11756585 MF:C10H10N2O MW:174.1992 |
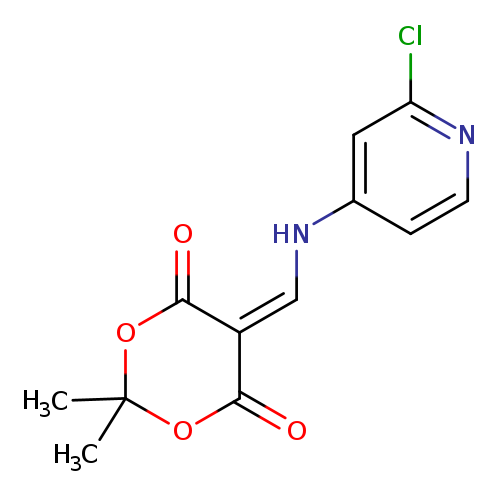
5-{[(2-chloropyridin-4-yl)amino]methylidene}-2,2-dimethyl-1,3-dioxane-4,6-dioneCatalog No.:AA01ACQ1 CAS No.:1018812-97-9 MDL No.:MFCD26936115 MF:C12H11ClN2O4 MW:282.6797 |
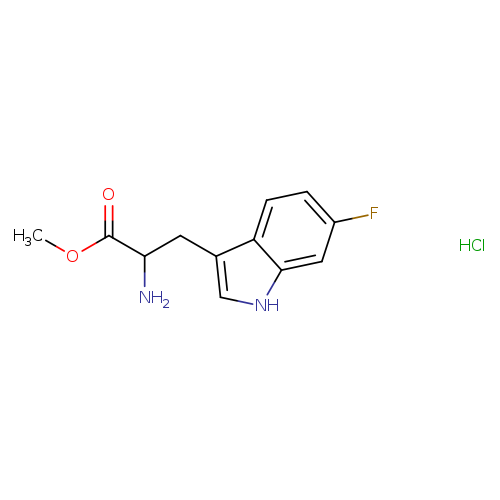
methyl 2-amino-3-(6-fluoro-1H-indol-3-yl)propanoate hydrochlorideCatalog No.:AA019Y9H CAS No.:1018815-57-0 MDL No.:MFCD22056390 MF:C12H14ClFN2O2 MW:272.7032 |
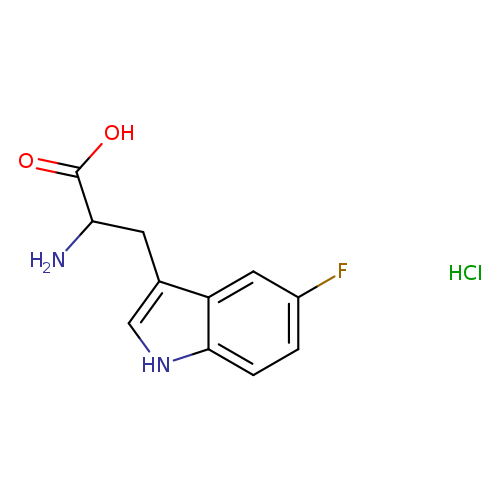
5-Fluoro D,L-Tryptophan HydrochlorideCatalog No.:AA01CB7M CAS No.:1018815-80-9 MDL No.:MFCD30720907 MF:C11H12ClFN2O2 MW:258.6766 |
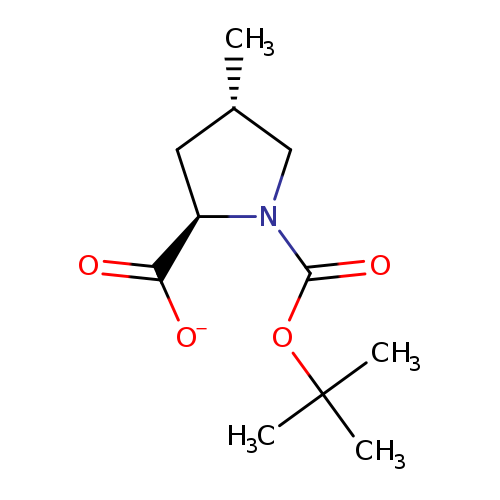
(2R,4S)-1-Boc-4-methylpyrrolidine-2-carboxylic acidCatalog No.:AA0005QU CAS No.:1018818-04-6 MDL No.:MFCD23105992 MF:C11H18NO4- MW:228.2649 |
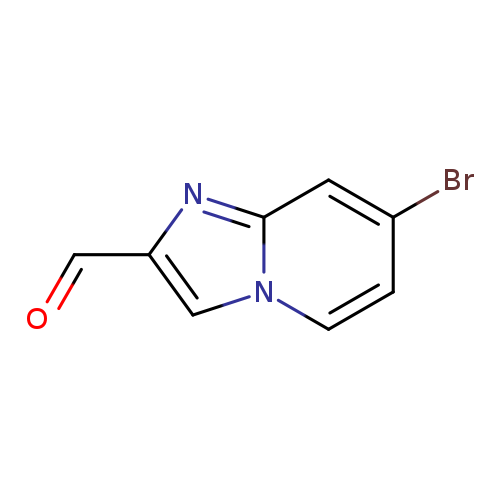
7-Bromoimidazo[1,2-a]pyridine-2-carbaldehydeCatalog No.:AA0005QR CAS No.:1018828-16-4 MDL No.:MFCD09995135 MF:C8H5BrN2O MW:225.0421 |
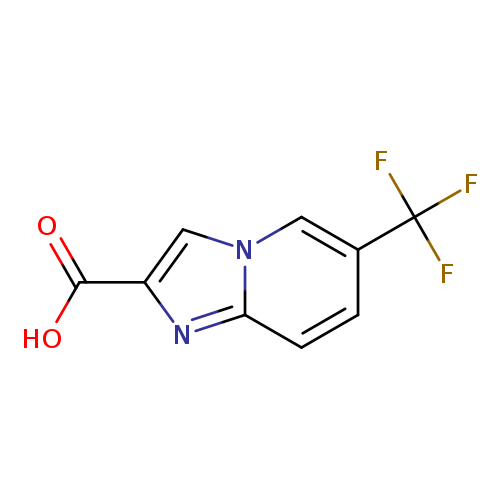
6-(Trifluoromethyl)imidazo[1,2-a]pyridine-2-carboxylic acidCatalog No.:AA0005QP CAS No.:1018828-69-7 MDL No.:MFCD22682819 MF:C9H5F3N2O2 MW:230.1434 |
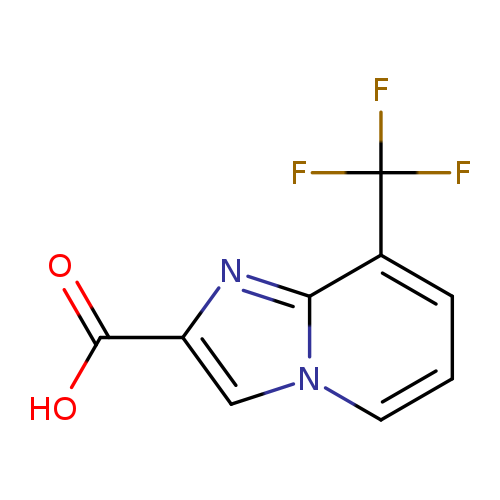
8-(Trifluoromethyl)imidazo[1,2-a]pyridine-2-carboxylic acidCatalog No.:AA0005QO CAS No.:1018828-72-2 MDL No.:MFCD22682817 MF:C9H5F3N2O2 MW:230.1434 |
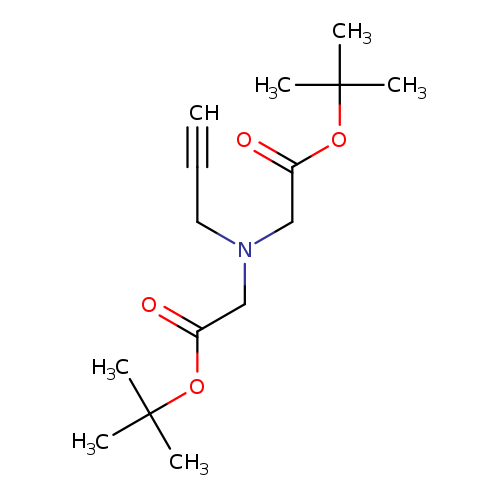
tert-butyl 2-{[2-(tert-butoxy)-2-oxoethyl](prop-2-yn-1-yl)amino}acetateCatalog No.:AA01E8QW CAS No.:1018831-90-7 MDL No.:MFCD30750012 MF:C15H25NO4 MW:283.3633 |
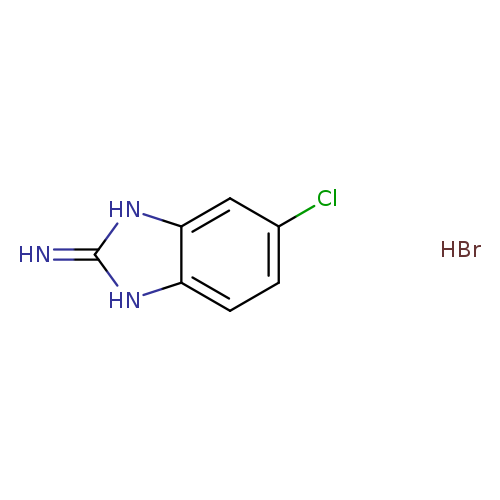
6-Chloro-1h-benzo[d]imidazol-2-amine hydrobromideCatalog No.:AA00H9KZ CAS No.:1018894-96-6 MDL No.:MFCD30185062 MF:C7H7BrClN3 MW:248.5076 |
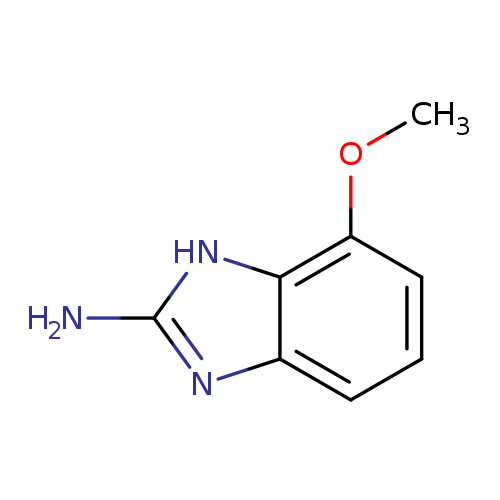
7-Methoxy-1H-benzo[d]imidazol-2-amineCatalog No.:AA0005RG CAS No.:1018895-06-1 MDL No.:MFCD16659610 MF:C8H9N3O MW:163.1766 |
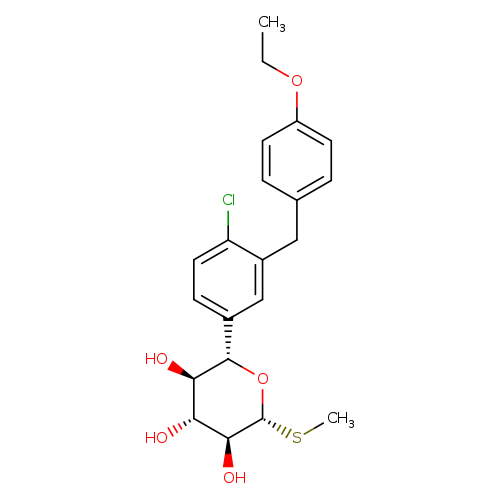
(2S,3R,4R,5S,6R)-2-(4-Chloro-3-(4-ethoxybenzyl)phenyl)-6-(methylthio)tetrahydro-2h-pyran-3,4,5-triolCatalog No.:AA0005RF CAS No.:1018899-04-1 MDL No.:MFCD22493506 MF:C21H25ClO5S MW:424.9382 |
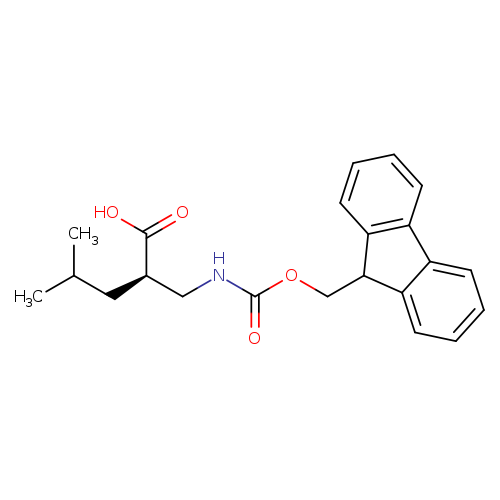
(R)-2-(((((9H-Fluoren-9-yl)methoxy)carbonyl)amino)methyl)-4-methylpentanoic acidCatalog No.:AA0005RE CAS No.:1018899-99-4 MDL No.:MFCD07372889 MF:C22H25NO4 MW:367.4382 |
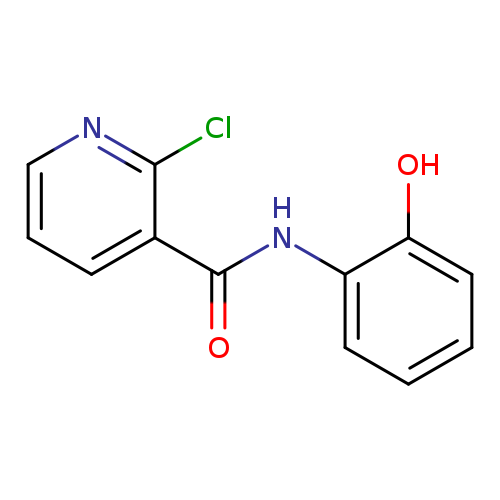
