2020-01-19 09:22:33
Kai-Xiong Ye+, Ting-Ting Fan+, Lawrence Jordan Keen, and Bing-Nan Han
1.Introduction
1.1Development of Marine Pigments
Natural pigments, a surprisingly indispensable part of human life, play an important role in food, cosmetic, medical treatment and textile industries. The first use of pigment can be dated back to ancient Egypt for mainly decorative applications. Due to the lack of natural pigments sources and the complexity of extraction process, they would not seek much attention until 1856, when Sir Willey Henry invented the first artificial colour-mauvine, after that began the age of synthetic pigments.[1] However, as people became more aware of the potential harmfulness of synthetic pigments, natural pigments return to the forefront.
In recent times, natural pigments are mainly extracted from some terrestrial plants, but their sources and production remain limited. Since the 1960’s, prostaglandin precursors have been found highly bioactive in the soft coral species Gorgonacea, leading many natural product chemists to move their research from the land to the sea. The oceans cover more than 70 percent of the earth‘s surface and are humanity‘s largest repository of resources. In fact, pigments were extracted from marine organisms in the early days, but due to underdeveloped science and technology of the time, people did not have a deep understanding of them. As an example, astaxanthin, first isolated from shrimp shells in 1930, has been subjected to extensive research since the 1980s for its obvious biological functions: enhancing immunity system, protecting the retina from radiation, antioxidization, anti-inflammation, and preventing blood LDL-cholesterol from oxidization. Artificial synthesis still dominates astaxanthin production, however, the green microalgae Haematococcus pluvialis and the red yeast Phaffia rhodozyma have begun to compete economically with synthetic astaxanthin.[2]
1.2Examples of Pigment Resources
In recent years, natural pigments from marine animals such as fish and invertebrates have attracted considerable attention, and a number of novel classes of natural pigments with potential economic importance have been reported.[3] Marine invertebrates, algae and microbes are the three main sources of marine pigments. Among marine animals, the pigments of astaxanthin that are produced by crustaceans and echino- chrome produced by echinoderms are known most widely. For marine plants, some algae can also produce pigments in addition to photosynthetic pigments produced by phytoplank- ton. Pigments from marine plants are divided into three categories: chlorophyll, carotenoid and phycobiliproteins. Among these pigments, chlorophyll, fucoxanthin, lutein and phycocyanin are best known. The pigments from the source of microorganisms are relatively abundant, and the microorgan- isms that can produce pigments mainly include bacteria, cyanobacteria, actinobacteria, yeast, and fungi. These marine microorganisms are isolated from different places, including algae, fishes, molluscs, sponges, mangroves, seawater, and sediments and the compounds of pigments are various, including carotenoids, indole derivatives, polyenes, alkaloids, peptides, macrolides, melanins, monascins, and terpenoids.[4]
Microorganisms have always been known to be an extremely diverse group and that is also seen in the wide array of colours they produce. Briefly, we list some example of different coloured pigments produced by a variety of micro- organisms: Miao et al extracted prodigiosin-like pigments from the marine bacteria Serratia proteamacula 657;[5] Ashley Franks et al. isolated a new yellow pigment from Pseudoalter- omonas tunicata;[6] several purple pigments were produced from Alteromonas species isolated from Kinko Bay in Kagoshima Prefecture, Japan;[7–9] melanin or melanin-like pigments were produced by marine bacterial strains Vibrio cholerae, Shewanella colwelliana, and Alteromonas nigrifa- ciens.[10–13]
2.Marine Animal Sources
Marine animals, especially those from tropical waters, are usually brilliantly coloured and that bright colouration is widespread in invertebrates. These spectacular natural colours are common among species inhabiting in shallow water areas, but also occur in animals living in deep dark areas where sunlight cannot reach them.[3] Natural pigments isolated from marine animals mainly include quinone, carotenoid and tetrapyrrole compounds, which are mainly derived from invertebrates such as echinoderms (sea urchins), molluscs (mussels) and crustaceans(crabs).
2.1 Echinochrome and Spinochromes
Echinochromes and spinochromes are the main natural pig- ments of quinones from marine sources as well as a group of 5,8-dihydroxy-1,4-naphthoquinones known as naphthazarins. Echinochrome is the name which was given by MacMunn [1885, 1889] to a pigment contained in the cells of the perivisceral fluid of Strongylocentrotus lividus, Amphidotus cordatus, E. sphaera and E. Esculentu.[14] It has been reported to have several protective biologic effects: including antiox- idant properties; scavenging reactive oxygen species (ROS); and chelating iron.[15–16]
Two new spinochromes, echinamines A (1) and echin- amines B (2), were isolated from the sea urchin Scaphechinus mirabilis at a depth of 5 m off Peter the Great Bay of the Japan Sea, in 2004.[17] Echinamines A was obtained as a dark brown powder, while B was obtained as dark brown needles. UV/vis spectra of Echinamines A and Echinamines B showed absorption maxima at lmax 274, 345, and 479 nm, corresponding to those for polyhydroxy- naphthazarines. Echinamines A and B were shown to have 50 % radical scavenging ability (EC50) at a concentration of 0.01 mM against 0.1 mM DPPH in ethanol. Lola Brasseur et al. extracted Echinochrome A and four Spinochromes (Spinochrome A, Spinochrome C, Spino- chrome D, Spinochrome E) (3–6) from four common normal sea urchins (Echinometra mathaei, Diadema savignyi, Trip- neustes gratilla and Toxopneustes pileolus) collected in the Indian Ocean reef shoals, in 2017.[18] All extracts showed antibacterial activity against at least one bacterial strain (E. coli, B. subtilis, V. aestuarianus, C. marina and S. oneidensis). Meanwhile, all isolated spinochromes showed high antioxidant effects and cytotoxic activity. Da-yong zhou et al. extracted Spinochrome A, Spinochrome C, Spinochrome E and Echin- amines A from sea urchins collected in the yellow sea of China and also extracted Spinochrome B (7).[19] It was found that the compound had the largest ultraviolet absorption peak at about 470 nm as well as having DPPH radical scavenging activity, Fe2 + chelating activity and reducing power.
2.2 Carotenoids
Carotenoids, commonly found in the yellow, orange or red pigments of higher plants, fungi and algae, are classed as antioxidant compounds that assume a key role in cell protection. In fact, the ability to quench singlet oxygen molecules, capture light and protect photosynthesis are the most relevant biological functions of carotenoids.[20] Studies have found that carotenoids can also be isolated from some marine animals. R. C. Symonds isolated two carotenoids from sea urchins Paracentrotus lividus collected at two locations along the shoreline of Bantry Bay.[21] The carotenoids were breakdown products of fucoxanthin (9); namely fucoxanthinol and amarouciaxanthin A. Mytiloxanthin (10) distributed in several marine invertebrates such as shellfish and tunicates. It was reported that mytiloxanthin was converted from fucox- anthin through a pinacol-like rearrangement. Mytiloxanthin presents as yellow and the maximum UV-VIS absorption values of the mytiloxanthin is 484 nm.[22] Takashi Maoka et al. synthesized mytiloxanthin and studied its anti-oxidative activities and found that the compound showed excellent scavenging activity for hydroxyl radicals.[23] Caroline Ute- rmann et al. isolated an Alloxanthin analogue (8) from mussels (Mytilus spp.) collected at an organic blue mussel farm located in KF (Baltic Sea, Germany).[24] Furthermore, Daniel Garama et, al. isolated seven carotenoids from sea urchins (Evechinus chloroticus) collected from Doubtful Sound, Fiordland (New Zealand).
2.3Other Pigments from Marine Animals
Another viable source of pigments is from marine inverte- brates found throughout the ocean. Caroline et al. isolated not only carotenoids but also two tetrapyrroles (Chlorophylone-a a(11) and Pyropheophorbide-a(12)) and a Porphyrin pigment Corallistin B (13) from mussels (Mytilus spp.) collected at an organic blue mussel farm located in KF (Baltic Sea, Germany).[24] Chlorophyllone-a (11) is a liposoluble pigment, that presents as a dark green colour and its maximum absorbance includes 408, 503, 534, 608, 665 nm. Interestingly it also has high antioxidant activity.[26] Pyropheophorbide-a
(12) is a seaweed chlorophyll derivative and presents as a liposoluble dark green pigment.[27] It is photosensitive and unstable under light conditions, therefore often chosen to be a photosensitizer. The absorption maximum of pyropheophor- bide a (12) is at 668 nm.[28] Plankinidine D (14) isolated from a Plakortis sp. sponge collected near the island of Rota, Northern Mariana Islands in 1996, is a polycyclic heteroar- omatic compound representing the first member of the pyrroloacridine class of marine alkaloids. Plankinidine D (14), a red-orange liposoluble solid and its maximum absorbance are 328, 388, 436, 514 nm. It can resist human colon cancer cells in vitro. In addition, studies have found that some deep- sea fish are the main sources of fluorescent pigments.[29–39]
R.H. Douglas et al. discovered that the deep-sea stomiid Malacosteus niger, which produces far red bioluminescence, has two visual pigments within its retina which form a rhodopsin/porphyropsin pigment pair with lmax values around 520 and 540 nm.[31] At the same time, they also found long- wave-sensitive visual pigments in its retina to enhance its sensitivity to longwave radiation such as its own biolumines- cence. These liposoluble pigments subsequently identify as a mixture of defarnesylated and demetallated derivatives of bacteriochlorophylls c (15) bacteriochlorophylls d (16) with lmax at 670 nm and found to be used as a photosensitiser to enhance its sensitivity to longwave radiation.
3.Marine Plant Sources
Pigments from marine plants are mainly found in phytoplank-ton and algae. The pigments are mainly divided into three groups: chlorophylls (a, b, c1, c2, c3, d), carotenoids (carotenes and their oxygenated derivatives known as xantho- phylls), and phycobiliproteins (allophycocyanins, phycocya- nins, phycoerythrins).
3.1Chlorophyll and its Derivatives
Chlorophylls are the famous photosynthetic greenish pigments found in algae, plants, and cyanobacteria. Chlorophylls contain a porphyrin ring and belong to a major class of tetrapyrroles.[33] Chlorophyll-a is present abundantly in the marine environment and always found in marine algae and cyanobacteria. Ruairi C. Robertson et al. isolated some anti- inflammatory lipids and chlorophyll-a (17) from three samples of red algae (Porphyra dioica, Palmaria palmata and Chondrus crispus) collected in Galway bay.[34] Chlorophyll-a is a blue/green colour pigment with maximum absorbance from 660 to 665 nm and plays an important role in marine plants photosynthesis.[35] Chlorophyll-b (18) is abundant in marine plankton, so it is one of the five most prominent biomarkers (The other four biomarkers are fucoxanthin, peridinin, 19’-hexanoyloxyfucoxanthin and alloxanthin).[36] Chlorophyll-b is a green/yellow colour pigment and its maximum absorbance is 642 to 652 nm. Chlorophyll-c(19–21) is an accessory blue-greenish colour pigment with maximum absorbance from 447 to 452 nm and it can be found in many algae (marine green Ulva intestinalis and brown macroalgae Sargassum angustifolium).[37] Chlorophyll-d (22) is commonly found in plants, algae and other cyanobacteria. Michael Ku¨ hl et al. isolated chlorophyll-d from the cyanobacteria, Acaryo- chloris marina, and found that the cyanobacterium uses chlorophyll-d as its principal light-harvesting pigment instead of chlorophyll-a.[38] Chlorophyll-d absorbs far-red light, at the 710 nm wavelength.[39] Y. Kamei et al. isolated a chlorophyl- c2 derivative from marine brown algae (Eisenia bicyclis) collected from the Japanese coastline.[40] The derivative named MC15(23) and found that the UV=VIS spectrum of MC15 in diethyl ether showed maximum absorption at 437 nm. MC15 showed similar antiviral activity against other salmonid enveloped viruses such as Paralichthys olivaceus virus and Oncorhynchus masou virus, and stability against any pH and temperatures up to 1008C.
3.2 Carotenoids
Carotenoids are the most common pigments in the marine environment. They are generally biosynthesized by all auto- trophic marine organisms; therefore, they can be easily found in marine plants. The most common carotenoids in marine plants include: fucoxanthin, astaxanthin, zeaxanthin, lutein, neoxanthinand violaxanthin.[41]
Fucoxanthin (9) is an orange-coloured pigment with the maximum absorption wavelength of at 447 nm, present in Chromophyta (Heterokontophyta or Ochrophyta), including brown seaweeds (Phaeophyceae) and diatoms (Bacillariophy- ta). Fucoxanthin was first isolated from the marine brown seaweeds Fucus, Dictyotaand Laminaria by Willsta¨tter and Page in 1914.[42] Fucoxanthin has been isolated from multiples sources each showing significant biological activity: such from Undaria pinnatifida exhibiting strong antiproliferative activity in 1999;[43] isolates from Hijrkia fusiformis displaying strong DPPH radical scavenging activity;[44] or efficient antioxidant properties in extracts from algae of the coast of Jeju Island, Korea in 2008.[48] Together, these suggest the potential of fucoxanthin as a candidate for medical benefits in a wide range of areas.[46]
Zeaxanthin (24), the yellow carotenoid can be found in marine algae, such as Spirulina and red algae.[47–49] It also can be isolated from the diatom (Phaeodactylum tricornutum). Zeaxanthin and lutein are xanthophylls considered to play a potential role in maintaining eye health.[50]
Waesarat Soontornchaiboon et al. isolated a red pigment violaxanthin (25) from microalgae (Chlorella ellipsoidea) obtained from the Daesang company with the maximum absorption wavelength at 441 nm.[51] Studies have shown that violaxanthin has strong antiproliferative activity against McF- 7 human breast cancer cells grown in vitro, which suggests that violaxanthin should be an active compound for induction of apoptosis.[52]
Weiqi fu et al. isolated the yellow pigment lutein (26) from the green microalgae (Dunaliella salina) obtained from the University of Texas at Austin.[53] Rhesa Pramudita Utomo et al. also isolated lutein from chlorella sp. with a maximum UV-VIS absorption value of 444 nm. Lutein has been described as a protective compound against the early stages of the atherosclerotic process and be widely consumed as food colourant in the last decade.[54]
Chiasa Uragami et al. obtained two neoxanthins (all-trans neoxanthin and 9’-cis neoxanthin) (27–28) from a siphonous green alga (Codium (C.) intricatum) isolated from Okinawa prefecture in Japan. These two compounds were then re- isolated from a siphonaceous marine green algae(Bryopsis corticulans)collected from intertidal zones around seashore of Qingdao city, China.[56] These two neoxanthins are liposoluble and present as green compounds, whilst showing moderate thermal stability, prone to oxidation and fading, and are easily isomerized under the action of heat, acid or light. Chiasa Uragami et al. found that these two neoxanthins are unstable under high-light conditions, when cultured under high irradi- ance, 9’-cis neoxanthin will convert to all-trans neoxanthin. The maximum ultraviolet absorption peaks of these two compounds in ethanol solution were 441 nm and 439 nm.[55]
Paulina Kuczynska et al. recorded that multiple carote- noids, including b-carotene (29), antheraxanthin (30), Diadinoxanthin(31)and Diatoxanthin(32), can be isolated from diatoms (Phaeodactylum tricornutum).[57] The maximum ultra- violet absorption of the four carotenoids was 453 nm, 446 nm, 448 nm and 454 nm, respectively. Duc Tranet al. isolated b- carotene from green algae (Dunaliella salina DCCBC15) provided by Dr. E.W. Polle, department of Biology, Brooklyn College of CUNY Brooklyn, NY (USA).[58] b-Carotene because of its nontoxic and antioxidative properties, have found applications in the food, drug and cosmetic industry.
Astaxanthin (33) a red liposoluble pigment, mainly found
in crustaceans (such as shrimp), algae (such as Haematococcus pluvialis and fungi) and Haematococcus pluvialis.[59–61] The compound is a ketone-type non-vitamin A protocarotenoid with various biological functions such as colouring, anti- oxidation, photoprotection, promotion of reproduction, enhancement of immunity, and maintenance of central nervous system health. The maximum UV-VIS absorption value of it in trichloromethane is 489 nm.[62–63] Due to the long conjugated unsaturated double bond in the molecular structure of astaxanthin its properties are extremely unstable. Oxygen, light, heat and metal ions can cause damage to astaxanthin, causing it to oxidize or degrade.[64] Astaxanthin possesses two identical asymmetric carbon atoms at C-3 and C-3 making three possible isomers with all-transconfiguration of the chain: 1.) (3S, 3s)-astaxanthin, isotated from lobster, salmon and the spider mite; 2.) (3R, 3R)-astaxanthin isolated from the red yeast Phaffia rhodozyma, which presents partial antioxidant activity; 3.) meso-form (3S, 3’R)-astaxanthin, which has not been found in nature. Canthaxanthin (34) is a natural diketo derivative of b- carotene primarily used by the food and feed industries.[65] Canthaxanthin, along with astaxanthin, lutein and b-carotene, was isolated from microalgae (Coelastrella sp. F50) collected in tropical Taiwan.[66] Canthaxanthin is used as a lipid-soluble natural pigment and has excellent antioxidant activity com- pared to carotenoids such as b-carotene.[67]
3.3 Phycobiliproteins
Phycobiliproteins are composed by a protein and a chromo- phore part (linked by covalent bonds) called phycobilin. Phycocyanin (35) from Arthrospira and phycoerythrin (36) from Porphyridium are two of the most known phycobilipro- teins.[68] The maximum ultraviolet absorption peaks of two phycobiliproteins were 540–570 nm and 610–620 nm, respec- tively. Owning to its strong fluorescence and easy combination with the isotope and biotin, phycobiliproteins has been developed as the fluorescence probe. With the development of single cell protein (SCP) from algae, phycobiliproteins has also been used widely in fields of the medicine, pharmaceut- ical, chemical industry, and medicinal diagnose.
4. Marine Microorganism Sources
Marine microorganisms, one of the main sources of natural pigments, are often isolated from algae, sponges, mangroves, and sediments. These pigments are mainly divided into indole derivatives (quinones and violacein), alkaloids (prodiginines and tambjamines), polyenes, macrolides, peptides, and terpe- noids.[4] Importantly, these compounds present different bio- logical properties.
4.1 Prodiginines
Bacteria as an important source of red pigments was first seen when prodigiosin compounds were first isolated from Serratia marcescens. The common aromatic chemical structure of these pigmented compounds was first named prodiginine by Gerber.[69] Prodiginines mainly includes compounds similar in chemical structures to compounds (37–39). A novel marine bacterium strain, Vibrio gazogenes, effectively produced prodiginine type pigments, including the typical prodigiosins compound 37 and 38 as well as two new prodiginines (40– 41).[70] These two new pigments had maximum absorption peaks at 533 nm and these colourants could dye wool, silk and synthetic fabrics such as polyester and polyacrylic and showed antibacterial properties against Escherichia coli and Staph- ylococcus aureus bacteria. Li Houjin et al. isolated two kinds of prodigiosin, prodigiosin A (42) and new derivative dimethyllinosin B (43) from the bacterial Pseudomonas sp. isolated from the surface of the sea water in Daya Bay, Guangdong province.[71] A marine bacterium (Serratia protea- macula 657) was screened out from 251 marine sponge- associated bacteria originated from San Juan Island and a prodigiosin-like pigment (44) with highly antitumor activity was isolated,[5] showing strong inhibition of HeLa and MGC803 cell lines. A gram-negative, red-pigment-producing marine bacterial strain (Zooshikella rubidus S1-1), was isolated from tidal flat sediment in Yellow Sea, Korea[73] with two red pigments (45–46) being extracted. These pigments are both prodigiosins and showed antimicrobial activity against Bacillus subtilis, Escheria coli, Salmonella serovar Typhimu- rium, Staphylococcus aureus, and Candida albicans. Interest- ingly they also both have anticancer activity to two human melanoma cell lines, SKMEL-28 and A375P.
4.2 Carotenoids
Carotenoids as terpenoids are also abundant in marine micro- organisms. Astaxanthin (33) can also be isolated from marine microorgannisms such as being produced by the marine bacterium, Paracoccus haeundaensis.
Two novel Carotenoids (47–48) were isolated from marine bacteria (Bacillus indicus HU36).[75] The maximum UV-VIS absorption values of the two carotenoids were 454 nm and 468 nm respectively. The carotenoids and phenolic antioxi- dants displayed synergistic activities in the inhibition of linoleic acid peroxidation induced by heme iron, but not by free iron.
4.3 Violacein
The violet pigment violacein is an indole derivative, predom- inantly isolated from bacteria of the genus Chromobacte- rium.[76] Violacein has a variety of biological activities, including antiviral, antibacterial, antiulcerogenic, antileishma- nial, and anticancer properties.[77–80] Hamilton and Austin first isolated violacein from marine bacteria (Chromobacterium marinum).[81] A marine bacterium (Pseudomonas sp.) was isolated from the surface of the sea water of Daya Bay in the south China sea.[82] The bacterium can produce the violacein Blue-1 (49). A series of anti-tumour activity experimental results showed that Blu-1 had a strong inhibition on tumour cell growth. The IC50 of against MCG803 and BEL7402 were
4.6 and 6.8 ug/ml respectively. Hideki Kobayashi et al. have purified a new violet pigment derived from Shewanella violacea DSS12 isolated from the deep-sea sediment of the Ryukyu Trench at a depth of 5,110 m.[83] The absorption maximum of this violet pigment (50) in THF was 616 nm and 636 nm in chloroform. The violet pigment of DSS12 was very stable and showed no antibiotic activity to Escherichia coli, but this was dependent on low permeation into cells because of the crystals’ insolubility in water or hydrocarbons.
