2020-01-14 09:04:16
Saumen Hajra,*[a] Subrata Maity,[a,b][‡] Sayan Roy,[a][‡] Ramkrishna Maity,[a] and Srikrishna Samanta
Introduction
Generation of all-carbon quaternary stereocentre always re- mains a demanding venture in organic synthesis.[1] In this con- text, development of quaternary centre particularly at the C3- position of oxindole captured considerable research enthusiasm of synthetic community because of the incredible prevalence of such molecules in numerous natural products. Among these, 3a-(3-indolyl)-hexahydropyrrolo-[2,3-b] indole[2] is a unique structural framework present in various bisindole alkaloids viz. gliocladine A, B, gliocladin C, bionectin A, B, leptosin D with tremendous biological and pharmacological activities. Similarly, 3-aryl oxindole[3] moiety is also existed in diazonamide A, azon- azine, naseseazine, XEN 907, XEN 402, displaying a wide range of biological activities (Figure 1). As a consequence, development of a unique synthetic strategy to avail 3,3-disubstituted oxindole is highly desirable.
A numerous synthetic strategy towards the construction of 3, 3-bisindole have been reported so far in literature. The examples of (i) Mukaiyama-aldol reaction of 3-(3-indolyl)-2-siloxy- indole with aldehyde,[4] (ii) acyl migration of indolyl carbon- ates,[5] (iii) Pd-catalysed allylic alkylation of 3-aryl-3-oxindoles with allenes,[6] (iv) organocatalytic conjugate addition of indoles to isatin derived nitroalkenes and α,ß-unsturated aldehydes,[7] (v) α-alkylation of carbonyl compounds with 3-hydroxy-3-indol- 3-yloxindoles,[8] (vi) Rh-catalysed multicomponent reaction of 3-diazooxindoles, indoles and aldehydes,[9] (vii) Cu-catalysed dearomatization of indoleacetamides with 3-indolylphenyliodo- nium salts[10] are worthy of attention. However the preparation of 3-aryl oxindole moiety is sparse in literature.[11–13] Only few reports are there such as (i) Hetero-Claisen reaction of nitrones with alkylarylketenes,[11] (ii) Cu catalysed dearomatisation of tryptamine derivative with aryl-iodonium salt,[12] (iii) Lewis acid catalysed F-C alkylation of 3-hydroxy-2-oxindole.[13] Thus con- struction of both these functionalities from a common precur- sor through a concise and divergent manner is very much necessary. A straightforward strategy for the synthesis of these sub- units could be a regioselective F-C reaction of easily accessible spiro-epoxyoxindole with heteroarenes and arenes. Our contin- uous research interest in exploring the reactivity of three mem- bered reactive intermediates[14] led us to envisage the Lewis acid catalysed F-C reaction of spiro-epoxyoxindoles with arenes as well as heteroarenes. After the affirmative presumption, re- cently, we have successfully reported the Lewis-acid catalysed efficient F-C reaction of spiro-epoxyoxindoles with indoles and arenes to obtain both the functionalities (Scheme 1).
Thus, spiro-epoxyoxindole has been emerged as a common precursor for the generation of all carbon quaternary centre prior to the synthesis of various indole alkaloids or their core structures. After this initial success, we are again exploring alter- native catalysts/solvents combination for the F-C reaction of spiro-epoxyoxindole. Eventually, we have observed that cata- lytic amount of Brønsted acid in organic solvent can also acti- vate the spiro-epoxide[16] and lead to regioselective ring open- ing at the tertiary centre by the carbon nucleophiles likely ind- oles and arenes. Moreover, we have recently reported catalyst free on-water ring opening reaction of spiro-aziridineoxindole and indole.[17] This result again encourages us to find an envi- ronment friendly greener approach for this ring-opening reac- tion. Herein we incorporated a detailed study of the Brønsted acid assisted regioselective F-C reaction of spiro-epoxides and comparison with the Lewis acid catalysed reactions along with its scopes and limitations.
Results and Discussion
As we mentioned earlier, epoxides are versatile and privileged framework to synthesis a various indole alkaloids with immense biological significance. Now regioselective ring opening at the tertiary centre of spiro-epoxides[15,18] with different carbon nu- cleophiles lead to the formation of all carbon quaternary centre. Friedel–Crafts reaction is one of such highly efficient atom-eco- nomic C-C bond forming reaction, although there are very few efficient approaches for intermolecular F-C reaction of epoxide.[19] Thus this method for the spiroepoxide demands thor- ough reconnaissance.
Reaction with Indoles
After initial success with Lewis acid, we tried to find out some other milder catalyst condition for this F-C reaction. During our research, we observed that the F-C reaction of spiro-epoxide was also proceeded well with the assistance of catalytic amount of Brønsted acid. To confirm it, we performed the model reac- tion between N-methyl spiro-epoxyoxindole 1a and indole em- ploying various Brønsted acids in different temperature. Our previous result with Lewis acid (Scheme 1) convinced us to per- form the reaction in DCE solvent. But unfortunately the reaction was not completed at 0 °C using 10 mol-% of ortho-phosphoric acid. Raising the temperature to 25 °C accomplished the F-C reaction with 81 % isolated yield of the regioselectively desired 3-(3-indolyl)-oxindole-3-methanol product (Table 1, entries 1 and 2). Next we changed the Brønsted acids to increase the yield of the desired product. But we were unfortunate to ac- quire significant amount of desired product with catalytic amount of benzoic acid, p-nitro benzoic acid, triflic acid (entries 3–5). Eventually, we were delighted to achieve the desired F-C product in presence of 10 mol-% of trifluoroacetic acid (TFA) with excellent isolated yield (92 %) within 6 h at 25 °C (entry 6). Further increase of the Brønsted acid did not affect the yield or time, on the contrary diminishing the amount of Brønsted acid decelerated the reaction with incomplete conversion. Thus 10 mol-% of TFA in DCE at 25 °C was the most accepted condition.
After successful optimisation, we wished to generalise the method. With this intention, we executed the F-C reaction with an assemblage of spiro-epoxyoxindoles and indoles. Almost all the cases, we achieved similar yield to that of our previously mentioned Lewis acid condition. Only electron withdrawing group on both spiro-epoxide as well as indole demanded more time for complete conversion (Scheme 2).