2-chloro-N-(2-hydroxyphenyl)pyridine-3-carboxamideCatalog No.:AA00IRCS CAS No.:10189-45-4 MDL No.:MFCD01568577 MF:C12H9ClN2O2 MW:248.6651 |

6,11-Dihydro-5h-pyrido[2,3-b][1,5]benzodiazepin-5-oneCatalog No.:AA0005RY CAS No.:10189-78-3 MDL No.:MFCD01568823 MF:C12H9N3O MW:211.2194 |
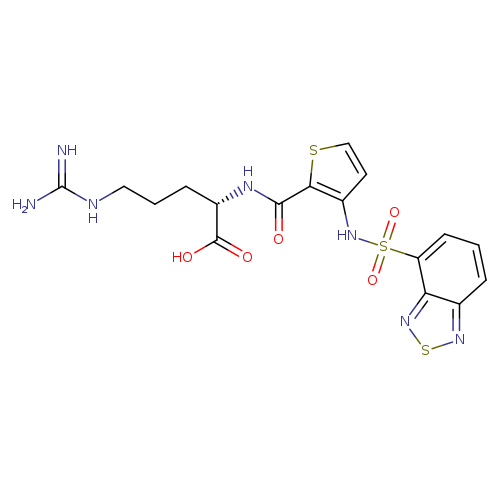
N2-[[3-[(2,1,3-Benzothiadiazol-4-ylsulfonyl)amino]-2-thienyl]carbonyl]-L-arginineCatalog No.:AA01ENKL CAS No.:1018927-63-3 MDL No.:MFCD18251520 MF:C17H19N7O5S3 MW:497.5717 |

Dwk-1339Catalog No.:AA0005RD CAS No.:1018946-38-7 MDL No.:MFCD22665705 MF:C20H22O4 MW:326.3863 |
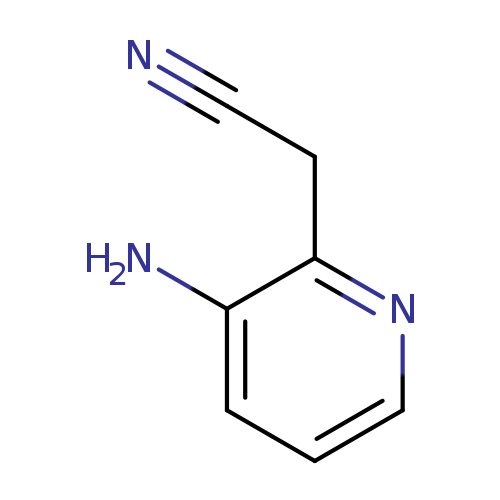
2-(3-Aminopyridin-2-yl)acetonitrileCatalog No.:AA01EN5B CAS No.:1018949-67-1 MDL No.:MFCD16608699 MF:C7H7N3 MW:133.1506 |
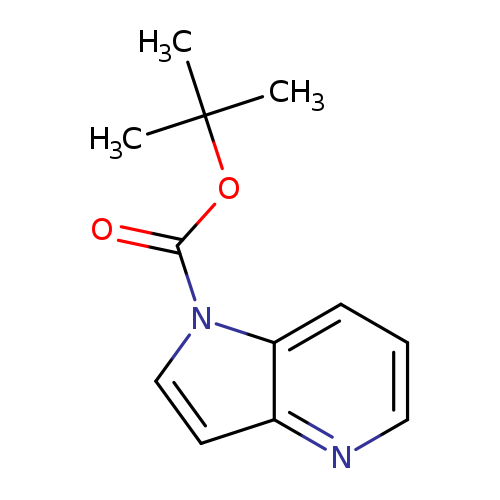
tert-Butyl 1h-pyrrolo[3,2-b]pyridine-1-carboxylateCatalog No.:AA009178 CAS No.:1018950-15-6 MDL No.:MFCD24387134 MF:C12H14N2O2 MW:218.2518 |
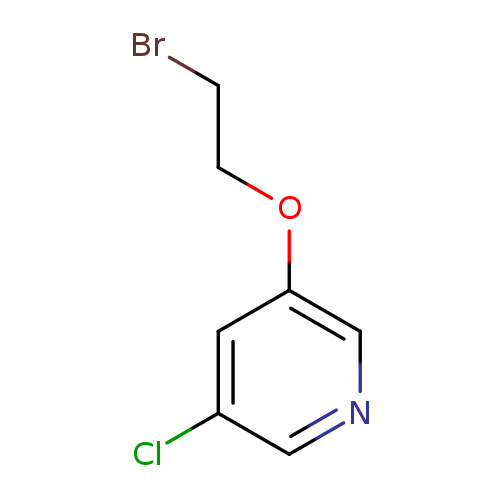
3-(2-bromoethoxy)-5-chloropyridineCatalog No.:AA01C727 CAS No.:1018974-74-7 MDL No.:MFCD20369541 MF:C7H7BrClNO MW:236.4936 |
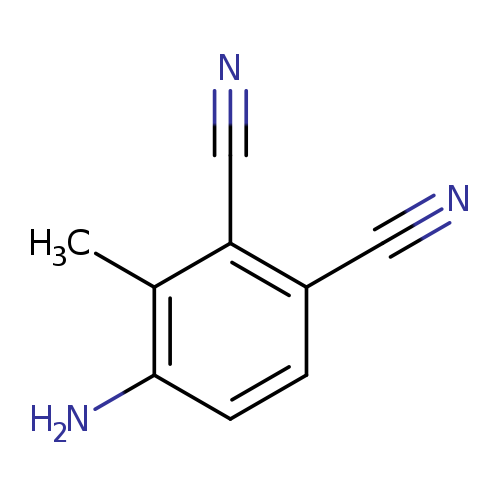
4-amino-3-methylbenzene-1,2-dicarbonitrileCatalog No.:AA01C3JV CAS No.:1018975-31-9 MDL No.:MFCD20720874 MF:C9H7N3 MW:157.1720 |
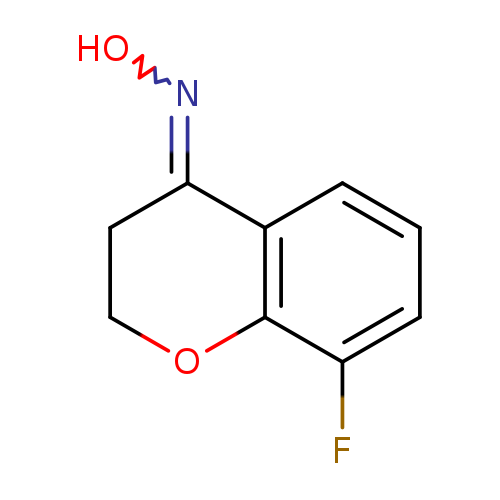
N-(8-fluoro-3,4-dihydro-2H-1-benzopyran-4-ylidene)hydroxylamineCatalog No.:AA01BCVM CAS No.:1018978-81-8 MDL No.:MFCD28118239 MF:C9H8FNO2 MW:181.1637 |
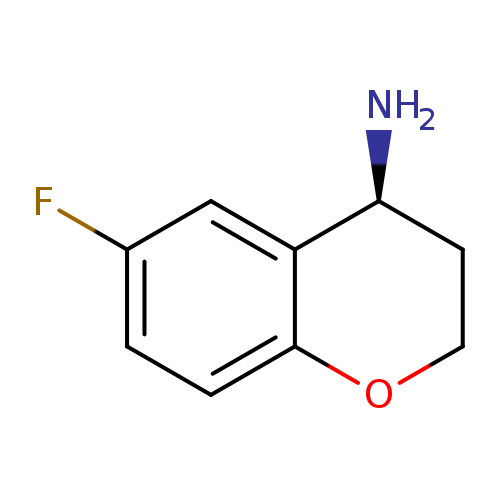
(S)-6-Fluorochroman-4-amineCatalog No.:AA0005S9 CAS No.:1018978-85-2 MDL No.:MFCD06761991 MF:C9H10FNO MW:167.1802 |
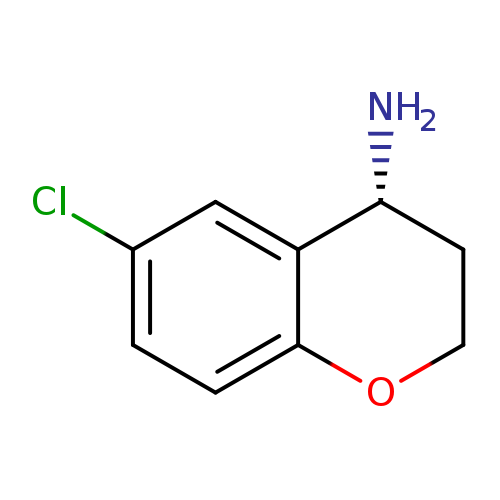
(4R)-6-Chlorochromane-4-ylamineCatalog No.:AA0005S8 CAS No.:1018978-86-3 MDL No.:MFCD06762096 MF:C9H10ClNO MW:183.6348 |
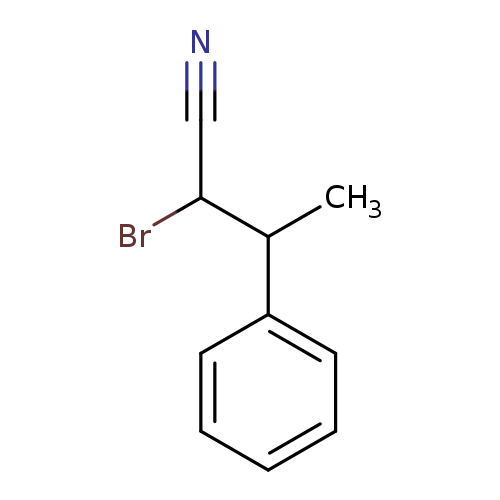
2-bromo-3-phenylbutanenitrile, Mixture of diastereomersCatalog No.:AA01C00J CAS No.:1018986-05-4 MDL No.:MFCD25986876 MF:C10H10BrN MW:224.0971 |
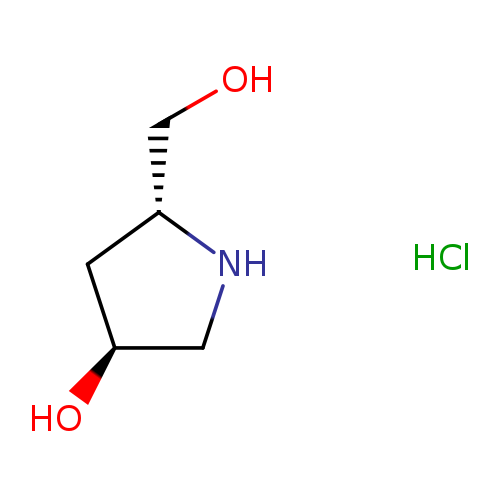
(3S,5R)-5-(Hydroxymethyl)pyrrolidin-3-ol hydrochlorideCatalog No.:AA0005S2 CAS No.:1018987-47-7 MDL No.:MFCD30719389 MF:C5H12ClNO2 MW:153.6073 |

N-Desmethyl-cis-tramadol HClCatalog No.:AA008SZJ CAS No.:1018989-94-0 MDL No.:MFCD03788593 MF:C15H24ClNO2 MW:285.8096 |
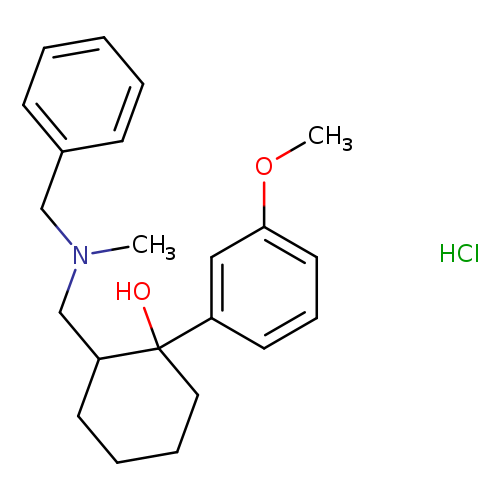
Cyclohexanol, 1-(3-methoxyphenyl)-2-[[methyl(phenylmethyl)amino]methyl]-, hydrochloride (1:1), (1R,2R)-rel-Catalog No.:AA0005S1 CAS No.:1018989-95-1 MDL No.:MFCD18385032 MF:C22H30ClNO2 MW:375.9321 |