4.4 Phenazine Compounds
Phenazines are redox-active, small nitrogen-containing aro- matic compounds produced by a diverse range of bacterial genera. Maskey et al. reported the isolation of two yellow pigments from the marine Pseudonocardia sp. B6273, a member of the Actinomycetes. Structural investigations identi- fied the two pigments as novel phenazostatin D(51) and phenazostatin B(52), and found to be inactive against the tested microorganisms, and methyl saphenate, a known phenazine antibiotic.[84] Phenazostatin B possessed a positive optical rotation at 365 nm. Li et al. also reported seven new oxidized and reduced phenazine-type pigments, dermacozines A–G (53–59) extracted from strains MT1.1 and MT1.2. All pigment molecules had the maximum ultraviolet absorption at 520 nm.[85] The bacteria were isolated from Mariana Trench sediment at a depth of 10898 m. Dermacozines F (58) and G(59) exhibited moderate cytotoxic activity against leukaemia cell line K562 with IC50 values of 9 and 7 mM, respectively, while the highest radical scavenger activity was observed for dermacozine C (50) with an IC50 value of 8.4 mM. The novel 5, 10-dihydrophencomycin methyl ester (60) and phencomycin(61) were isolated from an unidentified marine Streptomyces sp..[86] Compound (60) shows weak antibiotic activity against Escherichia coli and Bacillus subtilis.
4.5 Tambjamines
Tambjamines are alkaloids always isolated from various marine organisms like bryozoans, nudibranchs, and asci- dians.[87–88] A yellow pigment (62) was isolated from Pseudoal- teromonas tunicata and was identified as a new member of the tambjamine class of compounds.[89] P. tunicata has the highest and broadest range of biological activities which linked to the production of the yellow pigment.[90] David M. Pinkerton etal conducted a series of biological activity evaluation experi- ments on compond (62) showing antibacterial, antifungal effects and cytotoxic activity. The IC50 against human cancer cell lines HL-60 (promyelocytic leukaemia cells), MDA-MB-435 (derived from the M14 melanoma cell line), HCT-8 (ileocecal colorectal adenocarcinoma cell line), and SF-295 (glioblastoma cell line) as well as PBM cells are 0.69, 1.11, 0.98, 0.83, 0.64 mg/ml, respectively.
4.6 Quinones
Quinone derivatives range in colour from yellow to red, exhibit antiviral, anti-infection, antimicrobial, insecticidal, and anticancer activities, and have many commercial applications as natural and artificial dyes and pigments.[92] Two new anthracycline antibiotics, designated as himalomycin A (63) and B (64), were isolated from the culture broth of the marine Streptomyces sp. isolate B6921, derived from sandy sediment of a coastal site of Mauritius (Indian Ocean).[93] These two compounds are liposoluble yellow solid, with their maximum absorbance in MeOH at 254, 290, 441 and 256, 292, 434 nm, respectively. At concentrations of 50 mg/disk, compound 63 and 64 both exhibited strong antibacterial activity against Bacillus subtilis, Streptomyces viridochromogenes, Staphylo- coccus aureus and Escherichia coli.
Two novel pigmented antitumor, antibiotics, ellowish brown liposoluble solid chinikomycin A (65) and red liposoluble solid chinikomycin B (66) were isolated from a marine Streptomyces sp. Strain M045.[94] Chinikomycins A (65) and B (66) both exhibited moderate antitumor activity, Chinikomycins A (65) being significantly more potent. It selectively inhibited proliferation in cell lines of mammary cancer (MAXF 401NL, IC50 =2.41 mg/mL), melanoma(MEXF 462NL, IC50 =4.15 mg/mL) and renal cancer (RXF 944 L, IC50 = 4.02 mg/mL). The Chinikomycins B (66) showed selective antitumor activity against the mammary cancer cell line MAXF 401NL (IC50 = 3.04 mg/mL).
4.7 Azaphilones
Azaphilones are a family of fungal pigments characterized by a highly oxygenated pyrano-quinone bicyclic core. The coloured azaphilone derivatives are produced by species of ascomyceteous and basidiomyceteous fungi, including the genera Penicillium, Aspergillus, Chaetomium, Talaomyces, Emericella, Epicoccum, Pestalotiopsis, Phomopsis, Monascus and Hypoxylon.[95] One yellow new azaphilone (67), along with six known analogues (68–73), were isolated from the fungus Penicillium 303#, which was collected from sea water of Zhanjiang Mangrove National Nature Reserve in Guang- dong Province, China, in 2014. Evaluation of cytotoxicity of these compounds found that compound 69 showed potent cytotoxic activity with its IC50 values of 7.13 mM against MDA-MB-435.[96] Moreover, Weiyi Wang et al. isolated four new azaphilones, chaephilone C (74), chaetoviridides A–C (75–77) from a Chaetomium sp. strain NA-S01-R1 isolated from the seawater sample at a depth of 4050 m in the West Pacific Ocean in 2017.[95] Except chaephilone C is yellow, the other three compounds are all red. Through antibacterial assay and cytotoxicity assay found compounds 74, 76 and 77 displayed similar anti-MRSA activities in comparison to chloramphenicol and compounds 74 and 76 demonstrated relatively stronger cytotoxic activities than the other com- pounds against HeLa cell. In addition, compound 75 showed the most potent cytotoxic activities towards Hep G2 cell with IC50 below 5 mm 4.8 Other Pigments from Marine Bacteria Rui He et al. constructed a metagenomic library of the Japanese marine sponge discodermia calyx and screened out two clones producing porphyrins (78) as red pigments. The pigment has the UV absorption maximum at 405 nm.[97] The ability of porphyrins to generate reactive oxygen species had been utilized in cancer therapy. In addition, vitamin B12 composed of a porphyrin skeleton has been produced by a biotechnological process using bacteria.