Not only unprotected indoles, we also performed the reac- tion with N-protected indoles (Scheme 3). With N-methyl as well as N-benzyl indoles, we obtained regioselectively desired prod- uct 3,3-bisindole along with 15–20 % of 3,2-bisindole as a side product. But when we performed the same reaction with elec- tron withdrawing protecting group, viz. N-benzoyl indole we gained exclusively C(3)-C(3) product with very good (78 % to 82 %) isolated yield.
Method B: Optimisation of on-Water Reaction
Water has a long history as a reaction medium in organic chem- istry.[20] As a solvent, it possesses a very interesting property due to its extensive H-bonding ability. According to Jung- Marcus theory,[21] approximately 25 % of water molecules hav- ing free OH groups at the interface are available for potential H-bonding with the substrate on the surface. Recently, we dis- covered catalyst-free on-water regio- and stereoselective ring opening of spiroaziridine oxindoles with indoles.[17] This led us to presume that water can also activate spiro-epoxyoxindole similar to that of spiro-aziridine. But the extent of H-bonding is less in case of spiro-epoxide compare to spiroaziridine (Scheme 4).
This attributed to the incomplete reaction, alongwith sub- stantial amount of yield (Table 2, entry 1). To increase the H- bonding activity, we then planned to use different Brønsted acids as a promoter. We initiated our inspection with the model substrate N-methyl spiro-epoxyoxindole 1a with indole using TFA as a promoter in aqueous medium. While 0.5 equiv. of TFA yielded 41 % yield at 50 °C, 1.0 equiv. of TFA afforded up to 50–52 % yield (entries 2–4).
Employing ortho-phosphoric acid instead of TFA did not in- crease the yield (entries 5, 6). On the contrary, more acidic triflic acid reduced the yield of the desired product due to C3-proto- nation of indole (entry 7). Next we applied relatively less acidic acetic acid which led to the clean reaction profile. With
0.5 equiv. of acetic acid at 50 °C rendered 42 % yield, while increasing the temperature as well as amount of acetic acid up to 1.0 equiv. improved the yield up to 68 % (entries 8–10).
In contrast, a large excess of acetic acid (water/acetic acid = 9:1) led to messy reaction with reduced yield (entry 11). Thus
1.0 equiv. of acetic acid in water at 80 °C was the most suitable condition here.
With these optimised conditions in hand, we desired to ex- tend the substrate scope for the Brønsted acid promoted on- water ring opening reaction of spiro-epoxyoxindoles with indoles (Scheme 5). All the electron donating as well as electron withdrawing groups of spiro-epoxyoxindoles were well-toler- ated under optimised condition. All underwent smooth reac- tion rendering the desired 3, 3-bisindoles with moderate yield. It was also observed that all the spiro-epoxides irrespective of substitution or protection required longer times for completion than the previously described methods. Due to the nucleophilic character of water, we obtained a minor amount (15–20 %) of water adduct product in all the cases. Along with a minute amount of acetate adduct (<10 %) was also detected during the course of the reaction. These unwanted side reactions de- creased the overall yield of the desired 3,3 bisindole methanols.
We also unveiled the F-C reaction of spiro-epoxyoxindole with 3-substituted indole which might provide the bisindole with vicinal all-carbon quaternary centers. Consequently, com- pound 1c was treated with 3-methyl indole 4a under Brønsted acid catalysed condition in organic solvent (DCE) (Scheme 6). Interestingly, it afforded tetrahydrospirofuro-bisindole 5a hav- ing vicinal all- carbon quaternary centers as a diastereomeric mixture along with 2,3-bisindole 5a. It seemed epoxide open- ing with 3-methyl indole followed by intramolecular cyclization of the intermediate imine X afforded the tricyclic tetrahydro- furoindole core. Next we also examined the similar kind of reac- tion between N-benzyl spiro-epoxide 1b and 3-methyl N- benzoyl indole 4b under the same Method A. Consequently, both the reactions manifested identical outcome.
The protocol was further elaborated towards the reaction of trisubstituted spiro-epoxyoxindole (±)-6 (Scheme 7), having two possible reactive sites. Brønsted acid mediated SN1/SN2 type epoxide ring opening is expected to occur at less substituted benzylic center compare to more hindered pseudobenzylic po- sition of oxindole unit; moreover, the generation of carbo- cationic nature at α-to carbonyl is very unlikely. To explore the reactivity, trisubstituted spiro-epoxyoxindole (±)-6 was subjected under optimised conditions (Method A). As expected, it afforded compound (±)-7, generated from the less substituted benzylic position attack by indole as a major one compared to the more hindered C3-attack (±)-7. The reaction was found to be very slow with incomplete conversion.
Reaction with Arenes
Method A: Optimisation with Brønsted Acids in Organic Solvent
As depicted earlier, intermolecular F-C reaction of benzenoid arenes[22] at the tertiary carbon forming all carbon quaternary centre is still a demanding task. In this regards, we visualized that the reaction of benzenoid arenes, particularly phenols with the spiro-epoxides can solve the issue. Moreover this efficient methodology provides a direct access to 3-aryloxindole- methanols which can be further manipulated to synthesise dif- ferent scaffolds with paramount significance. As discussed ear- lier, we also want to find out some lenient catalysts other than metal triflates to perform the F-C reaction of spiro-epoxyox- indole with arenes.
As executed earlier, we also performed the model reaction between N-methyl spiro-epoxyoxindole 1a and p-cresol 8a us- ing various Brønsted acids in different temperature in DCE (Table 3). At the outset, the reaction was not completed at 0 °C using 10 mol-% of orthophosphoric acid. Raising the tempera- ture to 25 °C, accomplished the regioselective F-C reaction with 90 % isolated yield of the desired product (Table 3, entries 1 and 2). Next we applied catalytic amount of benzoic acid, p- nitrobenzoic acid, triflic acid and acetic acid both in 0 °C as well as 25 °C, but ended up with unsatisfied results (entries 3–7). Ultimately, by applying 10 mol-% of trifluoroacetic acid (TFA) at 0 °C, we were able to isolate the desired 3-aryl-3-hydroxymethyl oxindole product 9a with excellent isolated yield (92 %) within 4 h (entry 9). Raising the temperature to 25 °C did not show any further improvement. Thus once again TFA was emerged to be the best Brønsted acid catalyst for both the F-C reaction of spiro-epoxides.