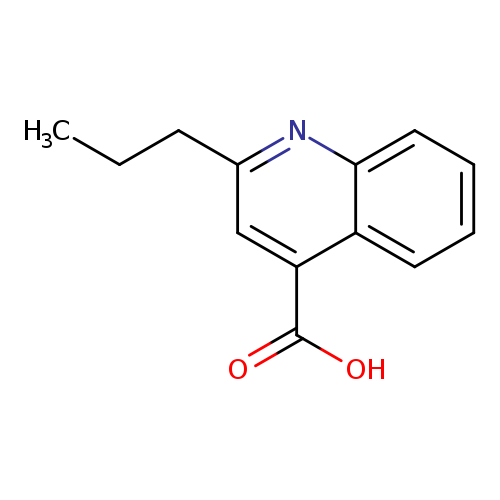
2-Propylquinoline-4-carboxylic acidCatalog No.:AA0005SP CAS No.:1019-03-0 MDL No.:MFCD01325647 MF:C13H13NO2 MW:215.2478 |
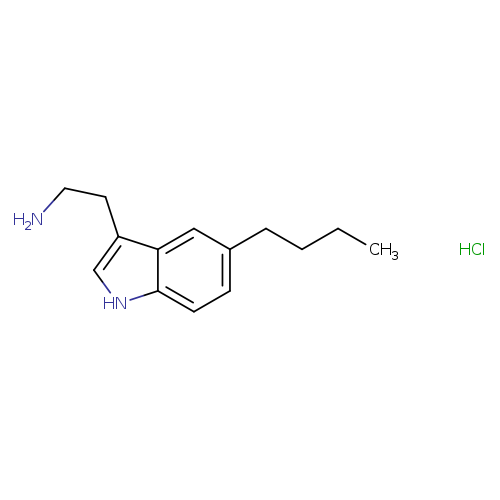
2-(5-Butyl-1h-indol-3-yl)ethanamine, HClCatalog No.:AA00H9L3 CAS No.:1019-47-2 MDL No.:MFCD00450783 MF:C14H21ClN2 MW:252.7829 |
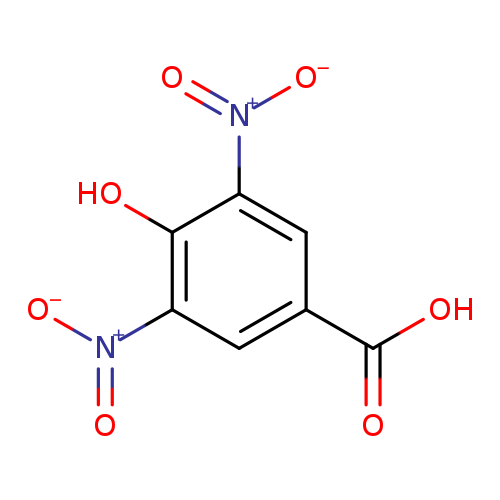
Benzoic acid, 4-hydroxy-3,5-dinitro-Catalog No.:AA0005SI CAS No.:1019-52-9 MDL No.:MFCD00017000 MF:C7H4N2O7 MW:228.1159 |
Acetaldehyde, 2-(2,4-dinitrophenyl)hydrazoneCatalog No.:AA0005SH CAS No.:1019-57-4 MDL No.:MFCD00191298 MF:C8H8N4O4 MW:224.1735 |
Bis(4-methylphenyl)chlorophosphineCatalog No.:AA0005TC CAS No.:1019-71-2 MDL No.:MFCD01630844 MF:C14H14ClP MW:248.6877 |
2-(4-Chlorophenyl)benzimidazoleCatalog No.:AA0005T8 CAS No.:1019-85-8 MDL No.:MFCD00224359 MF:C13H9ClN2 MW:228.6770 |
Ethyl 1-phenyl-1H-1,2,4-triazole-3-carboxylateCatalog No.:AA0005T4 CAS No.:1019-95-0 MDL No.:MFCD03837854 MF:C11H11N3O2 MW:217.2239 |
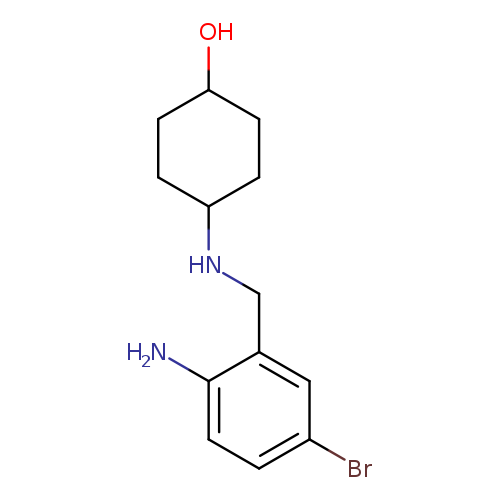
trans-4-[[(2-AMino-5-broMophenyl)Methyl]aMino]-cyclohexanolCatalog No.:AA008WAV CAS No.:101900-43-0 MDL No.:MFCD28143353 MF:C13H19BrN2O MW:299.2068 |
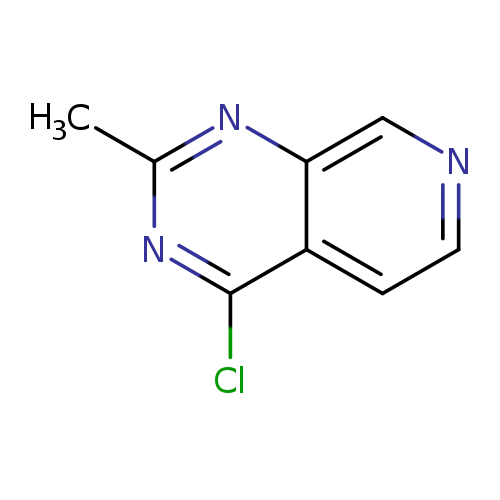
4-Chloro-2-methylpyrido[3,4-d]pyrimidineCatalog No.:AA0005T0 CAS No.:101900-98-5 MDL No.:MFCD19690424 MF:C8H6ClN3 MW:179.6063 |
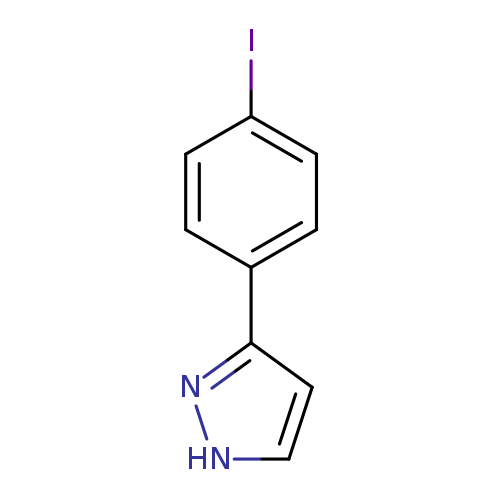
3-(4-iodophenyl)-1H-pyrazoleCatalog No.:AA01DUU7 CAS No.:1019007-92-1 MDL No.:MFCD04035693 MF:C9H7IN2 MW:270.0697 |
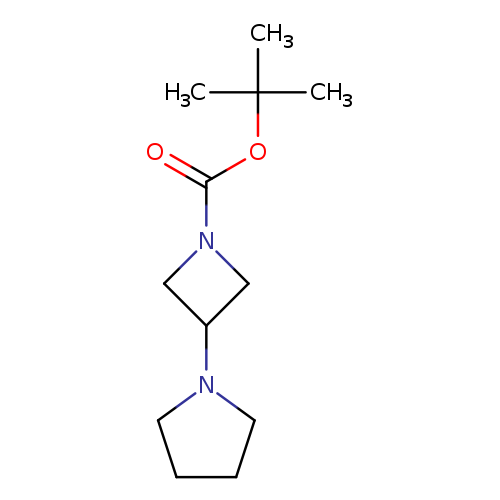
1-(1'-Boc-azetidin-3'-yl)pyrrolidineCatalog No.:AA008TUQ CAS No.:1019008-21-9 MDL No.:MFCD09860616 MF:C12H22N2O2 MW:226.3153 |
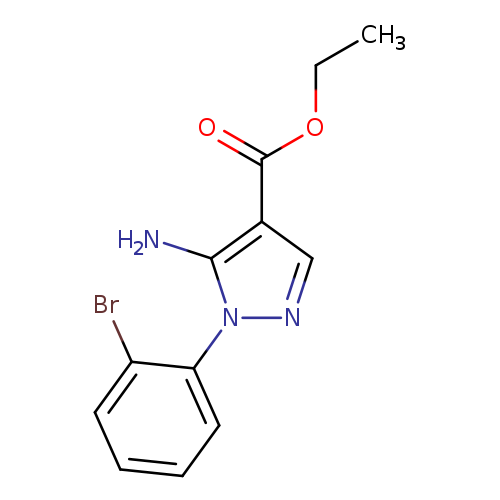
Ethyl 5-amino-1-(2-bromophenyl)-1h-pyrazole-4-carboxylateCatalog No.:AA0005ST CAS No.:1019009-68-7 MDL No.:MFCD06637280 MF:C12H12BrN3O2 MW:310.1466 |
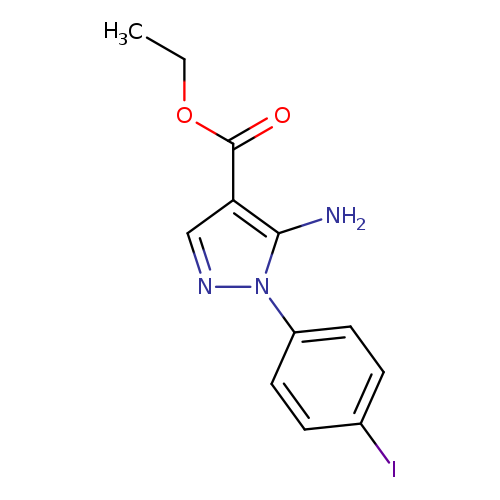
ethyl 5-amino-1-(4-iodophenyl)-1H-pyrazole-4-carboxylateCatalog No.:AA01FFMI CAS No.:1019009-81-4 MDL No.:MFCD06637284 MF:C12H12IN3O2 MW:357.1470 |
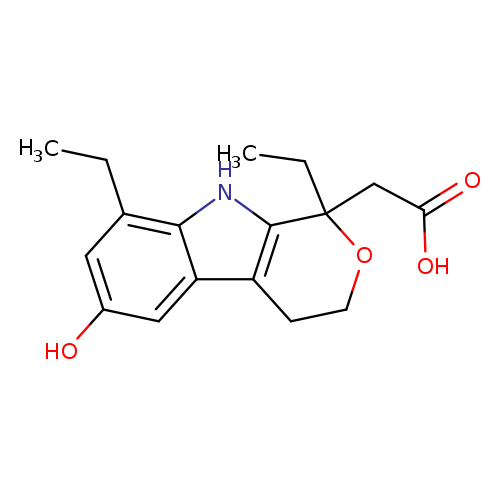
Pyrano[3,4-b]indole-1-acetic acid, 1,8-diethyl-1,3,4,9-tetrahydro-6-hydroxy-Catalog No.:AA0005SZ CAS No.:101901-06-8 MDL No.:MFCD01694076 MF:C17H21NO4 MW:303.3529 |
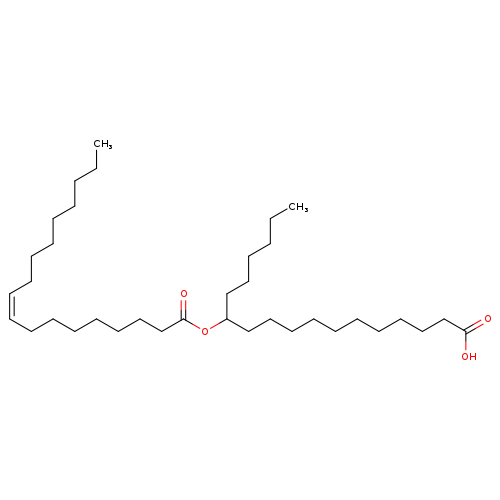