A yellow pigment (79) was isolated from marine bacterial strain Vibrio sp.,[98] isolated from water samples of the Arabian Sea. This pigment has antioxidant activity and shows significant inhibition of Gram-positive organisms (MIC=31.25 to 62.5 mg/ml) in the attribute biological properties against pathogenic bacteria. Scytonemin (80) is a yellowgreen pigment isolated from aquatic cyanobacteria and its maximum UV absorption at 370 nm.[99] Scytonemin is an important UV- radiation protective synthesised biomolecule. It protects bacteria by preventing about 85–90 % of all UV-light from entering through the cell membrane.[100] Grossart H P et al. extracted a blue pigment named Glaukothalin from Two g-Proteobacteria strains, isolated from the German Wadden Sea and the Øresund, Denmark, respectively. Glaukothalin is readily soluble in pyridine or HMPT, moderately soluble in DMSO, DMF or CHCl3, and insoluble in acetone, methanol, water (acidic or alkaline), benzene or cyclohexane. The UV spectrum of glaukothalin in CHCl3 exhibits characteristic absorption maxima at 636 nm (log e= 4.51), 582 nm (sh), 286 nm (sh), 241 nm. The molecular formula of Glaukothalin is C34H56N4O4, but the structure of this compound has not been reported so far.[101] AM13,1 strain, which was identified to belong to the Cytophaga/Flexibacteria cluster of North Sea bacteria, was found to produce yellow liposoluble tryptanthrin (81). This pigment is anti-fungal and anti-microbial and was found to show an antibacterial activity against Bacillus subtilis and demonstrated its hitherto unknown activity against dermatophytes. Its absorption maximum in MeOH were 248, 252, 277, 311, 328 nm and vis lmax at 387 nm.[102]
5.Future Prospects & Biotechnology
Although marine pigment sources are very extensive, some pigments are far from reaching the goal of mass production, especially those from marine animals. Use of microorganisms have advantages over marine animal sources due to simple culture conditions and rapid propagation; therefore, the pig- ment can be produced in high quantity in a short time by fermentation. In recent decades, scientists have made break- throughs in pigment production using genetic engineering and microbial fermentation. A novel gene involved in ketocom- pound biosynthesis, designated as crtW, was isolated from the marine bacteria Agrobacterium aurantiacum and Alcaligenes PC-1. When this gene was introduced into Escherichia coli, that accumulated b-carotene due to the Erwinia carotenogenic genes, the E. coli transformants synthesized canthaxanthin, one of ketocarotenoids.[103]
Carotenoids are the most omnipresent pigments in the oceans with important biological roles such as light capture and antioxidative activities and have made great achievements in biosynthesis. The early attempts led to the production of lycopene, b-carotene, and astaxanthin in Saccharomyces cerevisiae and Candida utilis by the expression of caroteno- genic enzymes from Pantoea ananatisi.[104–105] The lycopene synthetic pathway was engineered in Escherichia coli using the carotenoid genes (crtE, crtB and crtI) of Pantoea agglomerans and Pantoea ananatis. A two-fold higher lycopene production is obtained in E. coli by the expression of carotenogenic enzymes from P. agglomerans (27 mg/L) than from P. ananatis (12 mg/L).[106]
To improve the efficiency of carotenoid production, the biological system of the host organism also needs to be optimized. Direct efforts were focused on the modification of associated genes to these pathways. For example, deletion of pyruvate kinases PykFA can balance the availability of pyruvate and G3P for the MEP pathway, and increase lycopene production by 2.8-fold in E. coli.[107]
Astaxanthin, as previously mentioned, has multiple poten- tial uses due to its role as an antioxidant therefore it is under scrutiny by the biosynthetic industry. For example, an astaxanthin producing Saccharomyces cerevisiae strain was created by successively introducing the Haematococcus pluvialis b-carotenoid hydroxylase (crtZ) and ketolase (bkt) genes into a previously constructed b-carotene hyperproducer.
Through codon optimization, gene copy number adjustment and iron cofactor supplementation led to significant increase in the astaxanthin production, reaching up to 4.7 mg/g DCW in the shake-flask cultures which is the highest astaxanthin content in Saccharomyces cerevisiae reported to date.[108]