After fruitful Optimization, we wanted to unveil this method with a collection of spiro-epoxides and phenols (Scheme 8). In all cases, we obtained almost similar yield of the desired prod- uct to that of our previously reported Lewis acid catalysed re- sult.[15] Not only 4-substituted phenols but α-naphthol and re- sorcinol also rendered regioselectively desired ortho-arylated products. Moreover, we also performed the F-C reaction of spiro-epoxides with sesamol and 4-tBu phenol under this opti- mised Brønsted acid catalysed condition. These also afforded the regioselectively the desired products with high yields. The compound 9o, an advanced precursor, could be transformed to XEN907 in two steps.
We again continued the F-C reaction with electron rich benzenoid arenes like anisole, 1,2-dimethoxy, 1,3- dimethoxy, 1,4-dimethoxy, 1,2,3-trimethoxybenzenes, pyrrole, benzofuran in the presence of 10mol-% TFA (Figure 2). These arenes also reacted fluently under this condition to afford the regioselec- tively desired 3-aryl-3-hydroxymethyloxindoles with high yields. Whereas methoxyarenes bestowed exclusively 4-substituted products, other heteroarenes such as pyrrole, benzofuran re- acted through their usual reactive centres.
We further carried out the F-C reaction with 4-unsubstituted phenol, m-cresol and o-cresol (Scheme 9). Unlikely the Lewis acid condition, Brønsted acid catalysed F-C reactions provided the consistent para-arylated products with respect to hydroxyl group of phenols. This might be due to the fact, Lewis acid chelated between the phenol and spiro-epoxide (TS1; Scheme 10), hence phenol reacted through the proximal ortho- position, but the result was inconsistent with the change of the protecting group as well as the substitution on spiro-epoxides. In contrast, Brønsted acid can only activate the spiro-epoxide 1 by protonation (Scheme 8), thus phenol reacts with this acti- vated spiro-epoxide through the sterically less hindered para- position (TS2). Carbonyl group of carboxylic acid may act as a Lewis base, thus it might simultaneously activate both the spiro-epoxide and phenol through proton donation and proton acceptor, respectively. The phenol might, therefore, prefer to attack through para-position via macrocycle transition state TS3 (Scheme 11). The consistent result also ratified our assumption.
Method B: Optimisation of on-Water Reaction
As depicted earlier, we were again in search of environmentally sound condition for regioselective ring opening reaction of spiro- epoxyoxindole with arene.
In this regard, water has been considered as an advanta- geous solvent not only for its benign nature but also for its extensive H-bonding property. To the outset, we executed the ring opening reaction only in water but we were ended up with unsatisfactory result (Table 4, entry 1). We then anticipated that addition of different Brønsted acids along with water may in- crease the H-bonding capacity. We thus began our journey with the model substrate N-methyl spiro-epoxyoxindole with p- cresol employing TFA as a promoter in water. While 0.5 equiv. of TFA provided 50 % yield at 50 °C, 1 equiv. of TFA afforded up to 62 % yield (Table 4; entries 2, 3). Using ortho-phosphoric acid as well as highly acidic triflic acid did not increase the yield (entries 4, 5). Finally, we were privileged enough to apply relatively less acidic acetic acid which led to the clean reaction profile. While 0.5 equiv. acetic acid rendered 52 % yield, increas- ing the amount of acetic acid up to 1 equiv. improved the yield up to 79 % (entries 6, 7). On the other hand, large excess of acetic acid (water/acetic acid = 9:1) led to uncleaned reaction with less yield (entry 8). Thus 1 equiv. acetic acid in water at 50 °C was the most acceptable condition for water mediated Brønsted acid promoted F-C reaction of spiro-epoxides with arenes.
On the basis of the above stated optimisation, we wanted to extend the substrate scope with a collection of spiro-epoxyox- indoles and phenols (Scheme 12). All the reactions underwent in a smooth way under this optimized conditions irrespective of the protecting groups.
Moreover, electron donating as well as electron withdrawing substituents on spiro-epoxyoxindole did not impede the F-C reaction. In all cases good to moderate yield of the regioselec- tively desired ortho-arylated products was gained. All the spiro- epoxides took longer reaction times for completion than the method A. Unlike on-water reaction of indole, we did not ob- served any water adduct or acetate adduct. This might be due to the fact, these reactions required relatively lesser time in contrast to the previously described reactions with indoles.
Moreover, we again examined the F-C reaction with simple phenol under this condition. As expected, this results also mani- fested the consistent para-arylated product with respect to hydroxyl group of phenol. Here only these reactions took longer time than that of method A; while the yield was almost similar. Furthermore, the F-C reaction with phenol was elaborated with trisubstituted spiro-epoxyoxindole (±)-6 (Scheme 13), as we performed earlier with indole. Brønsted acid in organic sol- vent condition (Method A) was tested for the reaction. Unlike the reaction of indole (Scheme 5), here we ended up with an unexpected 3-aryl oxindole product 13. Possibly, the opening product 3-hydroxymethyl-3-aryl oxindole 12 underwent retro aldol kind of reaction to afford this interesting product 13.
2-Amino-5-chloropyridineCatalog No.:AA003310 CAS No.:1072-98-6 MDL No.:MFCD00006324 MF:C5H5ClN2 MW:128.5596 |
5-Fluoro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1h-indoleCatalog No.:AA0094QF CAS No.:1072009-08-5 MDL No.:MFCD16987816 MF:C14H17BFNO2 MW:261.0997 |
Benzyl n-methyl-n-(2-oxoethyl)carbamateCatalog No.:AA007T60 CAS No.:107201-33-2 MDL No.:MFCD14635609 MF:C11H13NO3 MW:207.2258 |
(R)-3-Aminotetrahydrofuran, HClCatalog No.:AA0039BC CAS No.:1072015-52-1 MDL No.:MFCD08461720 MF:C4H10ClNO MW:123.5813 |
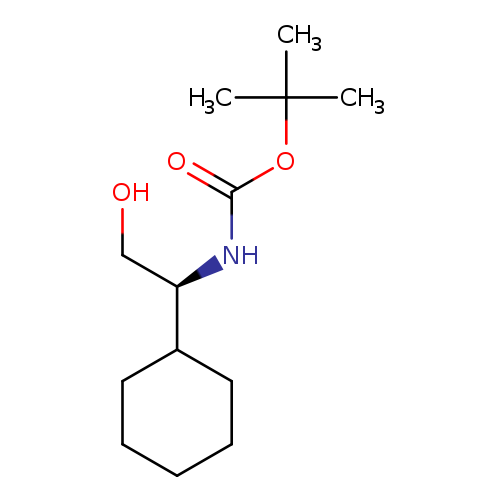
N-Boc-l-cyclohexylglycinolCatalog No.:AA003SR8 CAS No.:107202-39-1 MDL No.:MFCD04112591 MF:C13H25NO3 MW:243.3425 |
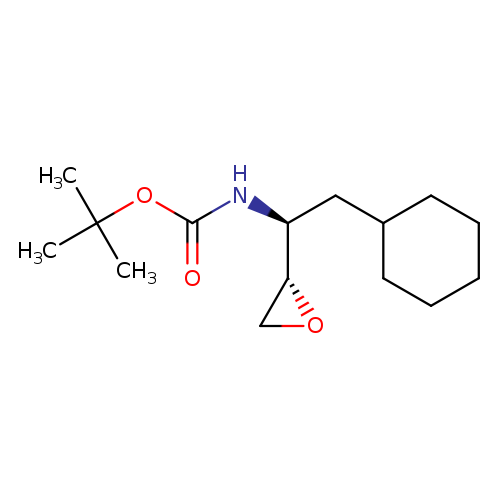
Erythro-n-boc-l-cyclohexylalanine epoxideCatalog No.:AA008R2J CAS No.:107202-62-0 MDL No.:MFCD08061628 MF:C15H27NO3 MW:269.3798 |
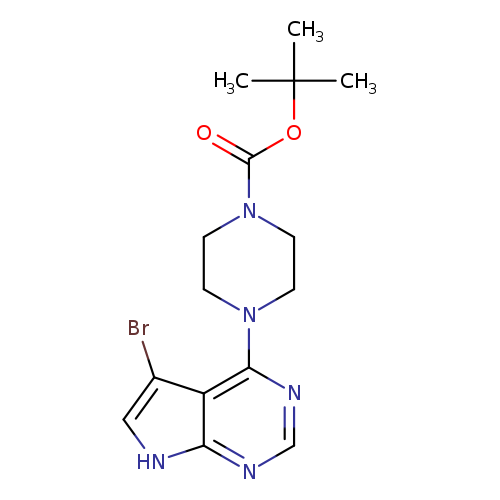
4-(4-Boc-1-piperazinyl)-5-bromo-7h-pyrrolo[2,3-d]pyrimidineCatalog No.:AA003JZG CAS No.:1072027-36-1 MDL No.:MFCD18642459 MF:C15H20BrN5O2 MW:382.2556 |
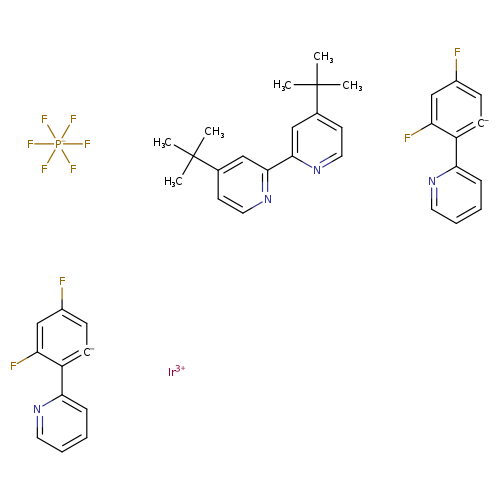