12-OAHSACatalog No.:AA008SQU CAS No.:101901-73-9 MDL No.: MF:C36H68O4 MW:564.9227 |
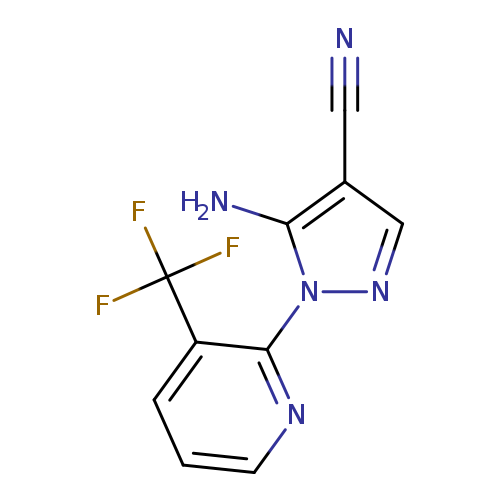
5-Amino-1-(3-(trifluoromethyl)pyridin-2-yl)-1H-pyrazole-4-carbonitrileCatalog No.:AA00ITLV CAS No.:1019010-69-5 MDL No.:MFCD06808548 MF:C10H6F3N5 MW:253.1833 |
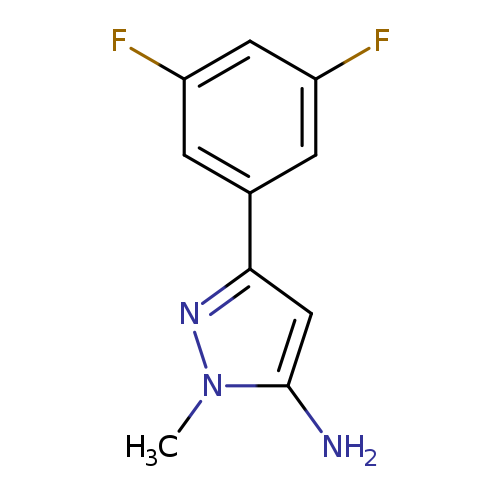
3-(3,5-difluorophenyl)-1-methyl-1H-pyrazol-5-amineCatalog No.:AA01BF0Z CAS No.:1019010-72-0 MDL No.:MFCD05664775 MF:C10H9F2N3 MW:209.1954 |
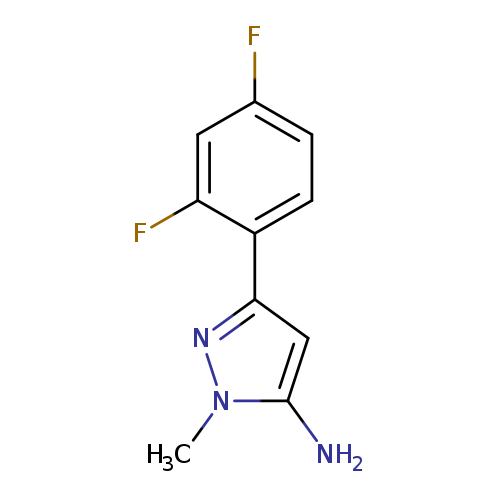
3-(2,4-difluorophenyl)-1-methyl-1H-pyrazol-5-amineCatalog No.:AA01BFOC CAS No.:1019011-31-4 MDL No.:MFCD05664865 MF:C10H9F2N3 MW:209.1954 |
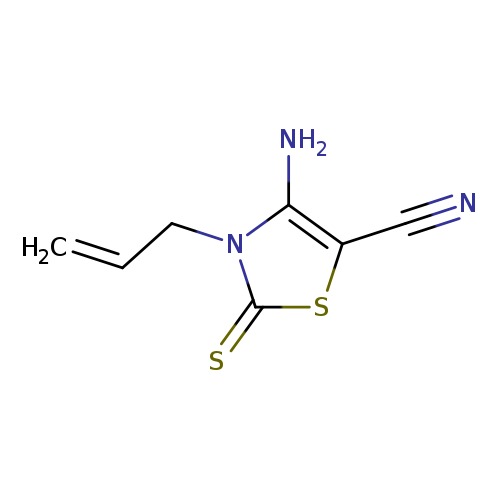
4-amino-3-(prop-2-en-1-yl)-2-sulfanylidene-2,3-dihydro-1,3-thiazole-5-carbonitrileCatalog No.:AA01A7H5 CAS No.:1019014-31-3 MDL No.:MFCD09861267 MF:C7H7N3S2 MW:197.2806 |
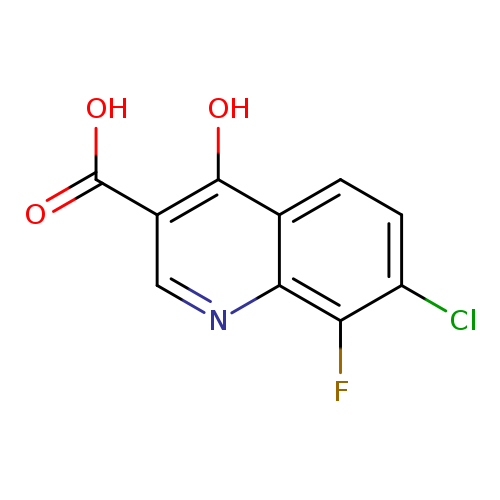
7-Chloro-8-fluoro-4-hydroxyquinoline-3-carboxylic acidCatalog No.:AA01AHHI CAS No.:1019015-91-8 MDL No.:MFCD09861319 MF:C10H5ClFNO3 MW:241.6030 |
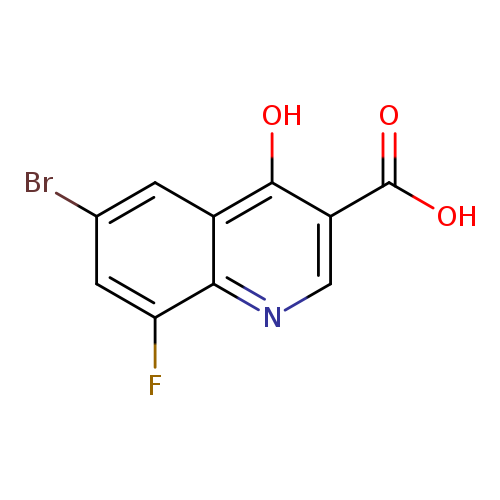
6-bromo-8-fluoro-4-hydroxyquinoline-3-carboxylic acidCatalog No.:AA01A954 CAS No.:1019016-07-9 MDL No.:MFCD09861321 MF:C10H5BrFNO3 MW:286.0540 |
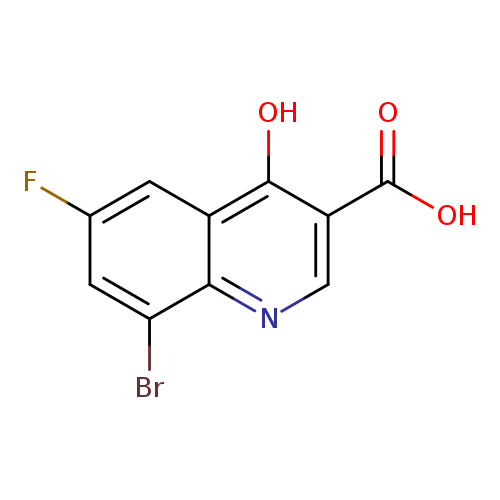
8-Bromo-6-fluoro-4-hydroxyquinoline-3-carboxylic acidCatalog No.:AA0005SS CAS No.:1019016-15-9 MDL No.:MFCD09861322 MF:C10H5BrFNO3 MW:286.0540 |
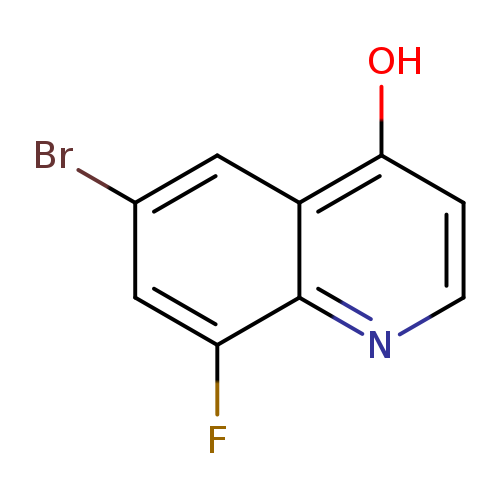
6-Bromo-8-fluoroquinolin-4-olCatalog No.:AA01DHBJ CAS No.:1019016-22-8 MDL No.:MFCD09861332 MF:C9H5BrFNO MW:242.0445 |
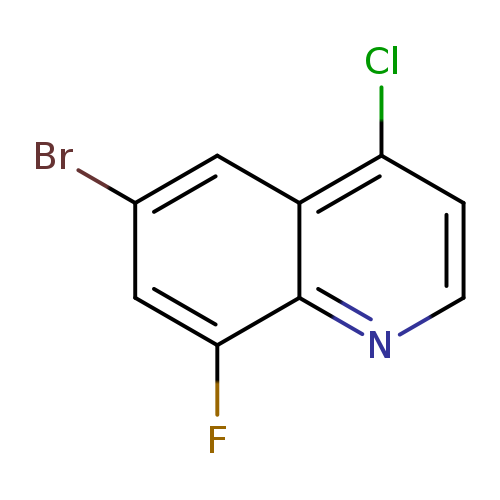
6-Bromo-4-chloro-8-fluoroquinolineCatalog No.:AA0005SR CAS No.:1019016-66-0 MDL No.:MFCD09861346 MF:C9H4BrClFN MW:260.4902 |
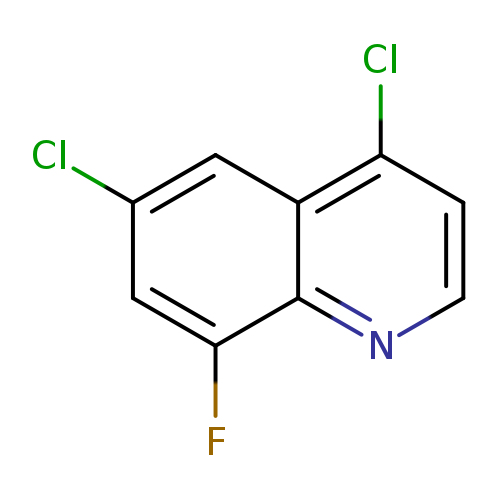
4,6-dichloro-8-fluoroquinolineCatalog No.:AA01AA8L CAS No.:1019016-87-5 MDL No.:MFCD09861349 MF:C9H4Cl2FN MW:216.0392 |
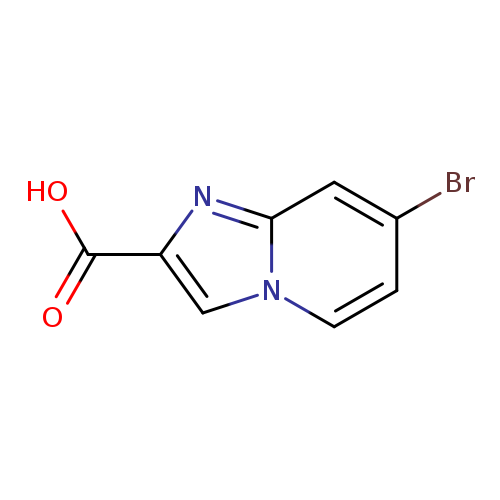
7-Bromoimidazo[1,2-a]pyridine-2-carboxylic acidCatalog No.:AA0005TR CAS No.:1019018-46-2 MDL No.:MFCD22989340 MF:C8H5BrN2O2 MW:241.0415 |
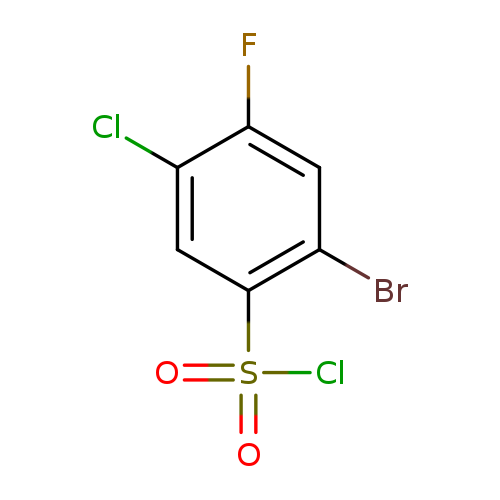
2-Bromo-5-chloro-4-fluorobenzenesulfonyl chlorideCatalog No.:AA01F92T CAS No.:1019018-84-8 MDL No.:MFCD09861353 MF:C6H2BrCl2FO2S MW:307.9523 |
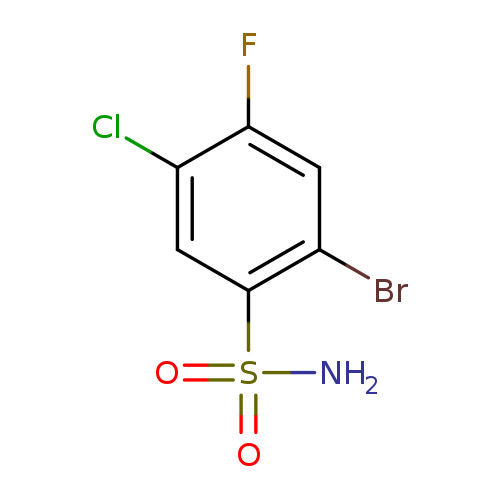
2-Bromo-5-chloro-4-fluorobenzenesulfonamideCatalog No.:AA01FAQH CAS No.:1019018-92-8 MDL No.:MFCD09861354 MF:C6H4BrClFNO2S MW:288.5219 |