Recently, the production of astaxanthin is mainly based on microbial fermentation, including yeast and algae. Interest- ingly, astaxanthin from yeast is 100 % right-handed (3R-3’R) with partial antioxidant activity, while astaxanthin from algae is 100 % left-handed (3S-3’S) with the strongest biological activity. Since 1990, Roche began a large-scale production of synthetic astaxanthin and practically fulfilled the world market for the pigment, estimated at 150–200 million dollars.
Due to harsh and extreme environments, marine organisms have developed unique adaptation mechanisms and metabolic pathways to survive. Most studies investigating marine organisms have shown that they have the excellent prospects in producing pigmented metabolites, which are found to have antibiotic, anticancer, and immunosuppressive activities. As these pigments have diverse and promising activities for different diseases, they can play an important role in both medical and agricultural research.
Overall, this review underscores the importance of finding new pigments in the vast oceans of the planet. These pigments have many bioactive properties and continue to provide promising avenues for basic science and applied biomedical research.
Acknowledgements
We acknowledged the grants supports from the National innovative and Entrepreneurship Training Program for College Students in China (201710338015), Agro-scientific Research in the Public Interest of Zhejiang province [grant number LGN18C190011], Science Foundation of Zhejiang Sci-Tech University [17042058-Y], 13th Five-year Plan Teaching Re- form Research in Higher Education of Zhejiang province [jg20180092] and Project for Jiaozhou Excellent Innovation Team [grant number 18-CX-1].We also acknowledged students of the Department of Development Technology of Marine Resources, College of Life Sciences, Zhejiang Sci-Tech University, Muhammad Ridwanur Rahman, Zengyi, Meng Fengbin, Ying Junjie, Ji Xiaofeng, Sun Shaokang, Li zhi, Song Jiaying, Meng Yuan, Wang Gaoyang, Zhang Shunli, Lei Yutong, Zhang Meiling, Yang Tianyong, Shi Xuhui, Huang Chaoyu, Ren Qingyu and Zhang Mengying, for their help in preparing this article.
4-Methyl-2-pentanolCatalog No.:AA0037XA CAS No.:108-11-2 MDL No.:MFCD00004550 MF:C6H14O MW:102.1748 |
Isovaleryl chlorideCatalog No.:AA0034ZN CAS No.:108-12-3 MDL No.:MFCD00000738 MF:C5H9ClO MW:120.5774 |
MalonamideCatalog No.:AA00351M CAS No.:108-13-4 MDL No.:MFCD00008034 MF:C3H6N2O2 MW:102.0919 |
1-(Dimethylamino)isopropylamineCatalog No.:AA003D0D CAS No.:108-15-6 MDL No.:MFCD00014812 MF:C5H14N2 MW:102.1781 |
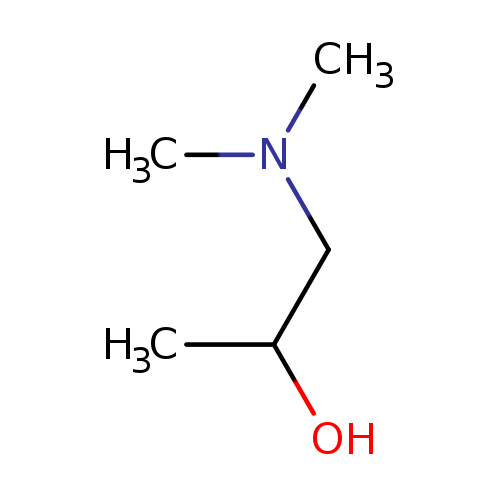
1-Dimethylamino-2-propanolCatalog No.:AA003AYT CAS No.:108-16-7 MDL No.:MFCD00004532 MF:C5H13NO MW:103.1628 |
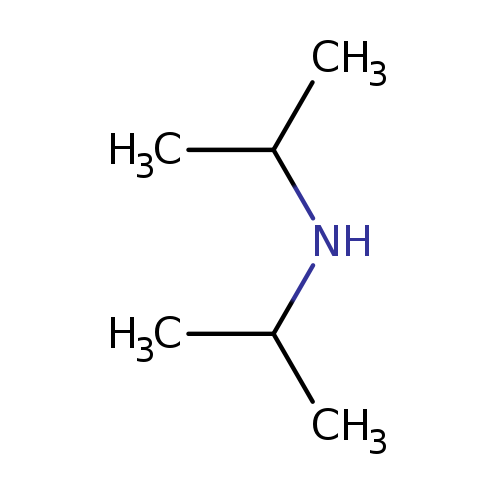
DiisopropylamineCatalog No.:AA0034O1 CAS No.:108-18-9 MDL No.:MFCD00008862 MF:C6H15N MW:101.1900 |
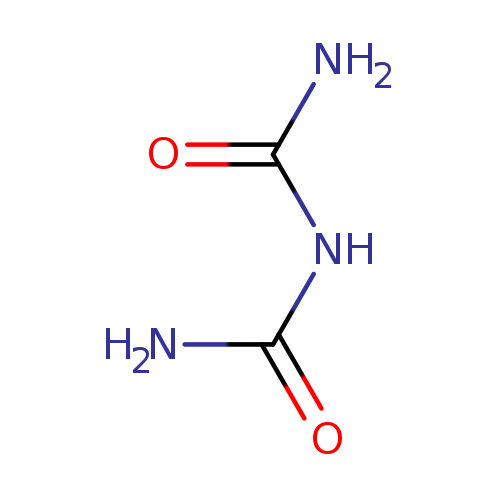
BiuretCatalog No.:AA003OBO CAS No.:108-19-0 MDL No.:MFCD00007946 MF:C2H5N3O2 MW:103.0800 |
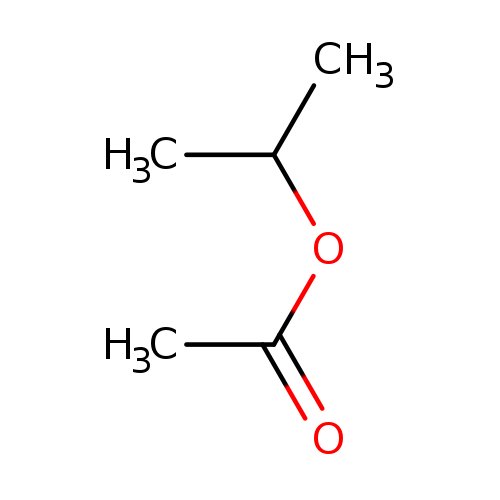
Isopropyl acetateCatalog No.:AA0035UT CAS No.:108-21-4 MDL No.:MFCD00008877 MF:C5H10O2 MW:102.1317 |
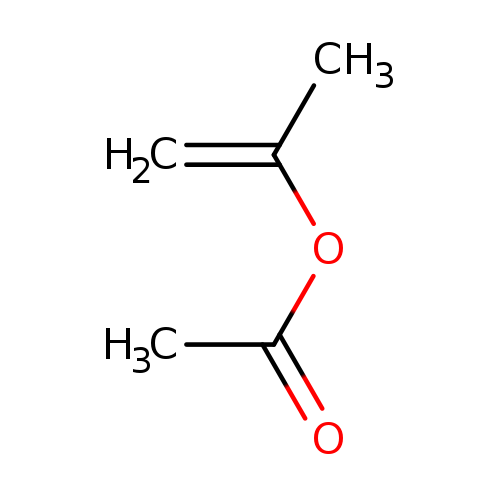
Prop-1-en-2-yl acetateCatalog No.:AA0034ZA CAS No.:108-22-5 MDL No.:MFCD08669885 MF:C5H8O2 MW:100.1158 |
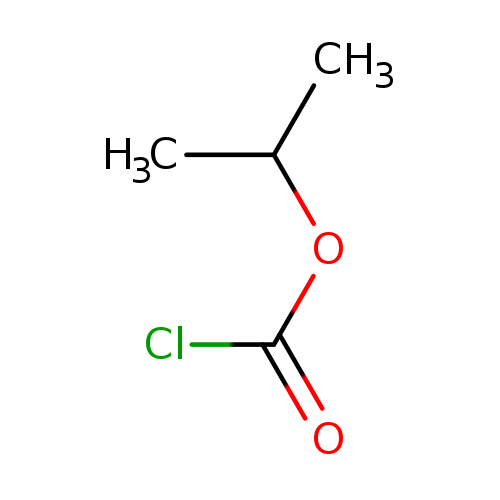
Isopropyl ChloroformateCatalog No.:AA007D0Q CAS No.:108-23-6 MDL No.:MFCD00075068 MF:C4H7ClO2 MW:122.5502 |
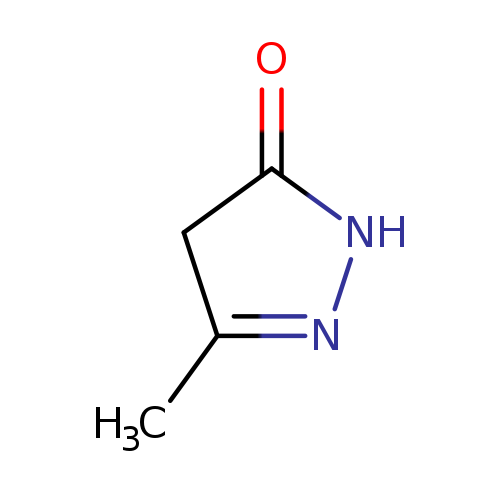
5-methyl-2,4-dihydropyrazol-3-oneCatalog No.:AA003JNK CAS No.:108-26-9 MDL No.:MFCD00020699 MF:C4H6N2O MW:98.1032 |
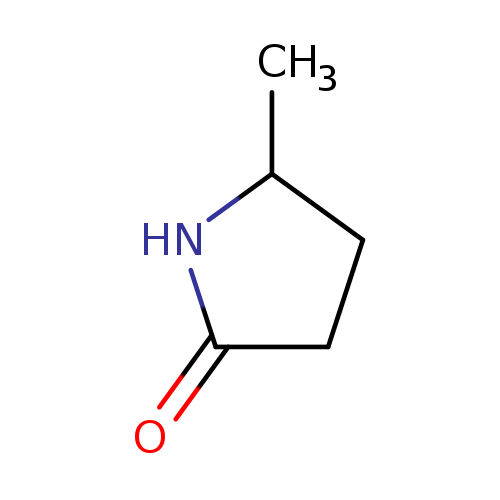
5-Methyl-2-pyrrolidoneCatalog No.:AA003485 CAS No.:108-27-0 MDL No.:MFCD00005273 MF:C5H9NO MW:99.1311 |
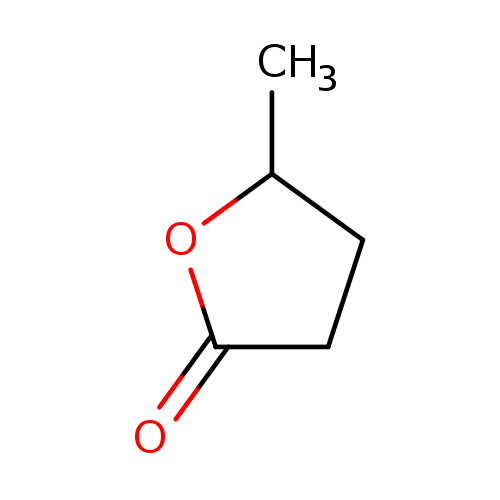
Gamma-valerolactoneCatalog No.:AA0034VY CAS No.:108-29-2 MDL No.:MFCD00005400 MF:C5H8O2 MW:100.1158 |
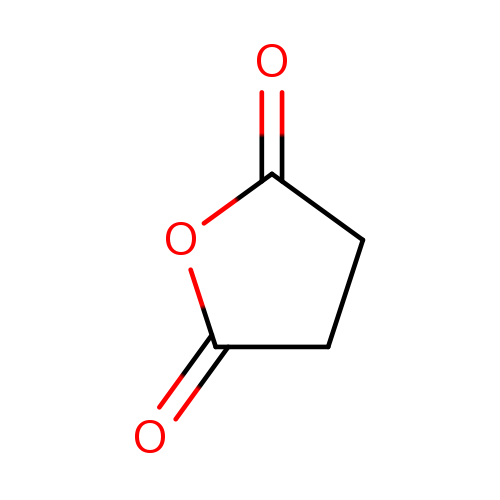
Succinic anhydrideCatalog No.:AA0035JQ CAS No.:108-30-5 MDL No.:MFCD00005525 MF:C4H4O3 MW:100.0728 |
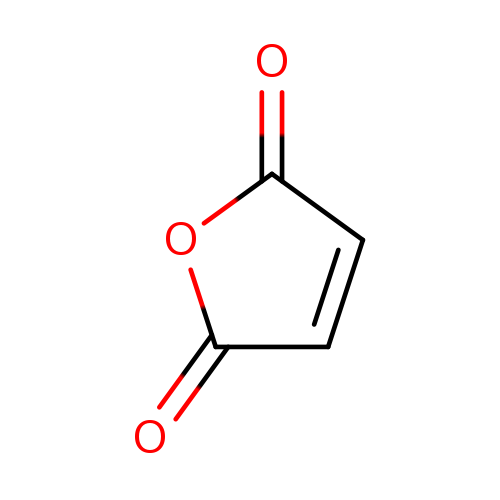
Maleic anhydrideCatalog No.:AA00351J CAS No.:108-31-6 MDL No.:MFCD00005518 MF:C4H2O3 MW:98.0569 |
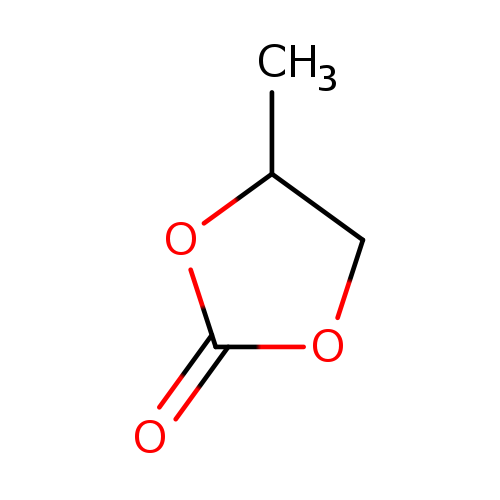
Propylene carbonateCatalog No.:AA0035GG CAS No.:108-32-7 MDL No.:MFCD00005385 MF:C4H6O3 MW:102.0886 |
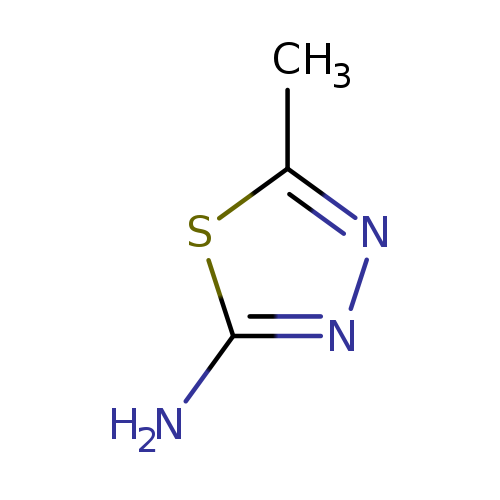
2-Amino-5-Methyl-1,3,4-thiadiazoleCatalog No.:AA0083LW CAS No.:108-33-8 MDL No.:MFCD00003110 MF:C3H5N3S MW:115.1569 |
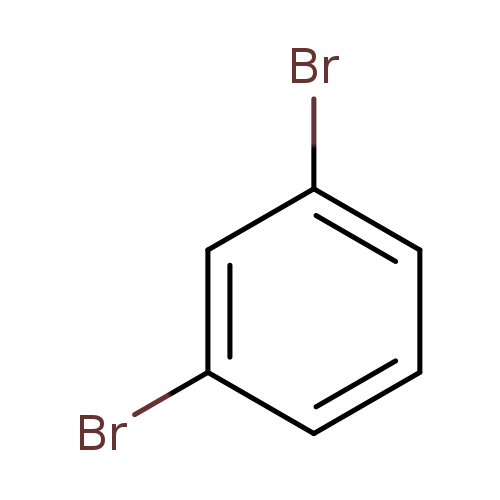
1,3-DibromobenzeneCatalog No.:AA0037X0 CAS No.:108-36-1 MDL No.:MFCD00000078 MF:C6H4Br2 MW:235.9040 |
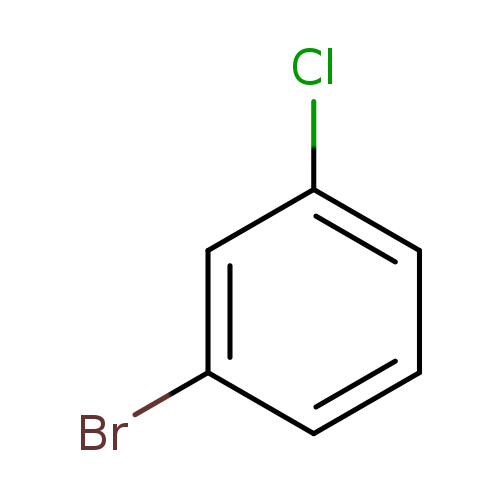
3-BromochlorobenzeneCatalog No.:AA0033K0 CAS No.:108-37-2 MDL No.:MFCD00000568 MF:C6H4BrCl MW:191.4530 |
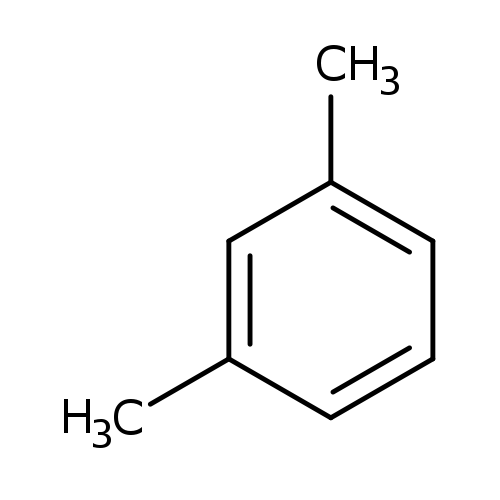
m-XyleneCatalog No.:AA00357A CAS No.:108-38-3 MDL No.:MFCD00008536 MF:C8H10 MW:106.1650 |
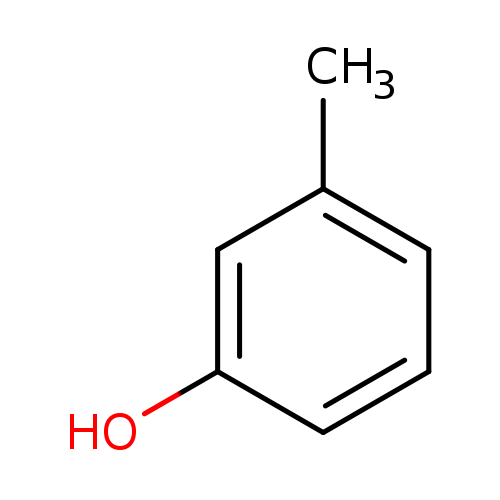