iridium(3+) ion bis(3,5-difluoro-2-(pyridin-2-yl)benzen-1-ide) 4-tert-butyl-2-(4-tert-butylpyridin-2-yl)pyridine hexafluoro-lambda5-phosphanuideCatalog No.:AA01EIFJ CAS No.:1072067-44-7 MDL No.:MFCD31657504 MF:C40H36F10IrN4P MW:985.9154 |
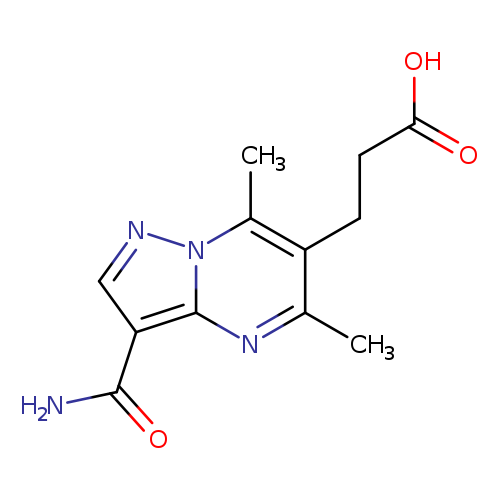
3-{3-carbamoyl-5,7-dimethylpyrazolo[1,5-a]pyrimidin-6-yl}propanoic acidCatalog No.:AA019UQC CAS No.:1072069-75-0 MDL No.:MFCD11857944 MF:C12H14N4O3 MW:262.2646 |
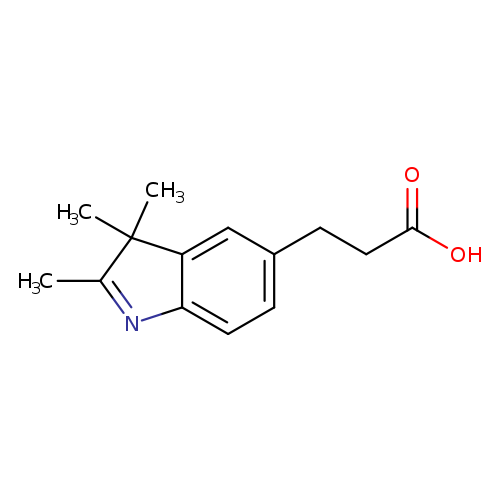
3-(2,3,3-trimethyl-3H-indol-5-yl)propanoic acidCatalog No.:AA00906R CAS No.:1072069-91-0 MDL No.:MFCD12922972 MF:C14H17NO2 MW:231.2903 |
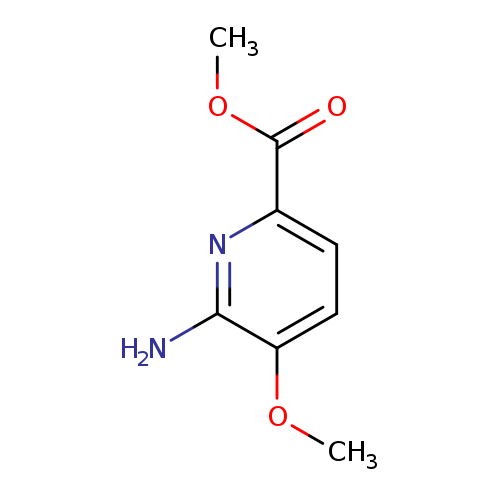
6-Amino-5-methoxy-pyridine-2-carboxylic acid methyl esterCatalog No.:AA0093KM CAS No.:1072077-54-3 MDL No.:MFCD18261109 MF:C8H10N2O3 MW:182.1766 |
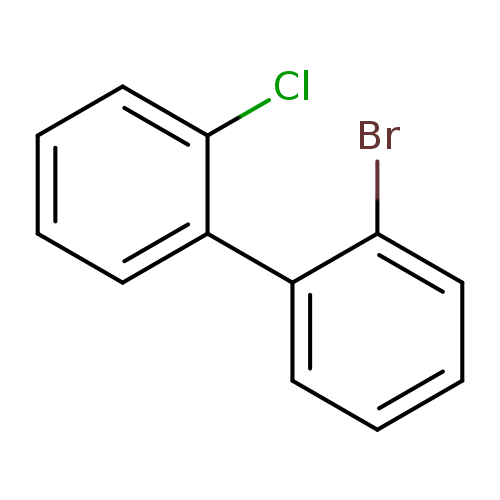
2-bromo-2-chloro-1,1-biphenylCatalog No.:AA019EQZ CAS No.:107208-70-8 MDL No.:MFCD00466291 MF:C12H8BrCl MW:267.5489 |
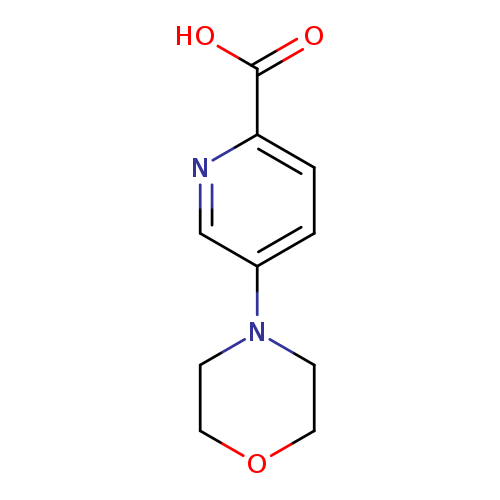
5-Morpholinopicolinic acidCatalog No.:AA00995G CAS No.:1072103-29-7 MDL No.:MFCD18251373 MF:C10H12N2O3 MW:208.2139 |
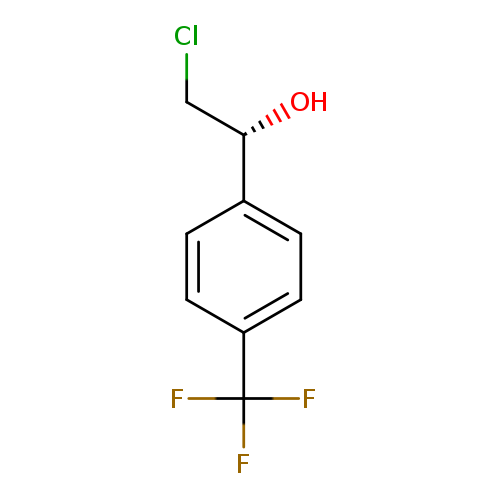
(1R)-2-chloro-1-[4-(trifluoromethyl)phenyl]ethan-1-olCatalog No.:AA01BCL2 CAS No.:1072105-58-8 MDL No.:MFCD24346567 MF:C9H8ClF3O MW:224.6074 |
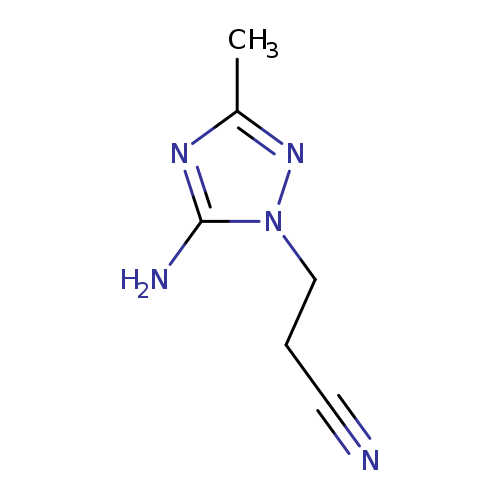
3-(5-AMINO-3-METHYL-1H-1,2,4-TRIAZOL-1-YL)PROPANENITRILECatalog No.:AA01EJML CAS No.:1072106-44-5 MDL No.:MFCD16845192 MF:C6H9N5 MW:151.1692 |
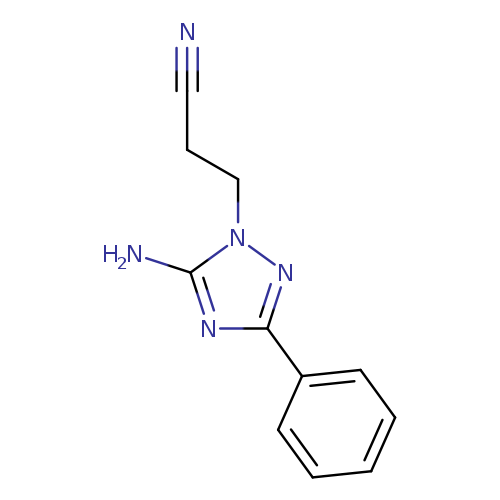
3-(5-amino-3-phenyl-1H-1,2,4-triazol-1-yl)propanenitrileCatalog No.:AA01EJQL CAS No.:1072106-46-7 MDL No.:MFCD24641918 MF:C11H11N5 MW:213.2385 |
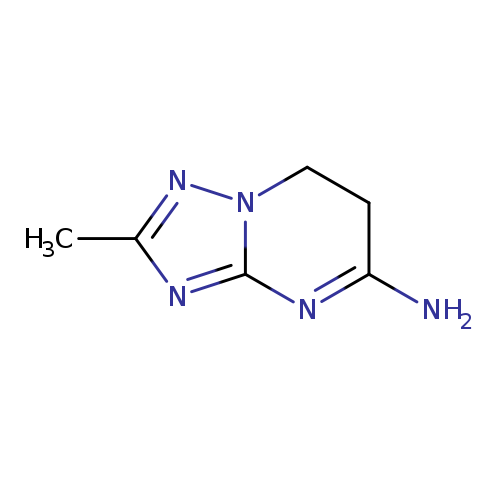
2-METHYL-6H,7H-[1,2,4]TRIAZOLO[1,5-A]PYRIMIDIN-5-AMINECatalog No.:AA01ELLE CAS No.:1072106-47-8 MDL No.:MFCD24641915 MF:C6H9N5 MW:151.1692 |
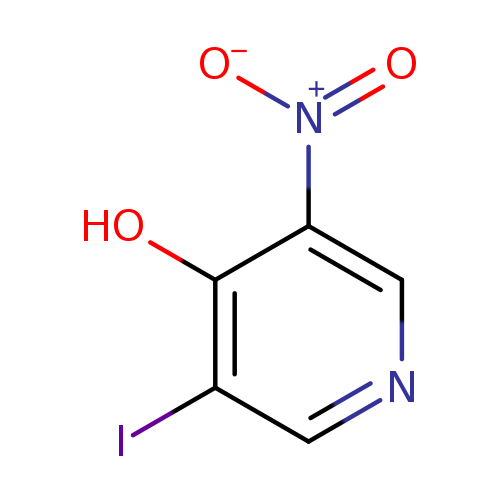
3-Iodo-5-nitropyridin-4-olCatalog No.:AA008SAD CAS No.:1072140-97-6 MDL No.:MFCD11052833 MF:C5H3IN2O3 MW:265.9934 |
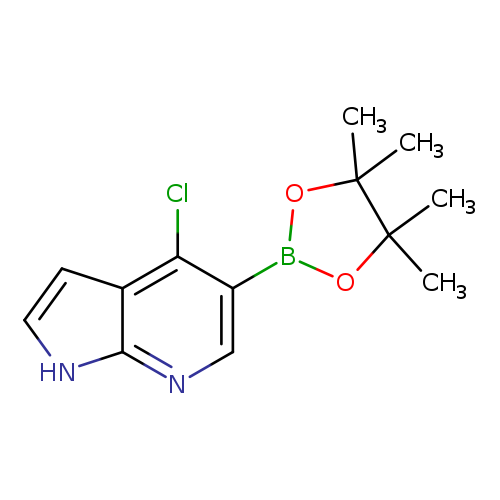
4-Chloro-1h-pyrrolo[2,3-b]pyridine-5-boronic acid pinacol esterCatalog No.:AA008U3S CAS No.:1072145-24-4 MDL No.:MFCD09965897 MF:C13H16BClN2O2 MW:278.5423 |
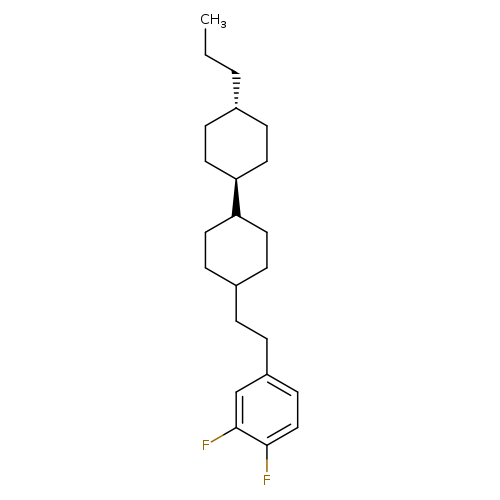
1,2-Difluoro-4-[2-[(trans,trans)-4'-propyl[1,1'-bicyclohexyl]-4-yl]ethyl]benzeneCatalog No.:AA0094QB CAS No.:107215-66-7 MDL No.:MFCD28963786 MF:C23H34F2 MW:348.5129 |
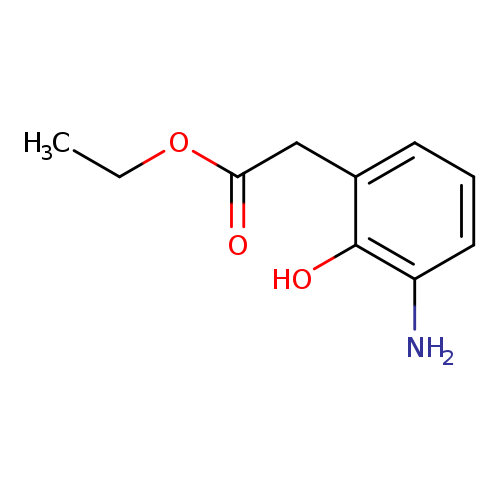
Ethyl 2-(3-amino-2-hydroxyphenyl)acetateCatalog No.:AA01B8DM CAS No.:1072150-23-2 MDL No.:MFCD24642393 MF:C10H13NO3 MW:195.2151 |
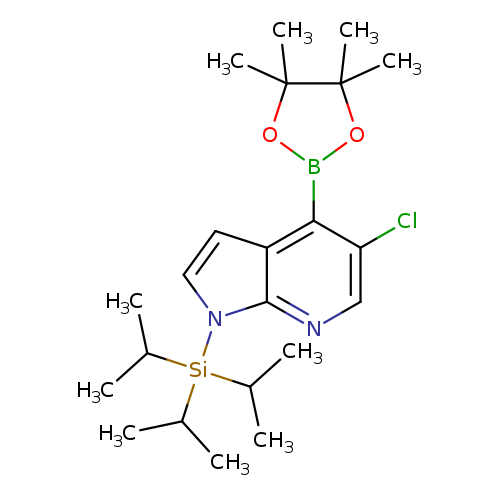
5-Chloro-1-(triisopropylsilyl)-1h-pyrrolo[2,3-b]pyridine-4-boronic acid pinacol esterCatalog No.:AA007ARF CAS No.:1072152-34-1 MDL No.:MFCD09965900 MF:C22H36BClN2O2Si MW:434.8829 |
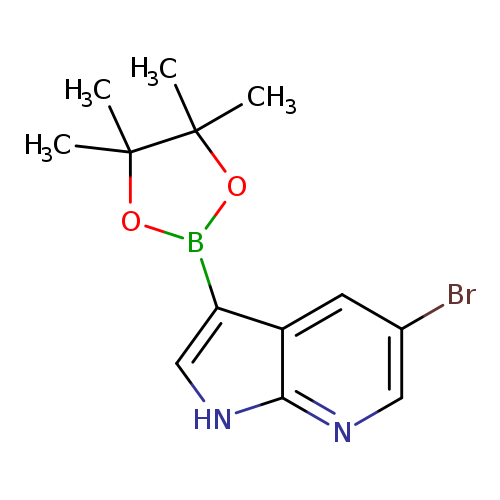
5-Bromo-3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1h-pyrrolo[2,3-b]pyridineCatalog No.:AA0094GZ CAS No.:1072152-50-1 MDL No.:MFCD10574985 MF:C13H16BBrN2O2 MW:322.9933 |
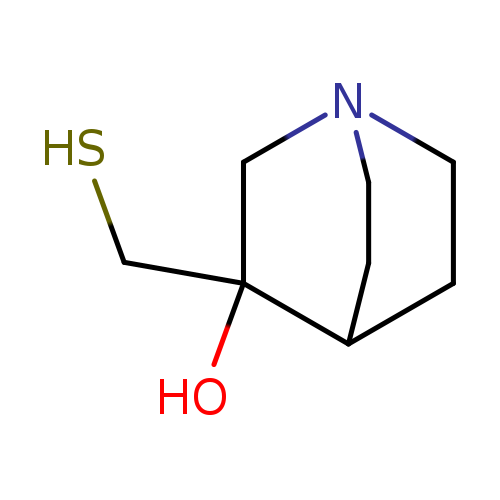
rac3-Hydroxy-3-mercaptomethylquinuclidineCatalog No.:AA01DZFL CAS No.:107220-26-8 MDL No.:MFCD11042346 MF:C8H15NOS MW:173.2758 |
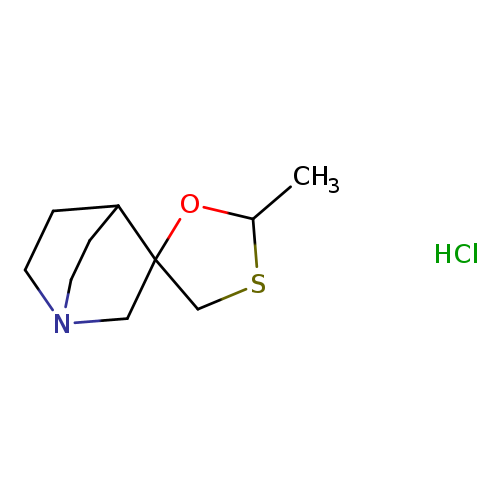
Cevimeline hydrochlorideCatalog No.:AA007ARD CAS No.:107220-28-0 MDL No.:MFCD01961045 MF:C10H18ClNOS MW:235.7740 |
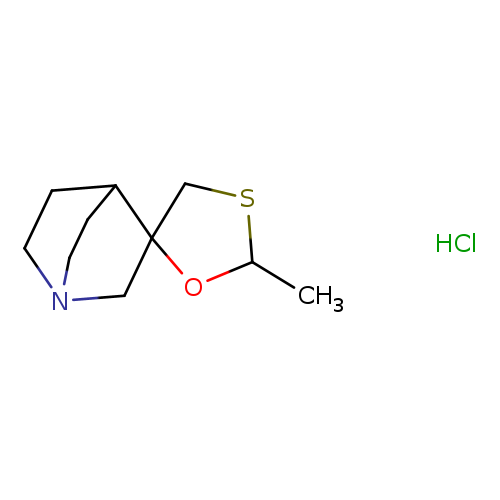
trans-CevimelineHydrochlorideCatalog No.:AA01DZFM CAS No.:107220-29-1 MDL No.:MFCD01961046 MF:C10H18ClNOS MW:235.7740 |
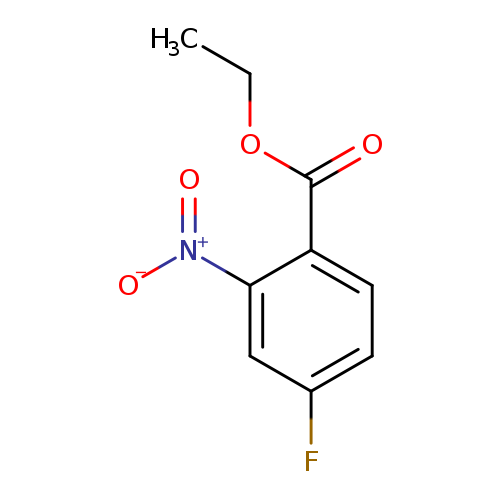
Ethyl 4-fluoro-2-nitrobenzoateCatalog No.:AA009NXM CAS No.:1072207-10-3 MDL No.:MFCD10566267 MF:C9H8FNO4 MW:213.1625 |
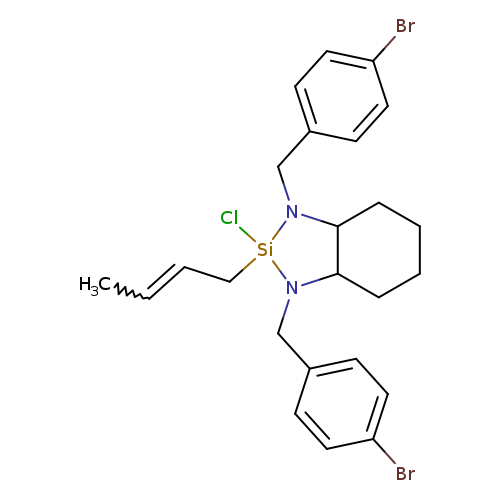
(S,S)-1,3-Bis(4-bromobenzyl)-2-chlorooctahydro-2-(2E)-crotyl-1H-1,3,2-benzodiazasiloleCatalog No.:AA003CM4 CAS No.:1072220-37-1 MDL No.:MFCD18632567 MF:C24H29Br2ClN2Si MW:568.8470 |
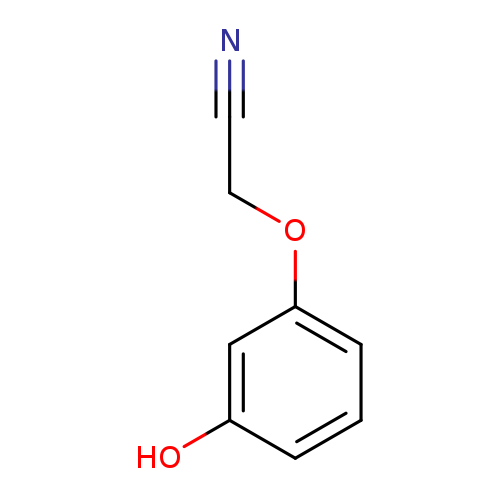
2-(3-Hydroxyphenoxy)acetonitrileCatalog No.:AA00HAVG CAS No.:107223-69-8 MDL No.:MFCD11181964 MF:C8H7NO2 MW:149.1467 |
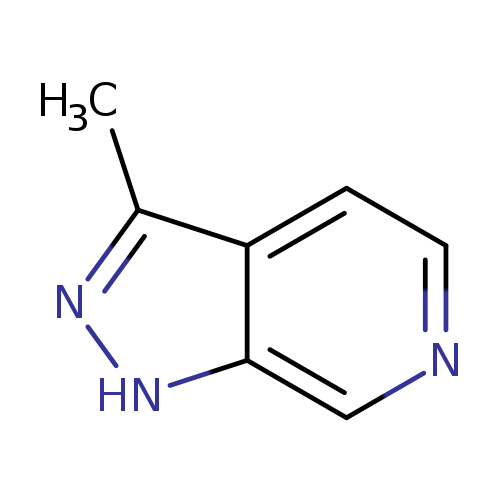
3-Methyl-1H-pyrazolo[3,4-c]pyridineCatalog No.:AA0082CB CAS No.:1072249-89-8 MDL No.:MFCD13191779 MF:C7H7N3 MW:133.1506 |
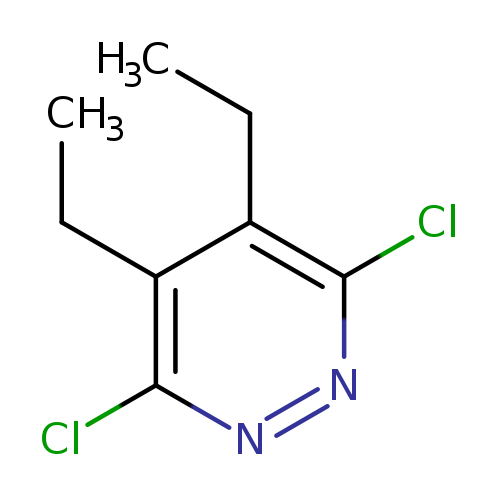
3,6-Dichloro-4,5-diethylpyridazineCatalog No.:AA007AR7 CAS No.:107228-53-5 MDL No.:MFCD17014878 MF:C8H10Cl2N2 MW:205.0844 |
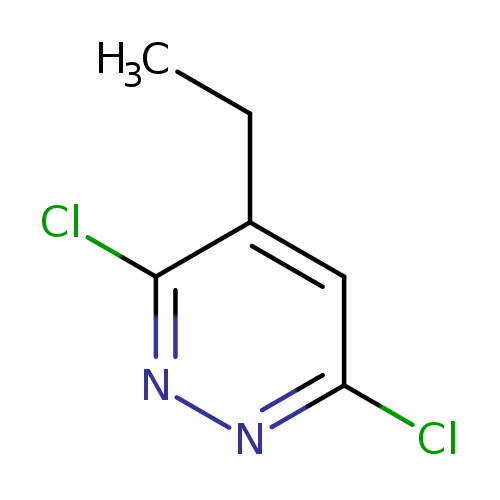
3,6-Dichloro-4-ethylpyridazineCatalog No.:AA0037VU CAS No.:107228-54-6 MDL No.:MFCD17014877 MF:C6H6Cl2N2 MW:177.0312 |
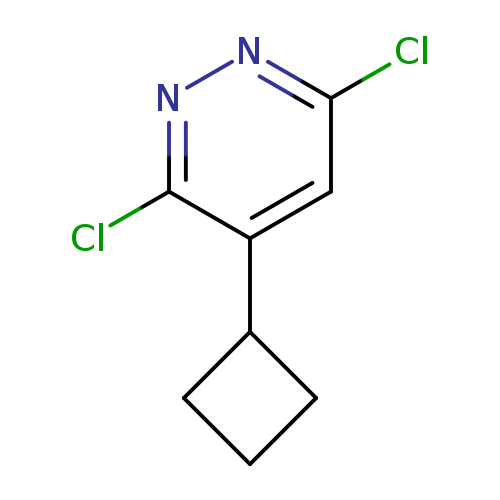
3,6-Dichloro-4-cyclobutylpyridazineCatalog No.:AA00HAVH CAS No.:107228-57-9 MDL No.:MFCD25955161 MF:C8H8Cl2N2 MW:203.0685 |
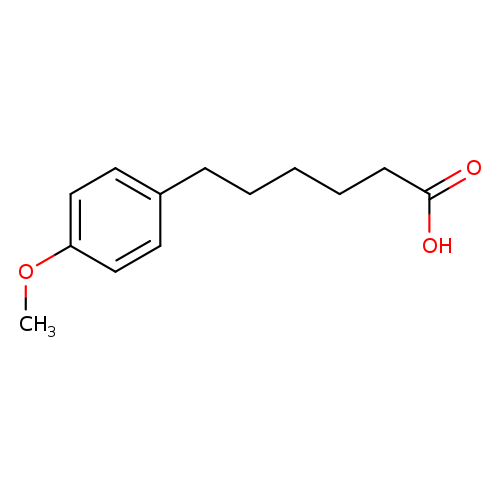
6-(4-Methoxyphenyl)hexanoic acidCatalog No.:AA01AA0M CAS No.:107228-87-5 MDL No.:MFCD12172613 MF:C13H18O3 MW:222.2802 |
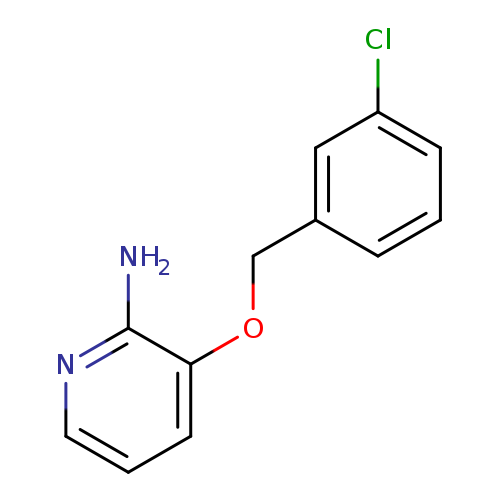
2-Pyridinamine, 3-[(3-chlorophenyl)methoxy]-Catalog No.:AA0082C5 CAS No.:107229-62-9 MDL No.:MFCD11195108 MF:C12H11ClN2O MW:234.6815 |
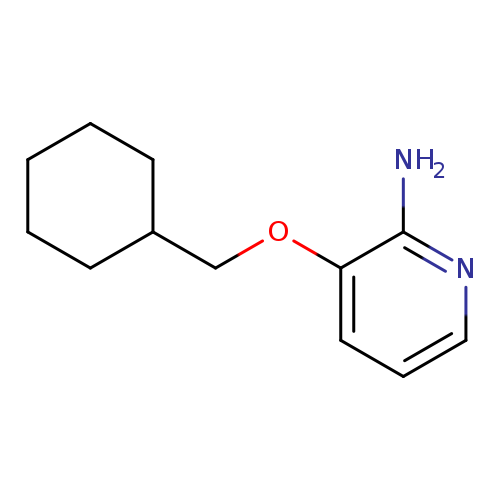
3-(cyclohexylmethoxy)pyridin-2-amineCatalog No.:AA01A8DY CAS No.:107229-69-6 MDL No.:MFCD10689932 MF:C12H18N2O MW:206.2841 |
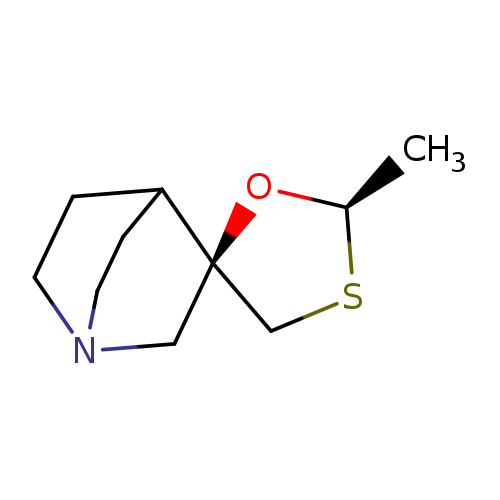
CevimelineCatalog No.:AA0082C4 CAS No.:107233-08-9 MDL No.:MFCD22577792 MF:C10H17NOS MW:199.3131 |
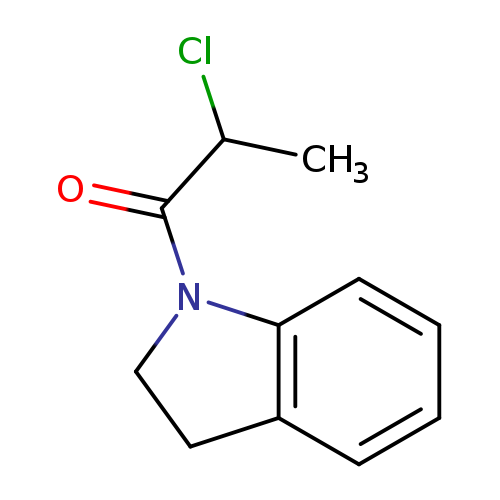
1-(2-Chloropropanoyl)indolineCatalog No.:AA009PNU CAS No.:107236-27-1 MDL No.:MFCD05263162 MF:C11H12ClNO MW:209.6721 |
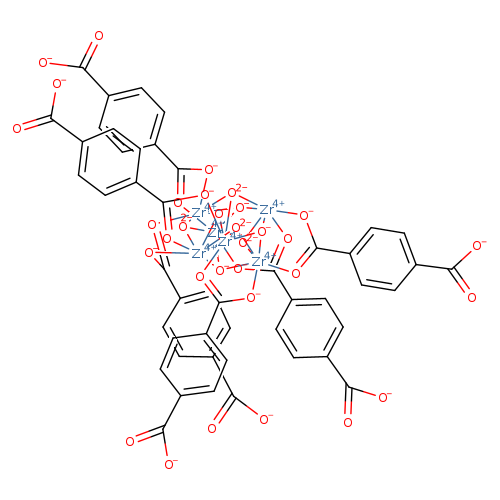
Zirconium 1,4-dicarboxybenzene MOF (UiO-66)Catalog No.:AA01FOVW CAS No.:1072413-89-8 MDL No.:MFCD29037163 MF:C48H24O32Zr6 MW:1660.0290 |
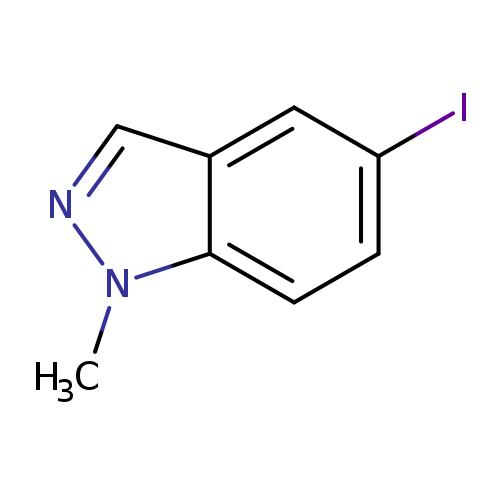
5-Iodo-1-methyl-1h-indazoleCatalog No.:AA007AR0 CAS No.:1072433-59-0 MDL No.:MFCD11977535 MF:C8H7IN2 MW:258.0590 |
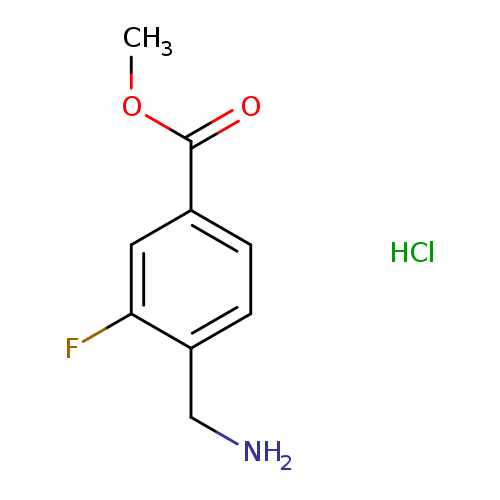
methyl 4-(aminomethyl)-3-fluorobenzoate hydrochlorideCatalog No.:AA01B6DP CAS No.:1072438-51-7 MDL No.:MFCD17171306 MF:C9H11ClFNO2 MW:219.6405 |
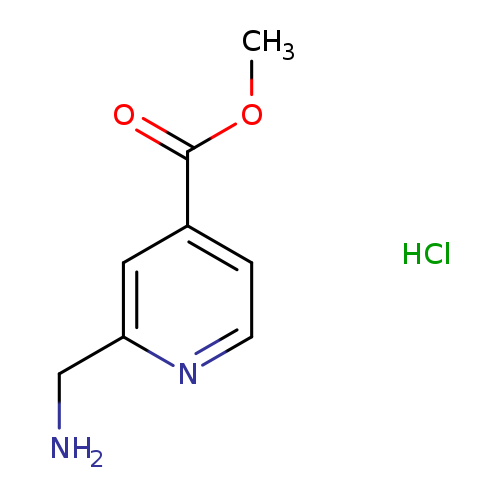
Methyl 2-(aminomethyl)pyridine-4-carboxylate, HClCatalog No.:AA007T5E CAS No.:1072438-54-0 MDL No.:MFCD09878665 MF:C8H11ClN2O2 MW:202.6381 |
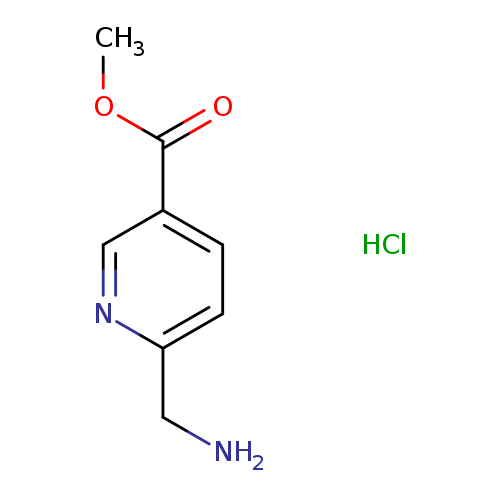
Methyl 6-(aminomethyl)nicotinate hydrochlorideCatalog No.:AA008ZD7 CAS No.:1072438-56-2 MDL No.:MFCD11045450 MF:C8H11ClN2O2 MW:202.6381 |
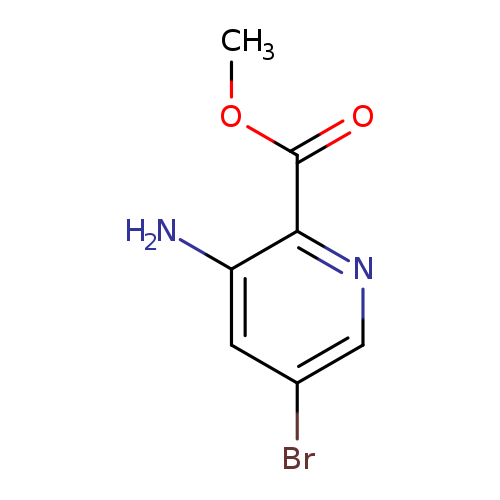
Methyl 3-amino-5-bromopicolinateCatalog No.:AA008U25 CAS No.:1072448-08-8 MDL No.:MFCD17015929 MF:C7H7BrN2O2 MW:231.0467 |
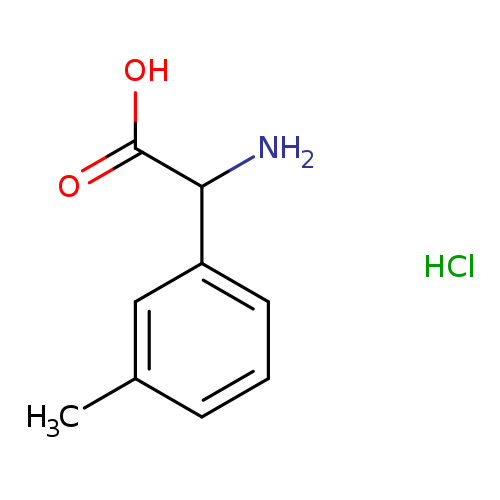