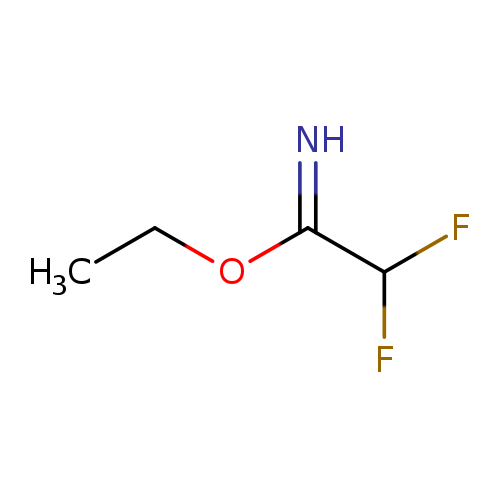
Ethyl 2,2-difluoroethanecarboximidateCatalog No.:AA01DUU8 CAS No.:1019019-37-4 MDL No.:MFCD09996088 MF:C4H7F2NO MW:123.1013 |
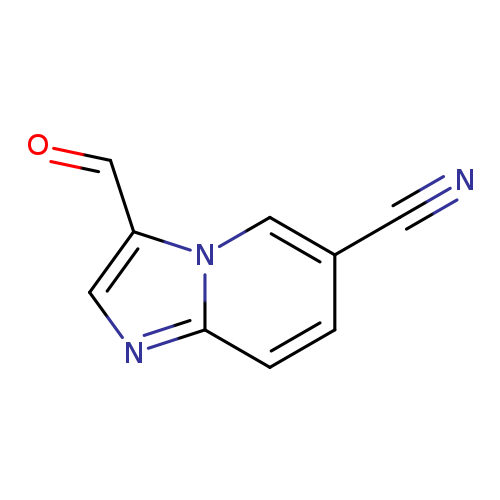
3-Formylimidazo[1,2-a]pyridine-6-carbonitrileCatalog No.:AA01AJ3R CAS No.:1019019-92-1 MDL No.:MFCD09994345 MF:C9H5N3O MW:171.1555 |
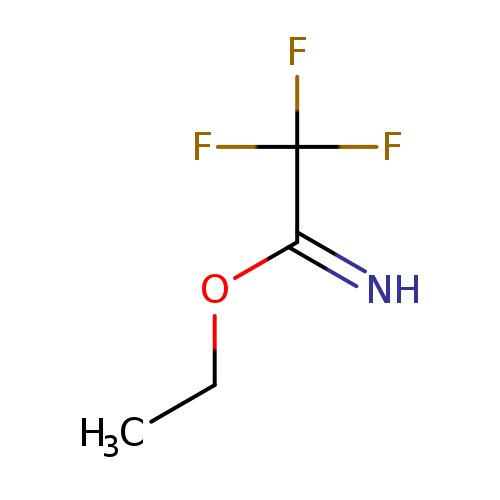
ethyl 2,2,2-trifluoroethanecarboximidateCatalog No.:AA01BDXE CAS No.:1019019-98-7 MDL No.:MFCD09996103 MF:C4H6F3NO MW:141.0917 |
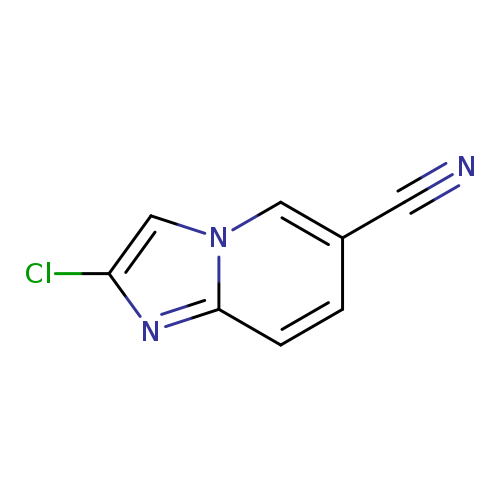
2-Chloroimidazo[1,2-a]pyridine-6-carbonitrileCatalog No.:AA0005TQ CAS No.:1019020-03-1 MDL No.:MFCD09994269 MF:C8H4ClN3 MW:177.5905 |
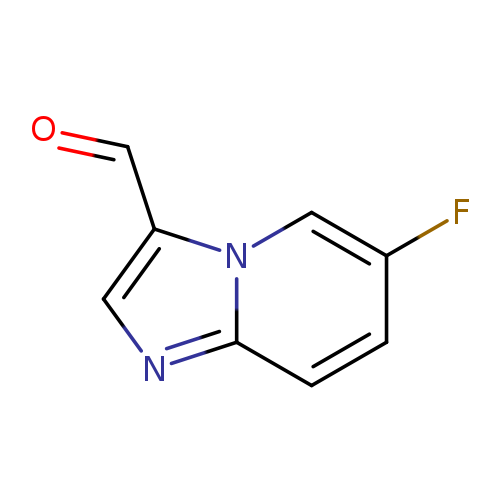
6-fluoroimidazo[1,2-a]pyridine-3-carbaldehydeCatalog No.:AA0005TP CAS No.:1019020-06-4 MDL No.:MFCD09994347 MF:C8H5FN2O MW:164.1365 |
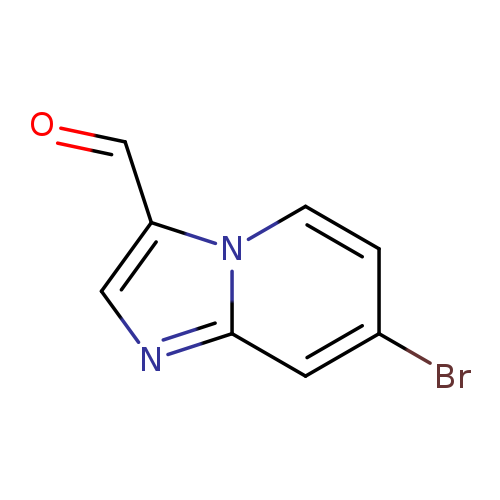
7-Bromoimidazo[1,2-a]pyridine-3-carbaldehydeCatalog No.:AA0005TO CAS No.:1019020-14-4 MDL No.:MFCD09994348 MF:C8H5BrN2O MW:225.0421 |
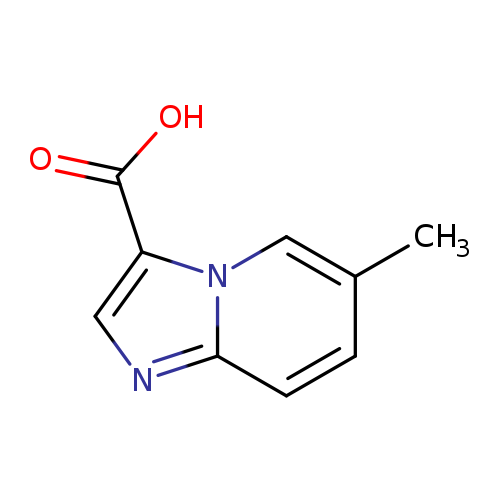
6-Methylimidazo[1,2-a]pyridine-3-carboxylic acidCatalog No.:AA0005TN CAS No.:1019021-64-7 MDL No.:MFCD09994698 MF:C9H8N2O2 MW:176.1720 |
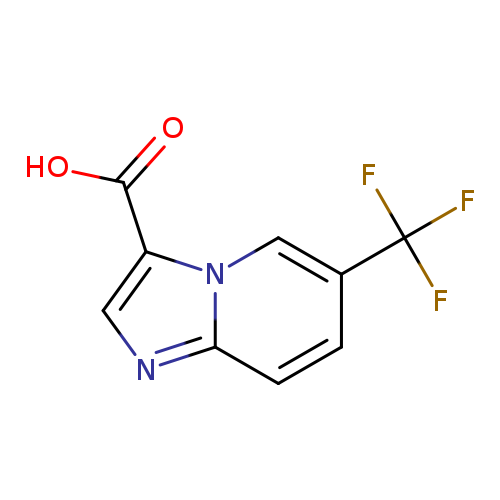
6-(Trifluoromethyl)imidazo[1,2-a]pyridine-3-carboxylic acidCatalog No.:AA0005TL CAS No.:1019021-78-3 MDL No.:MFCD09994700 MF:C9H5F3N2O2 MW:230.1434 |
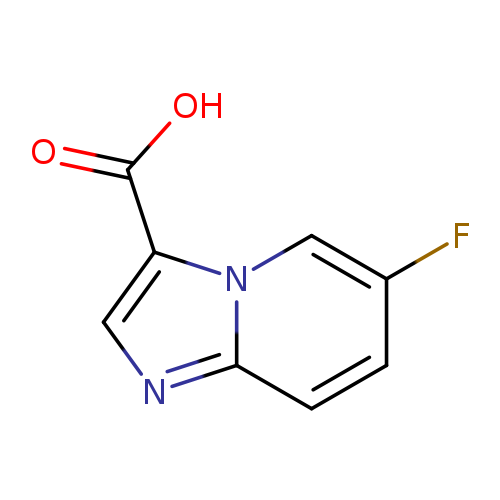
6-Fluoroimidazo[1,2-a]pyridine-3-carboxylic acidCatalog No.:AA0005TK CAS No.:1019021-85-2 MDL No.:MFCD09994701 MF:C8H5FN2O2 MW:180.1359 |
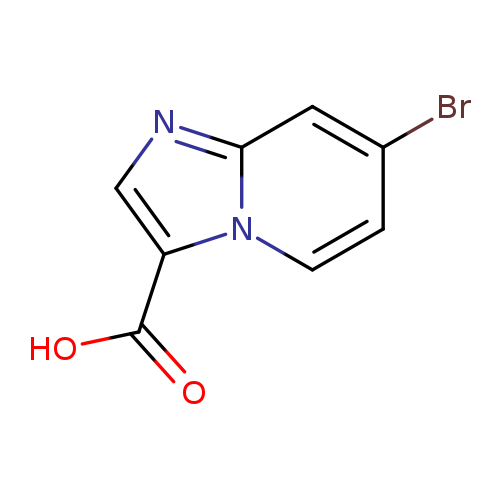
7-Bromoimidazo[1,2-a]pyridine-3-carboxylic acidCatalog No.:AA0005TI CAS No.:1019021-93-2 MDL No.:MFCD09994702 MF:C8H5BrN2O2 MW:241.0415 |
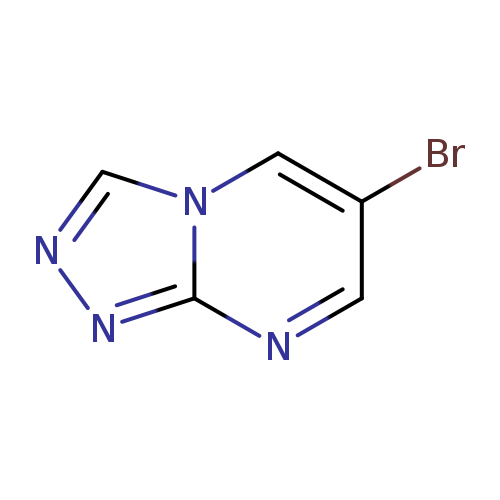
6-Bromo[1,2,4]triazolo[4,3-a]pyrimidineCatalog No.:AA008ZCW CAS No.:1019023-12-1 MDL No.:MFCD09994391 MF:C5H3BrN4 MW:199.0081 |
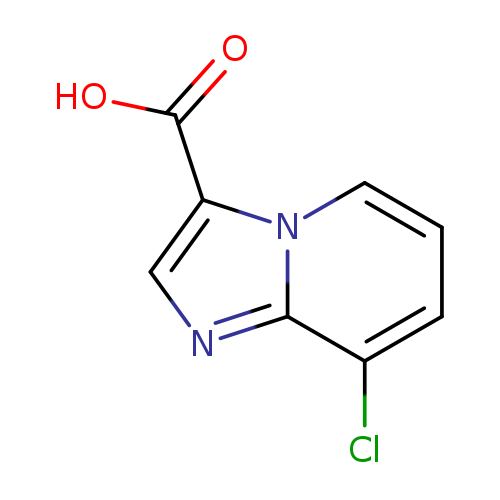
8-Chloroimidazo[1,2-a]pyridine-3-carboxylic acidCatalog No.:AA01C9E6 CAS No.:1019023-77-8 MDL No.:MFCD09994725 MF:C8H5ClN2O2 MW:196.5905 |
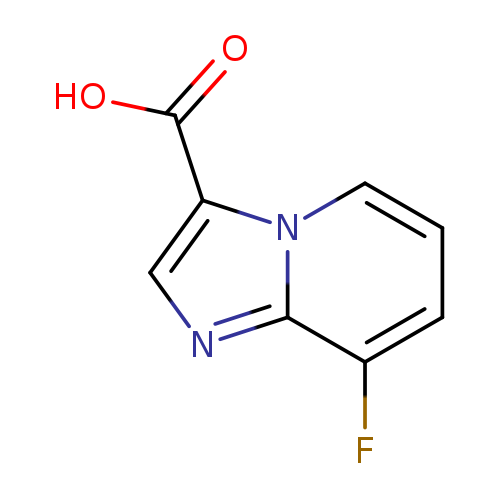
8-Fluoroimidazo[1,2-a]pyridine-3-carboxylic acidCatalog No.:AA0098YM CAS No.:1019023-85-8 MDL No.:MFCD09994726 MF:C8H5FN2O2 MW:180.1359 |
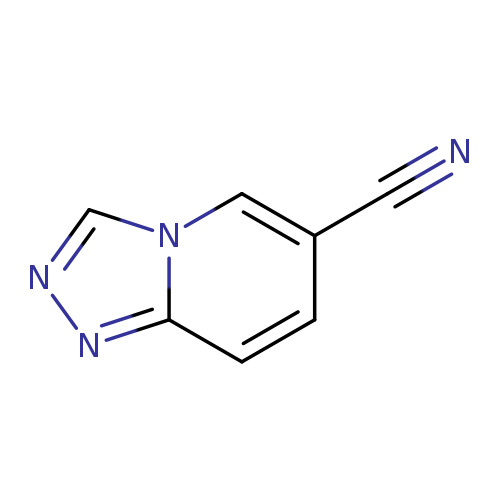
[1,2,4]Triazolo[4,3-a]pyridine-6-carbonitrileCatalog No.:AA0005TH CAS No.:1019024-64-6 MDL No.:MFCD09994414 MF:C7H4N4 MW:144.1335 |