M-CresolCatalog No.:AA003RDT CAS No.:108-39-4 MDL No.:MFCD00002302 MF:C7H8O MW:108.1378 |
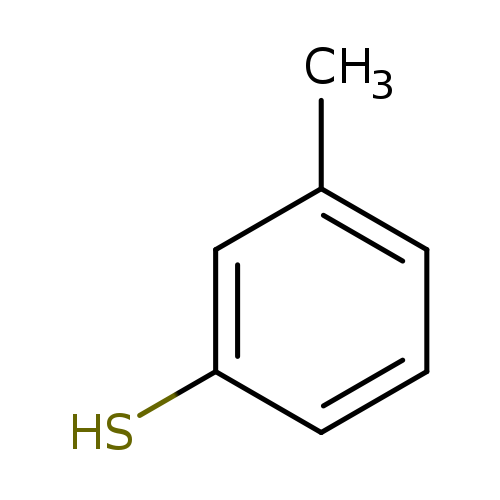
M-ThiocresolCatalog No.:AA003JNT CAS No.:108-40-7 MDL No.:MFCD00004843 MF:C7H8S MW:124.2034 |
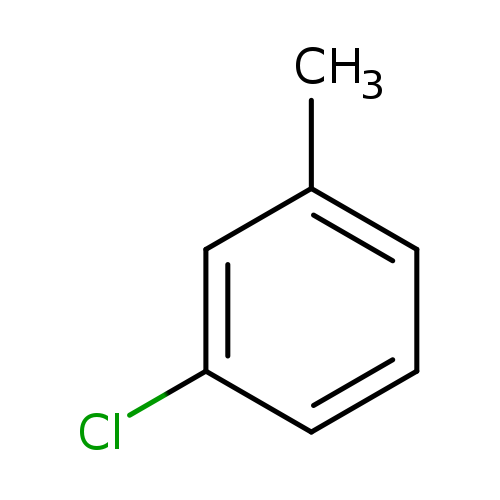
3-ChlorotolueneCatalog No.:AA0033LW CAS No.:108-41-8 MDL No.:MFCD00000595 MF:C7H7Cl MW:126.5835 |
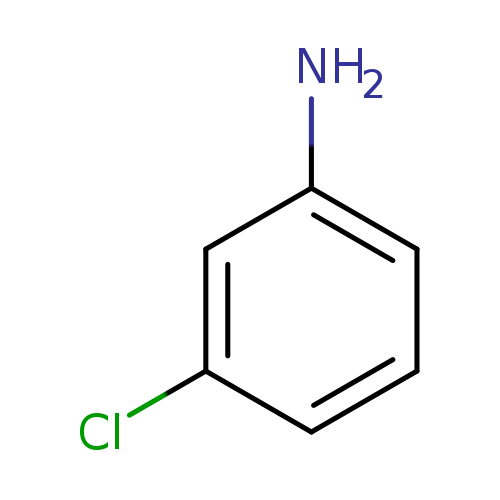
3-ChloroanilineCatalog No.:AA0033LF CAS No.:108-42-9 MDL No.:MFCD00007765 MF:C6H6ClN MW:127.5715 |
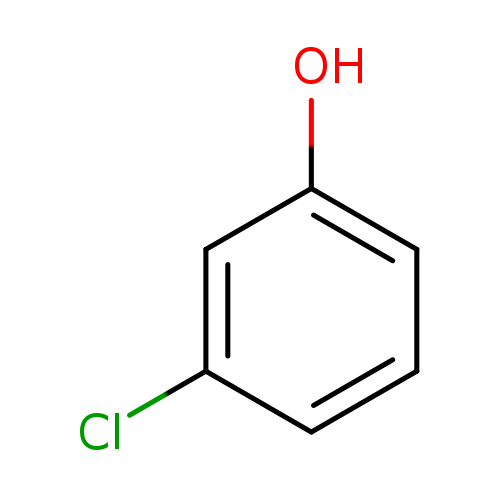
3-ChlorophenolCatalog No.:AA0037WY CAS No.:108-43-0 MDL No.:MFCD00048654 MF:C6H5ClO MW:128.5563 |
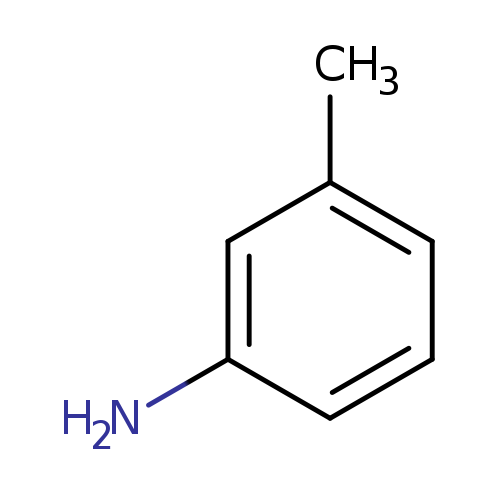
3-ToluidineCatalog No.:AA0033PJ CAS No.:108-44-1 MDL No.:MFCD00007808 MF:C7H9N MW:107.1531 |
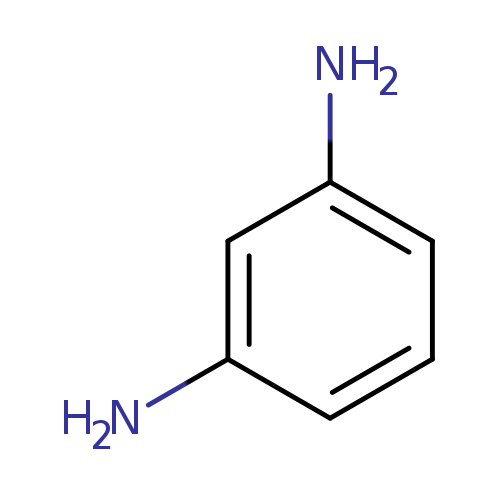
m-PhenylenediamineCatalog No.:AA003S3K CAS No.:108-45-2 MDL No.:MFCD00007799 MF:C6H8N2 MW:108.1411 |
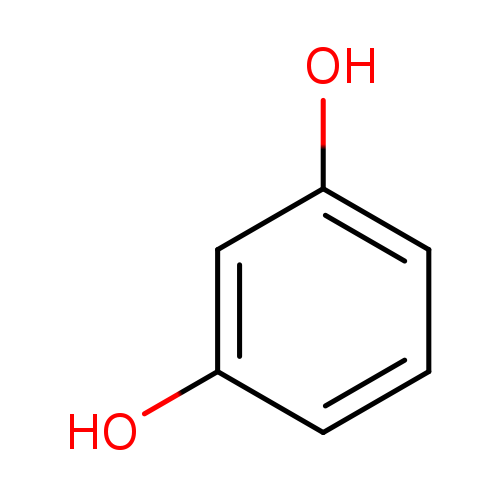
ResorcinolCatalog No.:AA0032IX CAS No.:108-46-3 MDL No.:MFCD00002269 MF:C6H6O2 MW:110.1106 |
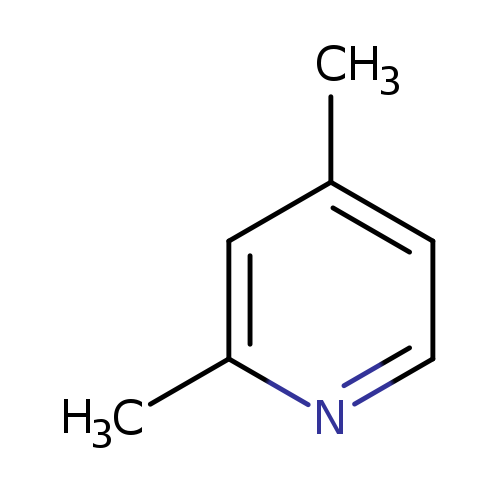
2,4-LutidineCatalog No.:AA0038WI CAS No.:108-47-4 MDL No.:MFCD00006337 MF:C7H9N MW:107.1531 |
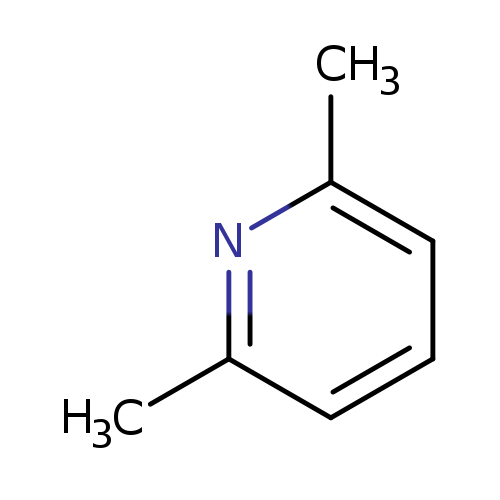
2,6-DimethylpyridineCatalog No.:AA0032ZC CAS No.:108-48-5 MDL No.:MFCD00006345 MF:C7H9N MW:107.1531 |
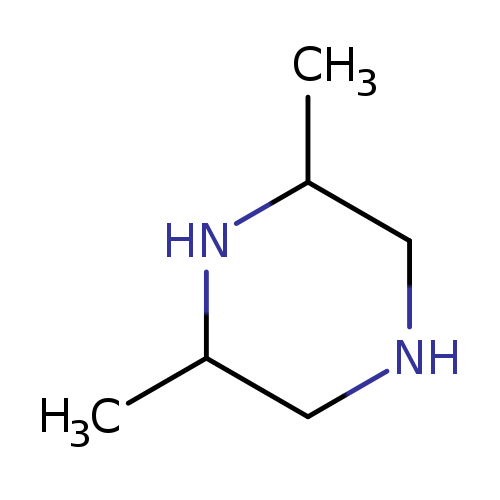
2,6-DimethylpiperazineCatalog No.:AA0035WQ CAS No.:108-49-6 MDL No.:MFCD00005956 MF:C6H14N2 MW:114.1888 |
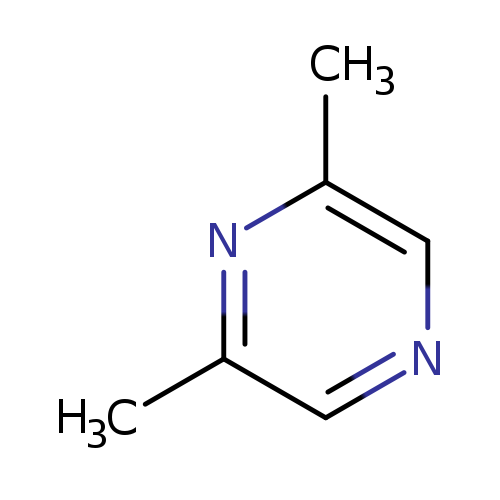
2,6-DimethylpyrazineCatalog No.:AA003G2Z CAS No.:108-50-9 MDL No.:MFCD00006148 MF:C6H8N2 MW:108.1411 |
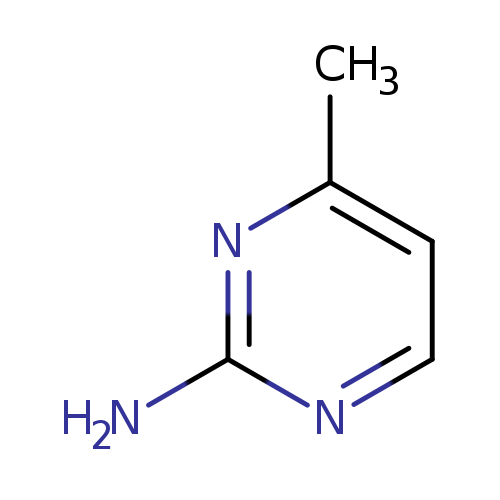
2-Amino-4-methylpyrimidineCatalog No.:AA003GCT CAS No.:108-52-1 MDL No.:MFCD00006101 MF:C5H7N3 MW:109.1292 |
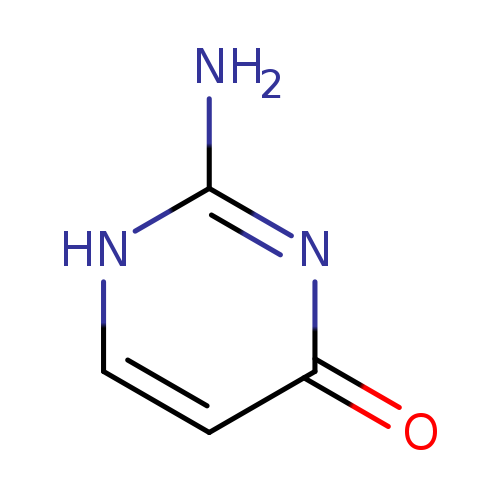
IsocytosineCatalog No.:AA003R0Z CAS No.:108-53-2 MDL No.:MFCD30183437 MF:C4H5N3O MW:111.1020 |
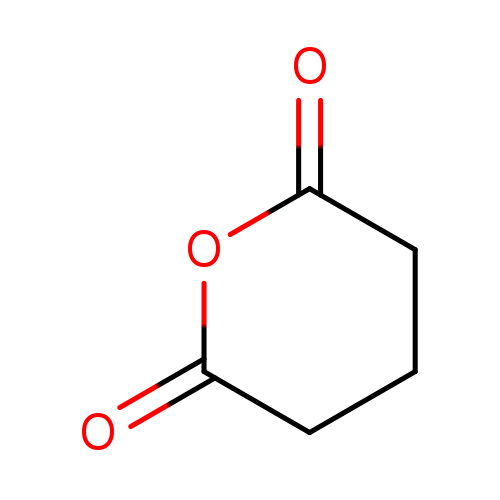
Glutaric anhydrideCatalog No.:AA0034W2 CAS No.:108-55-4 MDL No.:MFCD00006679 MF:C5H6O3 MW:114.0993 |
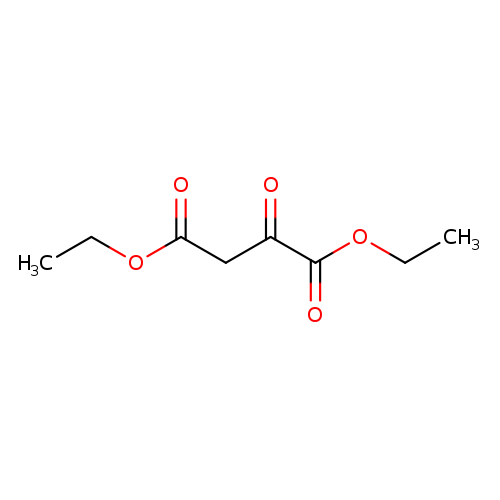
Diethyl 2-oxobutanedioateCatalog No.:AA0033CH CAS No.:108-56-5 MDL No.:MFCD00511083 MF:C8H12O5 MW:188.1779 |
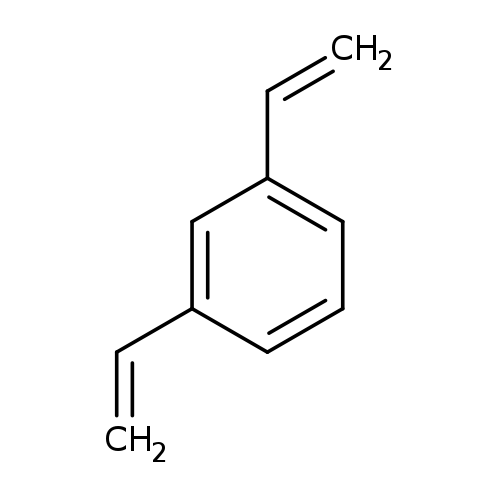
1,3-DivinylbenzeneCatalog No.:AA008SUI CAS No.:108-57-6 MDL No.:MFCD00008618 MF:C10H10 MW:130.1864 |
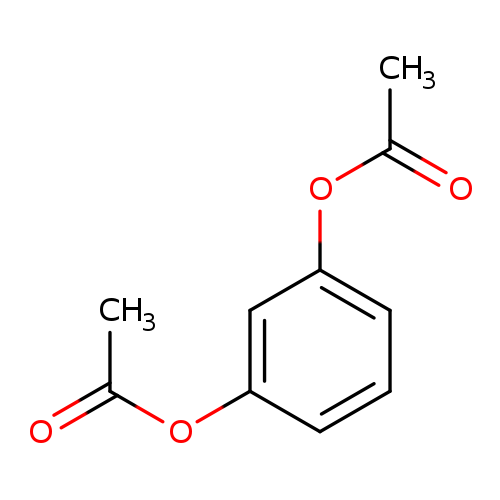
1,3-DiacetoxybenzeneCatalog No.:AA003DFR CAS No.:108-58-7 MDL No.:MFCD00008701 MF:C10H10O4 MW:194.1840 |
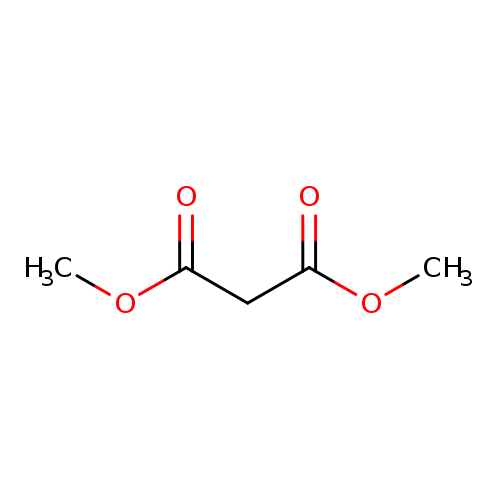
Dimethyl malonateCatalog No.:AA003PJE CAS No.:108-59-8 MDL No.:MFCD00008460 MF:C5H8O4 MW:132.1146 |
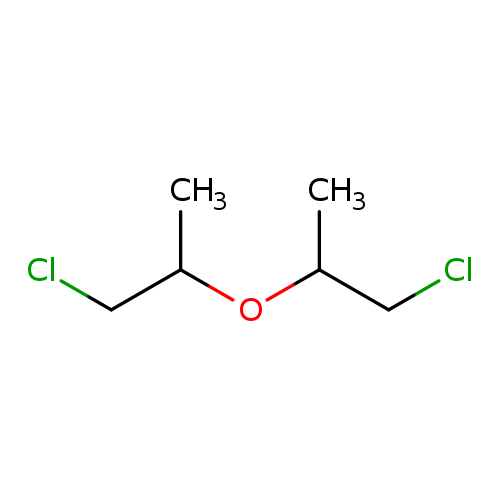
2,2'-Dichlorodiisopropyl etherCatalog No.:AA009NQW CAS No.:108-60-1 MDL No.:MFCD00152274 MF:C6H12Cl2O MW:171.0649 |
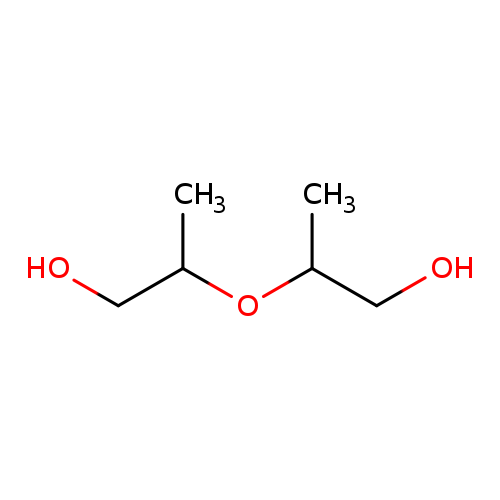
1-Propanol,2,2'-oxybis-Catalog No.:AA0083BU CAS No.:108-61-2 MDL No.:MFCD09751401 MF:C6H14O3 MW:134.1736 |
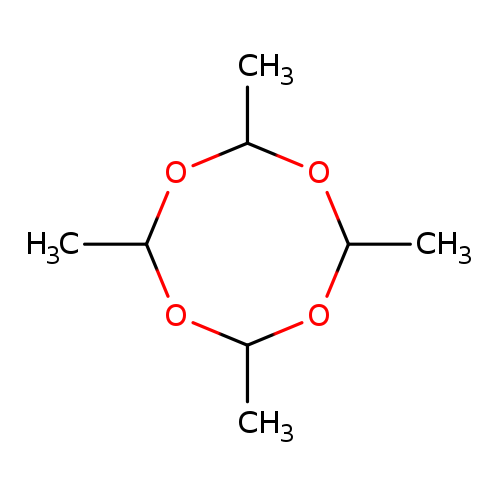
1,3,5,7-Tetroxocane, 2,4,6,8-tetramethyl-Catalog No.:AA00ILGO CAS No.:108-62-3 MDL No.:MFCD00071549 MF:C8H16O4 MW:176.2102 |
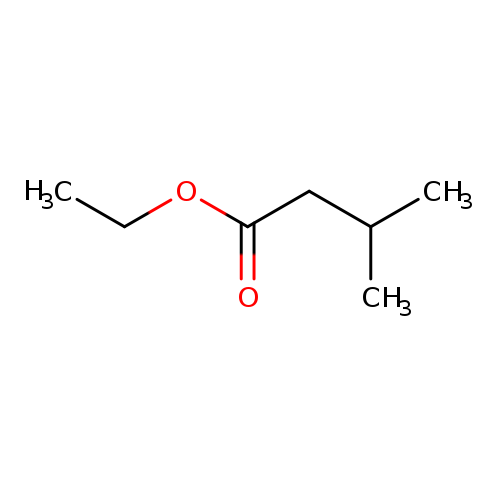
Ethyl isovalerateCatalog No.:AA0034TL CAS No.:108-64-5 MDL No.:MFCD00009203 MF:C7H14O2 MW:130.1849 |
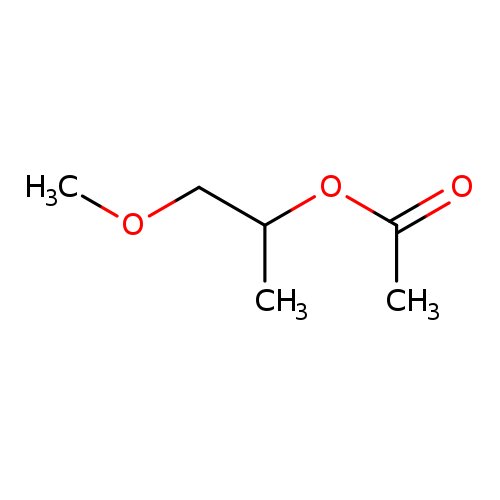
1-Methoxy-2-propyl acetateCatalog No.:AA003891 CAS No.:108-65-6 MDL No.:MFCD00038500 MF:C6H12O3 MW:132.1577 |
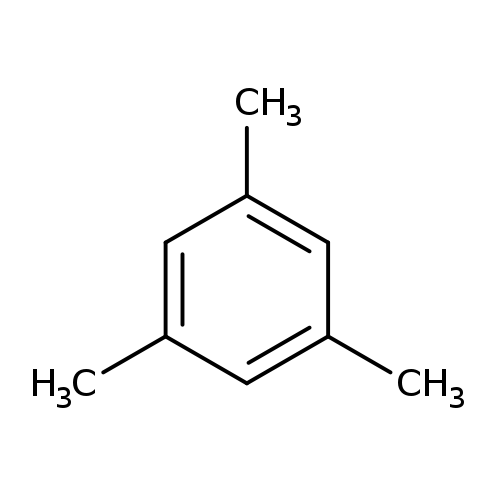
MesityleneCatalog No.:AA003REC CAS No.:108-67-8 MDL No.:MFCD00008538 MF:C9H12 MW:120.1916 |
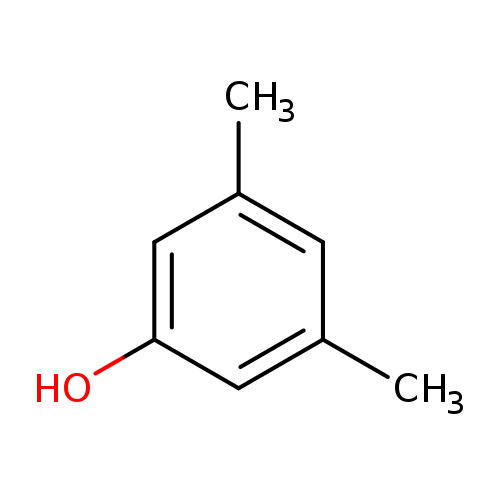
3,5-DimethylphenolCatalog No.:AA00385J CAS No.:108-68-9 MDL No.:MFCD00002307 MF:C8H10O MW:122.1644 |
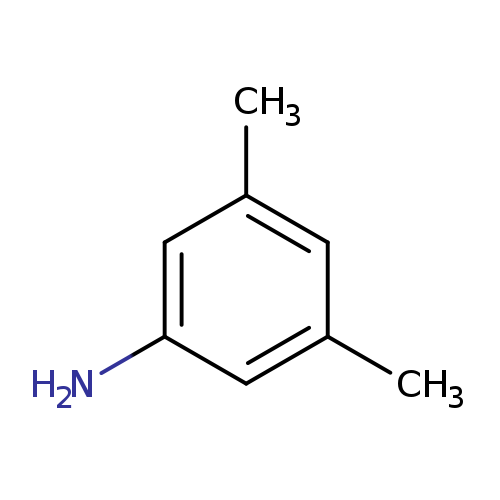
3,5-DimethylanilineCatalog No.:AA003ILX CAS No.:108-69-0 MDL No.:MFCD00007813 MF:C8H11N MW:121.1796 |
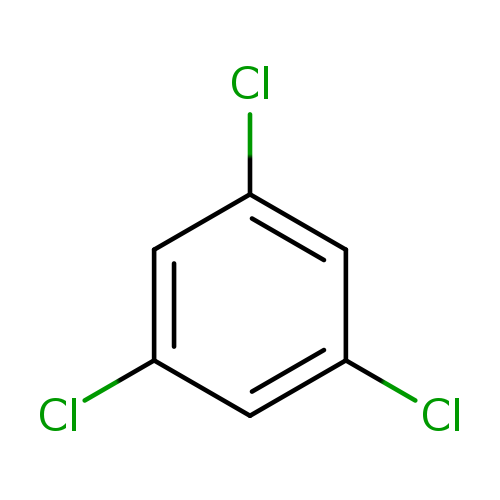
1,3,5-TrichlorobenzeneCatalog No.:AA00837J CAS No.:108-70-3 MDL No.:MFCD00000585 MF:C6H3Cl3 MW:181.4470 |
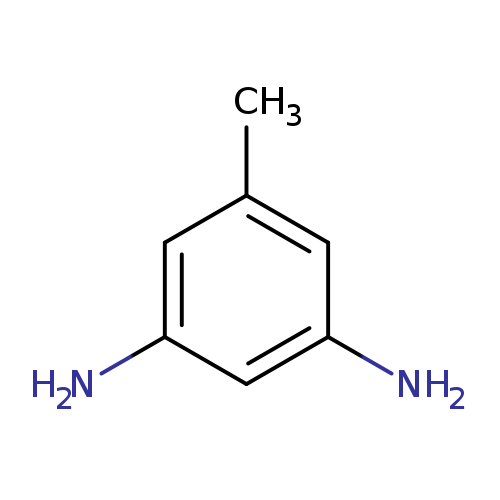
5-Methylbenzene-1,3-diamineCatalog No.:AA008YIR CAS No.:108-71-4 MDL No.:MFCD00136015 MF:C7H10N2 MW:122.1677 |
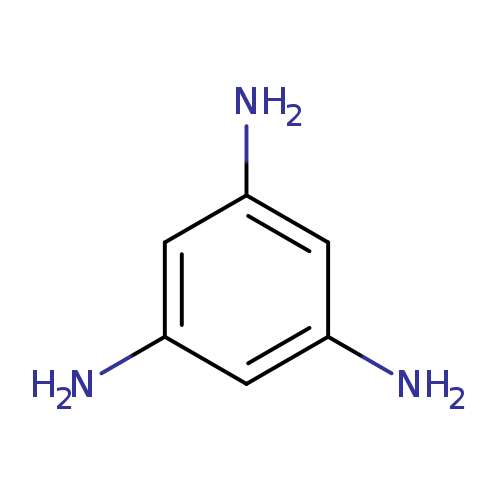
Benzene-1,3,5-triamineCatalog No.:AA007U1S CAS No.:108-72-5 MDL No.:MFCD01658125 MF:C6H9N3 MW:123.1558 |
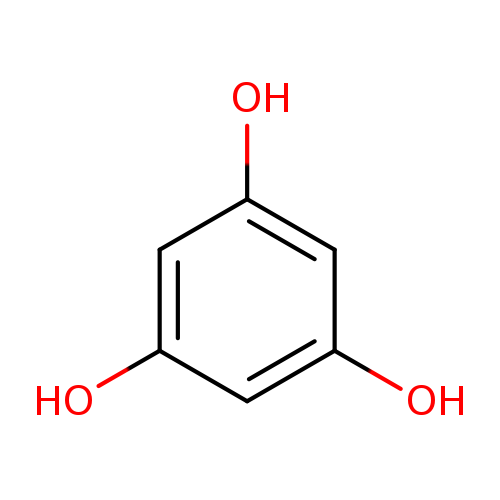
PhloroglucinolCatalog No.:AA003TNZ CAS No.:108-73-6 MDL No.:MFCD00149090 MF:C6H6O3 MW:126.1100 |
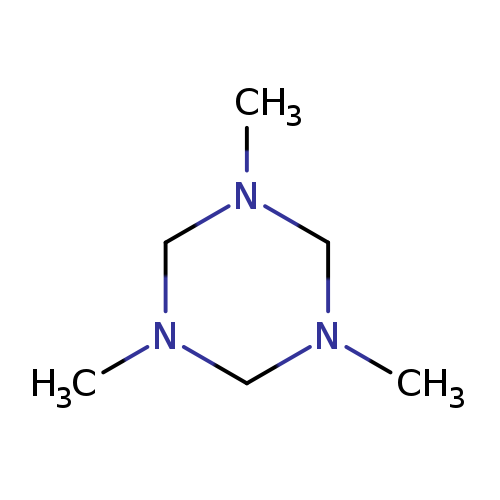
1,3,5-Trimethyl-1,3,5-triazinaneCatalog No.:AA003DDV CAS No.:108-74-7 MDL No.:MFCD00006057 MF:C6H15N3 MW:129.2034 |
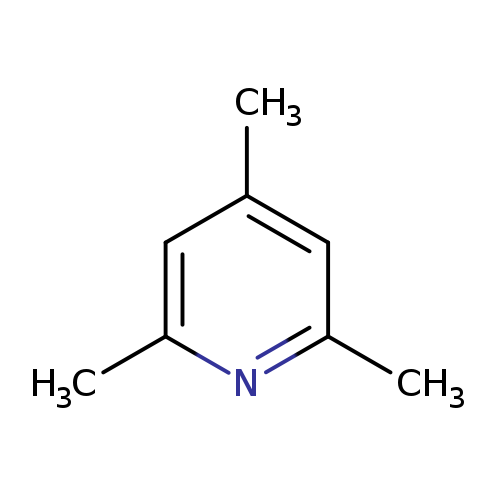
2,4,6-TrimethylpyridineCatalog No.:AA0032VN CAS No.:108-75-8 MDL No.:MFCD13175297 MF:C8H11N MW:121.1796 |
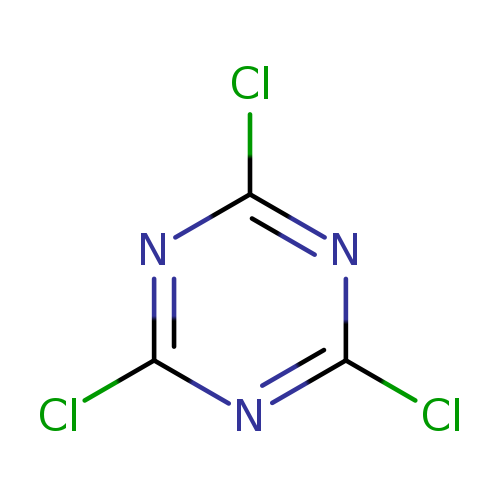
Cyanuric chlorideCatalog No.:AA0037WK CAS No.:108-77-0 MDL No.:MFCD00006046 MF:C3Cl3N3 MW:184.4112 |
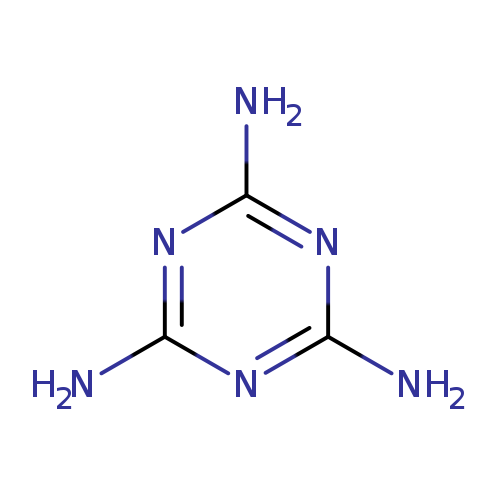
1,3,5-Triazine-2,4,6-triamineCatalog No.:AA00HAZF CAS No.:108-78-1 MDL No.:MFCD00006055 MF:C3H6N6 MW:126.1199 |
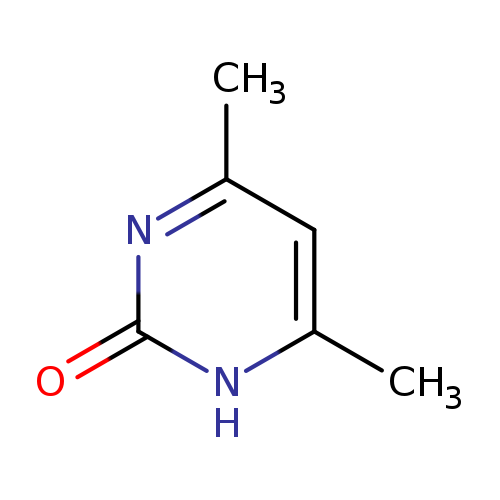