2-Amino-2-(3-methylphenyl)acetic acid HClCatalog No.:AA0082C0 CAS No.:1072449-62-7 MDL No.:MFCD06410554 MF:C9H12ClNO2 MW:201.6501 |
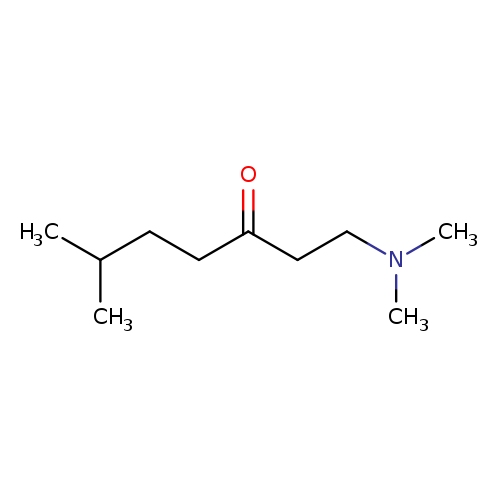
1-(Dimethylamino)-6-methylheptan-3-oneCatalog No.:AA00HAVI CAS No.:107245-26-1 MDL No.:MFCD22683883 MF:C10H21NO MW:171.2798 |

monoMETHACRYLOXYPROPYL FUNCTIONAL POLYTRIFLUOROPROPYLMETHYLSILOXANE, symmetric, 50-70 cStCatalog No.:AA01EIBU CAS No.:1072456-00-8 MDL No.: MF: MW: |
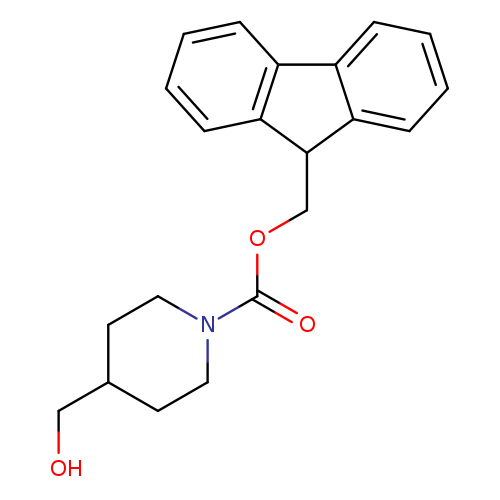
1-Fmoc-4-(hydroxymethyl)piperidineCatalog No.:AA00995K CAS No.:1072502-03-4 MDL No.:MFCD02094489 MF:C21H23NO3 MW:337.4122 |
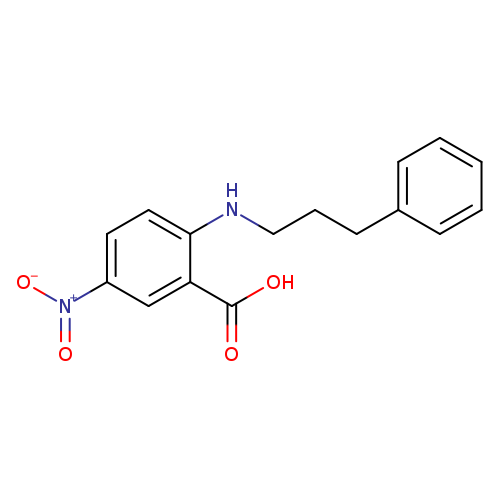
5-Nitro-2-((3-phenylpropyl)amino)benzoic acidCatalog No.:AA00385A CAS No.:107254-86-4 MDL No.:MFCD00153851 MF:C16H16N2O4 MW:300.3092 |
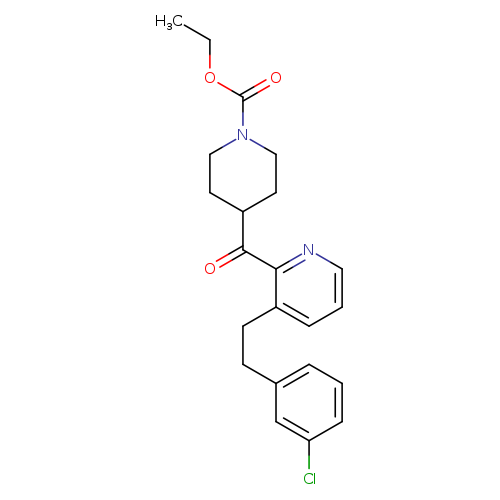
4-[[3-[2-(3-Chlorophenyl)ethyl]-2-pyridinyl]carbonyl]-1-piperidinecarboxylic AcidCatalog No.:AA008W2U CAS No.:107256-32-6 MDL No.:MFCD16251304 MF:C22H25ClN2O3 MW:400.8985 |
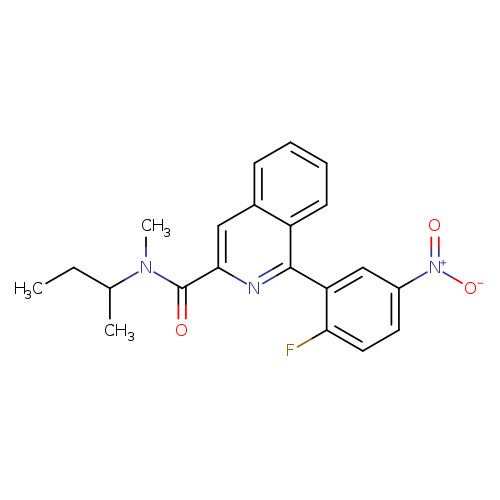
Pk14105Catalog No.:AA003TQE CAS No.:107257-28-3 MDL No.:MFCD28969041 MF:C21H20FN3O3 MW:381.4002 |
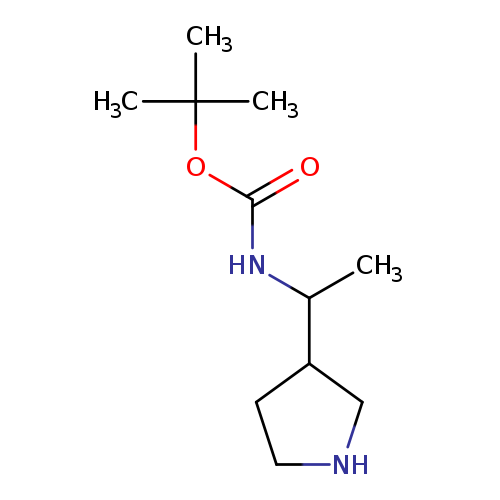
3-(1-(Boc-amino)ethyl)-pyrrolidineCatalog No.:AA00HAVK CAS No.:107258-90-2 MDL No.:MFCD18073167 MF:C11H22N2O2 MW:214.3046 |
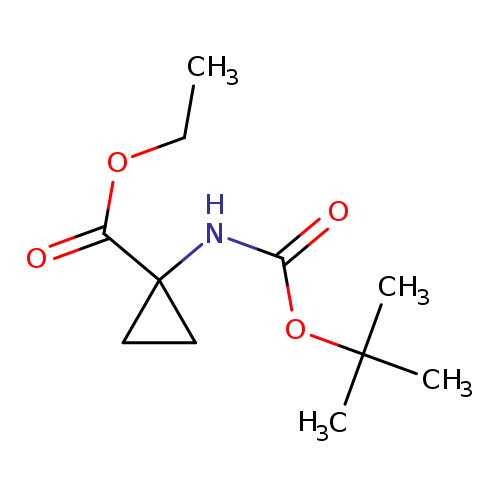
Ethyl 1-(boc-amino)cyclopropanecarboxylateCatalog No.:AA003PXD CAS No.:107259-05-2 MDL No.:MFCD11845623 MF:C11H19NO4 MW:229.2729 |
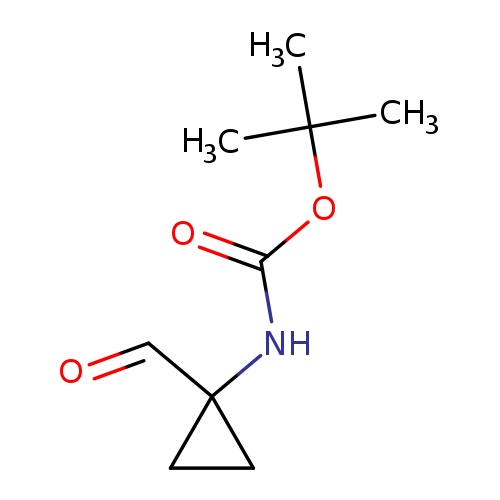
(1-Formyl-cyclopropyl)-carbamic acid tert-butyl esterCatalog No.:AA0037VS CAS No.:107259-06-3 MDL No.:MFCD10697899 MF:C9H15NO3 MW:185.2203 |
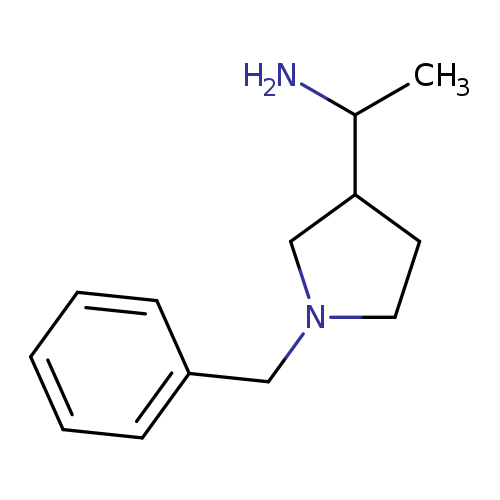
1-(1-Benzylpyrrolidin-3-yl)ethanamineCatalog No.:AA01A52R CAS No.:107259-16-5 MDL No.:MFCD22057269 MF:C13H20N2 MW:204.3113 |
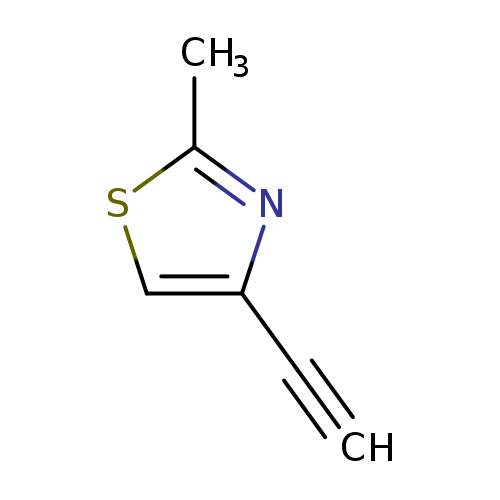
4-Ethynyl-2-methylthiazoleCatalog No.:AA008U6X CAS No.:107263-89-8 MDL No.:MFCD18207414 MF:C6H5NS MW:123.1756 |
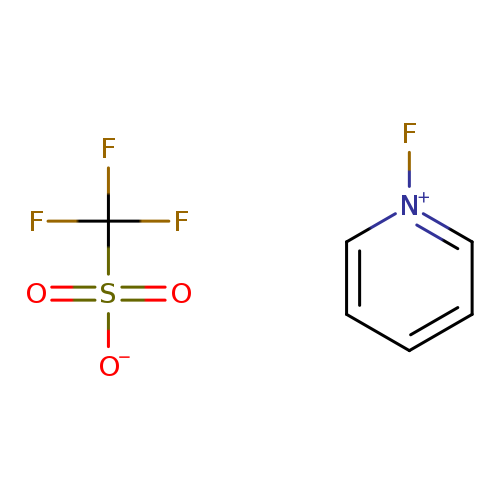
1-Fluoropyridinium TriflateCatalog No.:AA003AZ0 CAS No.:107263-95-6 MDL No.:MFCD00013458 MF:C6H5F4NO3S MW:247.1674 |
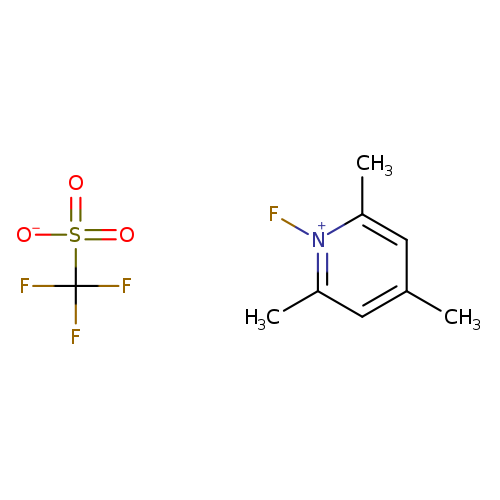
1-Fluoro-2,4,6-trimethylpyridin-1-ium trifluoromethanesulfonateCatalog No.:AA003E92 CAS No.:107264-00-6 MDL No.:MFCD00067525 MF:C9H11F4NO3S MW:289.2472 |
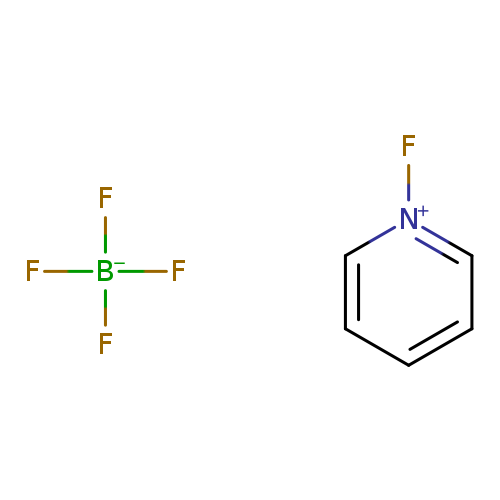
1-Fluoropyridinium tetrafluoroborateCatalog No.:AA003E9M CAS No.:107264-09-5 MDL No.:MFCD00153176 MF:C5H5BF5N MW:184.9029 |
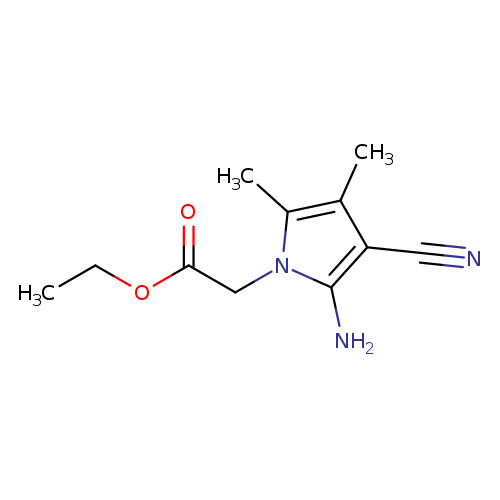
ethyl 2-(2-amino-3-cyano-4,5-dimethyl-1H-pyrrol-1-yl)acetateCatalog No.:AA00IYYI CAS No.:107267-88-9 MDL No.:MFCD04126142 MF:C11H15N3O2 MW:221.2557 |
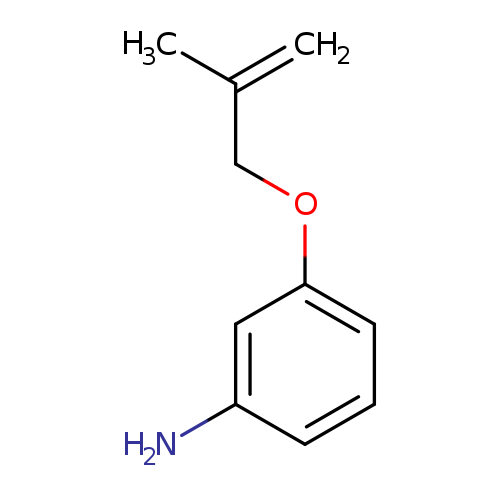
3-((2-Methylallyl)oxy)anilineCatalog No.:AA0082BS CAS No.:107268-37-1 MDL No.:MFCD06801092 MF:C10H13NO MW:163.2163 |
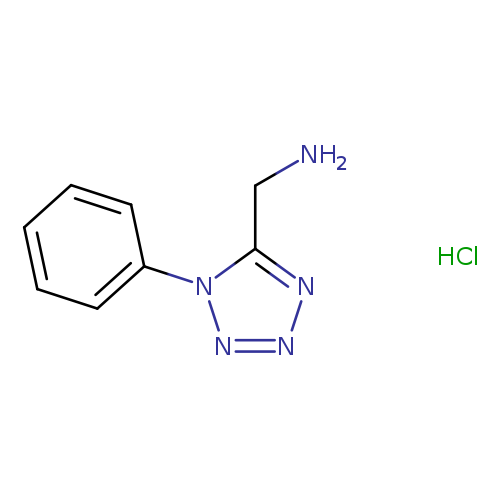
IFLAB-BB F1703-0109Catalog No.:AA008X8V CAS No.:107269-65-8 MDL No.:MFCD06623794 MF:C8H10ClN5 MW:211.6515 |
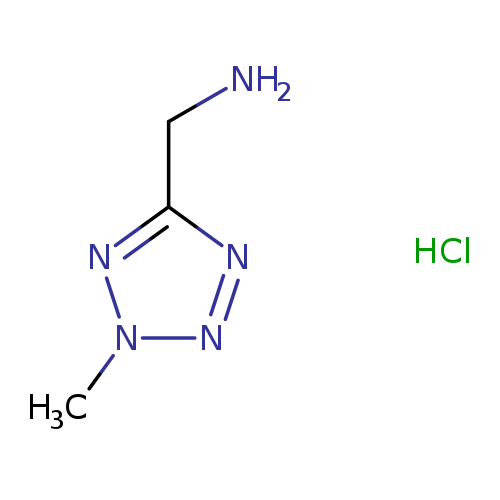
(2-methyl-2H-1,2,3,4-tetrazol-5-yl)methanamine hydrochlorideCatalog No.:AA01BFEC CAS No.:107269-68-1 MDL No.:MFCD28118594 MF:C3H8ClN5 MW:149.5821 |
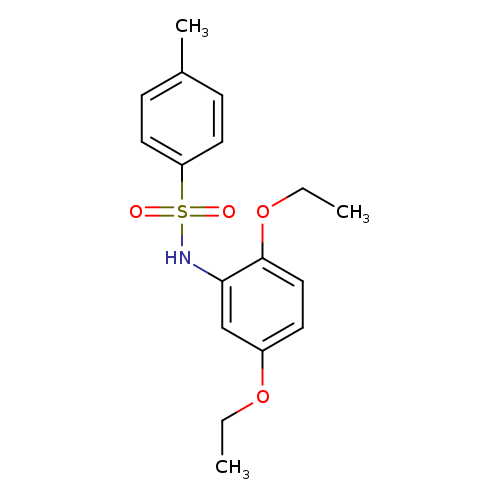
N-(2,5-Diethoxyphenyl)-4-methyl-benzenesulfonamideCatalog No.:AA019EHW CAS No.:107276-46-0 MDL No.:MFCD10170348 MF:C17H21NO4S MW:335.4179 |
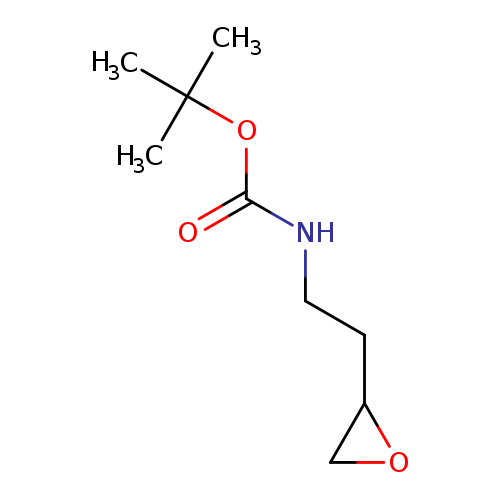
tert-Butyl n-[2-(oxiran-2-yl)ethyl]carbamateCatalog No.:AA01BCJZ CAS No.:1072793-83-9 MDL No.:MFCD24465622 MF:C9H17NO3 MW:187.2362 |
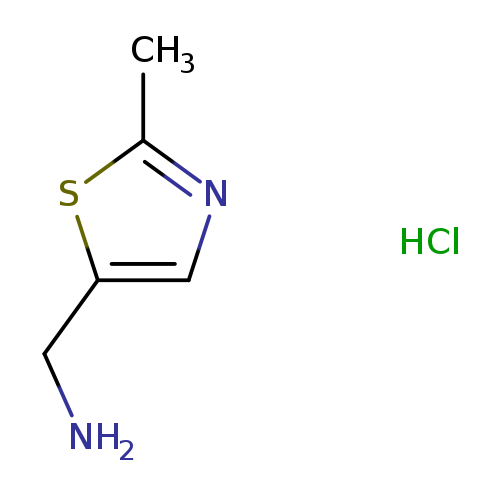
(2-Methylthiazol-5-yl)methanamine hydrochlorideCatalog No.:AA00HAVO CAS No.:1072806-59-7 MDL No.:MFCD11506341 MF:C5H9ClN2S MW:164.6564 |
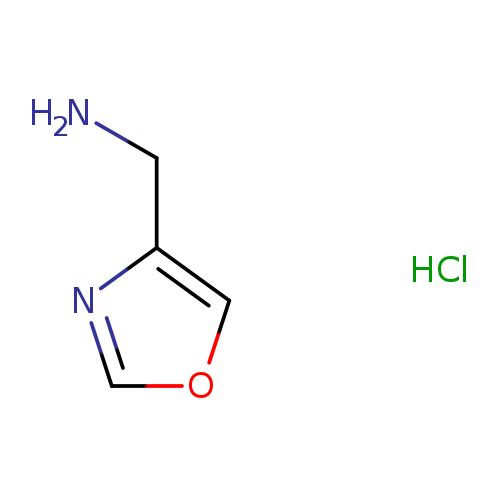
4-Oxazolemethanamine hydrochlorideCatalog No.:AA0038W5 CAS No.:1072806-60-0 MDL No.:MFCD08437648 MF:C4H7ClN2O MW:134.5642 |
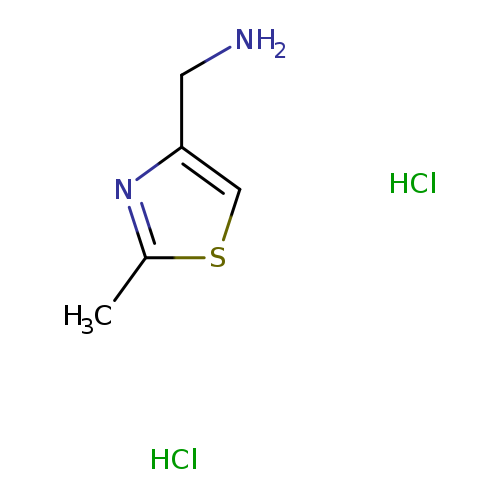
(2-Methylthiazol-4-yl)methanamine dihydrochlorideCatalog No.:AA0092J6 CAS No.:1072806-63-3 MDL No.:MFCD11506341 MF:C5H10Cl2N2S MW:201.1173 |
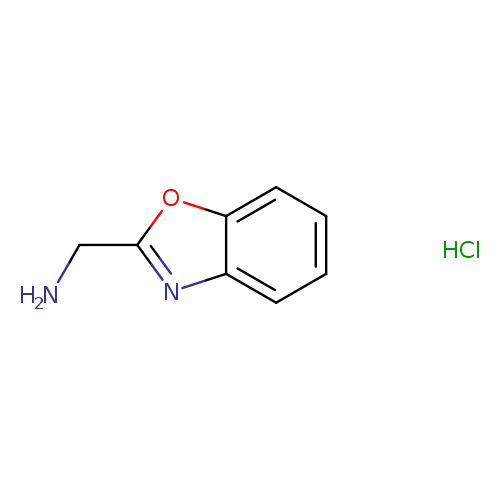
1,3-Benzoxazol-2-ylmethylamine hydrochlorideCatalog No.:AA008U23 CAS No.:1072806-65-5 MDL No.:MFCD09763577 MF:C8H9ClN2O MW:184.6229 |
2-(Aminomethyl)-1h-indole methanesulphonateCatalog No.:AA00982T CAS No.:1072806-66-6 MDL No.:MFCD03756929 MF:C10H14N2O3S MW:242.2948 |
4-Methoxyindole-2-boronic acid pinacol esterCatalog No.:AA007AQU CAS No.:1072811-21-2 MDL No.:MFCD11858387 MF:C15H20BNO3 MW:273.1352 |
4-Methylindole-2-boronic acid pinacol esterCatalog No.:AA0090CW CAS No.:1072811-23-4 MDL No.:MFCD11858397 MF:C15H20BNO2 MW:257.1358 |
4-Methoxycarbonylindole-2-boronic acid pinacol esterCatalog No.:AA007T51 CAS No.:1072811-67-6 MDL No.:MFCD11858381 MF:C16H20BNO4 MW:301.1453 |
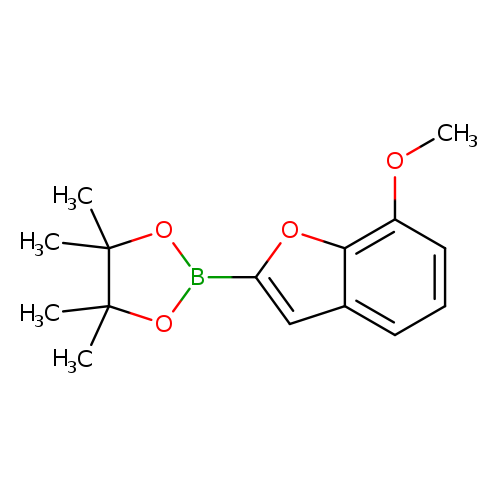
2-(7-Methoxybenzofuran-2-yl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolaneCatalog No.:AA00HAVU CAS No.:1072811-86-9 MDL No.:MFCD18383011 MF:C15H19BO4 MW:274.1200 |
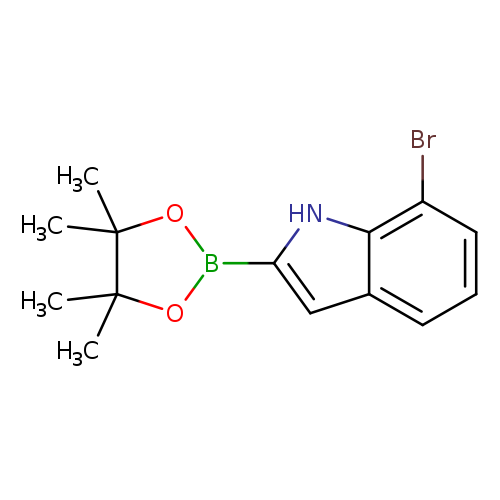
7-Bromoindole-2-boronic acid pinacol esterCatalog No.:AA0090DA CAS No.:1072812-23-7 MDL No.:MFCD11858379 MF:C14H17BBrNO2 MW:322.0053 |
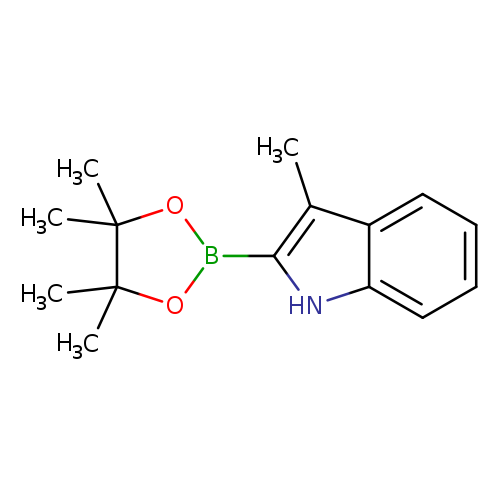
3-Methyl-2-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1h-indoleCatalog No.:AA0082BP CAS No.:1072812-35-1 MDL No.:MFCD16995768 MF:C15H20BNO2 MW:257.1358 |
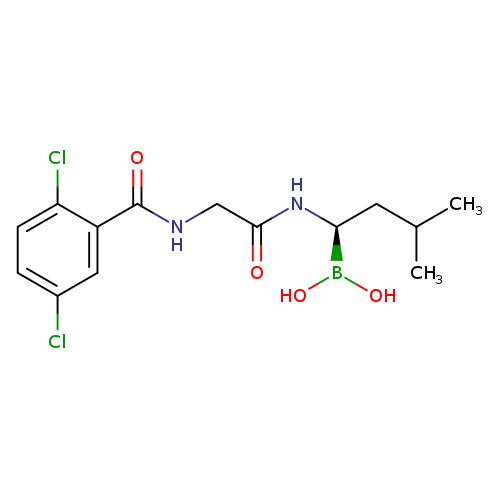
[(1R)-1-{2-[(2,5-dichlorophenyl)formamido]acetamido}-3-methylbutyl]boronic acidCatalog No.:AA0037VR CAS No.:1072833-77-2 MDL No.:MFCD18251438 MF:C14H19BCl2N2O4 MW:361.0287 |
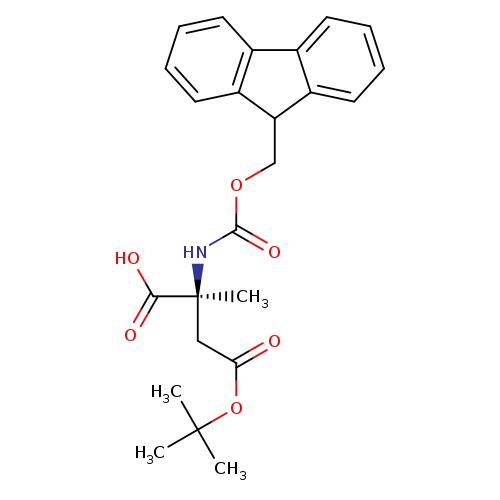
(S)-Fmoc-2-amino-2-methyl-succinic acid-4-tert-butyl esterCatalog No.:AA008Z6E CAS No.:1072845-47-6 MDL No.:MFCD12031692 MF:C24H27NO6 MW:425.4743 |
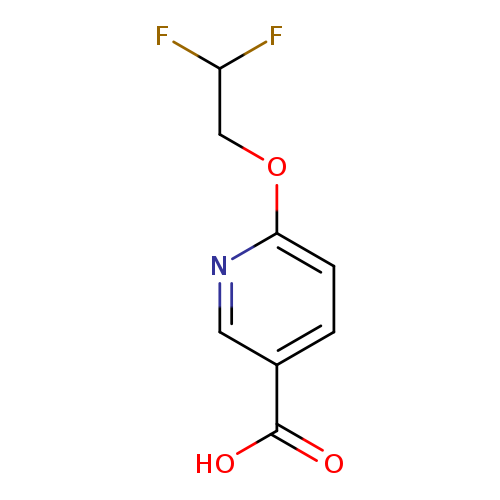
6-(2,2-Difluoroethoxy)pyridine-3-carboxylic acidCatalog No.:AA01A364 CAS No.:1072855-41-4 MDL No.:MFCD23701786 MF:C8H7F2NO3 MW:203.1429 |
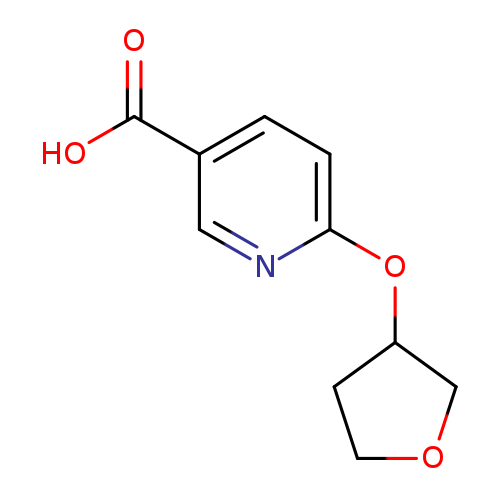
6-((Tetrahydrofuran-3-yl)oxy)nicotinic acidCatalog No.:AA01FLYZ CAS No.:1072855-68-5 MDL No.:MFCD18259826 MF:C10H11NO4 MW:209.1986 |
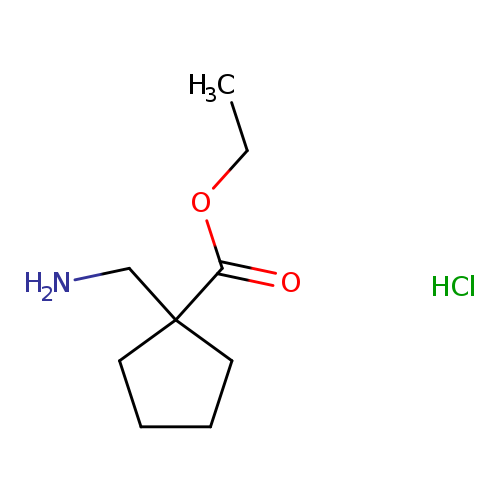
ethyl 1-(aminomethyl)cyclopentane-1-carboxylate hydrochlorideCatalog No.:AA01E7A0 CAS No.:1072856-85-9 MDL No.:MFCD30752272 MF:C9H18ClNO2 MW:207.6977 |
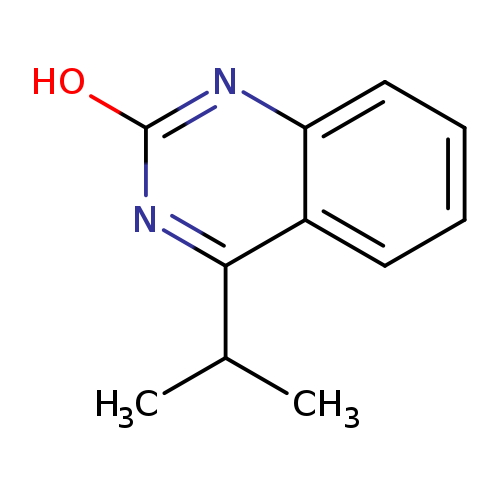
4-Isopropylquinazolin-2(1H)-oneCatalog No.:AA019EKE CAS No.:107289-03-2 MDL No.:MFCD24559421 MF:C11H12N2O MW:188.2258 |
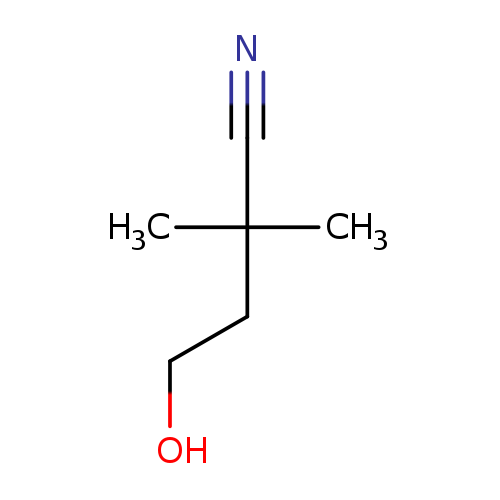