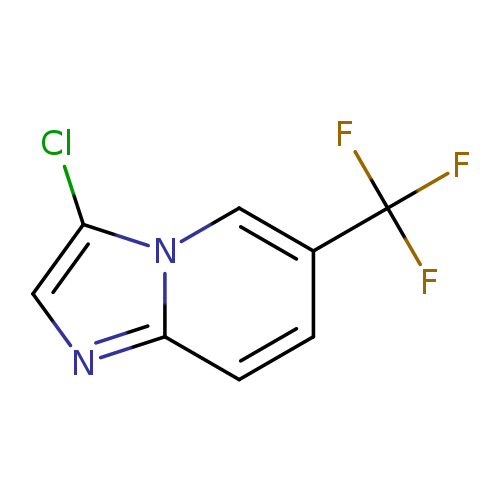
3-Chloro-6-(trifluoromethyl)imidazo[1,2-a]pyridineCatalog No.:AA00ITWT CAS No.:1019027-76-9 MDL No.:MFCD09994776 MF:C8H4ClF3N2 MW:220.5790 |
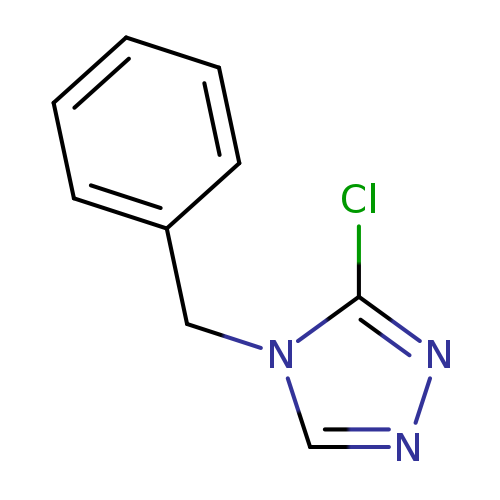
4-benzyl-3-chloro-4H-1,2,4-triazoleCatalog No.:AA01C70H CAS No.:1019031-93-6 MDL No.:MFCD09995772 MF:C9H8ClN3 MW:193.6329 |
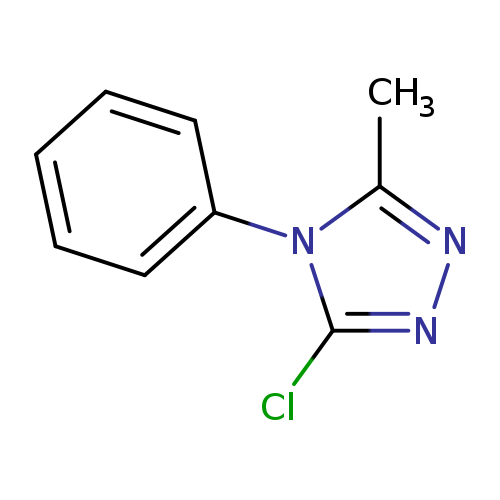
3-chloro-5-methyl-4-phenyl-4H-1,2,4-triazoleCatalog No.:AA019MKL CAS No.:1019032-00-8 MDL No.:MFCD09995773 MF:C9H8ClN3 MW:193.6329 |
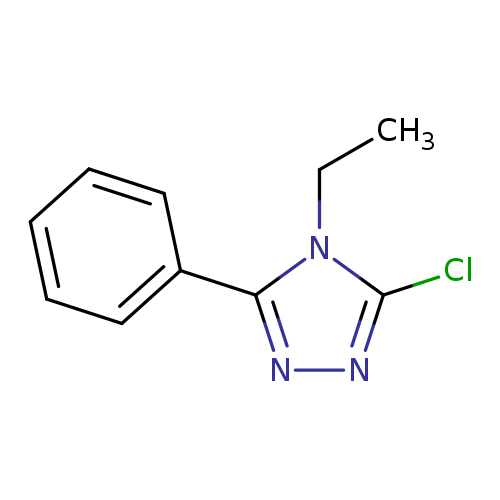
3-chloro-4-ethyl-5-phenyl-4H-1,2,4-triazoleCatalog No.:AA019MO8 CAS No.:1019032-87-1 MDL No.:MFCD09995790 MF:C10H10ClN3 MW:207.6595 |
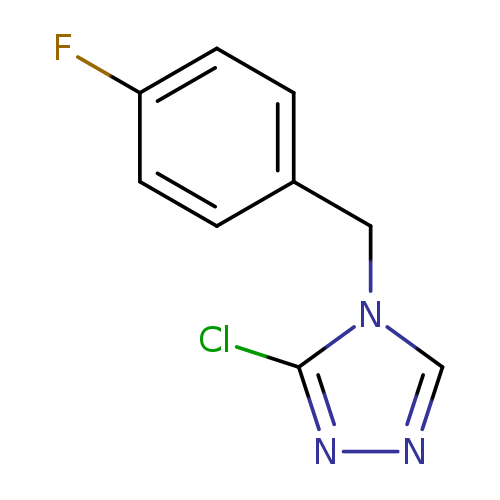
3-chloro-4-(4-fluorobenzyl)-4H-1,2,4-triazoleCatalog No.:AA01BJ5E CAS No.:1019033-05-6 MDL No.:MFCD09995796 MF:C9H7ClFN3 MW:211.6234 |

4-Chloro-5h-pyrrolo[3,2-d]pyrimidine-7-carboxylic acidCatalog No.:AA0005TE CAS No.:1019056-31-5 MDL No.:MFCD09999221 MF:C7H4ClN3O2 MW:197.5786 |
1-(4-Fluorophenyl)-2-piperazin-1-ylethanone dihydrochlorideCatalog No.:AA00VT6F CAS No.:1019058-85-5 MDL No.:MFCD09971581 MF:C12H17Cl2FN2O MW:295.1806 |
4-Trifluoromethylphenylglyoxal hydrateCatalog No.:AA0005TU CAS No.:101906-05-2 MDL No.:MFCD08705869 MF:C9H7F3O3 MW:220.1453 |
2-chloro-N-[1-(4-fluorophenyl)-3-methyl-1H-pyrazol-5-yl]acetamideCatalog No.:AA019PMY CAS No.:1019067-51-6 MDL No.:MFCD09802028 MF:C12H11ClFN3O MW:267.6866 |
1,4-dibromo-2,5-diethoxybenzeneCatalog No.:AA01C4V2 CAS No.:101907-55-5 MDL No.:MFCD08696083 MF:C10H12Br2O2 MW:324.0091 |
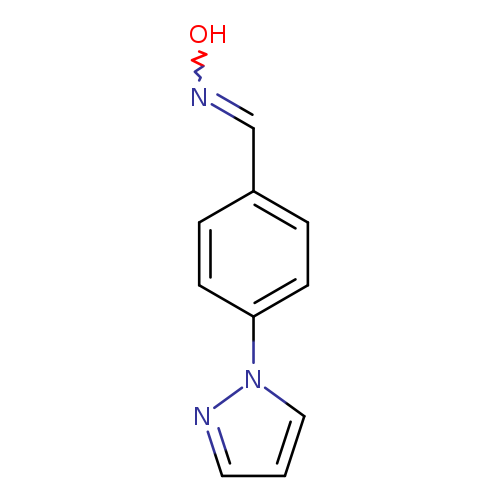
N-{[4-(1H-pyrazol-1-yl)phenyl]methylidene}hydroxylamineCatalog No.:AA019KRQ CAS No.:1019070-96-2 MDL No.:MFCD09403774 MF:C10H9N3O MW:187.1980 |
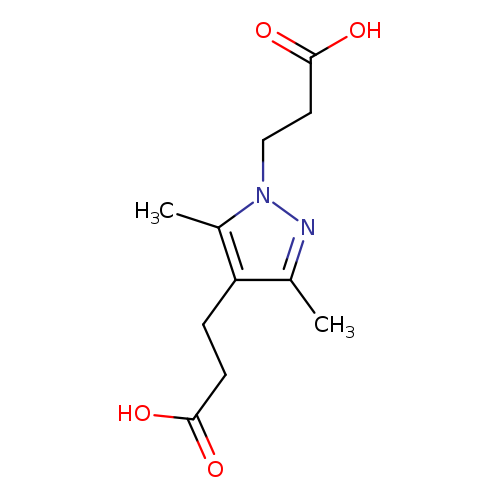
3-[4-(2-carboxyethyl)-3,5-dimethyl-1H-pyrazol-1-yl]propanoic acidCatalog No.:AA019KSK CAS No.:1019071-94-3 MDL No.:MFCD09802060 MF:C11H16N2O4 MW:240.2557 |
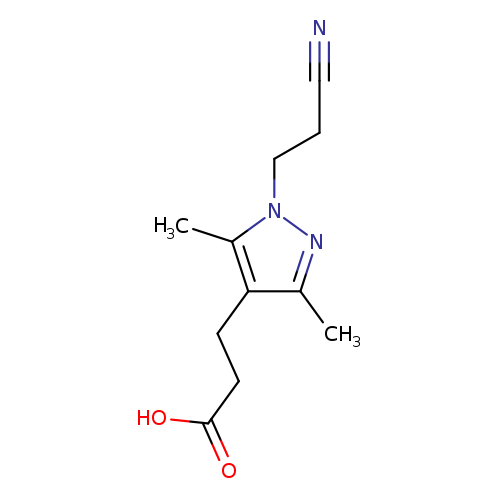
3-[1-(2-Cyanoethyl)-3,5-dimethyl-1h-pyrazol-4-yl]propanoic acidCatalog No.:AA019KYR CAS No.:1019074-26-0 MDL No.:MFCD09051597 MF:C11H15N3O2 MW:221.2557 |
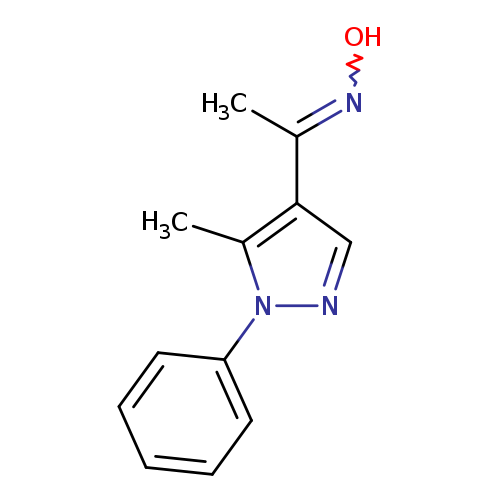
N-[1-(5-methyl-1-phenyl-1H-pyrazol-4-yl)ethylidene]hydroxylamineCatalog No.:AA019KTM CAS No.:1019075-43-4 MDL No.:MFCD09802068 MF:C12H13N3O MW:215.2511 |