4,6-Dimethyl-2-hydroxypyrimidineCatalog No.:AA003HBL CAS No.:108-79-2 MDL No.:MFCD00006072 MF:C6H8N2O MW:124.1405 |
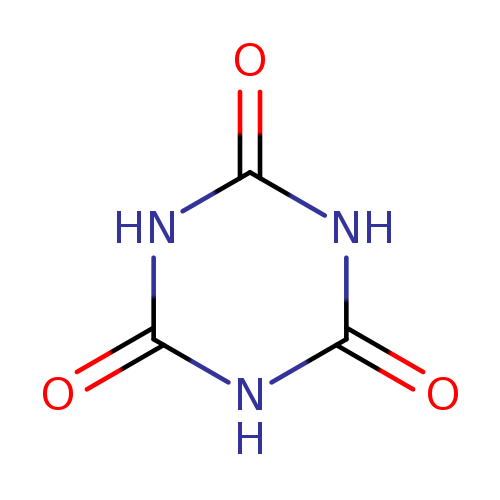
Cyanuric acidCatalog No.:AA0034KF CAS No.:108-80-5 MDL No.:MFCD00082990 MF:C3H3N3O3 MW:129.0742 |
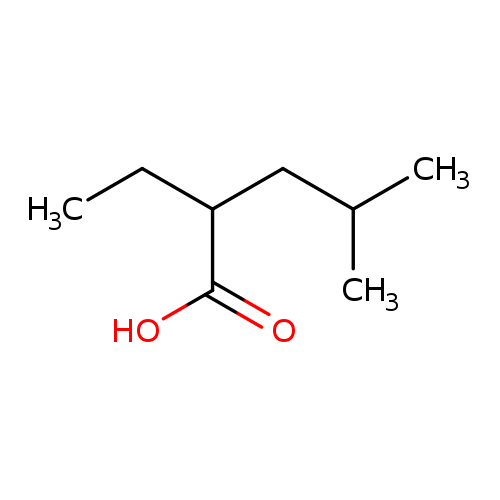
Pentanoic acid,2-ethyl-4-methyl-Catalog No.:AA0082ZE CAS No.:108-81-6 MDL No.:MFCD12794527 MF:C8H16O2 MW:144.2114 |
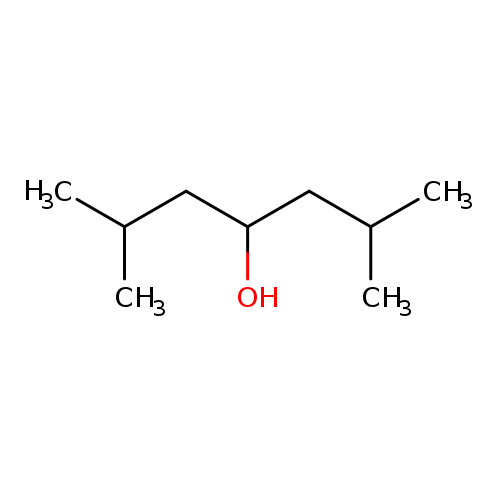
2,6-Dimethyl-4-heptanolCatalog No.:AA003PGQ CAS No.:108-82-7 MDL No.:MFCD00008944 MF:C9H20O MW:144.2545 |
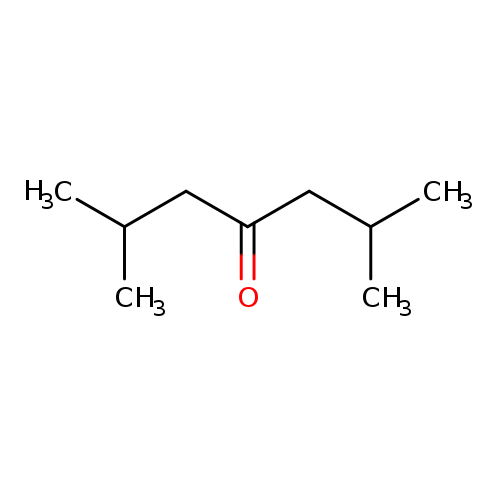
2,6-Dimethyl-4-heptanoneCatalog No.:AA0032Z1 CAS No.:108-83-8 MDL No.:MFCD00008940 MF:C9H18O MW:142.2386 |
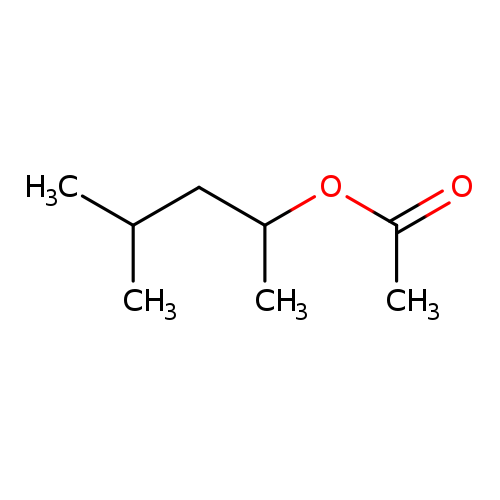
1,3-DIMETHYLBUTYL ACETATECatalog No.:AA003DHL CAS No.:108-84-9 MDL No.:MFCD00053723 MF:C8H16O2 MW:144.2114 |
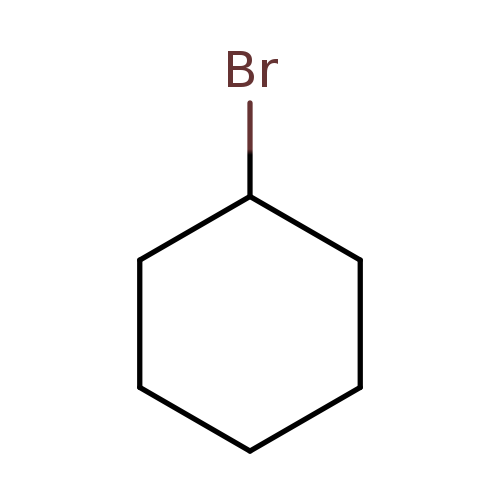
BromocyclohexaneCatalog No.:AA003OHM CAS No.:108-85-0 MDL No.:MFCD00003819 MF:C6H11Br MW:163.0555 |
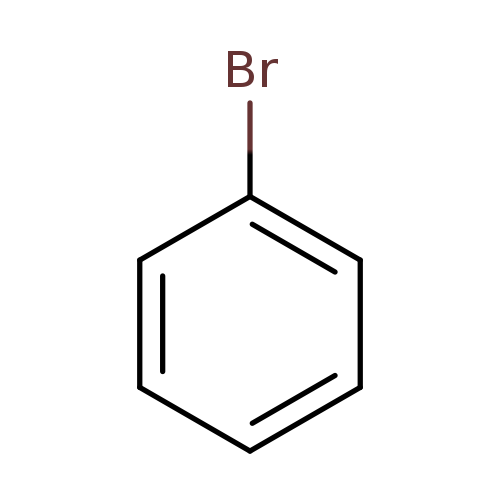
BromobenzeneCatalog No.:AA0034HN CAS No.:108-86-1 MDL No.:MFCD00000055 MF:C6H5Br MW:157.0079 |
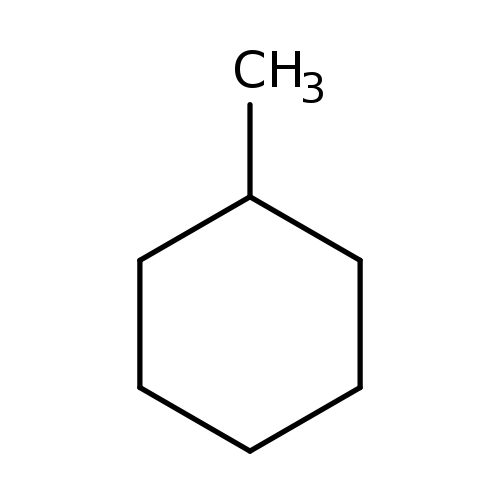
MethylcyclohexaneCatalog No.:AA00356N CAS No.:108-87-2 MDL No.:MFCD00001497 MF:C7H14 MW:98.1861 |
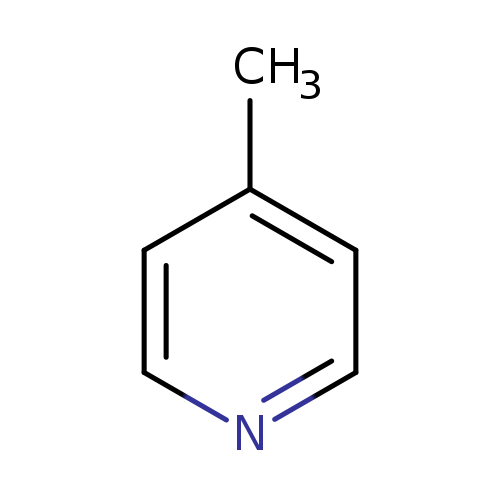
4-MethylpyridineCatalog No.:AA0037WF CAS No.:108-89-4 MDL No.:MFCD00006440 MF:C6H7N MW:93.1265 |
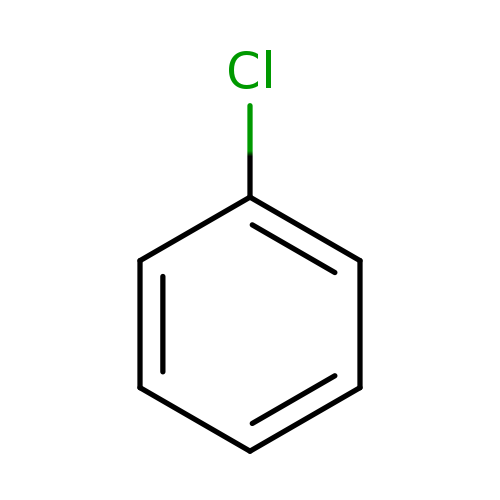
ChlorobenzeneCatalog No.:AA0034IM CAS No.:108-90-7 MDL No.:MFCD00000530 MF:C6H5Cl MW:112.5569 |
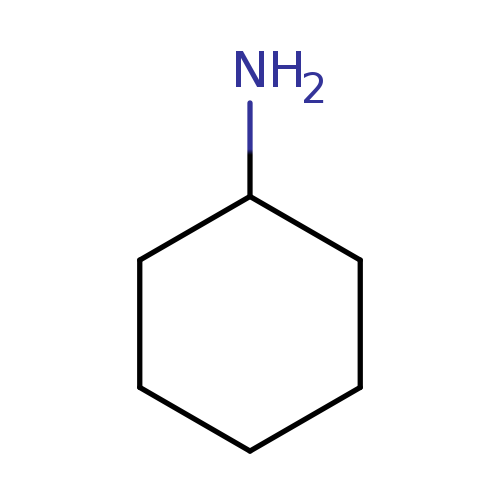
CyclohexylamineCatalog No.:AA003P2M CAS No.:108-91-8 MDL No.:MFCD00001486 MF:C6H13N MW:99.1741 |
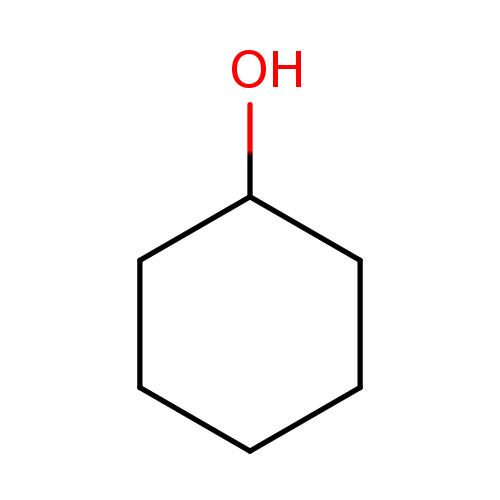
CyclohexanolCatalog No.:AA003P21 CAS No.:108-93-0 MDL No.:MFCD00003855 MF:C6H12O MW:100.1589 |
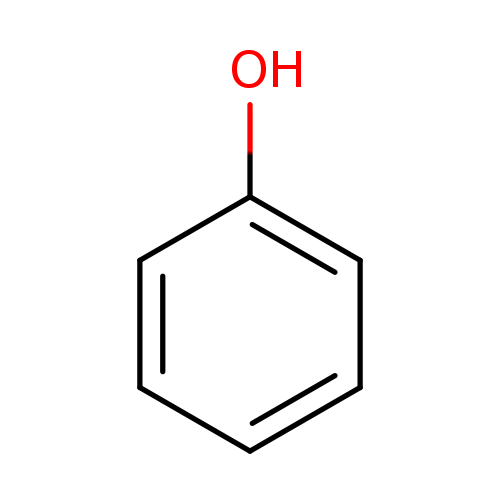
PhenolCatalog No.:AA0035E2 CAS No.:108-95-2 MDL No.:MFCD00002143 MF:C6H6O MW:94.1112 |
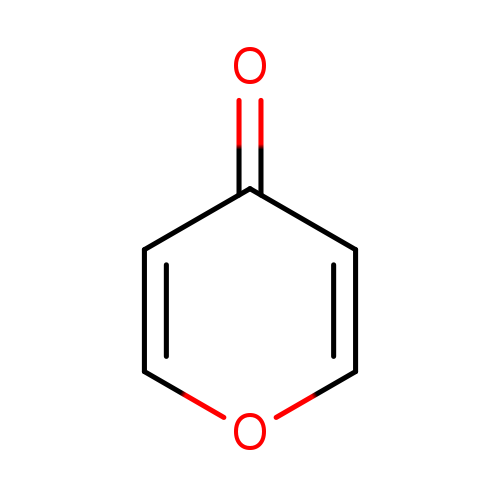
4H-Pyran-4-oneCatalog No.:AA003LED CAS No.:108-97-4 MDL No.:MFCD00006576 MF:C5H4O2 MW:96.0841 |
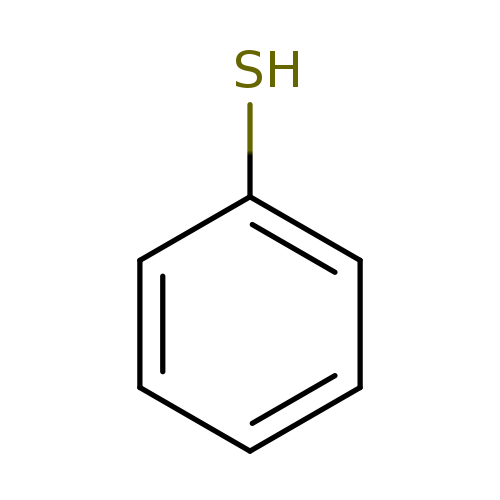
ThiophenolCatalog No.:AA0098HF CAS No.:108-98-5 MDL No.:MFCD00004826 MF:C6H6S MW:110.1768 |
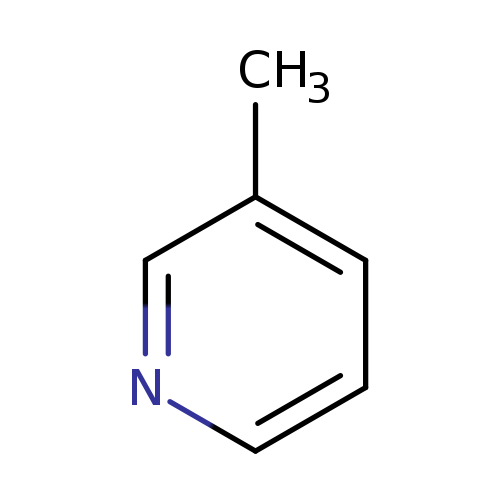
3-MethylpyridineCatalog No.:AA0033OQ CAS No.:108-99-6 MDL No.:MFCD00006402 MF:C6H7N MW:93.1265 |
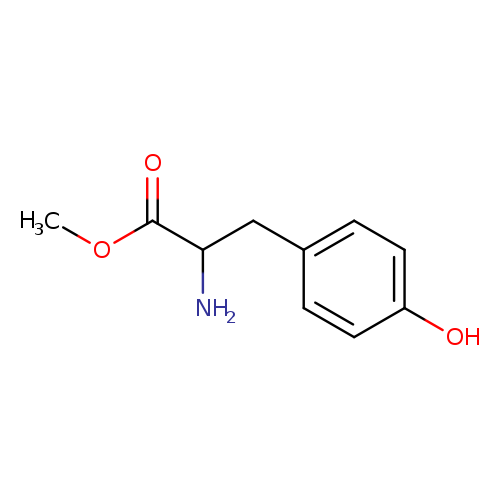
L-Tyrosine methyl esterCatalog No.:AA00HAZG CAS No.:1080-06-4 MDL No.:MFCD00002392 MF:C10H13NO3 MW:195.2151 |
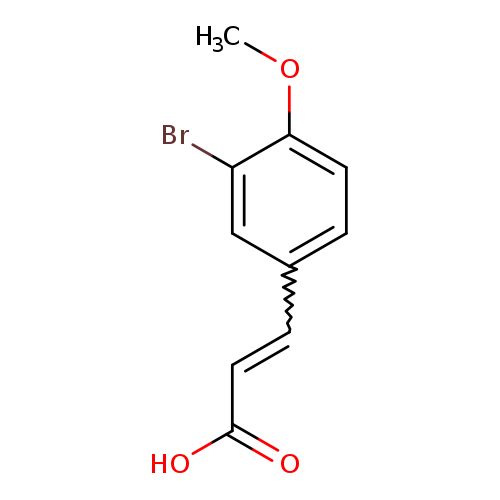
3-Bromo-4-methoxycinnamic acidCatalog No.:AA007DLV CAS No.:1080-07-5 MDL No.:MFCD01848832 MF:C10H9BrO3 MW:257.0807 |
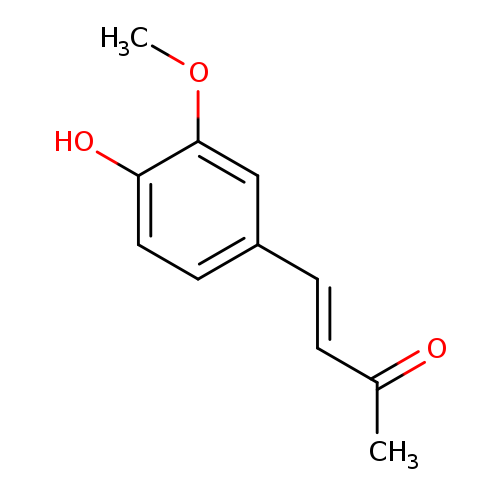
4-(4-Hydroxy-3-methoxyphenyl)-3-buten-2-oneCatalog No.:AA003K14 CAS No.:1080-12-2 MDL No.:MFCD00012210 MF:C11H12O3 MW:192.2112 |
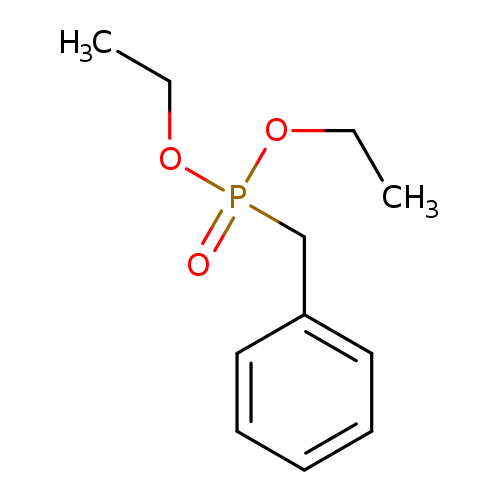
Diethyl benzylphosphonateCatalog No.:AA003PDK CAS No.:1080-32-6 MDL No.:MFCD00009078 MF:C11H17O3P MW:228.2246 |
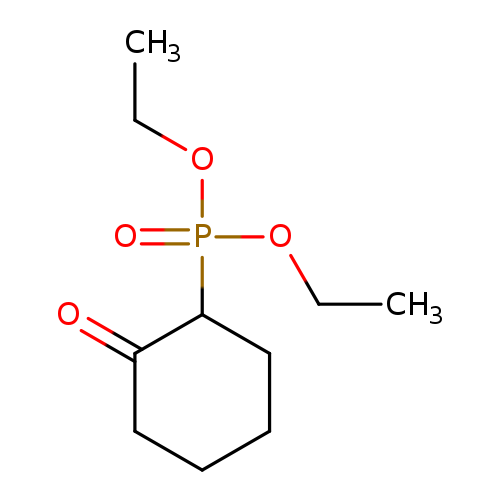
diethyl (2-oxocyclohexyl)phosphonateCatalog No.:AA007V5T CAS No.:1080-41-7 MDL No.:MFCD20922537 MF:C10H19O4P MW:234.2292 |
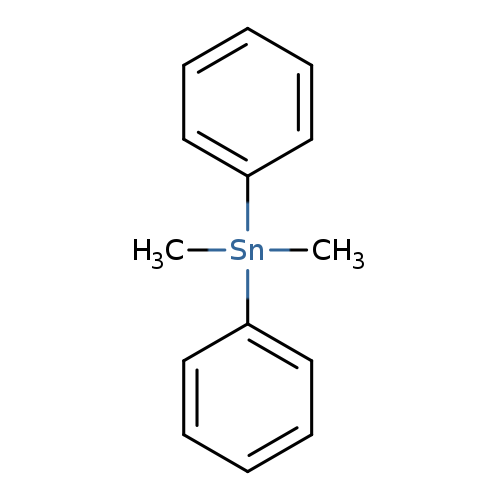
DimethyldiphenylstannaneCatalog No.:AA003PKF CAS No.:1080-43-9 MDL No.:MFCD00058812 MF:C14H16Sn MW:302.9778 |
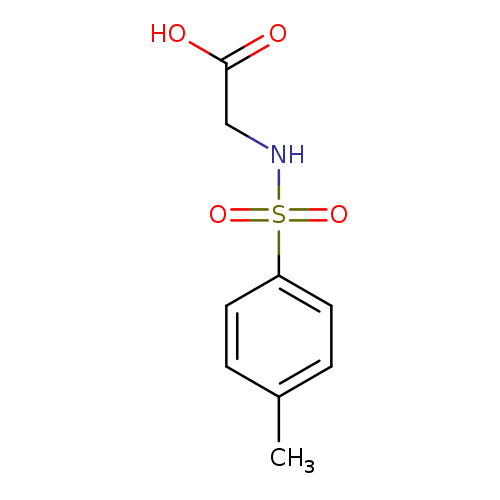
N-P-TosylglycineCatalog No.:AA00844A CAS No.:1080-44-0 MDL No.:MFCD03092531 MF:C9H11NO4S MW:229.2529 |
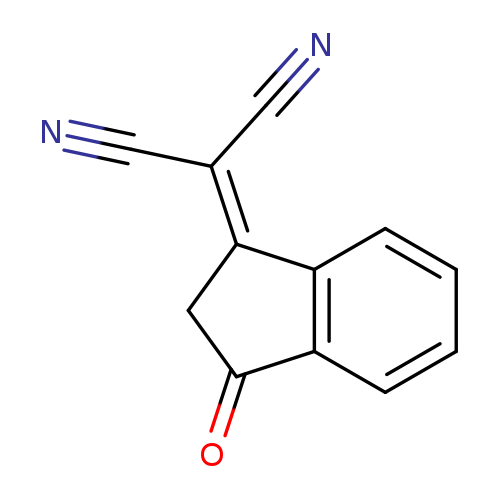
3-(Dicyanomethylidene)indan-1-oneCatalog No.:AA0033E2 CAS No.:1080-74-6 MDL No.:MFCD00143117 MF:C12H6N2O MW:194.1888 |
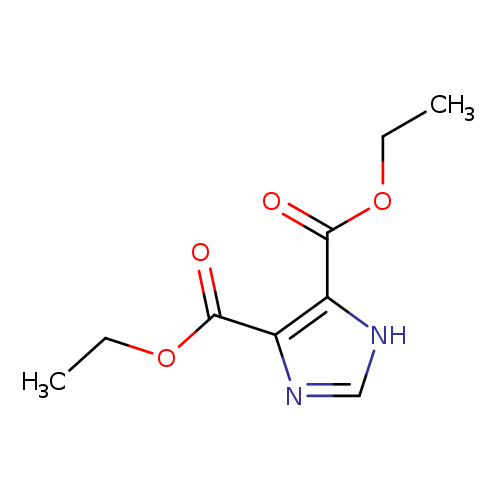
Diethyl 4,5-imidazole-1H-4,5-dicarboxylateCatalog No.:AA007V5L CAS No.:1080-79-1 MDL No.:MFCD00723829 MF:C9H12N2O4 MW:212.2026 |
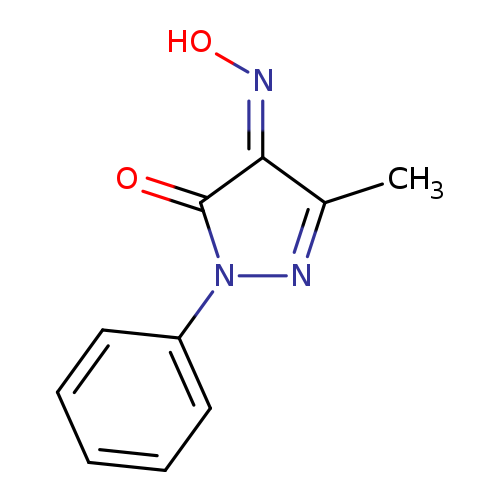
5-Methyl-2-phenyl-2h-pyrazole-3,4-dione 4-oximeCatalog No.:AA008Y28 CAS No.:1080-89-3 MDL No.:MFCD00448519 MF:C10H9N3O2 MW:203.1974 |
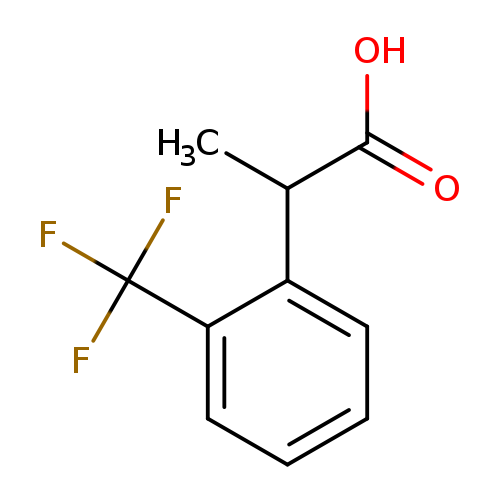
2-[2-(trifluoromethyl)phenyl]propanoic acidCatalog No.:AA019PO6 CAS No.:1080023-02-4 MDL No.:MFCD07787628 MF:C10H9F3O2 MW:218.1725 |
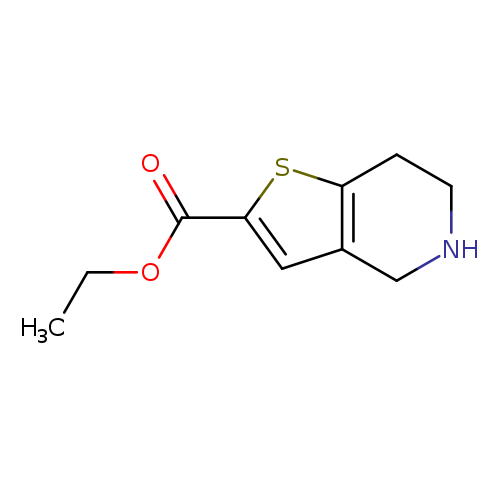
Ethyl 4,5,6,7-tetrahydrothieno[3,2-c]pyridine-2-carboxylateCatalog No.:AA007DLX CAS No.:1080026-94-3 MDL No.:MFCD10566074 MF:C10H13NO2S MW:211.2807 |
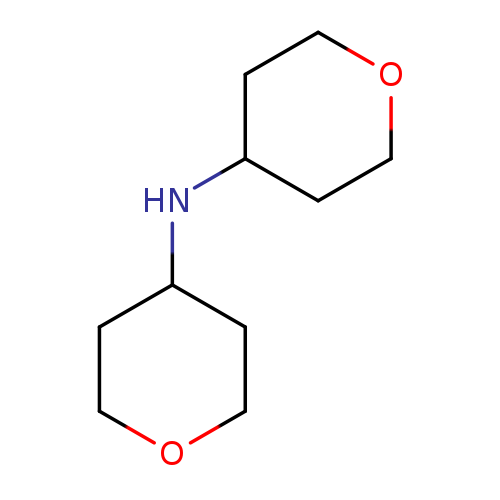
Di(tetrahydropyran-4-yl)amineCatalog No.:AA003O76 CAS No.:1080028-76-7 MDL No.:MFCD11841081 MF:C10H19NO2 MW:185.2634 |
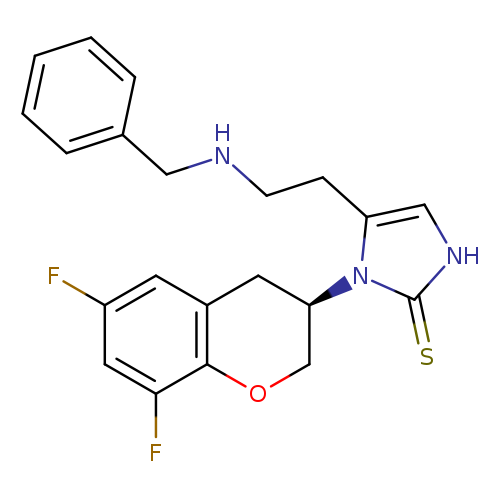
ZamicastatCatalog No.:AA01FNSH CAS No.:1080028-80-3 MDL No.:MFCD28502160 MF:C21H21F2N3OS MW:401.4727 |
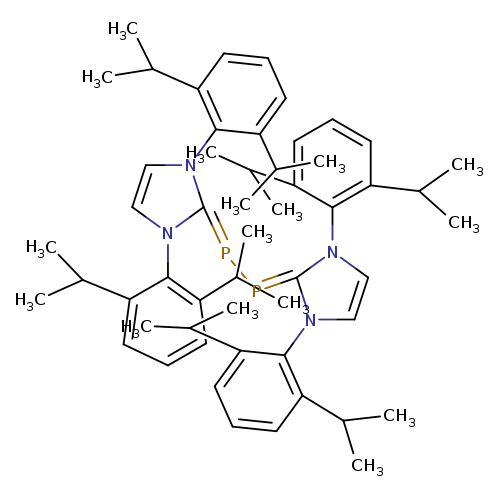
1,2-Bis[1,3-bis(2,6-di-i-propylphenyl)iMidazol-2-ylidene]diphosphineCatalog No.:AA008WXY CAS No.:1080030-13-2 MDL No.:MFCD23704802 MF:C54H72N4P2 MW:839.1238 |
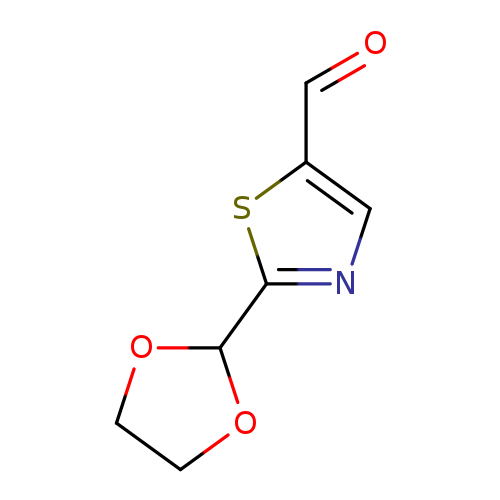
2-(1,3-dioxolan-2-yl)-1,3-thiazole-5-carbaldehydeCatalog No.:AA01BDWM CAS No.:1080060-60-1 MDL No.:MFCD24686876 MF:C7H7NO3S MW:185.2004 |
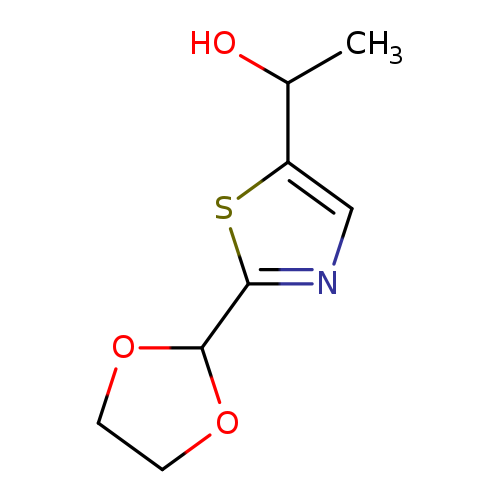