4-hydroxy-2,2-dimethylbutanenitrileCatalog No.:AA01B2LW CAS No.:107289-41-8 MDL No.:MFCD21185377 MF:C6H11NO MW:113.1576 |
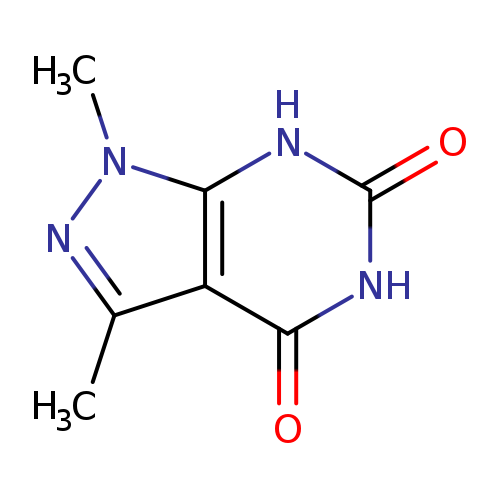
1,3-Dimethyl-1H-pyrazolo[3,4-d]pyrimidine-4,6(5H,7H)-dioneCatalog No.:AA00IWWL CAS No.:1072895-79-4 MDL No.:MFCD18157655 MF:C7H8N4O2 MW:180.1640 |
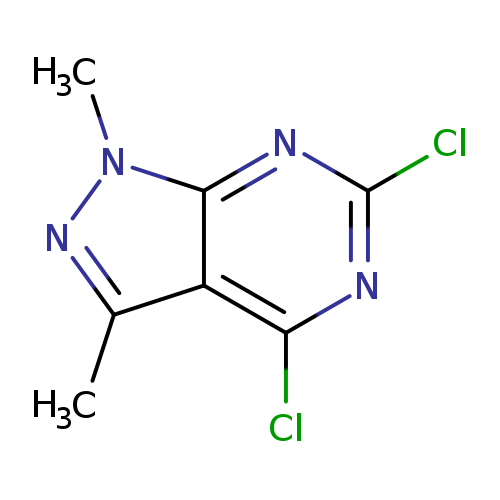
4,6-Dichloro-1,3-dimethyl-1h-pyrazolo[3,4-d]pyrimidineCatalog No.:AA008TZX CAS No.:1072895-86-3 MDL No.:MFCD11869749 MF:C7H6Cl2N4 MW:217.0553 |
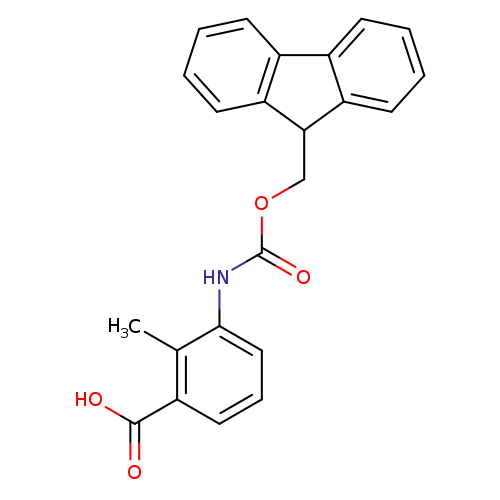
Fmoc-3-amino-2-methylbenzoic acidCatalog No.:AA008RU0 CAS No.:1072901-47-3 MDL No.:MFCD01861304 MF:C23H19NO4 MW:373.4013 |
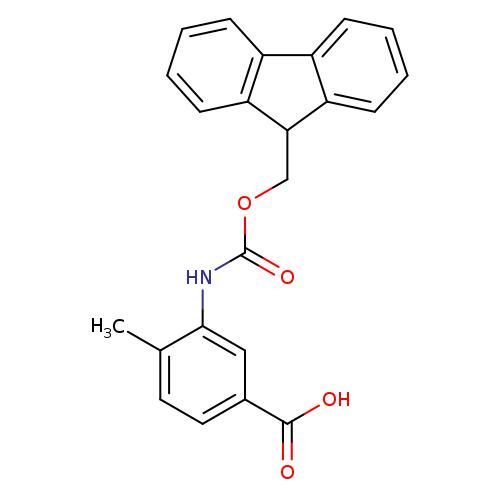
3-((((9H-Fluoren-9-yl)methoxy)carbonyl)amino)-4-methylbenzoic acidCatalog No.:AA008R2N CAS No.:1072901-59-7 MDL No.:MFCD01861379 MF:C23H19NO4 MW:373.4013 |
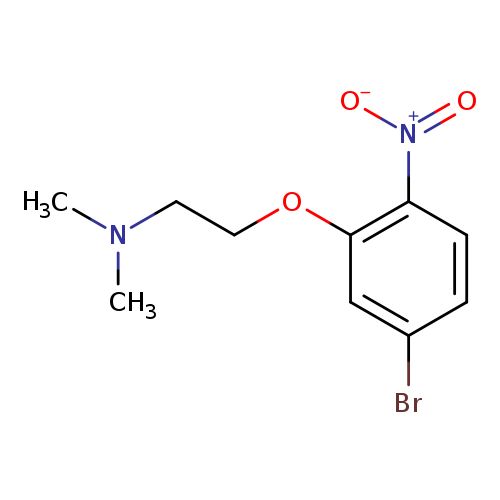
EthanaMine, 2-(5-broMo-2-nitrophenoxy)-N,N-diMethyl-Catalog No.:AA00995N CAS No.:1072906-04-7 MDL No.:MFCD11520820 MF:C10H13BrN2O3 MW:289.1258 |
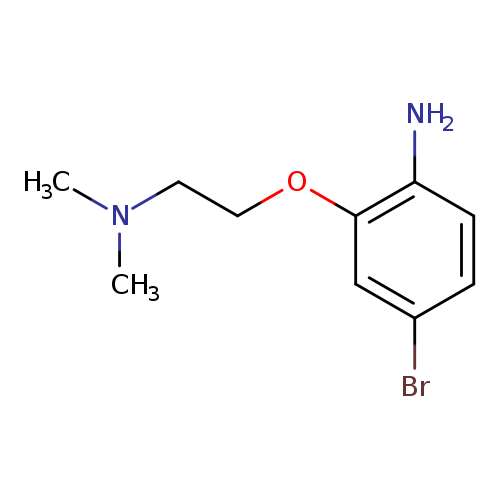
4-Bromo-2-(2-(dimethylamino)ethoxy)anilineCatalog No.:AA01DLLX CAS No.:1072906-05-8 MDL No.:MFCD11520819 MF:C10H15BrN2O MW:259.1429 |
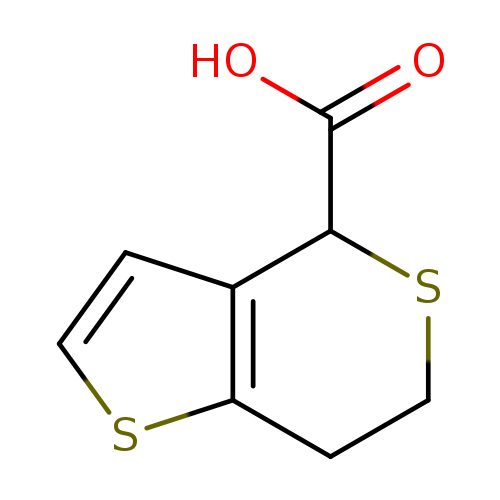
4H,6H,7H-Thieno[3,2-c]thiopyran-4-carboxylic acidCatalog No.:AA019SRC CAS No.:107292-02-4 MDL No.:MFCD20731128 MF:C8H8O2S2 MW:200.2779 |
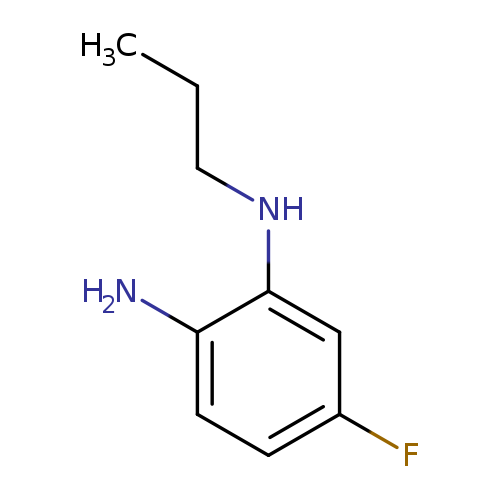
5-fluoro-1-N-propylbenzene-1,2-diamineCatalog No.:AA01A8RE CAS No.:1072928-93-8 MDL No.:MFCD16744197 MF:C9H13FN2 MW:168.2113 |
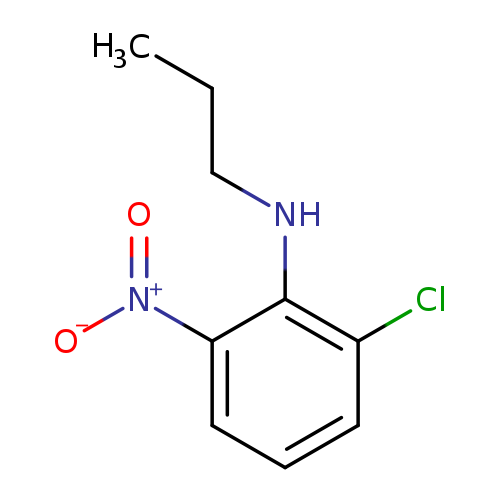
2-chloro-6-nitro-N-propylanilineCatalog No.:AA01AHV7 CAS No.:1072928-95-0 MDL No.:MFCD16672099 MF:C9H11ClN2O2 MW:214.6488 |
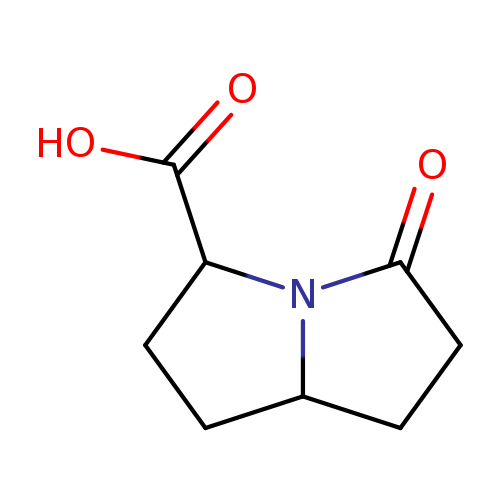
1H-Pyrrolizine-3-carboxylicacid,hexahydro-5-oxo-(9CI)Catalog No.:AA009Q1U CAS No.:107294-50-8 MDL No.:MFCD18810391 MF:C8H11NO3 MW:169.1778 |
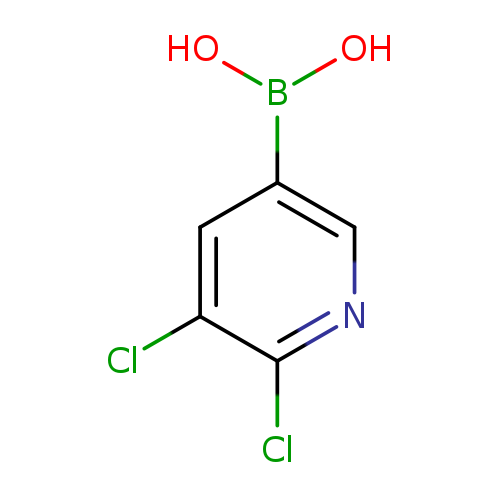
2,3-Dichloropyridine-5-boronic acidCatalog No.:AA008S47 CAS No.:1072944-15-0 MDL No.:MFCD08277308 MF:C5H4BCl2NO2 MW:191.8078 |
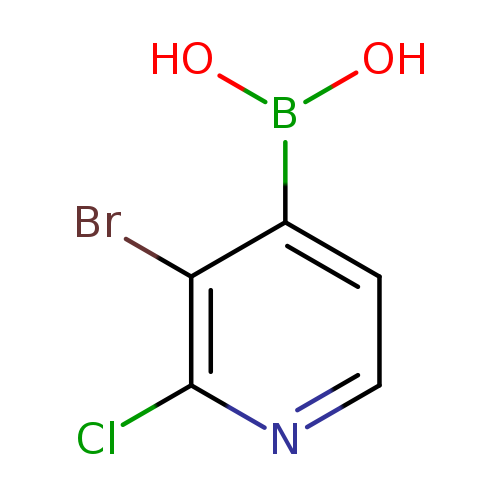
3-Bromo-2-chloropyridine-4-boronic acidCatalog No.:AA008R75 CAS No.:1072944-16-1 MDL No.:MFCD08458470 MF:C5H4BBrClNO2 MW:236.2588 |
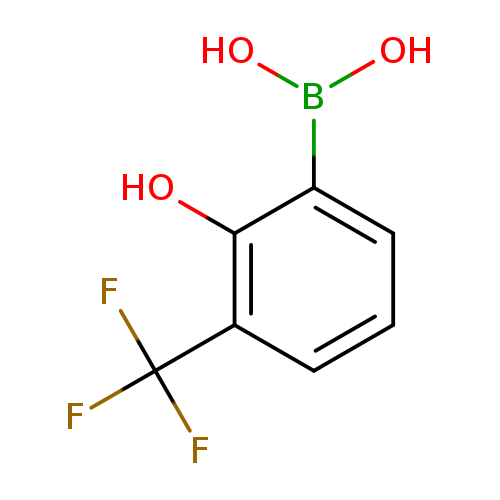
2-Hydroxy-3-(trifluoromethyl)phenylboronic acidCatalog No.:AA007T4V CAS No.:1072944-17-2 MDL No.:MFCD08689548 MF:C7H6BF3O3 MW:205.9269 |
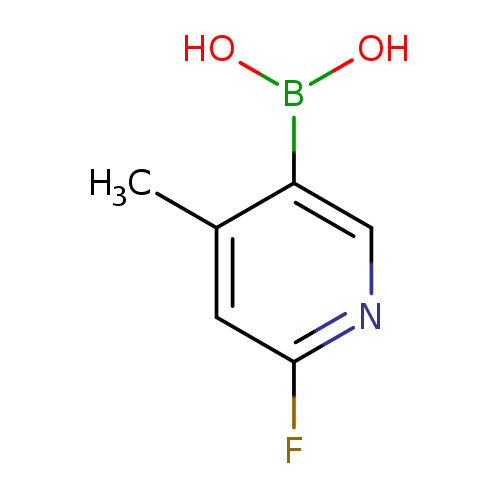
2-Fluoro-4-methylpyridine-5-boronic acidCatalog No.:AA007T4U CAS No.:1072944-18-3 MDL No.:MFCD08741406 MF:C6H7BFNO2 MW:154.9347 |
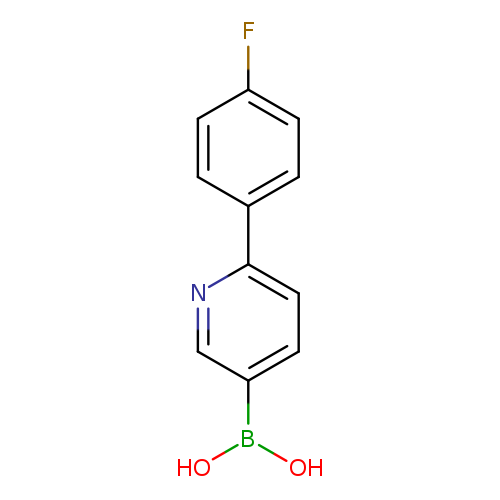
6-(4-Fluorophenyl)pyridine-3-boronic acidCatalog No.:AA003BVR CAS No.:1072944-20-7 MDL No.:MFCD09037487 MF:C11H9BFNO2 MW:217.0041 |
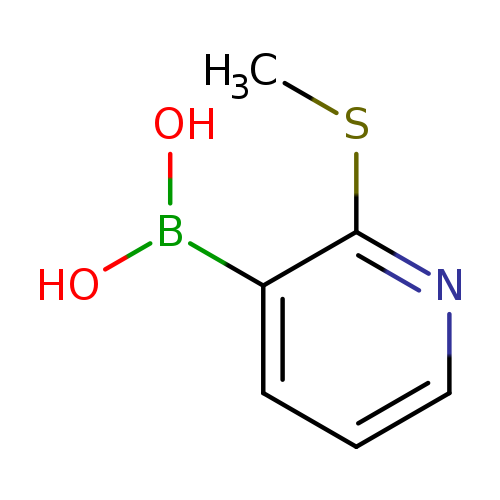
2-(Methylthio)pyridine-3-boronic acidCatalog No.:AA003BHF CAS No.:1072944-21-8 MDL No.:MFCD09037488 MF:C6H8BNO2S MW:169.0092 |
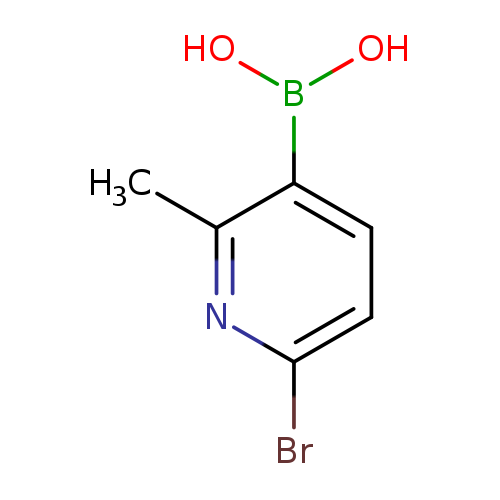
6-Bromo-2-methylpyridine-3-boronic acidCatalog No.:AA003BVZ CAS No.:1072944-22-9 MDL No.:MFCD09037490 MF:C6H7BBrNO2 MW:215.8403 |
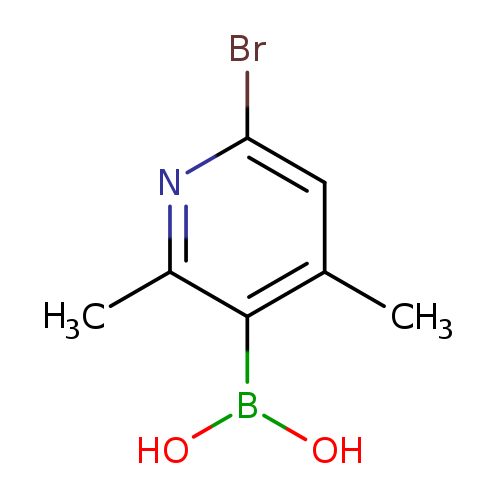
6-Bromo-2,4-dimethylpyridine-3-boronic acidCatalog No.:AA003BVV CAS No.:1072944-23-0 MDL No.:MFCD09037491 MF:C7H9BBrNO2 MW:229.8669 |
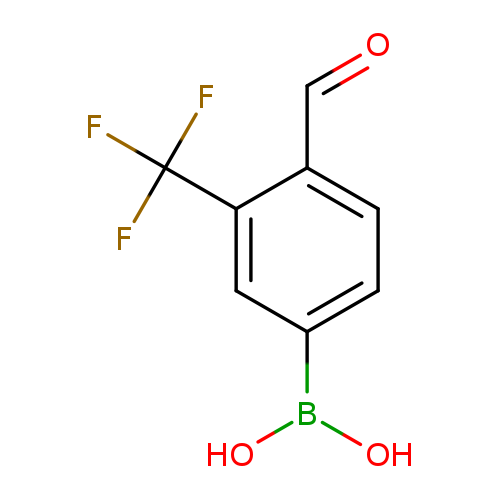
4-Formyl-3-(trifluoromethyl)phenylboronic acidCatalog No.:AA007T4T CAS No.:1072944-24-1 MDL No.:MFCD09037498 MF:C8H6BF3O3 MW:217.9376 |
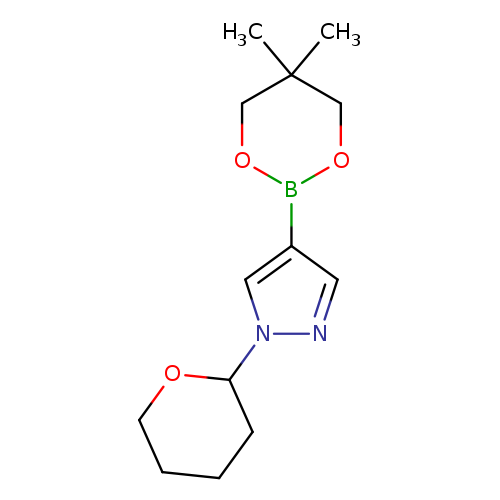
1-(2-Tetrahydropyranyl)-1H-pyrazole-4-boronic acid neopentyl glycol esterCatalog No.:AA003CSW CAS No.:1072944-26-3 MDL No.:MFCD09037503 MF:C13H21BN2O3 MW:264.1284 |
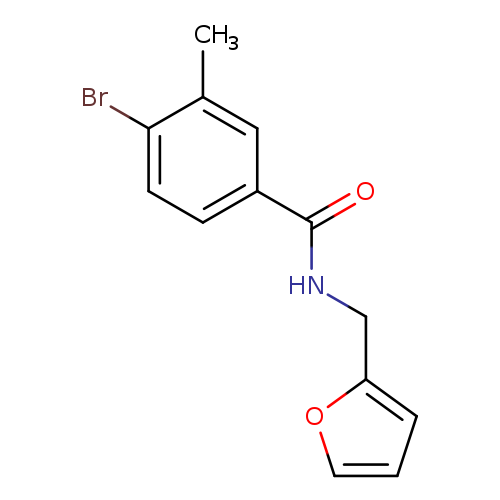
N-(furan-2-ylmethyl) 4-bromo-3-methylbenzamideCatalog No.:AA003KXO CAS No.:1072944-32-1 MDL No.:MFCD11504866 MF:C13H12BrNO2 MW:294.1439 |
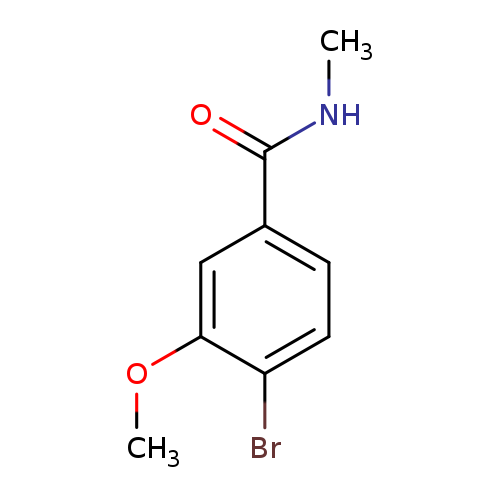
N-Methyl 4-bromo-3-methoxybenzamideCatalog No.:AA007T0J CAS No.:1072944-34-3 MDL No.:MFCD11504869 MF:C9H10BrNO2 MW:244.0852 |
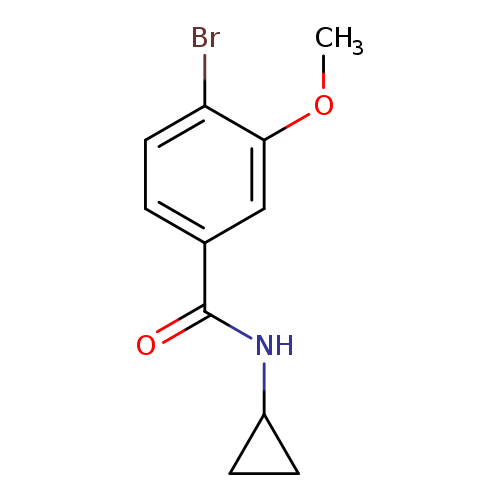
N-Cyclopropyl 4-bromo-3-methoxybenzamideCatalog No.:AA007T0I CAS No.:1072944-35-4 MDL No.:MFCD11504870 MF:C11H12BrNO2 MW:270.1225 |
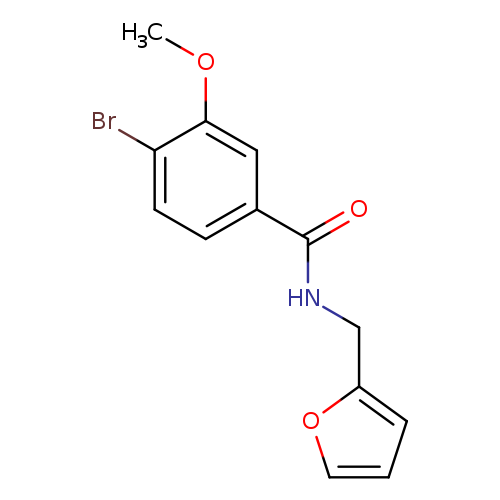
N-(Furan-2-ylmethyl) 4-bromo-3-methoxybenzamideCatalog No.:AA003KXN CAS No.:1072944-36-5 MDL No.:MFCD11504871 MF:C13H12BrNO3 MW:310.1433 |
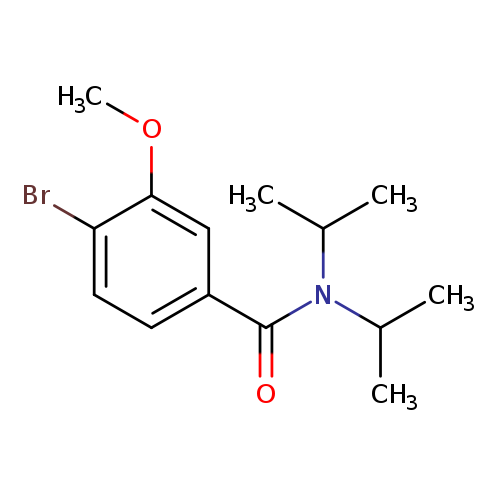
N,N-Diisopropyl 4-bromo-3-methoxybenzamideCatalog No.:AA0082BN CAS No.:1072944-37-6 MDL No.:MFCD11504872 MF:C14H20BrNO2 MW:314.2181 |
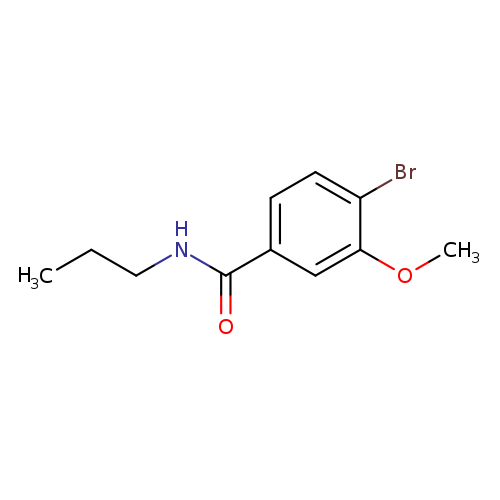
N-Propyl 4-bromo-3-methoxybenzamideCatalog No.:AA0082BM CAS No.:1072944-38-7 MDL No.:MFCD11504873 MF:C11H14BrNO2 MW:272.1384 |
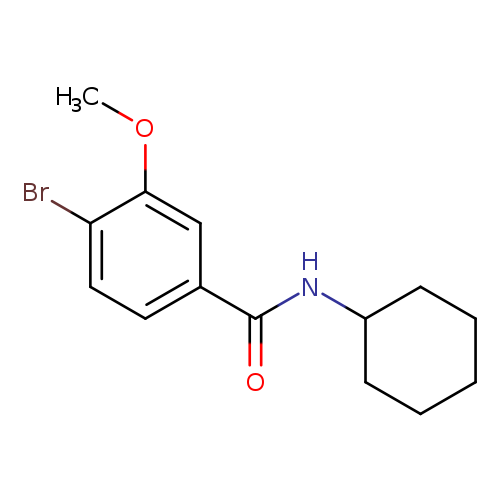
N-Cyclohexyl 4-bromo-3-methoxybenzamideCatalog No.:AA003SU3 CAS No.:1072944-40-1 MDL No.:MFCD11504875 MF:C14H18BrNO2 MW:312.2022 |
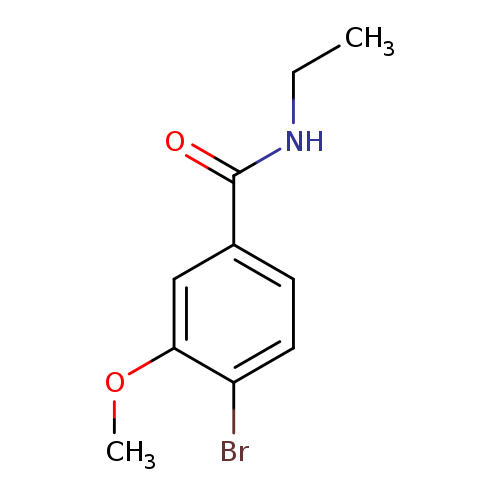
N-Ethyl 4-bromo-3-methoxybenzamideCatalog No.:AA0082BL CAS No.:1072944-41-2 MDL No.:MFCD11504876 MF:C10H12BrNO2 MW:258.1118 |
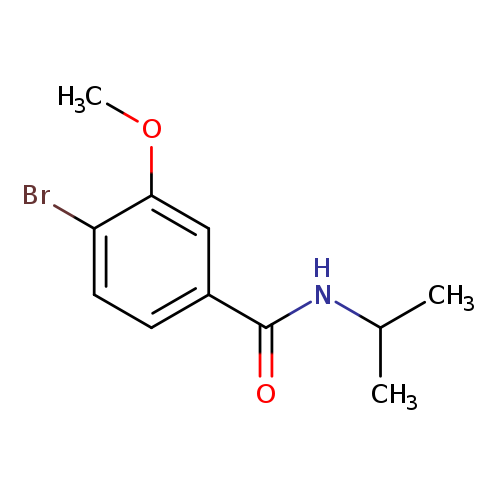
N-Isopropyl 4-bromo-3-methoxybenzamideCatalog No.:AA003T1R CAS No.:1072944-42-3 MDL No.:MFCD11504877 MF:C11H14BrNO2 MW:272.1384 |
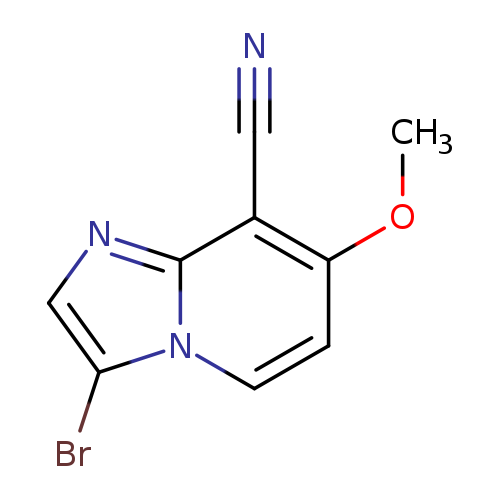
3-Bromo-7-methoxyimidazo[1,2-a]pyridine-8-carbonitrileCatalog No.:AA0082BK CAS No.:1072944-45-6 MDL No.:MFCD11504889 MF:C9H6BrN3O MW:252.0674 |
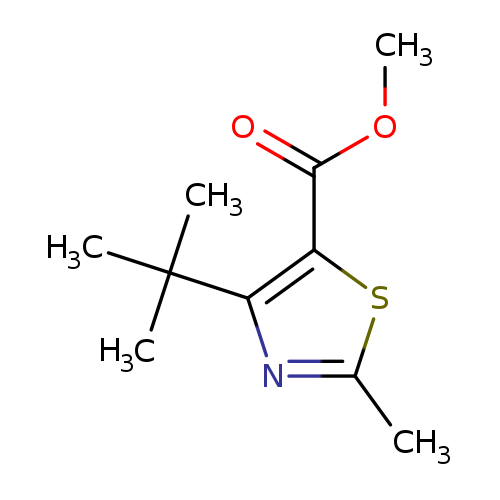
Methyl 4-tert-butyl-2-methylthiazole-5-carboxylateCatalog No.:AA007AP4 CAS No.:1072944-46-7 MDL No.:MFCD11504890 MF:C10H15NO2S MW:213.2966 |
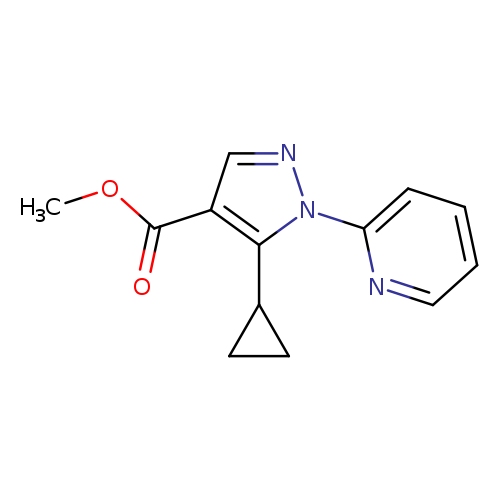
Methyl 5-cyclopropyl-1-(pyridin-2-yl)-1H-pyrazole-4-carboxylateCatalog No.:AA0082BJ CAS No.:1072944-47-8 MDL No.:MFCD11504891 MF:C13H13N3O2 MW:243.2612 |
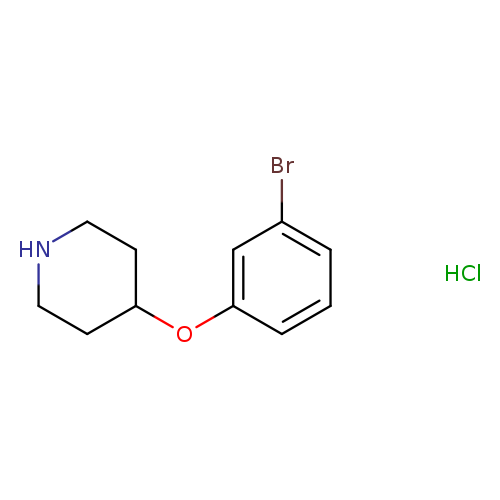
4-(3-Bromophenoxy)piperidine, HClCatalog No.:AA0090CO CAS No.:1072944-50-3 MDL No.:MFCD09879219 MF:C11H15BrClNO MW:292.5999 |
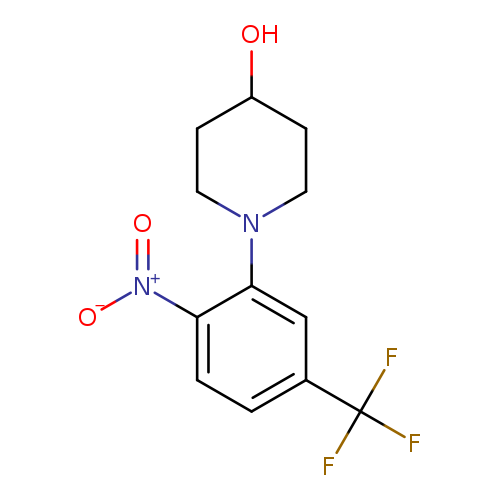
4-Hydroxy-1-(2-nitro-5-trifluoromethylphenyl)piperidineCatalog No.:AA0090CI CAS No.:1072944-51-4 MDL No.:MFCD11504892 MF:C12H13F3N2O3 MW:290.2384 |
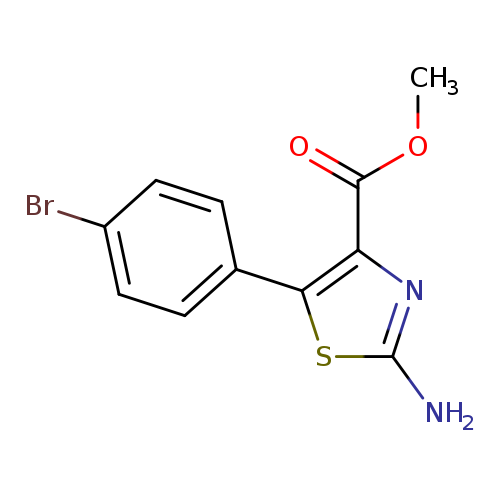
Methyl 2-amino-5-(4-bromophenyl)thiazole-4-carboxylateCatalog No.:AA0090D8 CAS No.:1072944-52-5 MDL No.:MFCD09999101 MF:C11H9BrN2O2S MW:313.1704 |
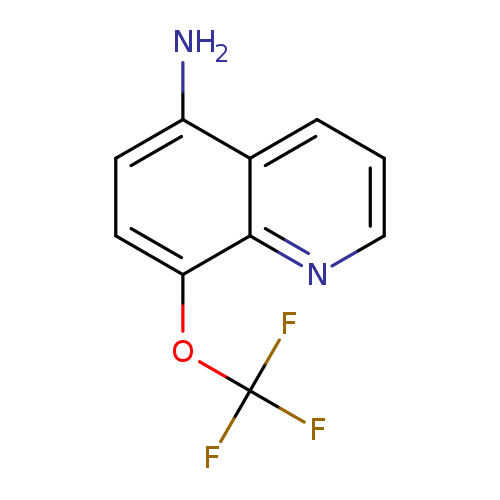
5-Amino-8-trifluoromethoxyquinolineCatalog No.:AA003MBB CAS No.:1072944-53-6 MDL No.:MFCD11504893 MF:C10H7F3N2O MW:228.1706 |
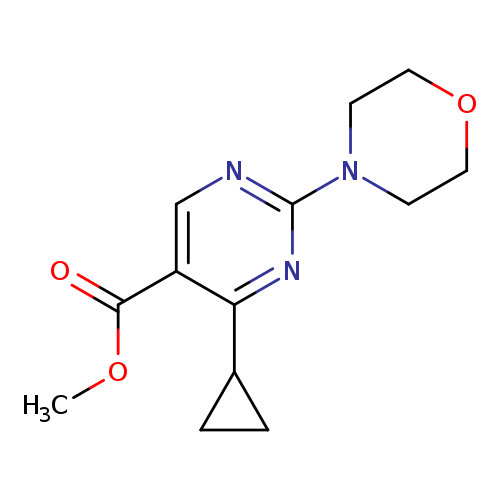
Methyl 4-cyclopropyl-2-morpholinopyrimidine-5-carboxylateCatalog No.:AA003RSD CAS No.:1072944-54-7 MDL No.:MFCD11504896 MF:C13H17N3O3 MW:263.2924 |