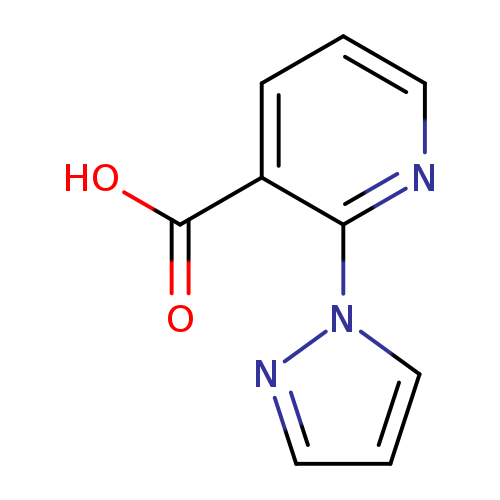
2-(1H-pyrazol-1-yl)pyridine-3-carboxylic acidCatalog No.:AA019KUN CAS No.:1019075-47-8 MDL No.:MFCD09802092 MF:C9H7N3O2 MW:189.1708 |
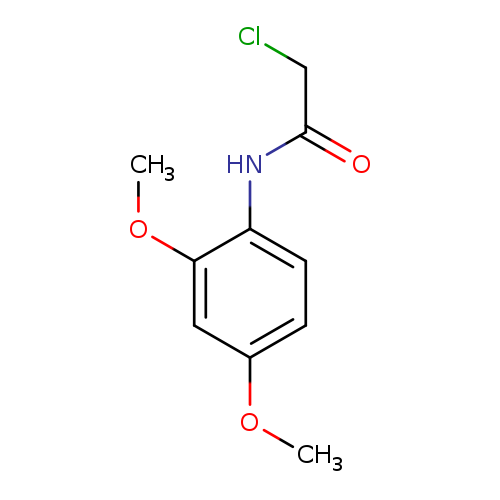
N1-(2,4-Dimethoxyphenyl)-2-chloroacetamideCatalog No.:AA0005UF CAS No.:101908-41-2 MDL No.:MFCD00750873 MF:C10H12ClNO3 MW:229.6602 |
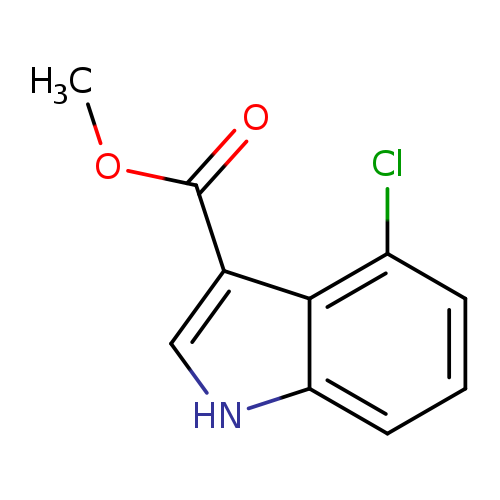
Methyl 4-chloro-1H-indole-3-carboxylateCatalog No.:AA0005UB CAS No.:101909-42-6 MDL No.:MFCD09841616 MF:C10H8ClNO2 MW:209.6290 |
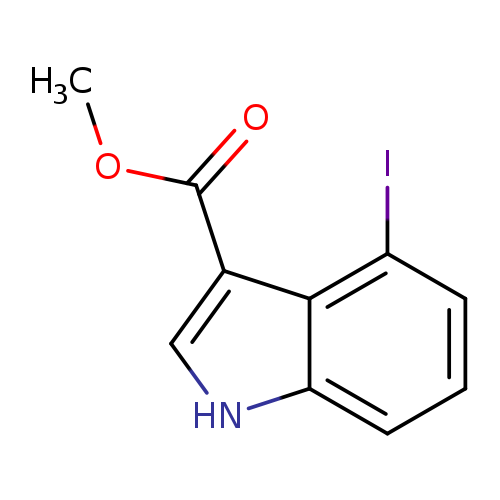
Methyl 4-iodo-1H-indole-3-carboxylateCatalog No.:AA0005U9 CAS No.:101909-44-8 MDL No.:MFCD04038681 MF:C10H8INO2 MW:301.0805 |
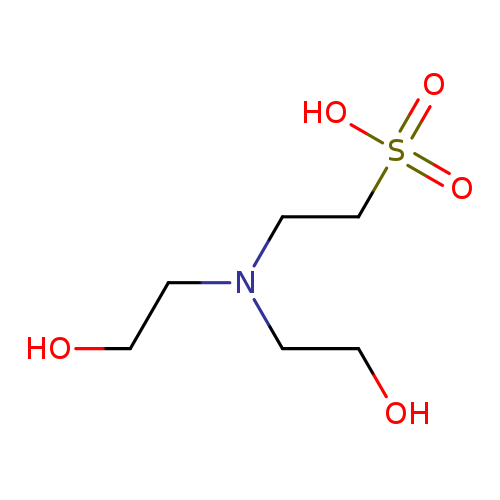
N,N-Bis(2-hydroxyethyl)-2-aminoethanesulphonic acidCatalog No.:AA0005UP CAS No.:10191-18-1 MDL No.:MFCD00007533 MF:C6H15NO5S MW:213.2520 |

3-Hydroxyhexanoic acidCatalog No.:AA0005UO CAS No.:10191-24-9 MDL No.:MFCD02259045 MF:C6H12O3 MW:132.1577 |
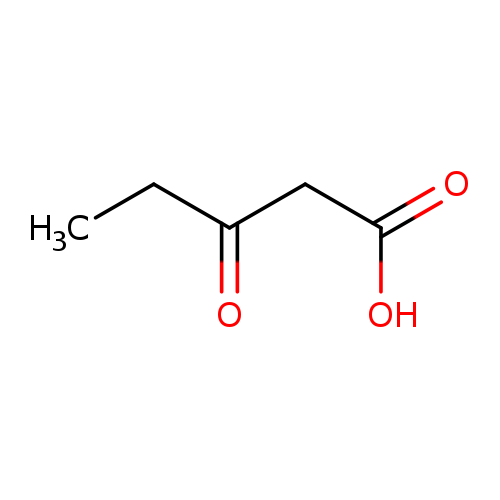
Pentanoic acid, 3-oxo-Catalog No.:AA0005UN CAS No.:10191-25-0 MDL No.:MFCD11040434 MF:C5H8O3 MW:116.1152 |
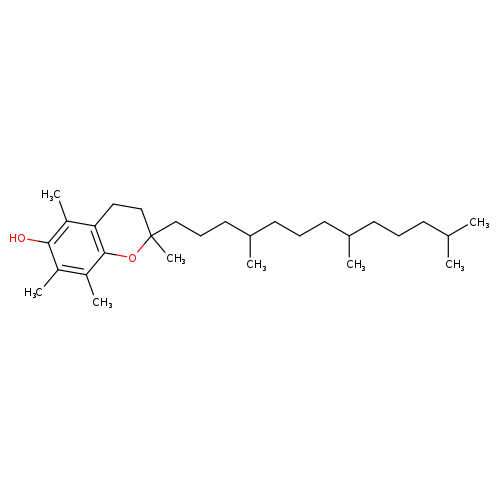
2,5,7,8-tetramethyl-2-(4,8,12-trimethyltridecyl)-3,4-dihydro-2H-1-benzopyran-6-olCatalog No.:AA0034Q8 CAS No.:10191-41-0 MDL No.:MFCD00072045 MF:C29H50O2 MW:430.7061 |
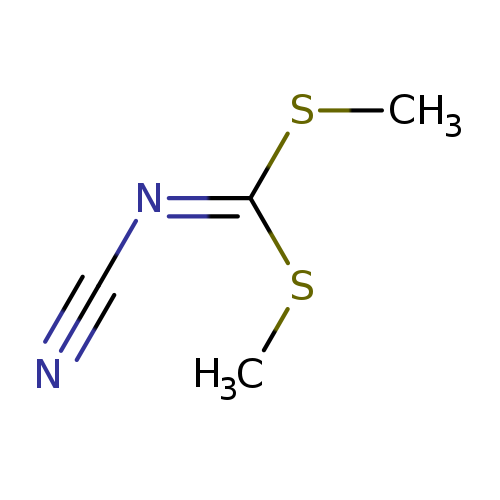
N-Cyanoimido-s,s-dimethyl-dithiocarbonateCatalog No.:AA003PJ2 CAS No.:10191-60-3 MDL No.:MFCD00009825 MF:C4H6N2S2 MW:146.2338 |
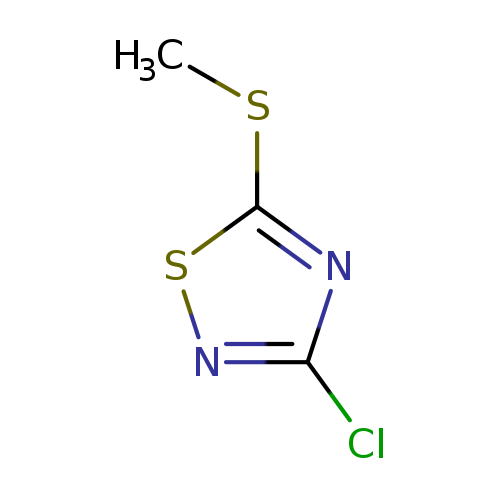
3-Chloro-5-methylthio-1,2,4-thiadiazoleCatalog No.:AA0005UH CAS No.:10191-90-9 MDL No.:MFCD06658049 MF:C3H3ClN2S2 MW:166.6523 |
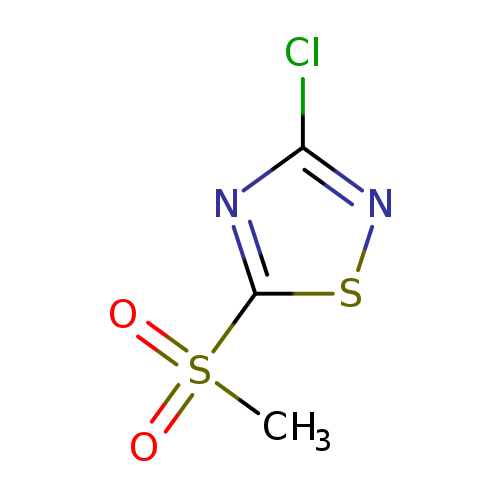
3-chloro-5-methanesulfonyl-1,2,4-thiadiazoleCatalog No.:AA01EIHX CAS No.:10191-91-0 MDL No.:MFCD31629214 MF:C3H3ClN2O2S2 MW:198.6511 |

Benzenemethanol, α-pentyl-3-(2-quinolinylmethoxy)-Catalog No.:AA0005U8 CAS No.:101910-24-1 MDL No.:MFCD00211150 MF:C22H25NO2 MW:335.4394 |
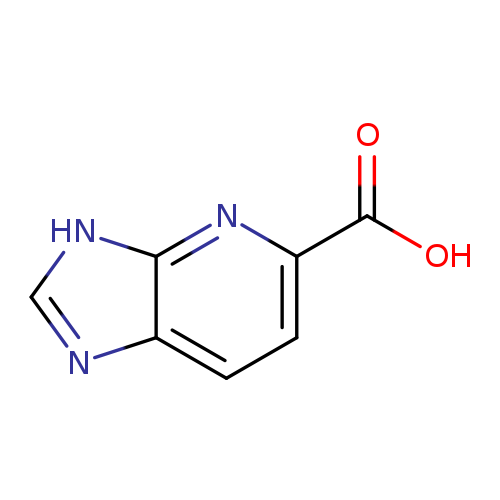
3H-Imidazo[4,5-b]pyridine-5-carboxylic acidCatalog No.:AA0005U5 CAS No.:1019108-05-4 MDL No.:MFCD09863858 MF:C7H5N3O2 MW:163.1335 |
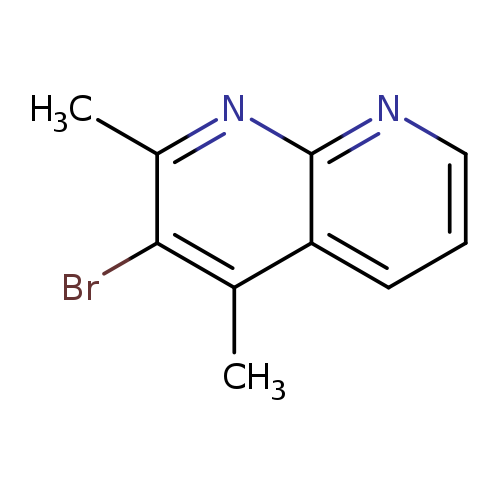
3-Bromo-2,4-dimethyl-1,8-naphthyridineCatalog No.:AA01ARBL CAS No.:1019108-06-5 MDL No.:MFCD09863865 MF:C10H9BrN2 MW:237.0959 |
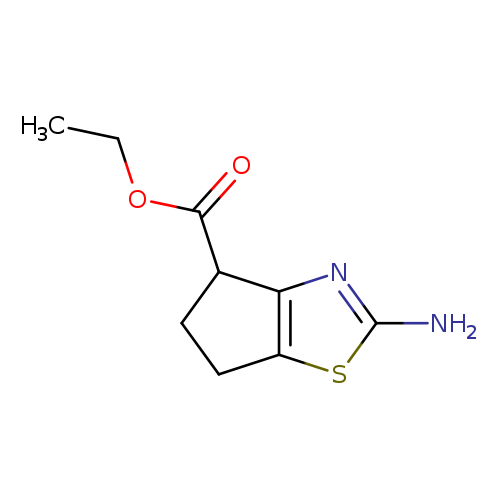