1-[2-(1,3-dioxolan-2-yl)-1,3-thiazol-5-yl]ethan-1-olCatalog No.:AA01BDEF CAS No.:1080060-61-2 MDL No.:MFCD28118249 MF:C8H11NO3S MW:201.2428 |
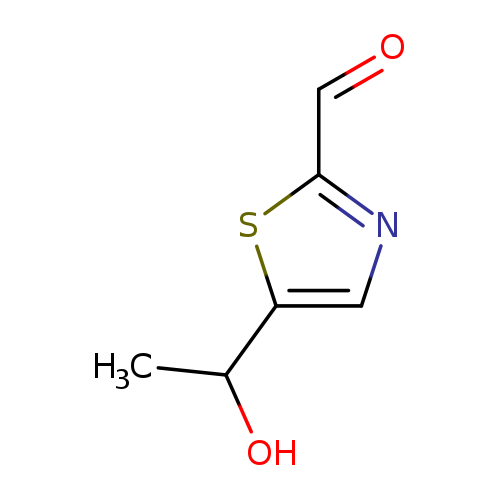
5-(1-Hydroxyethyl)-1,3-thiazole-2-carbaldehydeCatalog No.:AA01BF2Z CAS No.:1080060-62-3 MDL No.:MFCD24686875 MF:C6H7NO2S MW:157.1903 |
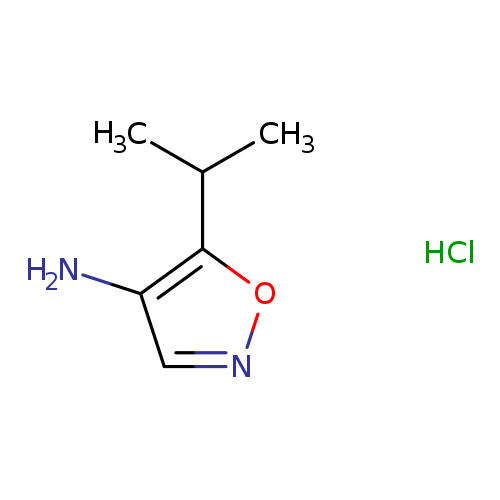
5-(propan-2-yl)-1,2-oxazol-4-amine hydrochlorideCatalog No.:AA01E8QQ CAS No.:1080063-94-0 MDL No.:MFCD30730716 MF:C6H11ClN2O MW:162.6173 |
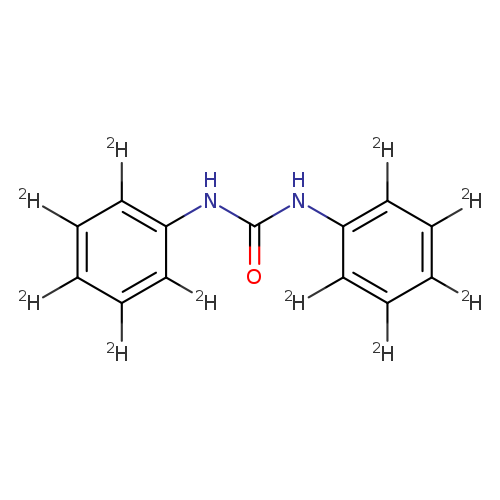
N,N′-DIPHENYLUREA D10Catalog No.:AA008RCH CAS No.:108009-46-7 MDL No.:MFCD08273812 MF:C13H2D10N2O MW:222.3088 |
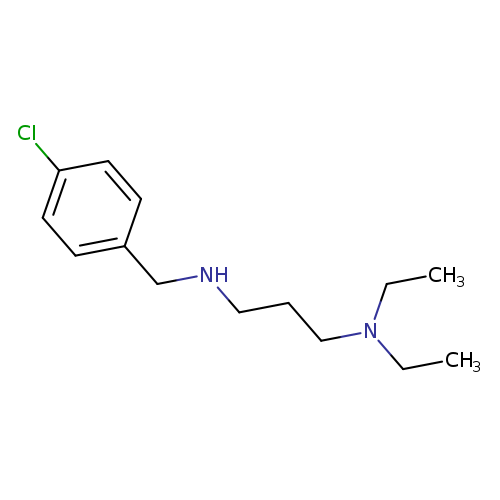
[(4-chlorophenyl)methyl][3-(diethylamino)propyl]amineCatalog No.:AA01C33C CAS No.:108010-87-3 MDL No.:MFCD03832191 MF:C14H23ClN2 MW:254.7988 |
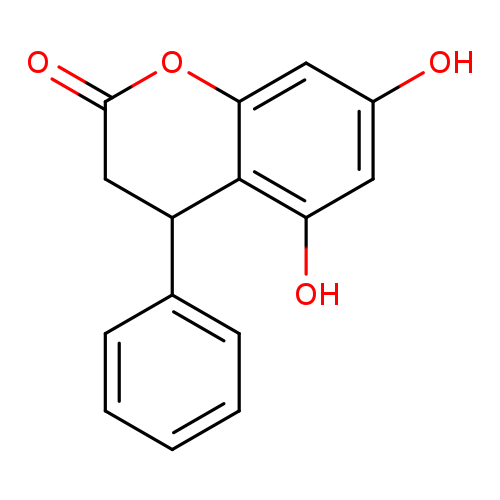
2H-1-Benzopyran-2-one, 3,4-dihydro-5,7-dihydroxy-4-phenyl-Catalog No.:AA00844D CAS No.:108013-15-6 MDL No.:MFCD00138911 MF:C15H12O4 MW:256.2534 |
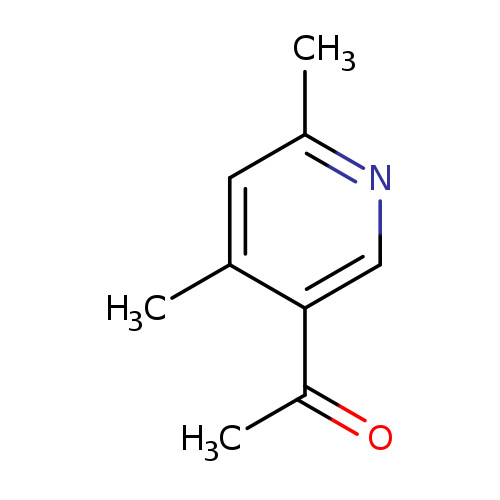
1-(4,6-Dimethylpyridin-3-yl)ethanoneCatalog No.:AA00HAZJ CAS No.:108028-67-7 MDL No.:MFCD09263834 MF:C9H11NO MW:149.1897 |
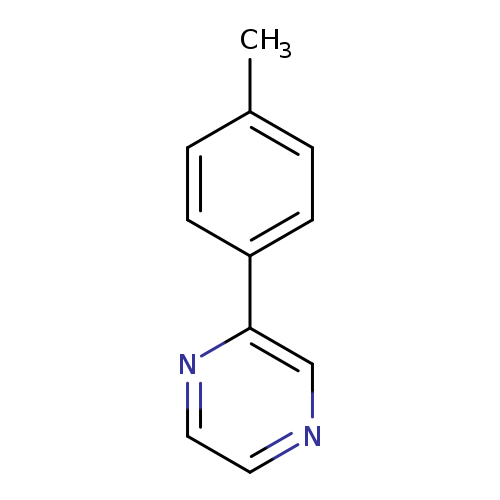
2-(4-Methylphenyl)pyrazineCatalog No.:AA01EMTI CAS No.:108030-80-4 MDL No.:MFCD24849202 MF:C11H10N2 MW:170.2105 |
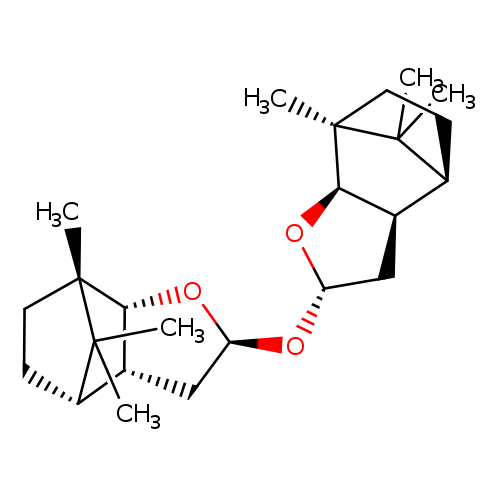
(-)-NOES REAGENTCatalog No.:AA003B8X CAS No.:108031-79-4 MDL No.:MFCD00151131 MF:C24H38O3 MW:374.5567 |
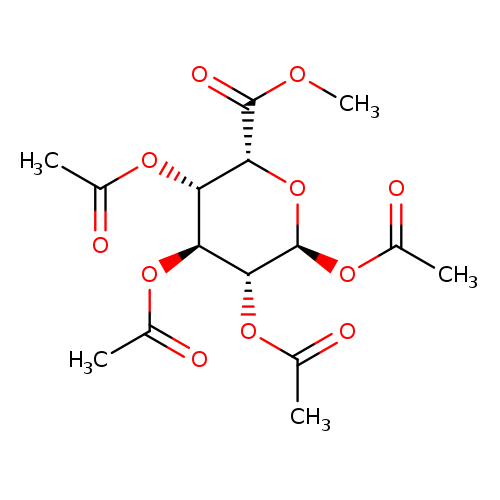
Methyl 1,2,3,4-Tetra-O-acetyl-α-L-idopyranuronateCatalog No.:AA008VXR CAS No.:108032-41-3 MDL No.:MFCD09750788 MF:C15H20O11 MW:376.3127 |
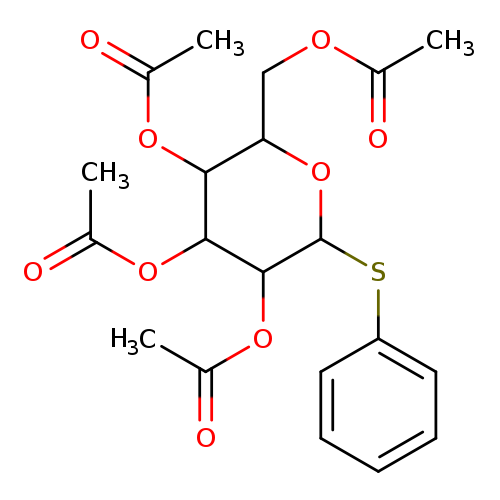
Phenyl 2,3,4,6-Tetra-O-acetyl-1-thio-a-D-mannopyranosideCatalog No.:AA008VNR CAS No.:108032-93-5 MDL No.:MFCD01862644 MF:C20H24O9S MW:440.4642 |
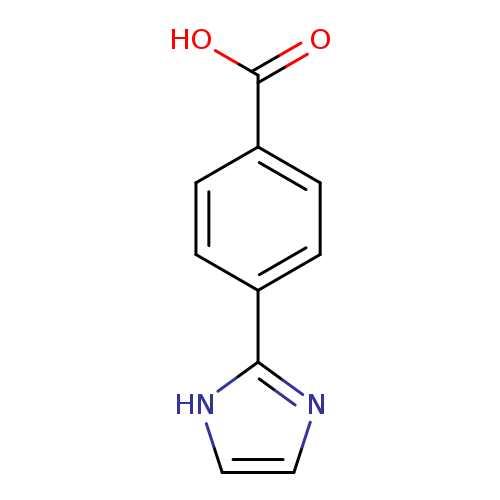
4-(1H-Imidazol-2-yl)benzoic acidCatalog No.:AA003JX8 CAS No.:108035-45-6 MDL No.:MFCD00456057 MF:C10H8N2O2 MW:188.1827 |
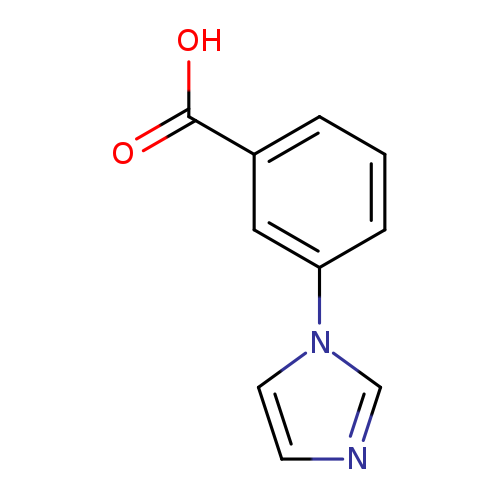
3-(1H-Imidazol-1-yl)benzoic acidCatalog No.:AA003HWS CAS No.:108035-47-8 MDL No.:MFCD06659077 MF:C10H8N2O2 MW:188.1827 |
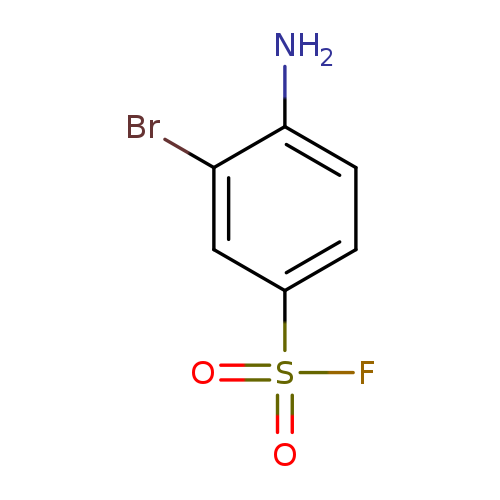
4-amino-3-bromobenzene-1-sulfonyl fluorideCatalog No.:AA01EITF CAS No.:108045-28-9 MDL No.:MFCD00085900 MF:C6H5BrFNO2S MW:254.0768 |
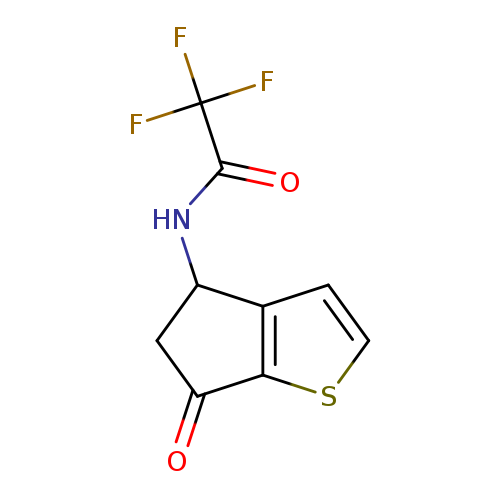
2,2,2-Trifluoro-N-(6-oxo-5,6-dihydro-4H-cyclopenta[b]thiophen-4-yl)acetamideCatalog No.:AA008ROO CAS No.:108046-14-6 MDL No.:MFCD03012801 MF:C9H6F3NO2S MW:249.2096 |
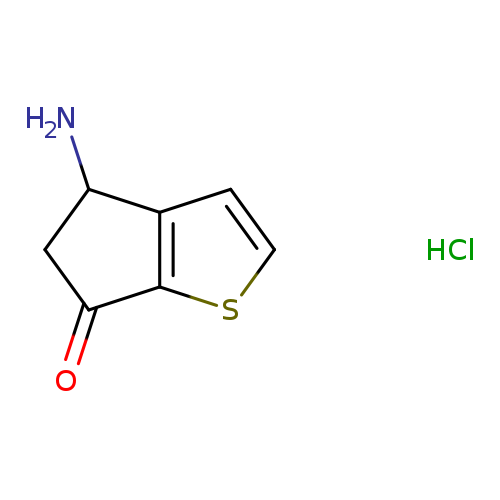
4-Amino-4H-cyclopenta[b]thiophen-6(5H)-one hydrochlorideCatalog No.:AA007V5S CAS No.:108046-16-8 MDL No.:MFCD03791250 MF:C7H8ClNOS MW:189.6625 |
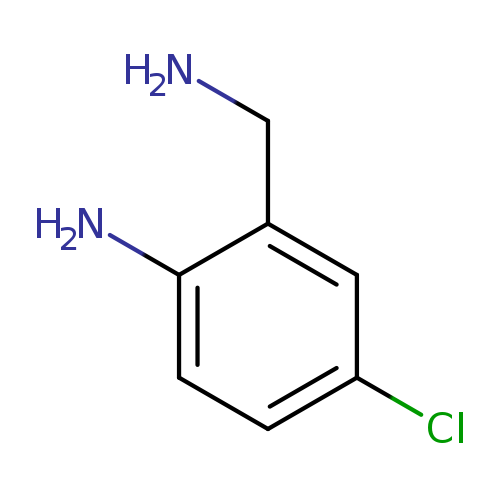
2-Aminomethyl-4-chloro-phenylamineCatalog No.:AA0083VS CAS No.:108047-39-8 MDL No.:MFCD06212615 MF:C7H9ClN2 MW:156.6128 |
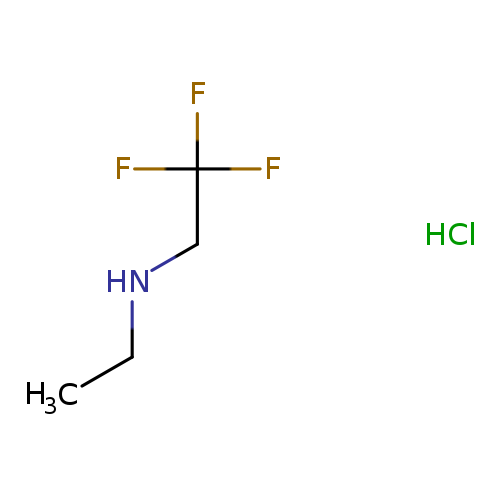
N-Ethyl-2,2,2-trifluoroethanamine hydrochlorideCatalog No.:AA00J1QY CAS No.:1080474-98-1 MDL No.:MFCD09971665 MF:C4H9ClF3N MW:163.5692 |
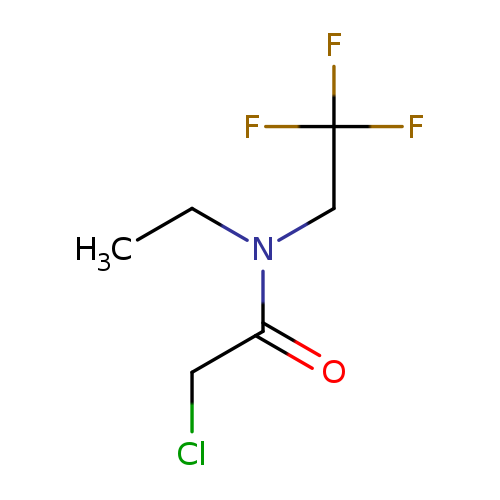
2-chloro-n-ethyl-n-(2,2,2-trifluoroethyl)acetamideCatalog No.:AA01BXV2 CAS No.:1080474-99-2 MDL No.:MFCD16058712 MF:C6H9ClF3NO MW:203.5900 |
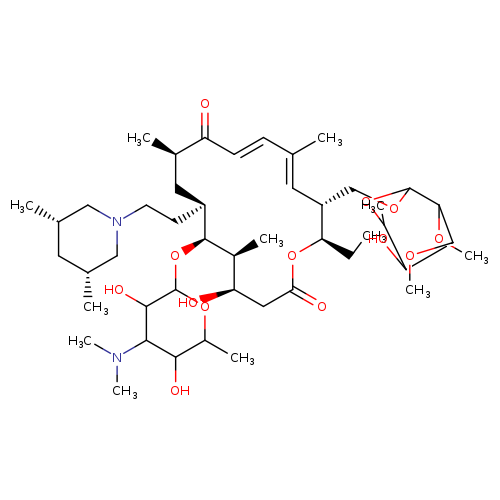
TilmicosinCatalog No.:AA0083VP CAS No.:108050-54-0 MDL No.:MFCD00864842 MF:C46H80N2O13 MW:869.1330 |
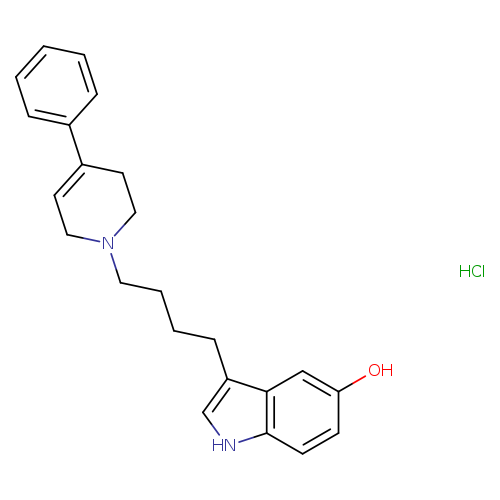
RoxindoleHydrochlorideCatalog No.:AA01DZGG CAS No.:108050-82-4 MDL No.: MF:C23H27ClN2O MW:382.9263 |
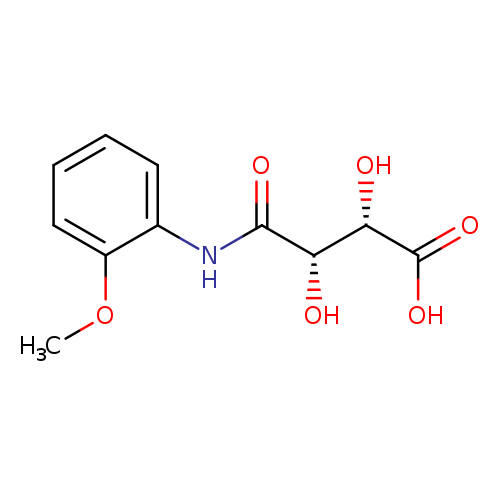
(2S,3S)-2,3-dihydroxy-3-[(2-methoxyphenyl)carbamoyl]propanoic acidCatalog No.:AA00J076 CAS No.:108052-07-9 MDL No.:MFCD28369728 MF:C11H13NO6 MW:255.2240 |
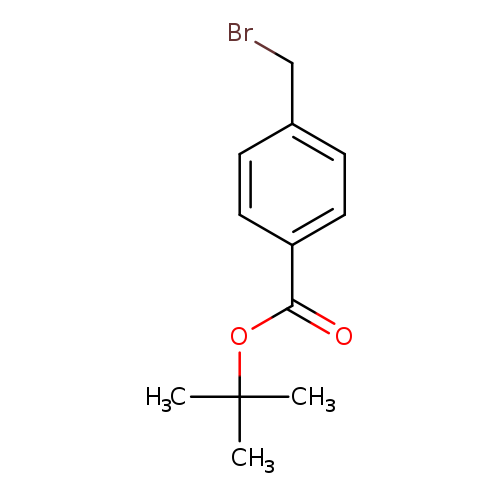
t-Butyl 4-bromomethylbenzoateCatalog No.:AA003K3T CAS No.:108052-76-2 MDL No.:MFCD04973450 MF:C12H15BrO2 MW:271.1503 |

Mirtazapine N-Glucuronide (Mixture of Diastereomers) Contains Unknown InorganicsCatalog No.:AA01CBGT CAS No.:1080533-15-8 MDL No.:MFCD24386435 MF:C23H27N3O6 MW:441.4770 |
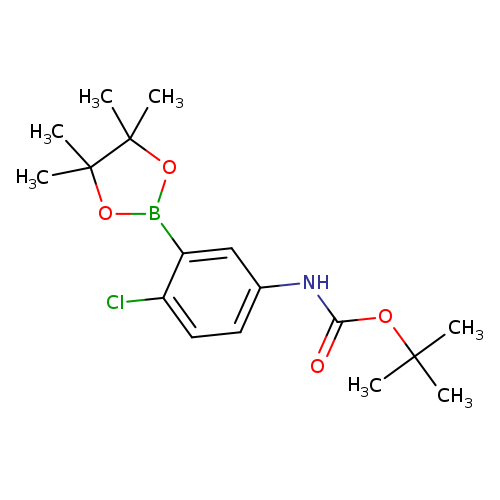
5-BOC-Amino-2-chlorophenylboronic acid pinacol esterCatalog No.:AA009474 CAS No.:1080573-28-9 MDL No.:MFCD23379564 MF:C17H25BClNO4 MW:353.6487 |
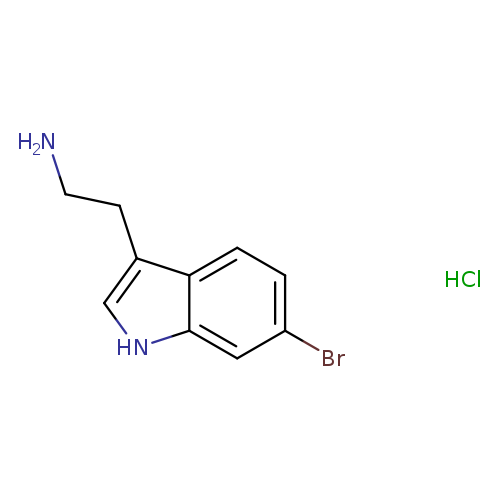
2-(6-Bromo-1h-indol-3-yl)ethanamine hydrochlorideCatalog No.:AA007V5O CAS No.:108061-77-4 MDL No.:MFCD11109987 MF:C10H12BrClN2 MW:275.5727 |
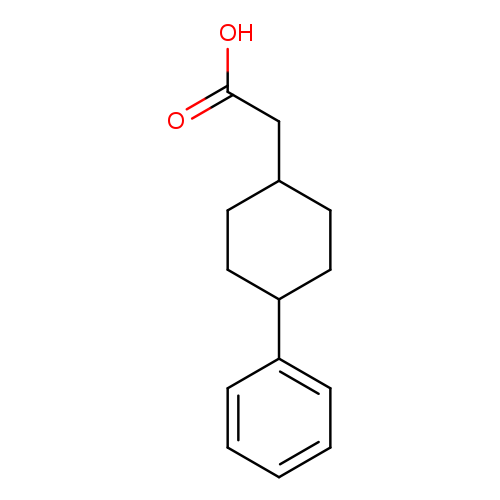
2-(4-Phenylcyclohexyl)acetic acidCatalog No.:AA009A1S CAS No.:108062-27-7 MDL No.:MFCD03414265 MF:C14H18O2 MW:218.2915 |
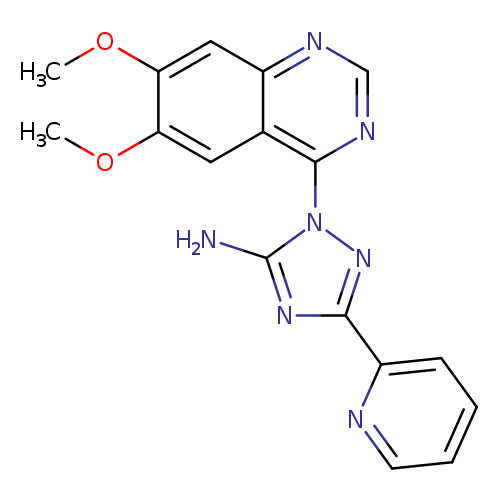
Cp-466722Catalog No.:AA008TCB CAS No.:1080622-86-1 MDL No.:MFCD17392566 MF:C17H15N7O2 MW:349.3467 |
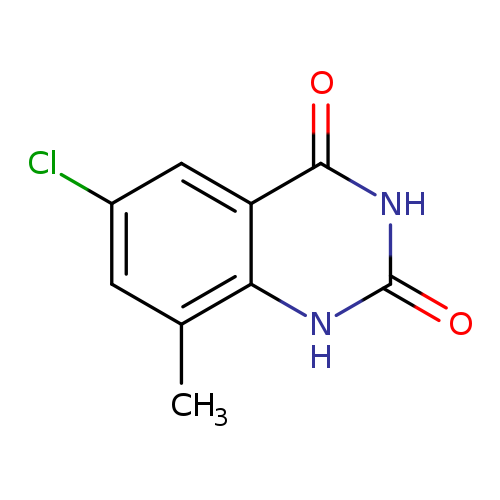
6-chloro-8-methyl-1,2,3,4-tetrahydroquinazoline-2,4-dioneCatalog No.:AA00ISLE CAS No.:1080622-94-1 MDL No.:MFCD24614691 MF:C9H7ClN2O2 MW:210.6171 |
4-Terphenyl boronic acid pinacol esterCatalog No.:AA0091YO CAS No.:1080632-76-3 MDL No.:MFCD16294429 MF:C24H25BO2 MW:356.2651 |