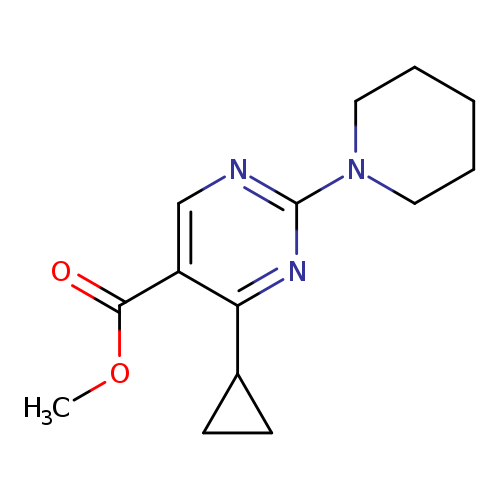
Methyl 4-cyclopropyl-2-(piperidin-1-yl)pyrimidine-5-carboxylateCatalog No.:AA003RSC CAS No.:1072944-55-8 MDL No.:MFCD11504897 MF:C14H19N3O2 MW:261.3196 |
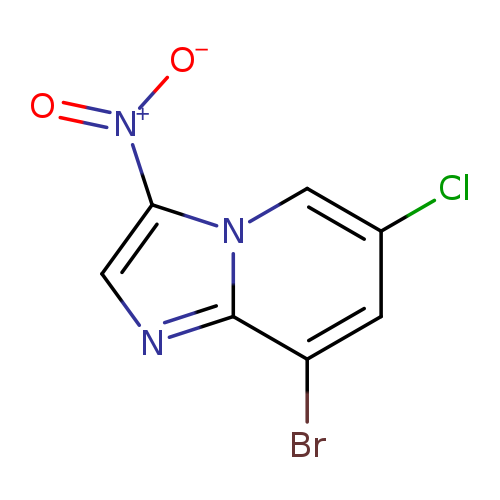
8-Bromo-6-chloro-3-nitroimidazo[1,2-a]pyridineCatalog No.:AA003NF2 CAS No.:1072944-56-9 MDL No.:MFCD11504898 MF:C7H3BrClN3O2 MW:276.4746 |
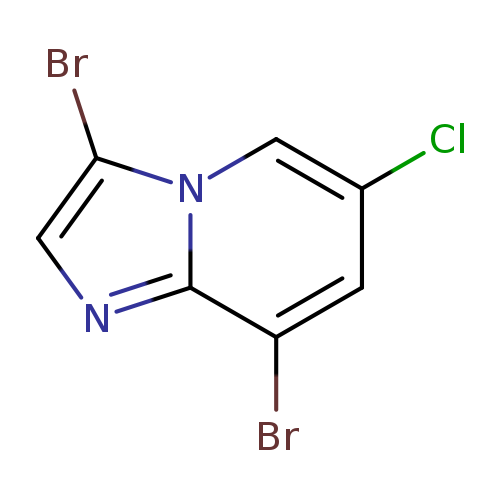
3,8-Dibromo-6-chloroimidazo[1,2-a]pyridineCatalog No.:AA003IOA CAS No.:1072944-57-0 MDL No.:MFCD11504899 MF:C7H3Br2ClN2 MW:310.3731 |
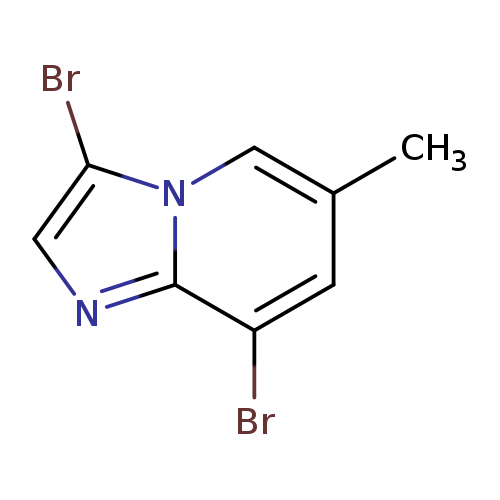
3,8-Dibromo-6-methylimidazo[1,2-a]pyridineCatalog No.:AA0084ZH CAS No.:1072944-58-1 MDL No.:MFCD11504900 MF:C8H6Br2N2 MW:289.9546 |
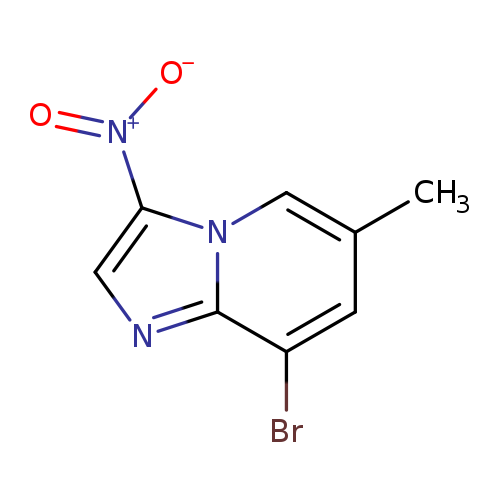
8-Bromo-6-methyl-3-nitroimidazo[1,2-a]pyridineCatalog No.:AA003NF5 CAS No.:1072944-59-2 MDL No.:MFCD11504901 MF:C8H6BrN3O2 MW:256.0561 |
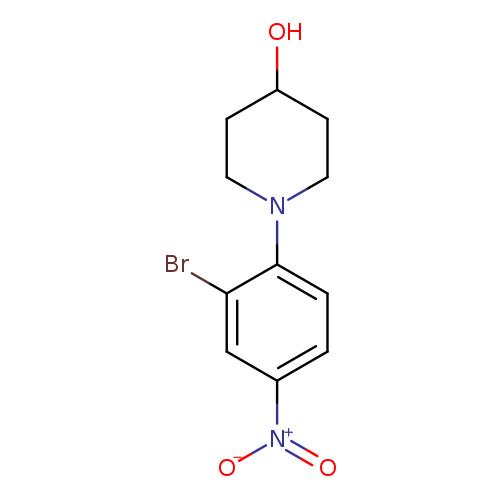
1-(2-Bromo-4-nitrophenyl)-4-hydroxypiperidineCatalog No.:AA003CQM CAS No.:1072944-61-6 MDL No.:MFCD11039803 MF:C11H13BrN2O3 MW:301.1365 |
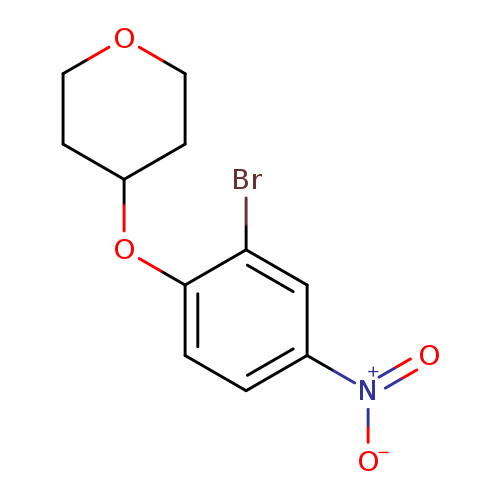
4-(2-Bromo-4-nitrophenoxy)tetrahydro-2H-pyranCatalog No.:AA003JWA CAS No.:1072944-62-7 MDL No.:MFCD11504903 MF:C11H12BrNO4 MW:302.1213 |
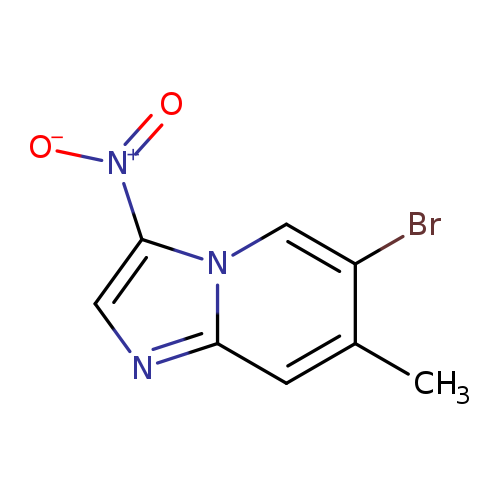
6-Bromo-7-methyl-3-nitroimidazo[1,2-a]pyridineCatalog No.:AA007WM6 CAS No.:1072944-64-9 MDL No.:MFCD11504905 MF:C8H6BrN3O2 MW:256.0561 |
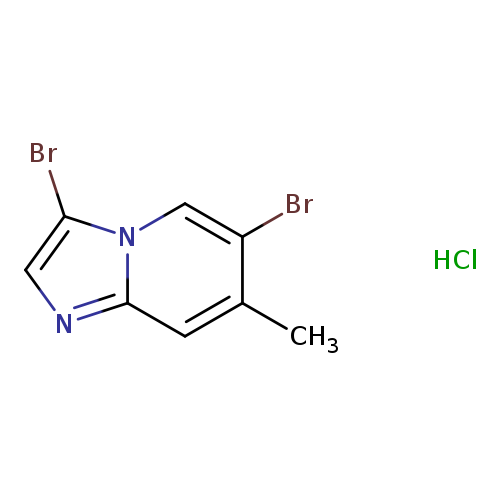
3,6-Dibromo-7-methylimidazo[1,2-a]pyridineCatalog No.:AA003INE CAS No.:1072944-65-0 MDL No.:MFCD11855860 MF:C8H7Br2ClN2 MW:326.4156 |
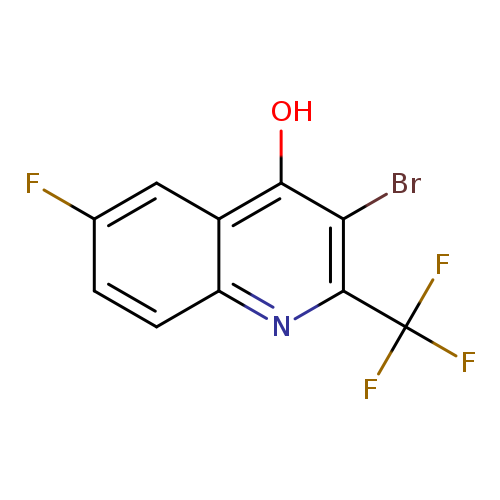
3-Bromo-6-fluoro-4-hydroxy-2-trifluoromethylquinolineCatalog No.:AA003J0T CAS No.:1072944-66-1 MDL No.:MFCD11504911 MF:C10H4BrF4NO MW:310.0425 |
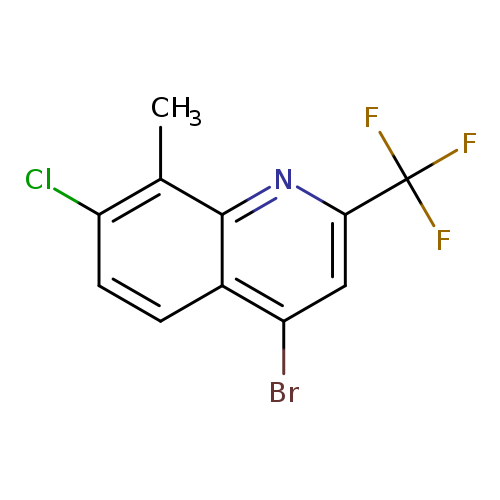
4-Bromo-7-chloro-8-methyl-2-(trifluoromethyl)quinolineCatalog No.:AA007ESJ CAS No.:1072944-67-2 MDL No.:MFCD11504913 MF:C11H6BrClF3N MW:324.5242 |
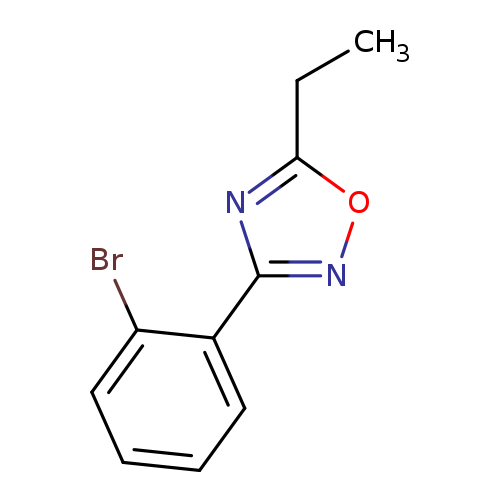
3-(2-Bromophenyl)-5-ethyl-1,2,4-oxadiazoleCatalog No.:AA003HXL CAS No.:1072944-70-7 MDL No.:MFCD11504917 MF:C10H9BrN2O MW:253.0953 |
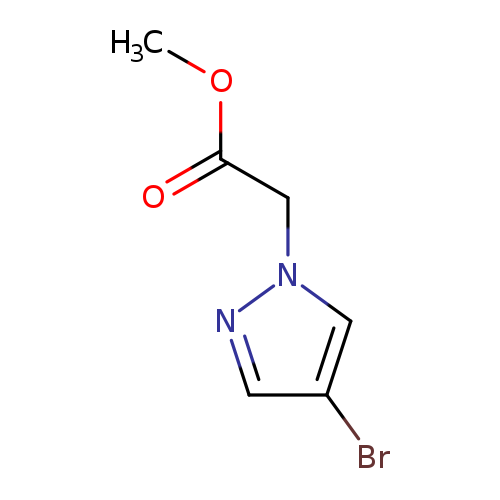
Methyl 2-(4-bromo-1H-pyrazol-1-yl)acetateCatalog No.:AA0084ZF CAS No.:1072944-71-8 MDL No.:MFCD09878400 MF:C6H7BrN2O2 MW:219.0360 |
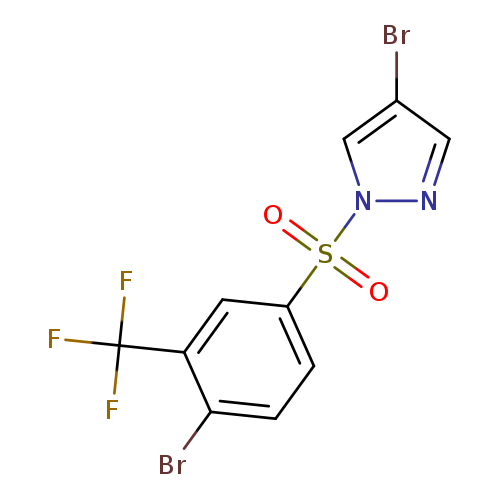
4-Bromo-1-(4-bromo-3-trifluoromethylphenylsulfonyl)pyrazoleCatalog No.:AA003KRO CAS No.:1072944-72-9 MDL No.:MFCD11053886 MF:C10H5Br2F3N2O2S MW:434.0271 |
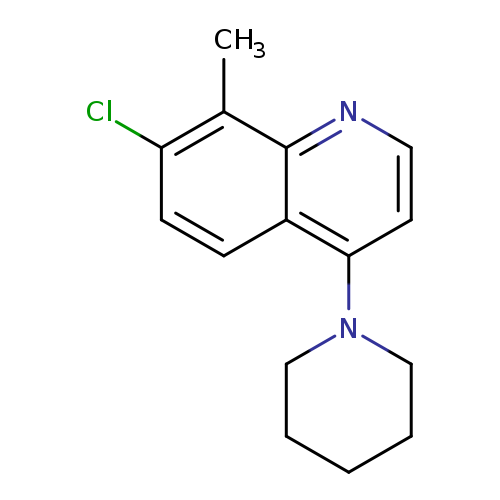
7-Chloro-8-methyl-4-(piperidin-1-yl)quinolineCatalog No.:AA003NBU CAS No.:1072944-74-1 MDL No.:MFCD11504919 MF:C15H17ClN2 MW:260.7619 |
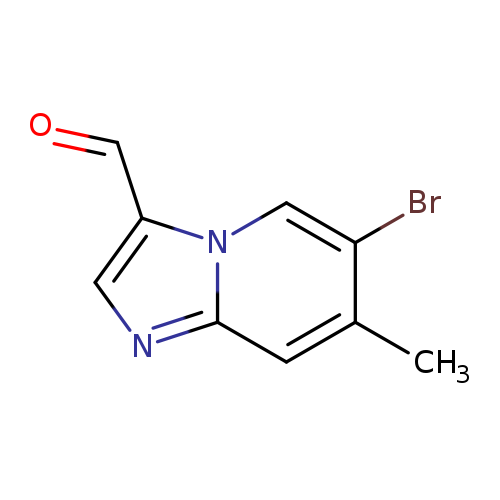
6-Bromo-7-methylimidazo[1,2-a]pyridine-3-carbaldehydeCatalog No.:AA007ESI CAS No.:1072944-75-2 MDL No.:MFCD11504923 MF:C9H7BrN2O MW:239.0687 |
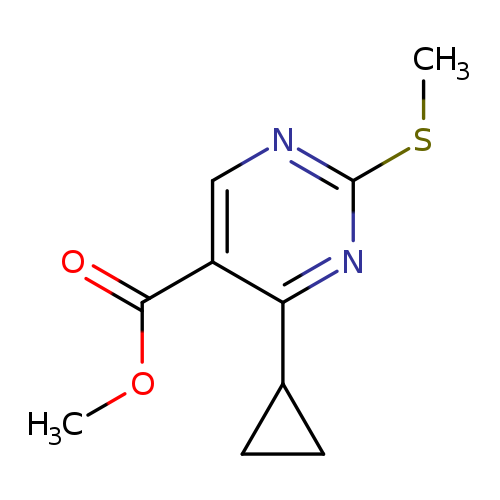
Methyl 4-cyclopropyl-2-(methylthio)pyrimidine-5-carboxylateCatalog No.:AA007ESH CAS No.:1072944-76-3 MDL No.:MFCD11504925 MF:C10H12N2O2S MW:224.2795 |
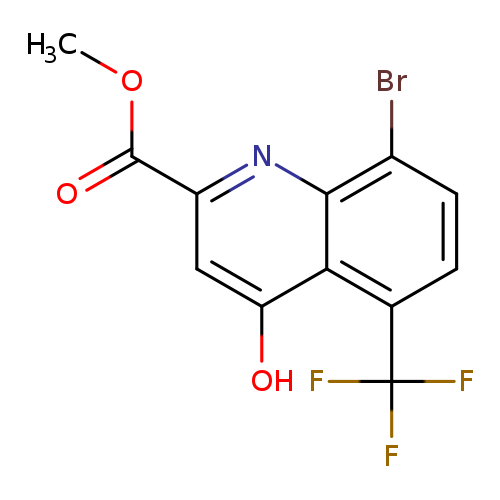
Methyl 8-bromo-4-hydroxy-5-trifluoromethylquinoline-2-carboxylateCatalog No.:AA003RW5 CAS No.:1072944-77-4 MDL No.:MFCD11504926 MF:C12H7BrF3NO3 MW:350.0881 |
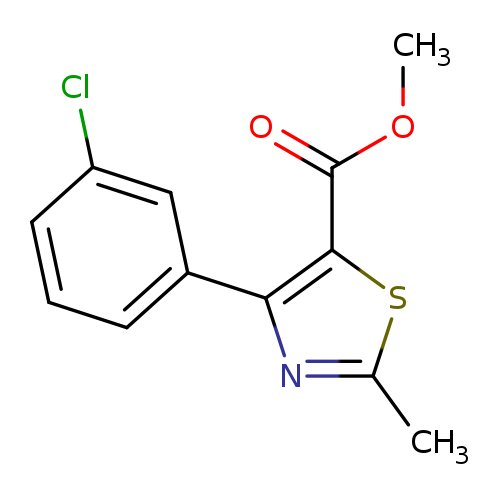
Methyl 4-(3-chlorophenyl)-2-methylthiazole-5-carboxylateCatalog No.:AA003RQR CAS No.:1072944-80-9 MDL No.:MFCD11504928 MF:C12H10ClNO2S MW:267.7313 |
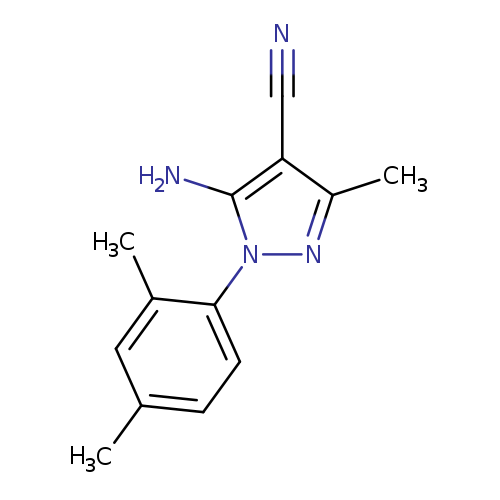
5-Amino-4-cyano-1-(2,4-dimethylphenyl)-3-methylpyrazoleCatalog No.:AA003M9B CAS No.:1072944-82-1 MDL No.:MFCD10699684 MF:C13H14N4 MW:226.2771 |
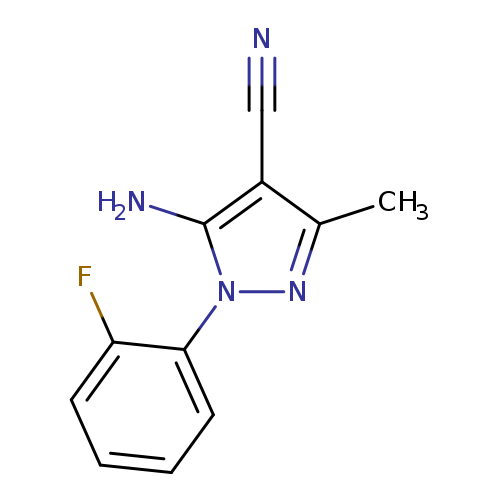
5-Amino-4-cyano-1-(2-fluorophenyl)-3-methylpyrazoleCatalog No.:AA003M9G CAS No.:1072944-83-2 MDL No.:MFCD10699687 MF:C11H9FN4 MW:216.2144 |
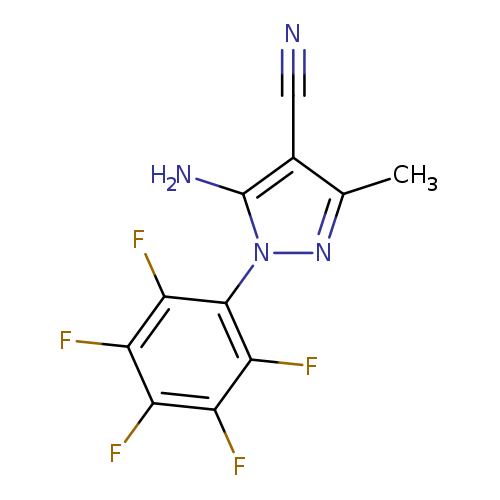
5-Amino-4-cyano-3-methyl-1-(perfluorophenyl)pyrazoleCatalog No.:AA007WM4 CAS No.:1072944-84-3 MDL No.:MFCD11053834 MF:C11H5F5N4 MW:288.1762 |
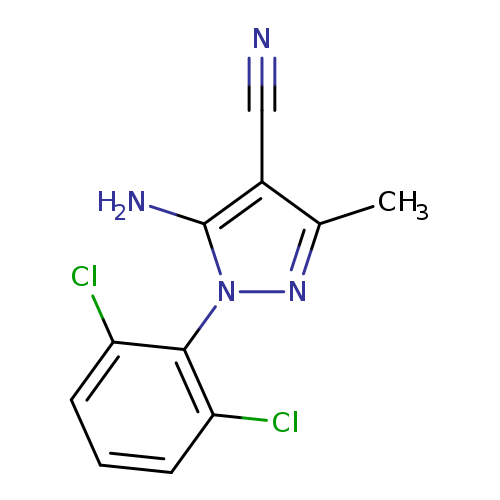
5-Amino-4-cyano-1-(2,6-dichlorophenyl)-3-methylpyrazoleCatalog No.:AA003M9E CAS No.:1072944-85-4 MDL No.:MFCD11053837 MF:C11H8Cl2N4 MW:267.1140 |
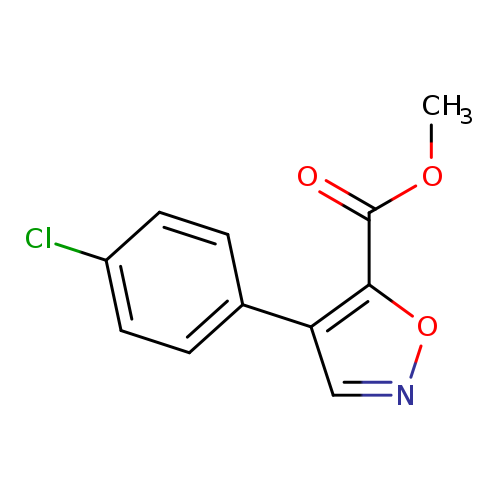
Methyl 4-(4-chlorophenyl)isoxazole-5-carboxylateCatalog No.:AA003RQV CAS No.:1072944-87-6 MDL No.:MFCD11504938 MF:C11H8ClNO3 MW:237.6391 |
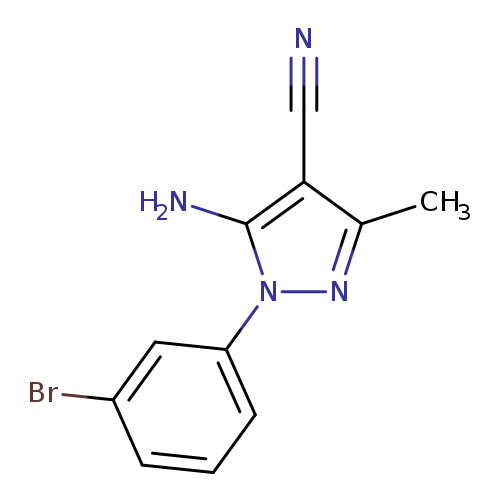
5-Amino-1-(3-bromophenyl)-4-cyano-3-methylpyrazoleCatalog No.:AA0090CL CAS No.:1072944-89-8 MDL No.:MFCD11504941 MF:C11H9BrN4 MW:277.1200 |
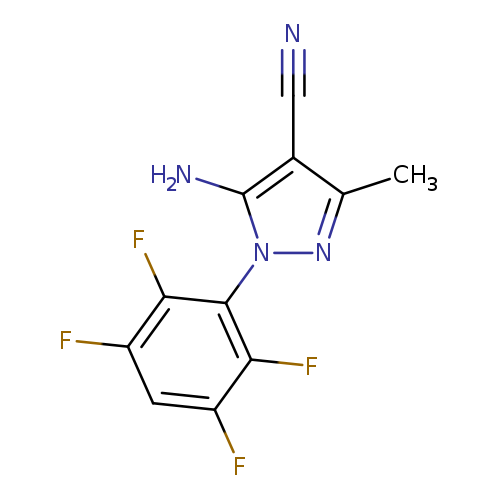
5-Amino-4-cyano-3-methyl-1-(2,3,5,6-tetrafluorophenyl)pyrazoleCatalog No.:AA003MAZ CAS No.:1072944-90-1 MDL No.:MFCD11504942 MF:C11H6F4N4 MW:270.1858 |
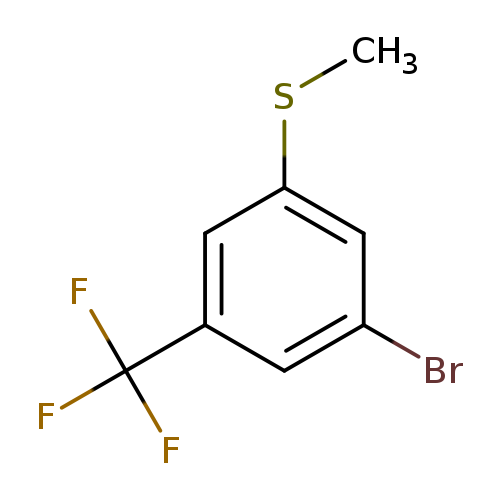
3-Bromo-5-trifluoromethylthioanisoleCatalog No.:AA003J0M CAS No.:1072944-92-3 MDL No.:MFCD11504946 MF:C8H6BrF3S MW:271.0974 |
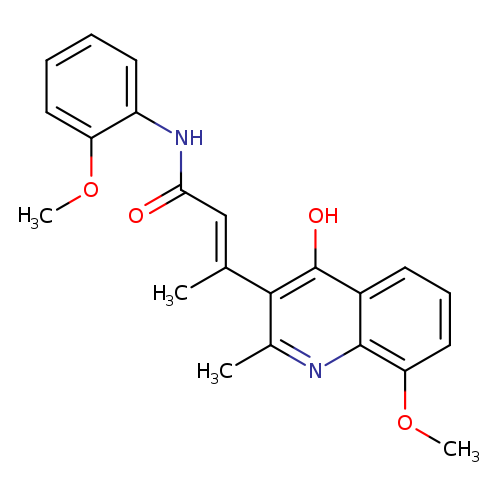
(E)-3-(4-Hydroxy-8-methoxy-2-methylquinolin-3-yl)-N-(2-methoxyphenyl)but-2-enamideCatalog No.:AA003BXP CAS No.:1072944-93-4 MDL No.:MFCD11504952 MF:C22H22N2O4 MW:378.4211 |
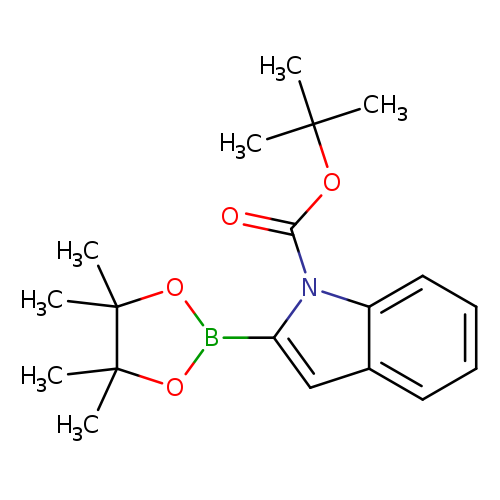
1-BOC-indole-2-boronic acid, pinacol esterCatalog No.:AA003UGC CAS No.:1072944-96-7 MDL No.:MFCD09838985 MF:C19H26BNO4 MW:343.2250 |
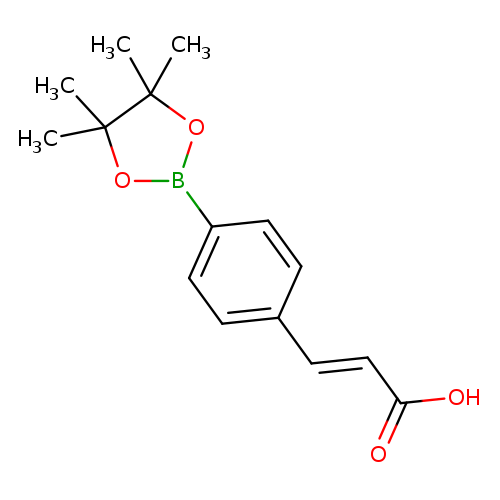
4-(E-2-Carboxyvinyl)phenylboronic acid pinacol esterCatalog No.:AA003BXW CAS No.:1072944-97-8 MDL No.:MFCD11053843 MF:C15H19BO4 MW:274.1200 |
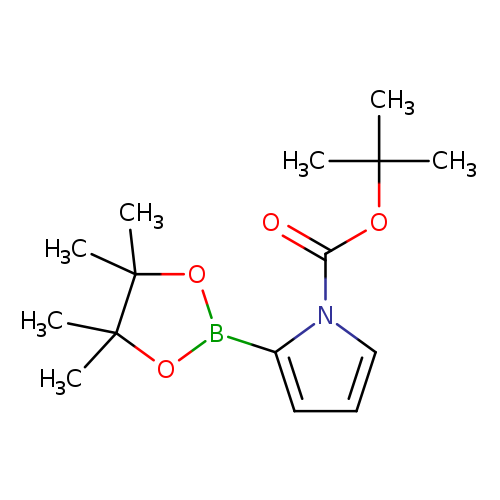
1-BOC-pyrrole-2-boronic acid, pinacol esterCatalog No.:AA007EPR CAS No.:1072944-98-9 MDL No.:MFCD11504960 MF:C15H24BNO4 MW:293.1664 |
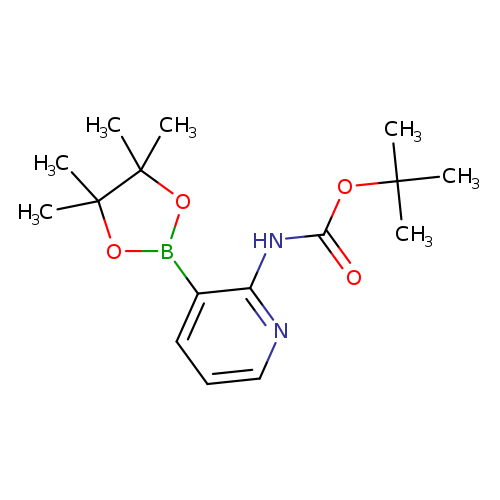
2-(N-Boc-amino)pyridine-3-boronic acid, pinacol esterCatalog No.:AA003UIC CAS No.:1072944-99-0 MDL No.:MFCD08063072 MF:C16H25BN2O4 MW:320.1917 |
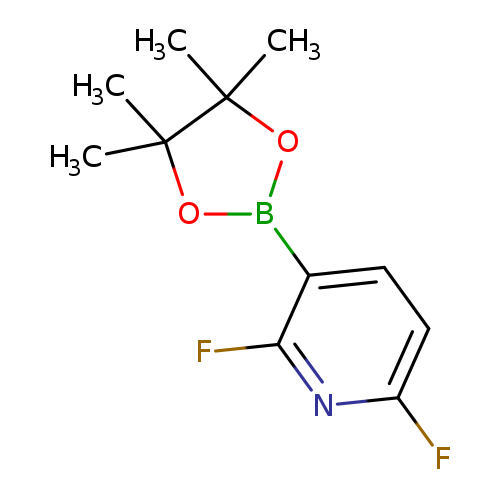
2,6-Difluoropyridin-3-ylboronic acid, pinacol esterCatalog No.:AA003G1B CAS No.:1072945-00-6 MDL No.:MFCD09037475 MF:C11H14BF2NO2 MW:241.0422 |
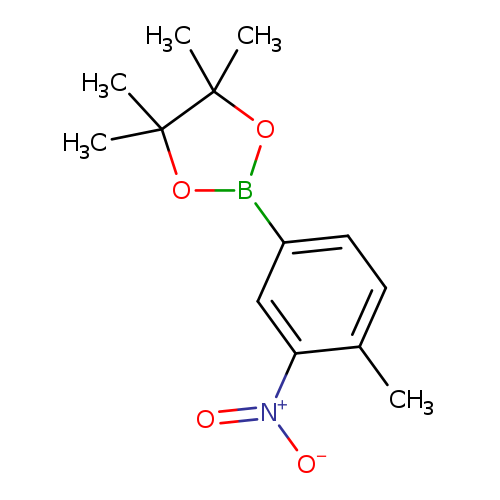
4-Methyl-3-nitrophenylboronic acid pinacol esterCatalog No.:AA008SFZ CAS No.:1072945-06-2 MDL No.:MFCD09027080 MF:C13H18BNO4 MW:263.0973 |
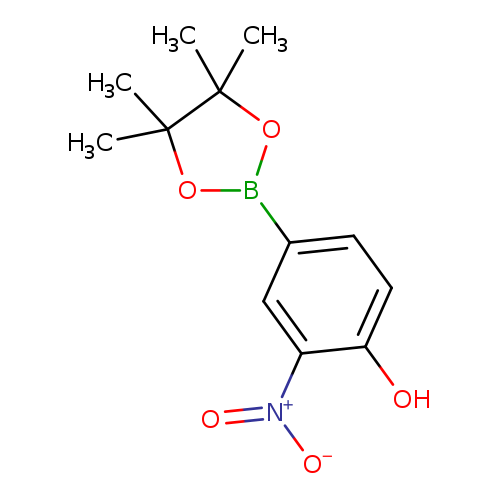
4-hydroxy-3-nitrophenylboronic acid, pinacol esterCatalog No.:AA003LFI CAS No.:1072945-08-4 MDL No.:MFCD08458188 MF:C12H16BNO5 MW:265.0701 |
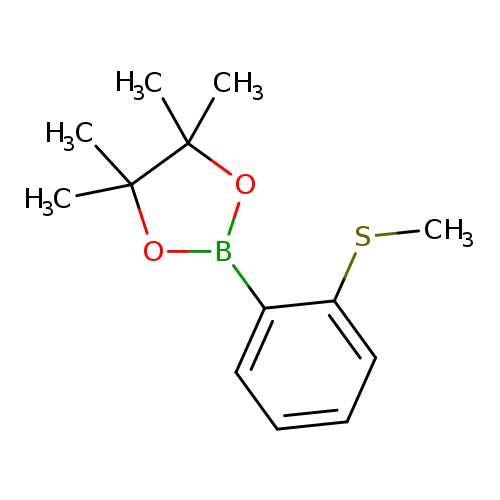
2-Methylthiophenylboronic acid, pinacol esterCatalog No.:AA008S48 CAS No.:1072945-09-5 MDL No.:MFCD05155219 MF:C13H19BO2S MW:250.1648 |