Ethyl 2-amino-5,6-dihydro-4H-cyclopenta[d]thiazole-4-carboxylateCatalog No.:AA0005U4 CAS No.:1019108-35-0 MDL No.:MFCD09864118 MF:C9H12N2O2S MW:212.2688 |
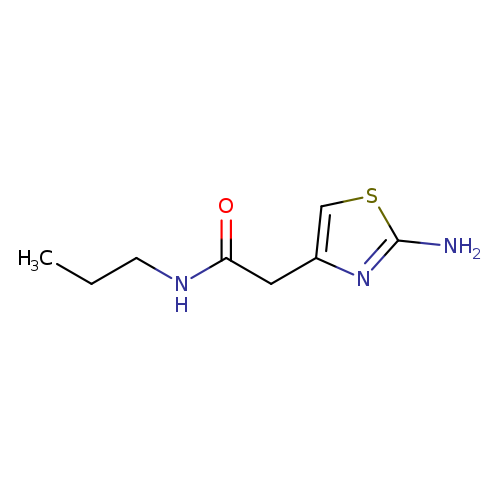
2-(2-aminothiazol-4-yl)-N-propylacetamideCatalog No.:AA01ARB9 CAS No.:1019108-37-2 MDL No.:MFCD09864128 MF:C8H13N3OS MW:199.2733 |
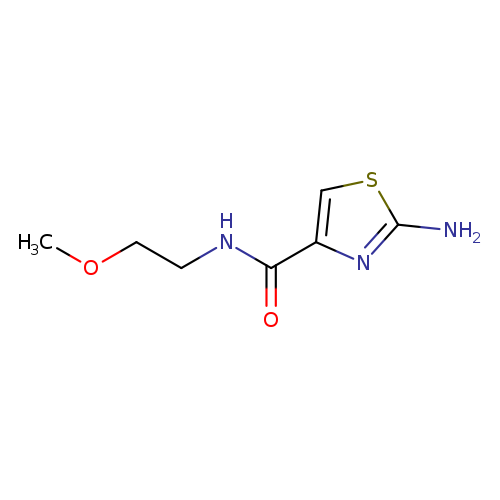
2-Amino-N-(2-methoxyethyl)-1,3-thiazole-4-carboxamideCatalog No.:AA01ARAN CAS No.:1019108-39-4 MDL No.:MFCD09864138 MF:C7H11N3O2S MW:201.2461 |
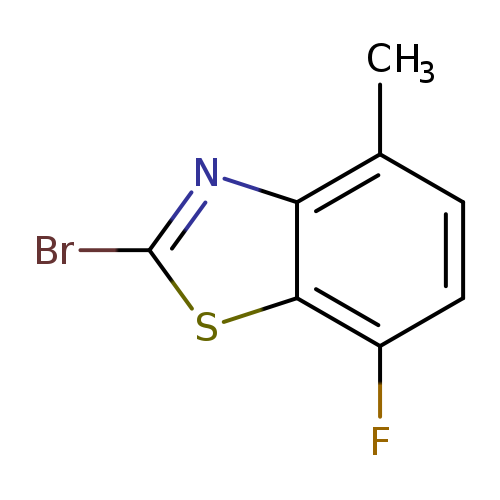
2-Bromo-7-fluoro-4-methylbenzothiazoleCatalog No.:AA0005U3 CAS No.:1019108-45-2 MDL No.:MFCD09749155 MF:C8H5BrFNS MW:246.0994 |
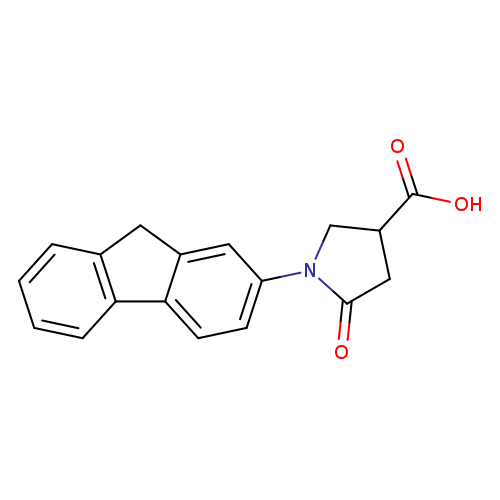
1-(9H-Fluoren-2-yl)-5-oxopyrrolidine-3-carboxylic acidCatalog No.:AA019WQT CAS No.:1019109-76-2 MDL No.:MFCD09815349 MF:C18H15NO3 MW:293.3166 |
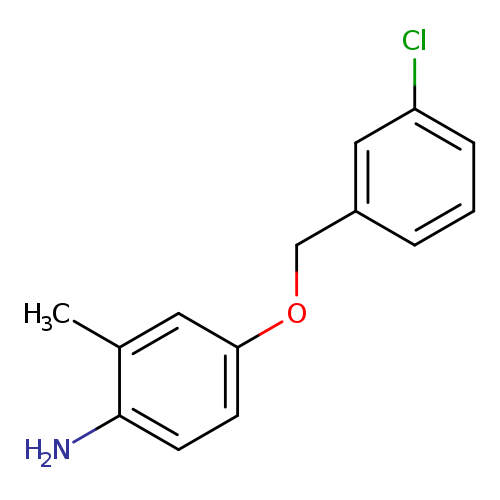
4-[(3-chlorophenyl)methoxy]-2-methylanilineCatalog No.:AA01AA7J CAS No.:1019110-00-9 MDL No.:MFCD09808863 MF:C14H14ClNO MW:247.7201 |
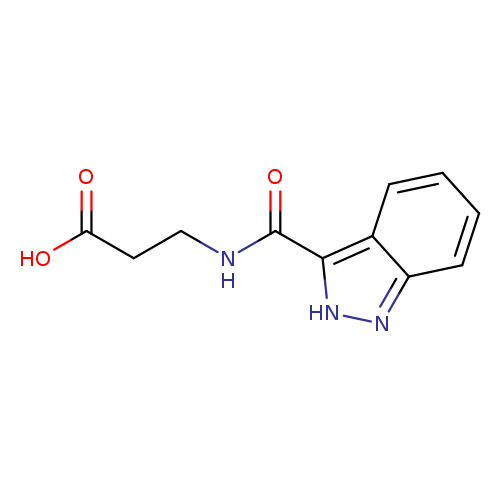
3-(2H-indazol-3-ylformamido)propanoic acidCatalog No.:AA01A9IW CAS No.:1019110-35-0 MDL No.:MFCD11126333 MF:C11H11N3O3 MW:233.2233 |
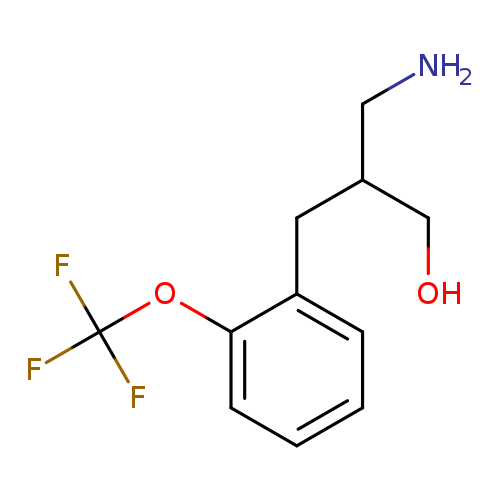
3-amino-2-{[2-(trifluoromethoxy)phenyl]methyl}propan-1-olCatalog No.:AA019SBT CAS No.:1019110-74-7 MDL No.:MFCD11126708 MF:C11H14F3NO2 MW:249.2296 |
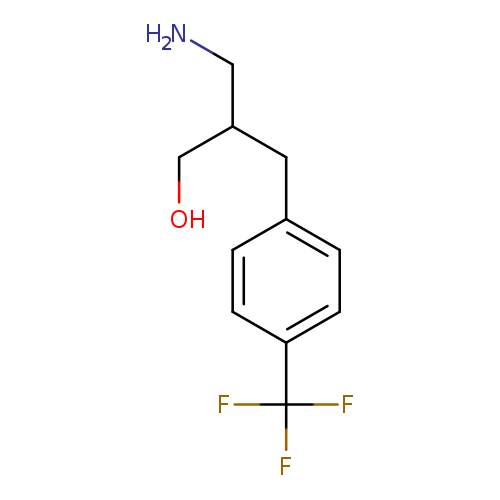
3-amino-2-{[4-(trifluoromethyl)phenyl]methyl}propan-1-olCatalog No.:AA019XU7 CAS No.:1019110-79-2 MDL No.:MFCD11126646 MF:C11H14F3NO MW:233.2302 |
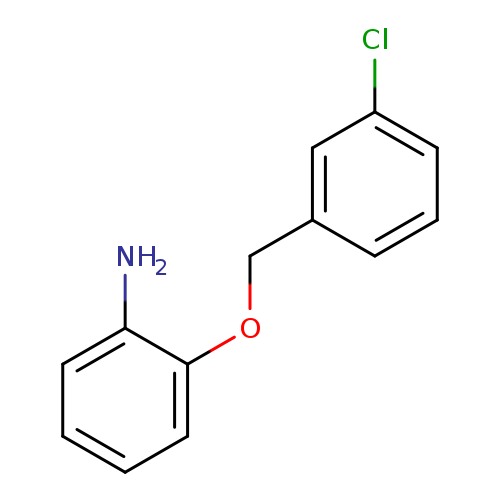
2-[(3-Chlorophenyl)methoxy]anilineCatalog No.:AA01AJKS CAS No.:1019111-09-1 MDL No.:MFCD09803910 MF:C13H12ClNO MW:233.6935 |
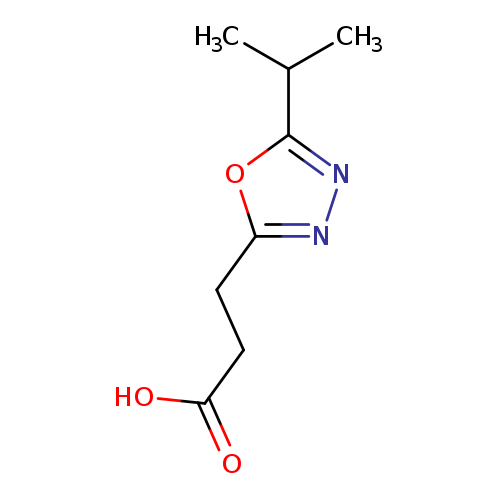
3-(5-Isopropyl-1,3,4-oxadiazol-2-yl)propanoic acidCatalog No.:AA019TH0 CAS No.:1019111-17-1 MDL No.:MFCD09880433 MF:C8H12N2O3 MW:184.1925 |
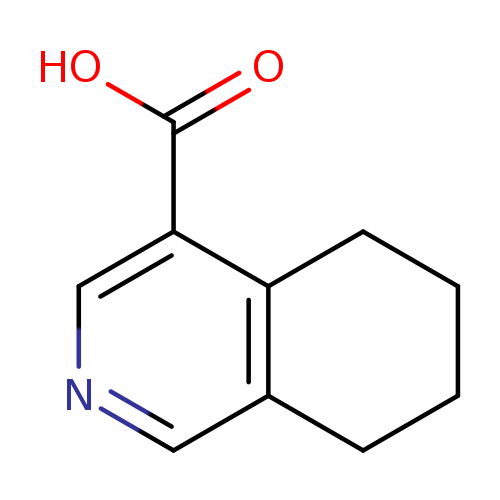
5,6,7,8-TETRAHYDROISOQUINOLINE-4-CARBOXYLIC ACIDCatalog No.:AA01DX45 CAS No.:1019111-31-9 MDL No.:MFCD09863909 MF:C10H11NO2 MW:177.1998 |
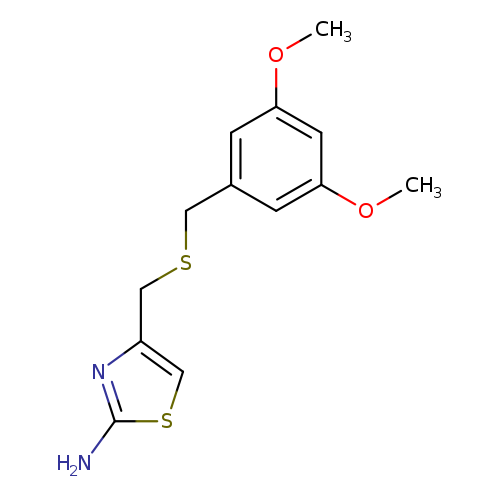
4-([(3,5-Dimethoxybenzyl)thio]methyl)-1,3-thiazol-2-amineCatalog No.:AA01ARAR CAS No.:1019111-59-1 MDL No.:MFCD09864142 MF:C13H16N2O2S2 MW:296.4083 |
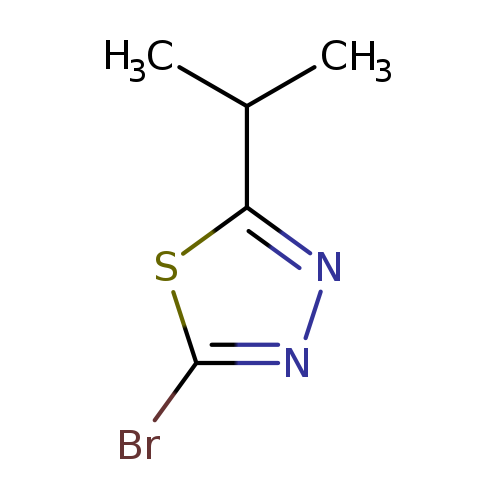
2-Bromo-5-isopropyl-1,3,4-thiadiazoleCatalog No.:AA0005V2 CAS No.:1019111-62-6 MDL No.:MFCD09965539 MF:C5H7BrN2S MW:207.0915 |