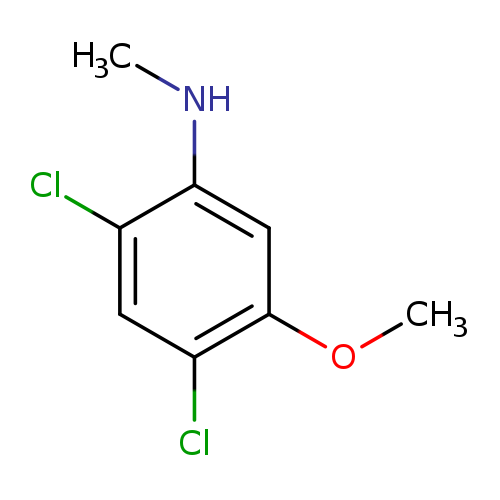
2,4-Dichloro-5-methoxy-N-methylanilineCatalog No.:AA007EPP CAS No.:1072945-55-1 MDL No.:MFCD11504829 MF:C8H9Cl2NO MW:206.0692 |
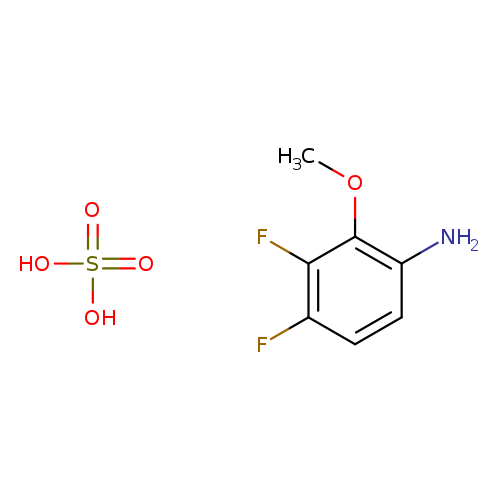
3,4-Difluoro-2-methoxyaniline sulfateCatalog No.:AA003IDY CAS No.:1072945-56-2 MDL No.:MFCD11053865 MF:C7H9F2NO5S MW:257.2119 |
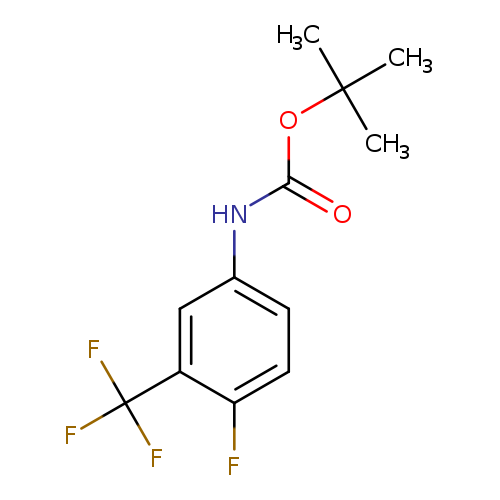
N-BOC-4-Fluoro-3-trifluoromethylanilineCatalog No.:AA007WM1 CAS No.:1072945-57-3 MDL No.:MFCD11504831 MF:C12H13F4NO2 MW:279.2307 |
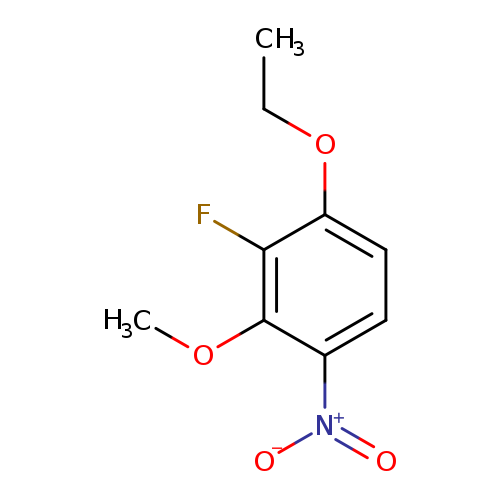
3-Ethoxy-2-fluoro-6-nitroanisoleCatalog No.:AA003JAQ CAS No.:1072945-58-4 MDL No.:MFCD11504833 MF:C9H10FNO4 MW:215.1784 |
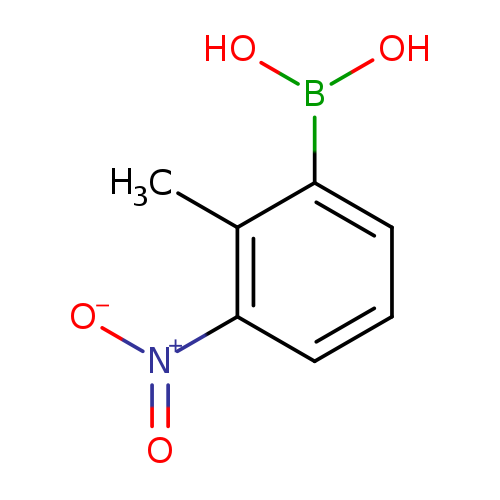
2-Methyl-3-nitrophenylboronic acidCatalog No.:AA00HAW2 CAS No.:1072945-60-8 MDL No.:MFCD06201025 MF:C7H8BNO4 MW:180.9537 |
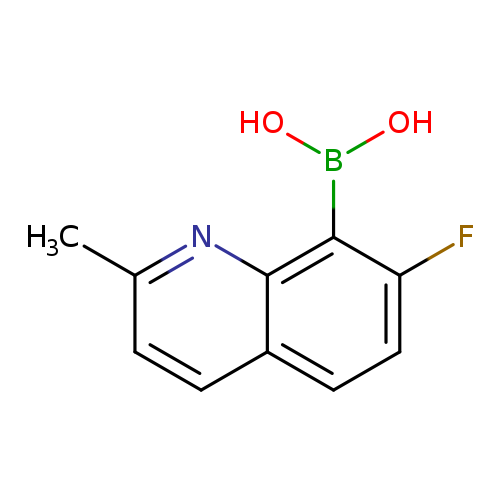
7-Fluoro-2-methylquinoline-8-boronic acidCatalog No.:AA003BW8 CAS No.:1072945-61-9 MDL No.:MFCD11504838 MF:C10H9BFNO2 MW:204.9934 |
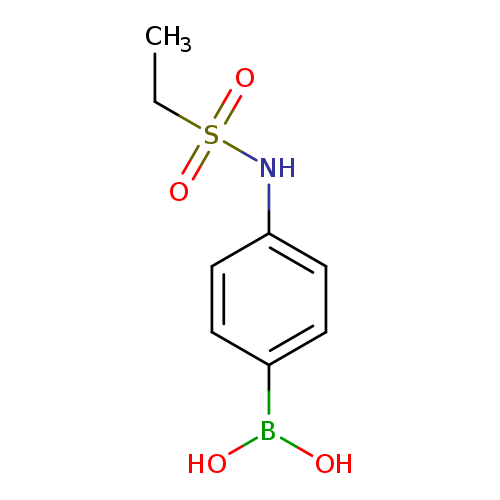
4-(Ethylsulfonamido)phenylboronic acidCatalog No.:AA007WM0 CAS No.:1072945-62-0 MDL No.:MFCD10699608 MF:C8H12BNO4S MW:229.0612 |
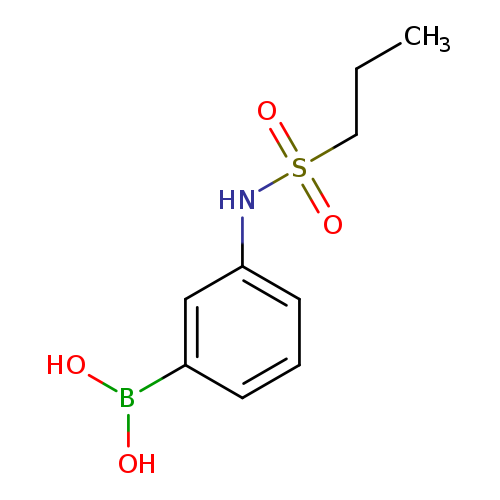
3-(Propylsulfonamido)phenylboronic acidCatalog No.:AA0084ZE CAS No.:1072945-64-2 MDL No.:MFCD10699610 MF:C9H14BNO4S MW:243.0878 |
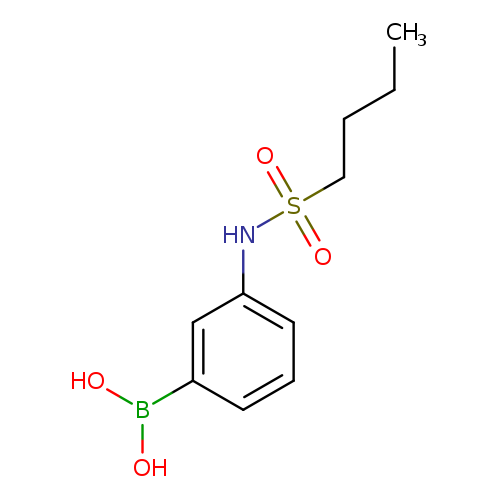
3-(Butylsulfonamido)phenylboronic acidCatalog No.:AA007WLZ CAS No.:1072945-65-3 MDL No.:MFCD10699611 MF:C10H16BNO4S MW:257.1143 |
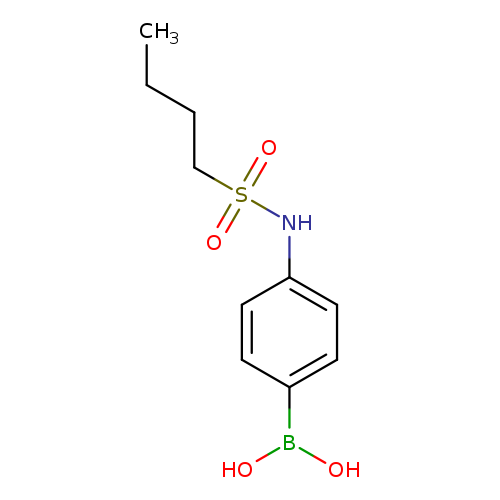
4-(Butylsulfonamido)phenylboronic acidCatalog No.:AA003K3Y CAS No.:1072945-66-4 MDL No.:MFCD10699612 MF:C10H16BNO4S MW:257.1143 |
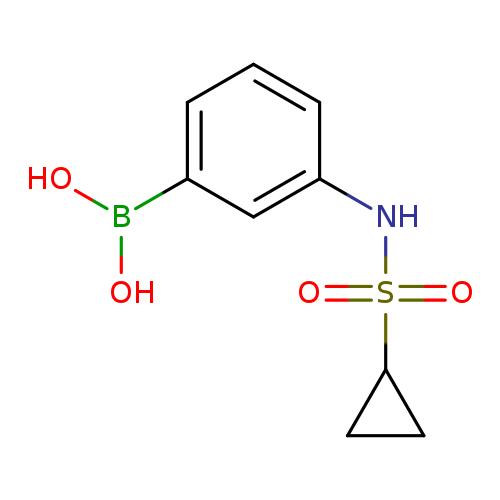
3-(Cyclopropanesulfonamido)phenylboronic acidCatalog No.:AA007EPO CAS No.:1072945-67-5 MDL No.:MFCD10699613 MF:C9H12BNO4S MW:241.0719 |
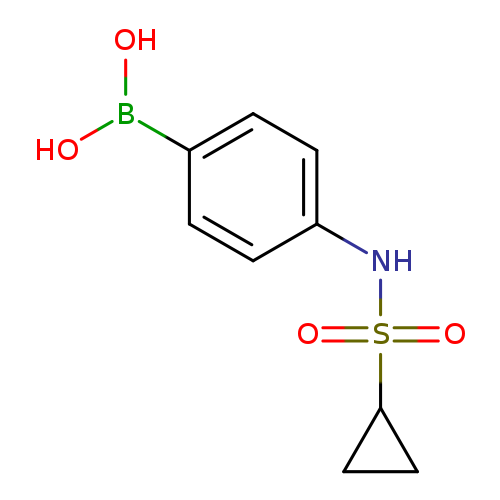
4-(Cyclopropanesulfonamido)phenylboronic acidCatalog No.:AA0090C6 CAS No.:1072945-68-6 MDL No.:MFCD10699614 MF:C9H12BNO4S MW:241.0719 |
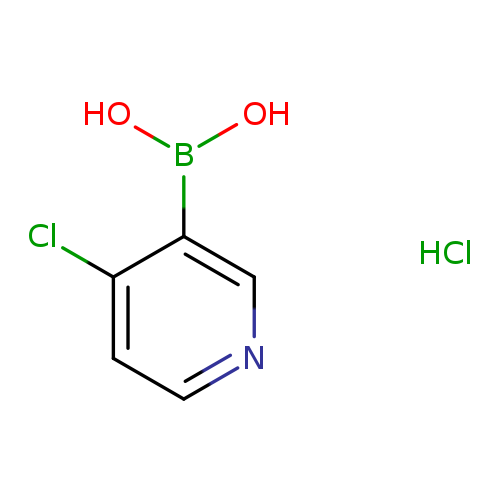
4-Chloropyridine-3-boronic acid , HClCatalog No.:AA007EPN CAS No.:1072945-69-7 MDL No.:MFCD09972093 MF:C5H6BCl2NO2 MW:193.8236 |
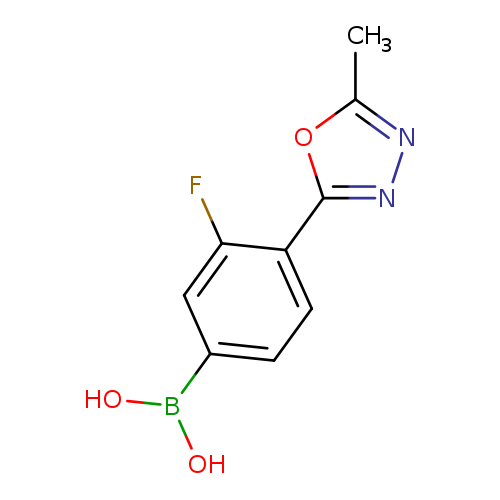
3-Fluoro-4-(5-methyl-1,3,4-oxadiazol-2-yl)phenylboronic acidCatalog No.:AA007WLY CAS No.:1072945-71-1 MDL No.:MFCD09972094 MF:C9H8BFN2O3 MW:221.9808 |
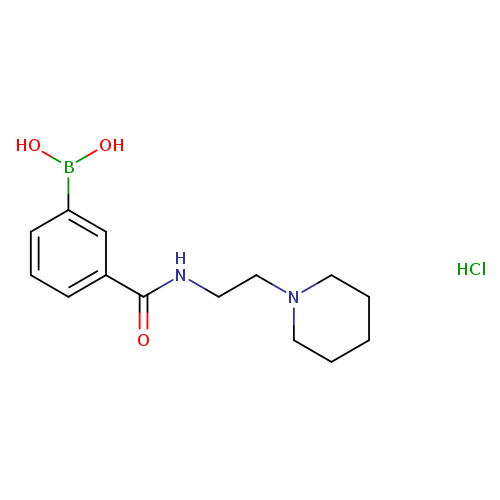
3-(2-(Piperidin-1-yl)ethylcarbamoyl)phenylboronic acid, HClCatalog No.:AA008SR9 CAS No.:1072945-72-2 MDL No.:MFCD11053816 MF:C14H22BClN2O3 MW:312.6001 |
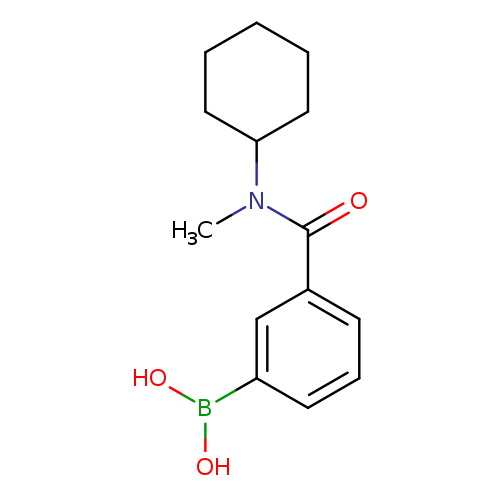
3-(Cyclohexyl(methyl)carbamoyl)phenylboronic acidCatalog No.:AA007EPM CAS No.:1072945-73-3 MDL No.:MFCD09972095 MF:C14H20BNO3 MW:261.1245 |
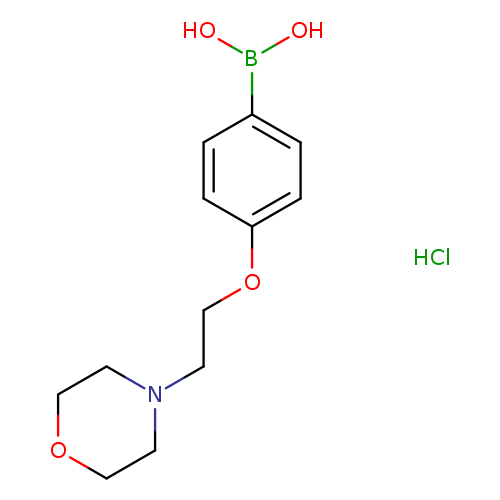
4-(2-Morpholinoethoxy)phenylboronic acid, HClCatalog No.:AA003JXH CAS No.:1072945-74-4 MDL No.:MFCD10699615 MF:C12H19BClNO4 MW:287.5476 |
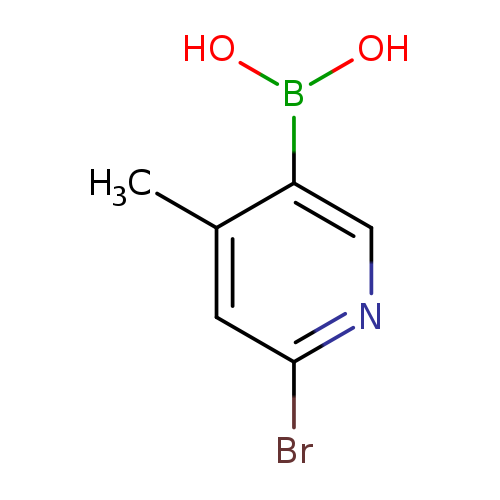
6-Bromo-4-methylpyridine-3-boronic acidCatalog No.:AA003N1O CAS No.:1072945-75-5 MDL No.:MFCD09037489 MF:C6H7BBrNO2 MW:215.8403 |
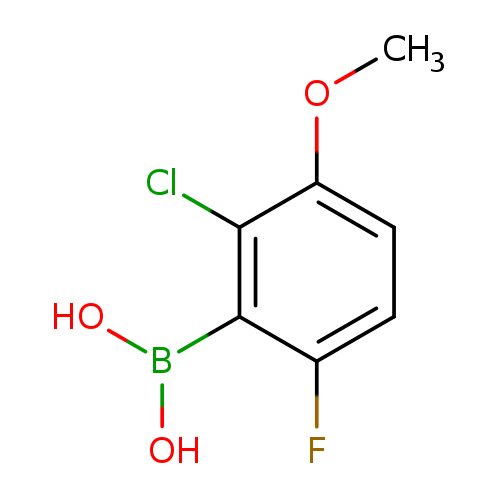
2-Chloro-6-fluoro-3-methoxyphenylboronic acidCatalog No.:AA0090CD CAS No.:1072945-77-7 MDL No.:MFCD09972096 MF:C7H7BClFO3 MW:204.3911 |
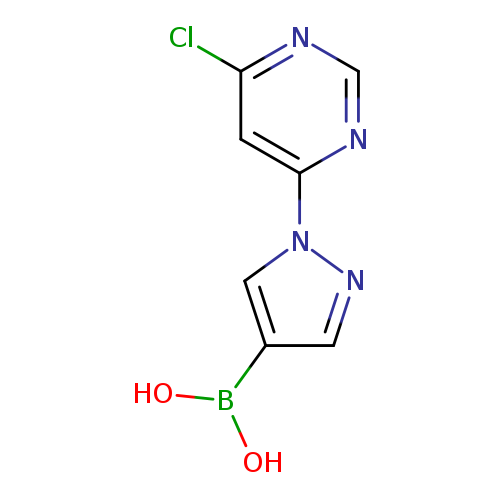
1-(6-Chloropyrimidin-4-yl)pyrazole-4-boronic acidCatalog No.:AA003CZ2 CAS No.:1072945-81-3 MDL No.:MFCD09972097 MF:C7H6BClN4O2 MW:224.4121 |
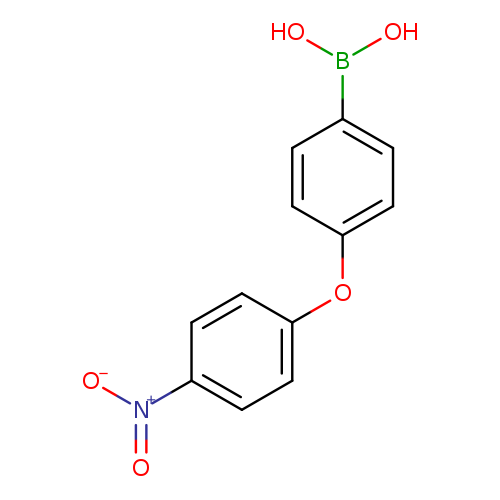
4-(4-Nitrophenoxy)phenylboronic acidCatalog No.:AA0090C5 CAS No.:1072945-82-4 MDL No.:MFCD10699618 MF:C12H10BNO5 MW:259.0225 |
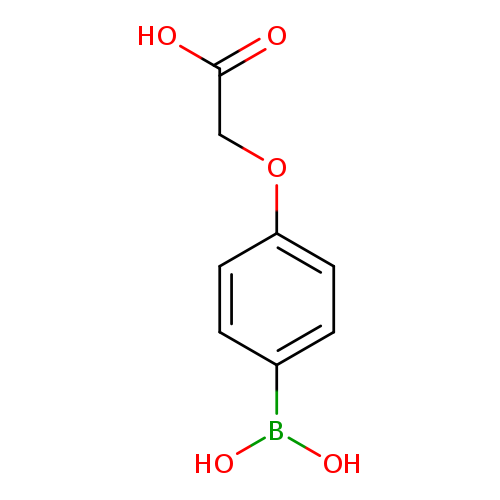
4-(Carboxymethoxy)phenylboronic acidCatalog No.:AA007WLX CAS No.:1072945-84-6 MDL No.:MFCD03425920 MF:C8H9BO5 MW:195.9651 |
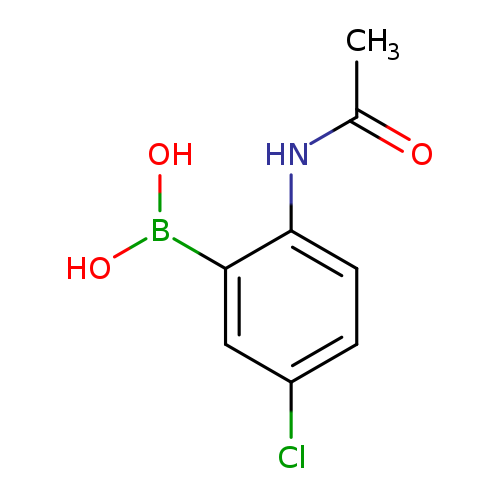
2-Acetamido-5-chlorophenylboronic acidCatalog No.:AA003BIM CAS No.:1072945-85-7 MDL No.:MFCD11504839 MF:C8H9BClNO3 MW:213.4260 |
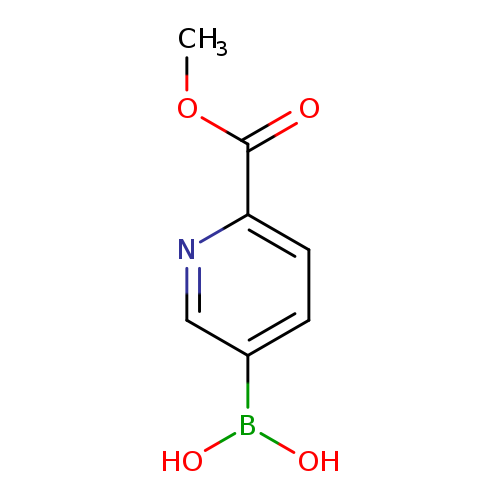
6-(Methoxycarbonyl)pyridine-3-boronic acidCatalog No.:AA007EPL CAS No.:1072945-86-8 MDL No.:MFCD09800559 MF:C7H8BNO4 MW:180.9537 |
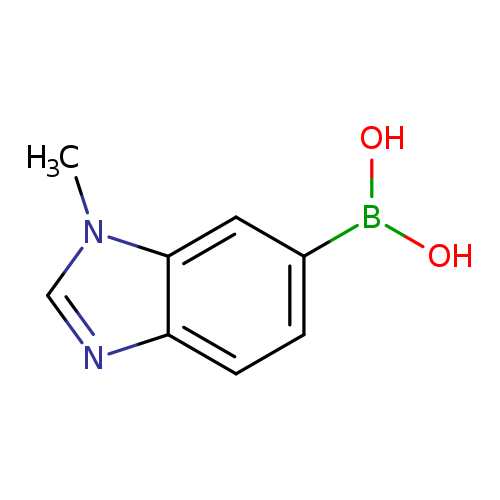
1-Methyl-1H-benzoimidazole-6-boronic acidCatalog No.:AA007WLW CAS No.:1072945-87-9 MDL No.:MFCD09972099 MF:C8H9BN2O2 MW:175.9803 |
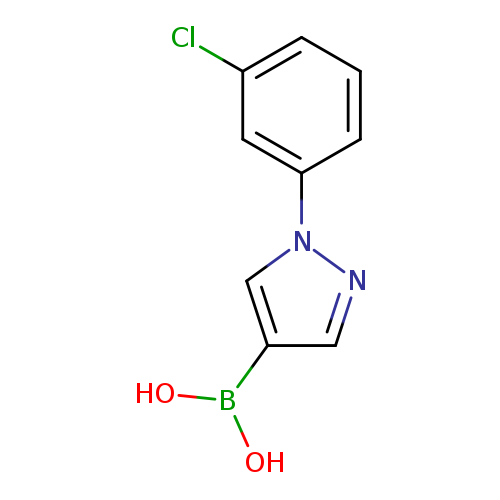
1-(3-Chlorophenyl)pyrazole-4-boronic acidCatalog No.:AA008SSG CAS No.:1072945-88-0 MDL No.:MFCD09878353 MF:C9H8BClN2O2 MW:222.4360 |
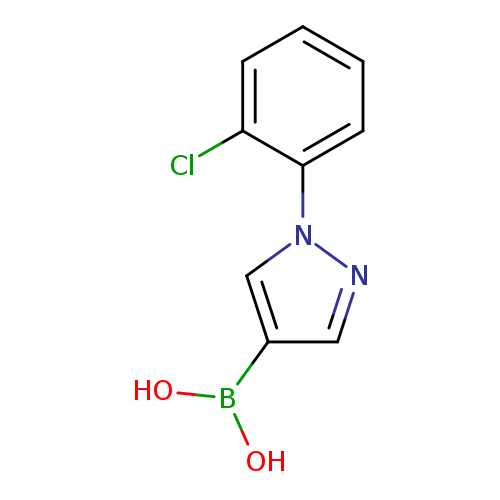
1-(2-Chlorophenyl)pyrazole-4-boronic acidCatalog No.:AA003CRB CAS No.:1072945-91-5 MDL No.:MFCD09972101 MF:C9H8BClN2O2 MW:222.4360 |
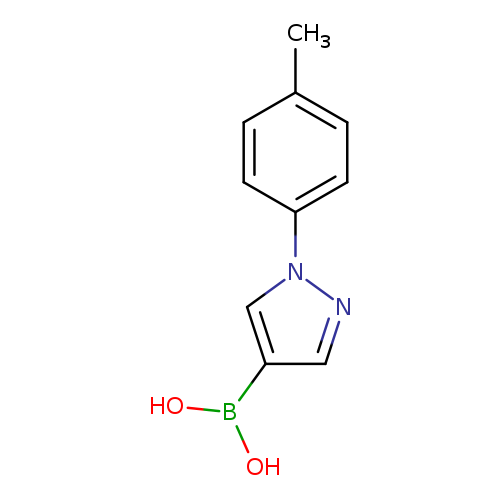
1-p-Tolylpyrazole-4-boronic acidCatalog No.:AA00386F CAS No.:1072945-92-6 MDL No.:MFCD09972102 MF:C10H11BN2O2 MW:202.0175 |
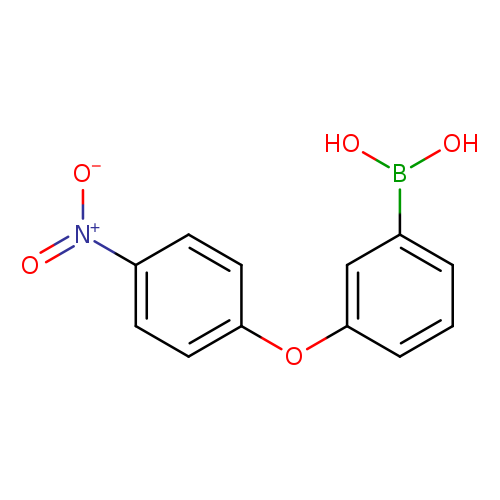
3-(4-Nitrophenoxy)phenylboronic acidCatalog No.:AA007EPK CAS No.:1072945-93-7 MDL No.:MFCD09972103 MF:C12H10BNO5 MW:259.0225 |
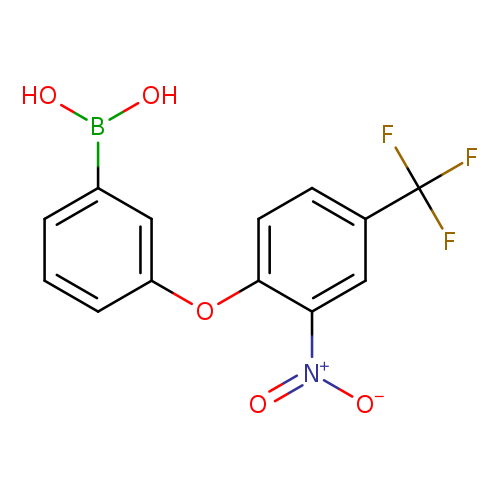
3-(2-Nitro-4-trifluoromethylphenoxy)phenylboronic acidCatalog No.:AA007WLV CAS No.:1072945-94-8 MDL No.:MFCD09972104 MF:C13H9BF3NO5 MW:327.0205 |
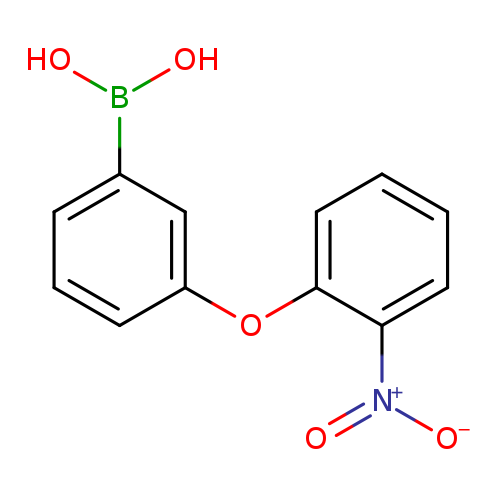
3-(2-Nitrophenoxy)phenylboronic acidCatalog No.:AA007EPJ CAS No.:1072945-95-9 MDL No.:MFCD09972105 MF:C12H10BNO5 MW:259.0225 |
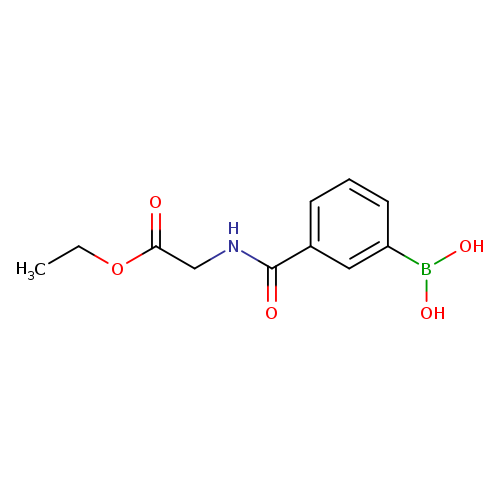
Ethyl (3-boronobenzoylamino)acetateCatalog No.:AA007EPI CAS No.:1072945-97-1 MDL No.:MFCD09878356 MF:C11H14BNO5 MW:251.0436 |
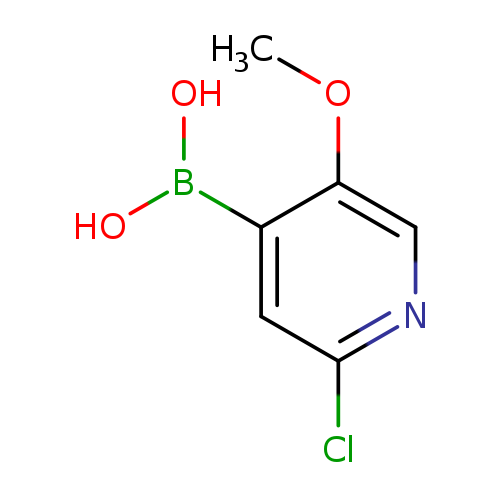
2-Chloro-5-methoxypyridine-4-boronic acidCatalog No.:AA003BK0 CAS No.:1072945-98-2 MDL No.:MFCD09972106 MF:C6H7BClNO3 MW:187.3887 |
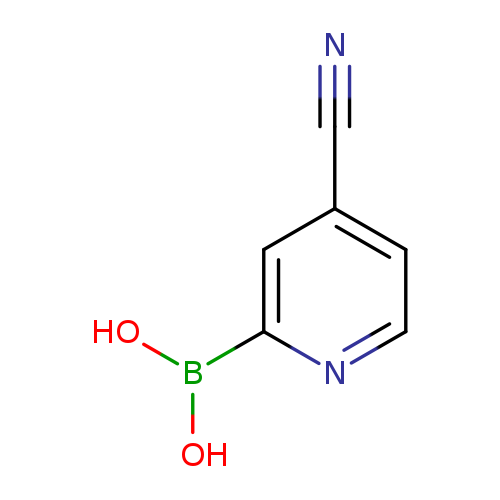
4-Cyanopyridine-2-boronic acidCatalog No.:AA003L6T CAS No.:1072946-01-0 MDL No.:MFCD08703250 MF:C6H5BN2O2 MW:147.9271 |
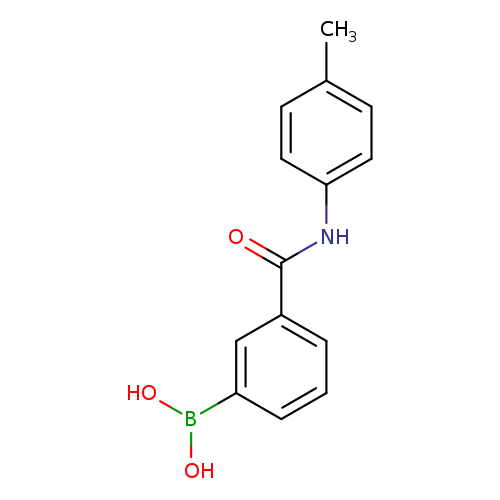
3-(p-Tolylcarbamoyl)phenylboronic acidCatalog No.:AA003I78 CAS No.:1072946-03-2 MDL No.:MFCD10699620 MF:C14H14BNO3 MW:255.0769 |
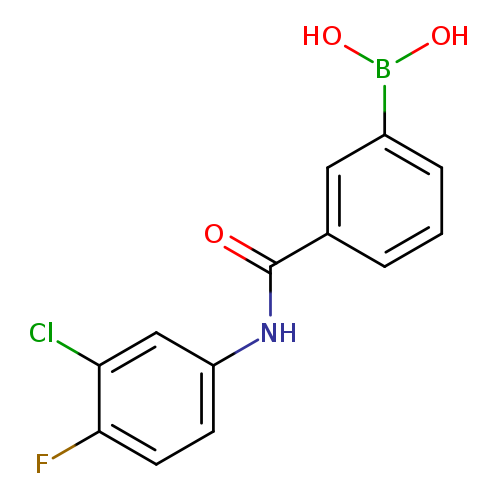
N-(3-Chloro-4-fluorophenyl) 3-boronobenzamideCatalog No.:AA003BN6 CAS No.:1072946-04-3 MDL No.:MFCD09972110 MF:C13H10BClFNO3 MW:293.4858 |
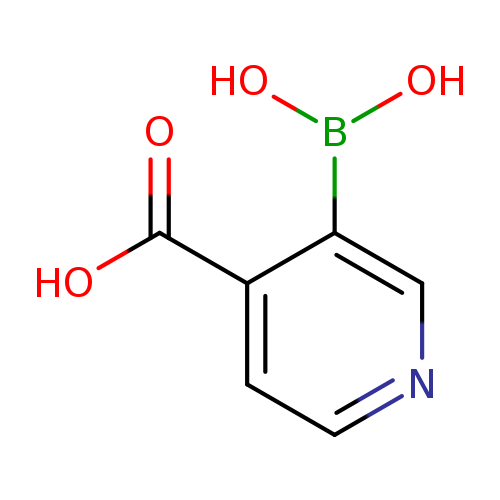
3-Boronoisonicotinic acidCatalog No.:AA003IWD CAS No.:1072946-05-4 MDL No.:MFCD10696649 MF:C6H6BNO4 MW:166.9271 |
3-(3-Isopropylthioureido)phenylboronic acidCatalog No.:AA0090CS CAS No.:1072946-07-6 MDL No.:MFCD09972112 MF:C10H15BN2O2S MW:238.1143 |