3-(2-Fluorophenyl)cyclobutan-1-oneCatalog No.:AA009OS4 CAS No.:1080636-31-2 MDL No.:MFCD11848521 MF:C10H9FO MW:164.1763 |
3-(2-Chlorophenyl)cyclobutanoneCatalog No.:AA00HAZM CAS No.:1080636-35-6 MDL No.:MFCD11848525 MF:C10H9ClO MW:180.6309 |
3-(2-methylphenyl)cyclobutan-1-oneCatalog No.:AA019V0X CAS No.:1080636-36-7 MDL No.:MFCD20233430 MF:C11H12O MW:160.2124 |
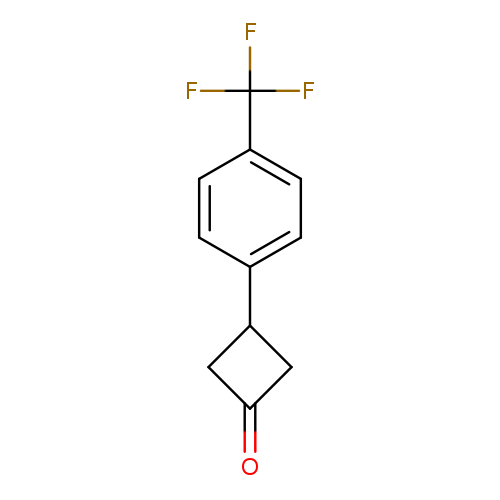
3-[4-(Trifluoromethyl)phenyl]cyclobutan-1-oneCatalog No.:AA01AJ99 CAS No.:1080636-43-6 MDL No.:MFCD18074125 MF:C11H9F3O MW:214.1838 |
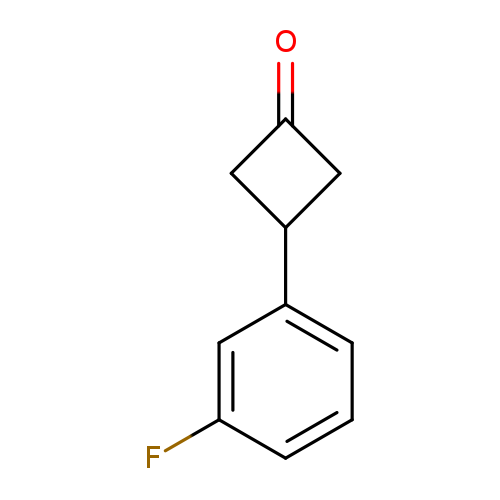
3-(3-fluorophenyl)cyclobutan-1-oneCatalog No.:AA019V0V CAS No.:1080636-45-8 MDL No.:MFCD11848522 MF:C10H9FO MW:164.1763 |
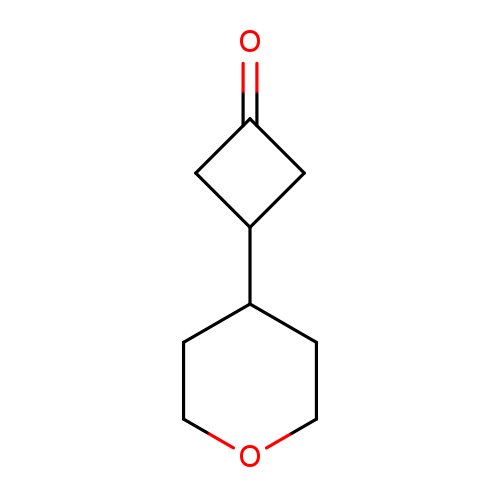
3-(OXAN-4-YL)CYCLOBUTAN-1-ONECatalog No.:AA01DX7A CAS No.:1080636-46-9 MDL No.:MFCD24712485 MF:C9H14O2 MW:154.2063 |
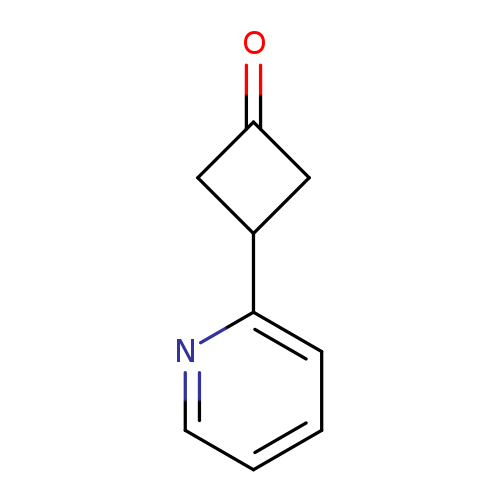
3-(Pyridin-2-yl)cyclobutan-1-oneCatalog No.:AA01BBKK CAS No.:1080636-50-5 MDL No.:MFCD14706866 MF:C9H9NO MW:147.1739 |
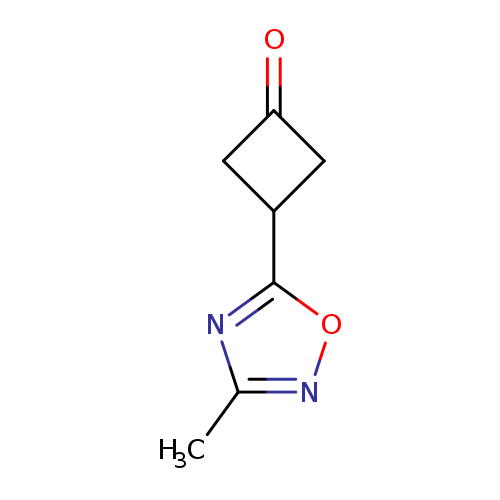
3-(3-METHYL-1,2,4-OXADIAZOL-5-YL)CYCLOBUTAN-1-ONECatalog No.:AA01DX7B CAS No.:1080636-56-1 MDL No.:MFCD24712043 MF:C7H8N2O2 MW:152.1506 |
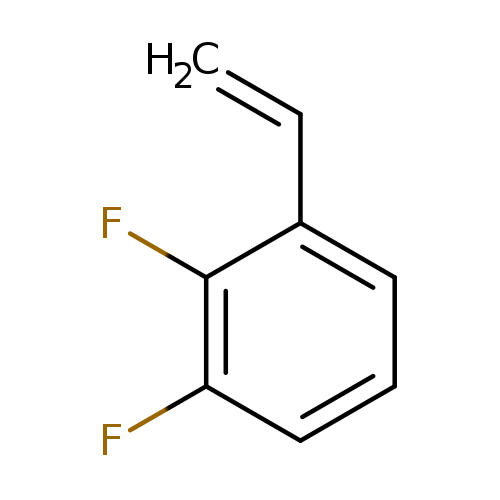
2,3-DifluorostyreneCatalog No.:AA00HAZN CAS No.:1080636-91-4 MDL No.:MFCD12827910 MF:C8H6F2 MW:140.1300 |
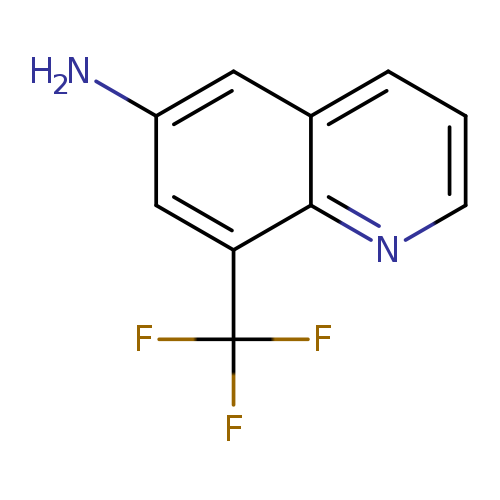
8-(Trifluoromethyl)quinolin-6-amineCatalog No.:AA0083VI CAS No.:1080640-91-0 MDL No.:MFCD11052592 MF:C10H7F3N2 MW:212.1712 |
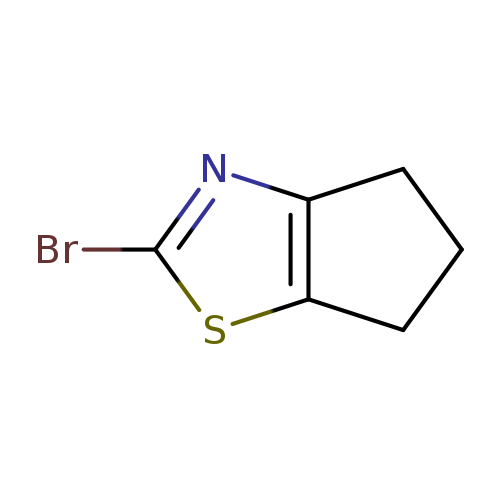
2-Bromo-5,6-dihydro-4h-cyclopenta[d]thiazoleCatalog No.:AA0083VH CAS No.:1080642-17-6 MDL No.:MFCD20921667 MF:C6H6BrNS MW:204.0875 |
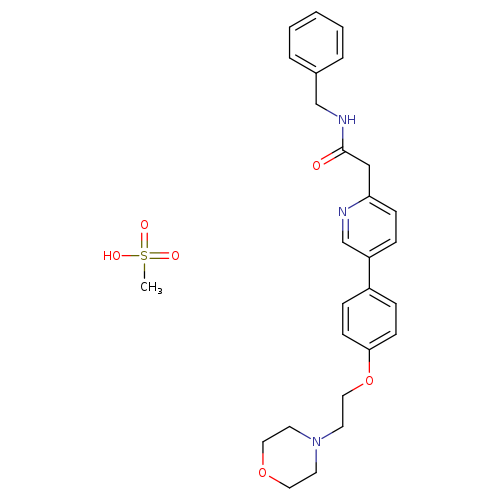
KX2-391 MesylateCatalog No.:AA01EO4R CAS No.:1080645-95-9 MDL No.:MFCD30496706 MF:C27H33N3O6S MW:527.6324 |
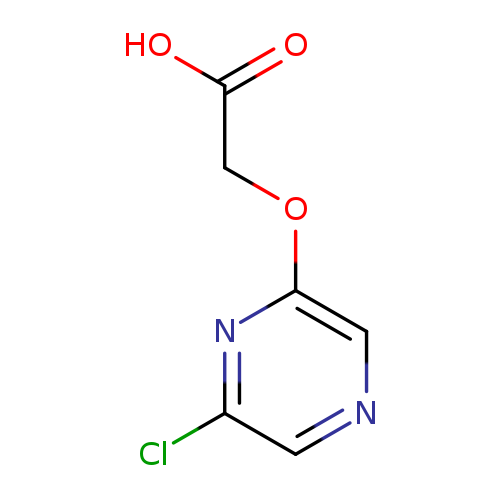
2-[(6-Chloropyrazin-2-yl)oxy]acetic acidCatalog No.:AA01AF4S CAS No.:1080650-29-8 MDL No.:MFCD20660485 MF:C6H5ClN2O3 MW:188.5685 |
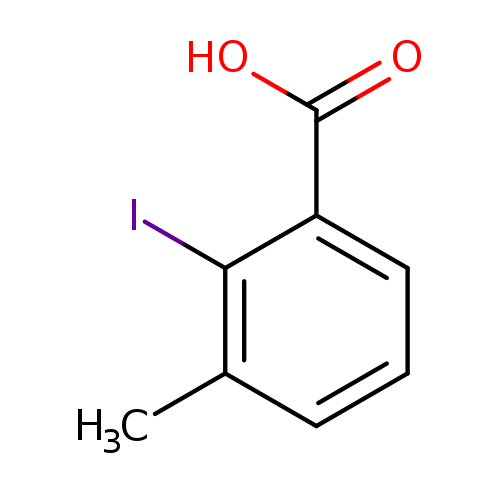
2-Iodo-3-methylbenzoic acidCatalog No.:AA003HDP CAS No.:108078-14-4 MDL No.:MFCD00079764 MF:C8H7IO2 MW:262.0444 |
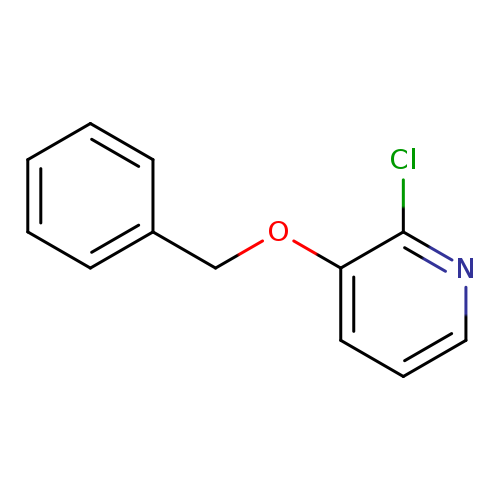
3-(Benzyloxy)-2-chloropyridineCatalog No.:AA008XNA CAS No.:108082-72-0 MDL No.:MFCD00661469 MF:C12H10ClNO MW:219.6669 |
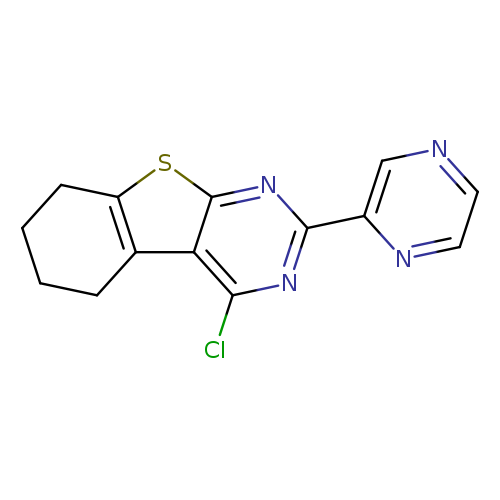
3-chloro-5-(pyrazin-2-yl)-8-thia-4,6-diazatricyclo[7.4.0.0,2,7]trideca-1(9),2,4,6-tetraeneCatalog No.:AA01A7V4 CAS No.:1080821-72-2 MDL No.:MFCD13805864 MF:C14H11ClN4S MW:302.7819 |
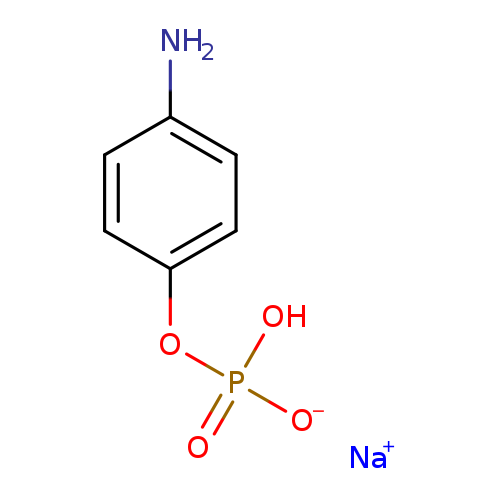
4-Aminophenyl phosphate monosodium salt hydrateCatalog No.:AA008SQZ CAS No.:108084-47-5 MDL No.:MFCD09841685 MF:C6H7NNaO4P MW:211.0876 |
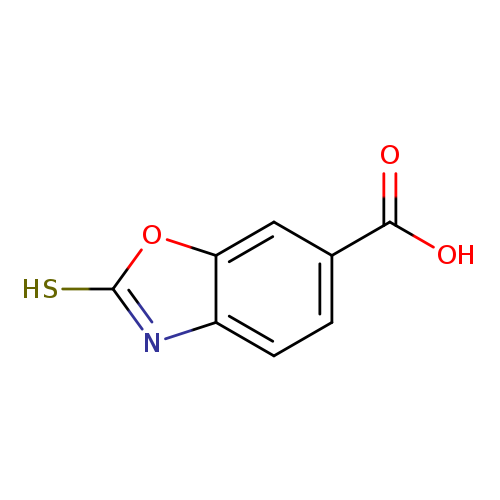
2-Mercaptobenzo[d]oxazole-6-carboxylic acidCatalog No.:AA01AJD8 CAS No.:108085-62-7 MDL No.:MFCD09259279 MF:C8H5NO3S MW:195.1952 |
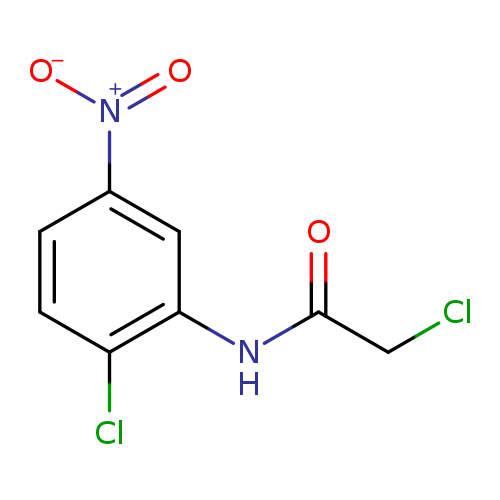
2-Chloro-n-(2-chloro-5-nitrophenyl)acetamideCatalog No.:AA007V5J CAS No.:108086-37-9 MDL No.:MFCD01357758 MF:C8H6Cl2N2O3 MW:249.0508 |
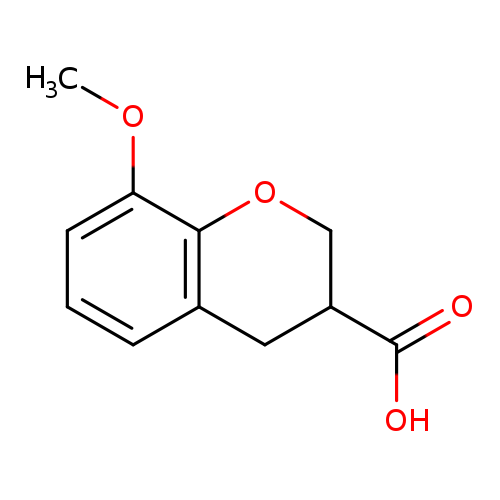
8-Methoxy-chroman-3-carboxylic acidCatalog No.:AA007D8U CAS No.:108088-19-3 MDL No.:MFCD04114622 MF:C11H12O4 MW:208.2106 |
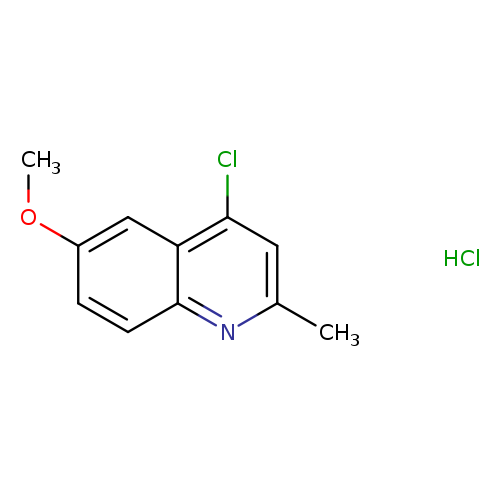
4-chloro-6-methoxy-2-methylquinoline hydrochlorideCatalog No.:AA01AHMA CAS No.:108096-99-7 MDL No.:MFCD00451825 MF:C11H11Cl2NO MW:244.1171 |
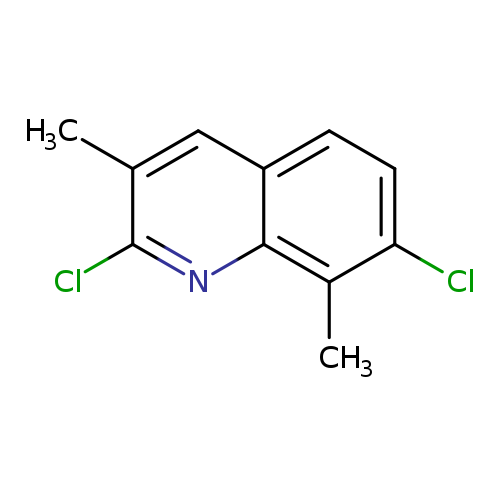
2,7-Dichloro-3,8-dimethylquinolineCatalog No.:AA007V5I CAS No.:108097-00-3 MDL No.:MFCD09787544 MF:C11H9Cl2N MW:226.1019 |
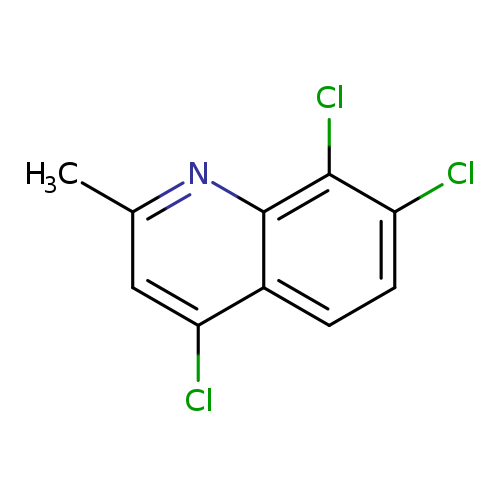
2-Methyl-4,7,8-trichloroquinolineCatalog No.:AA007V5H CAS No.:108097-02-5 MDL No.:MFCD00272309 MF:C10H6Cl3N MW:246.5203 |
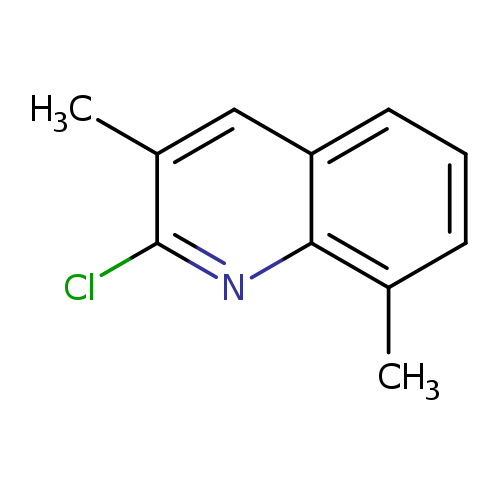
2-Chloro-3,8-dimethylquinolineCatalog No.:AA0083VA CAS No.:108097-04-7 MDL No.:MFCD07644546 MF:C11H10ClN MW:191.6568 |
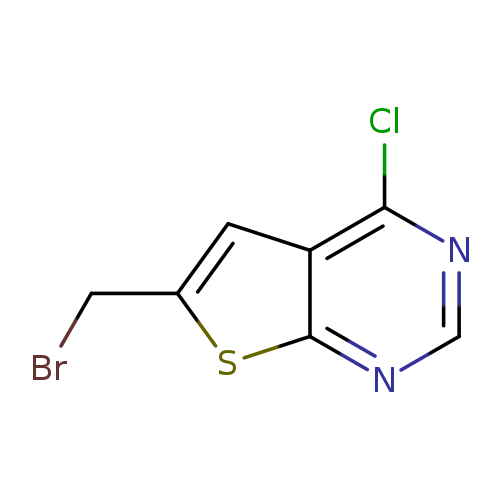
6-(BROMOMETHYL)-4-CHLOROTHIENO[2,3-D]PYRIMIDINECatalog No.:AA0094PF CAS No.:108099-55-4 MDL No.:MFCD11215541 MF:C7H4BrClN2S MW:263.5421 |
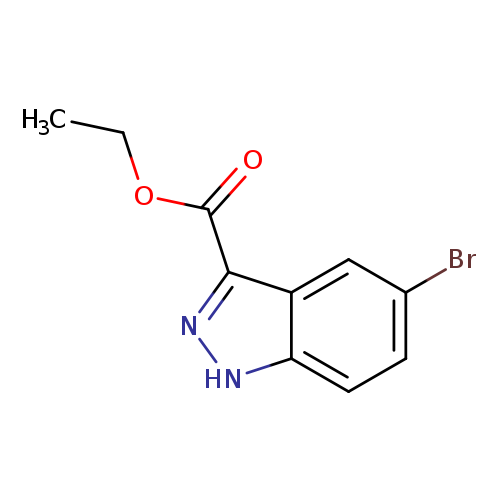
ethyl 5-bromo-1H-indazole-3-carboxylateCatalog No.:AA007D8P CAS No.:1081-04-5 MDL No.:MFCD05663980 MF:C10H9BrN2O2 MW:269.0947 |
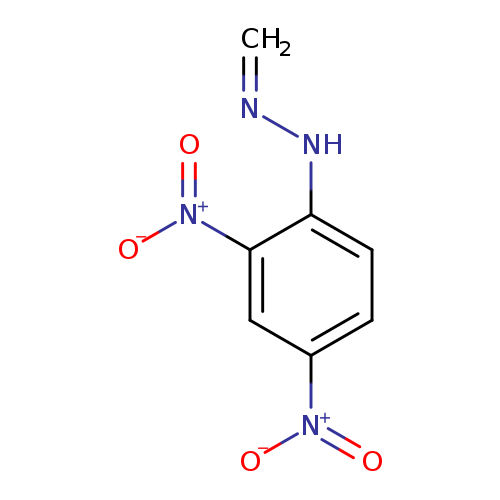
FORMALDEHYDE 2,4-DINITROPHENYLHYDRAZONECatalog No.:AA003QLS CAS No.:1081-15-8 MDL No.:MFCD00191364 MF:C7H6N4O4 MW:210.1469 |
2,2':5',2''-TErthiopheneCatalog No.:AA0035QH CAS No.:1081-34-1 MDL No.:MFCD00012167 MF:C12H8S3 MW:248.3869 |
4-Hydroxy-3-methoxyphenylpyruvic acidCatalog No.:AA003LF9 CAS No.:1081-71-6 MDL No.:MFCD00056707 MF:C10H10O5 MW:210.1834 |
N-[2-(4-Bromophenoxy)ethyl]pyrrolidineCatalog No.:AA003CPT CAS No.:1081-73-8 MDL No.:MFCD00003180 MF:C12H16BrNO MW:270.1655 |
1-PhenylnonaneCatalog No.:AA003EJU CAS No.:1081-77-2 MDL No.:MFCD00009575 MF:C15H24 MW:204.3511 |
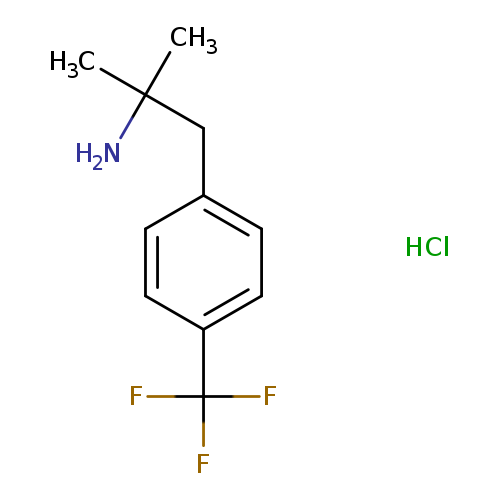
D-380Catalog No.:AA009MYR CAS No.:1081-78-3 MDL No.:MFCD01761830 MF:C11H15ClF3N MW:253.6917 |
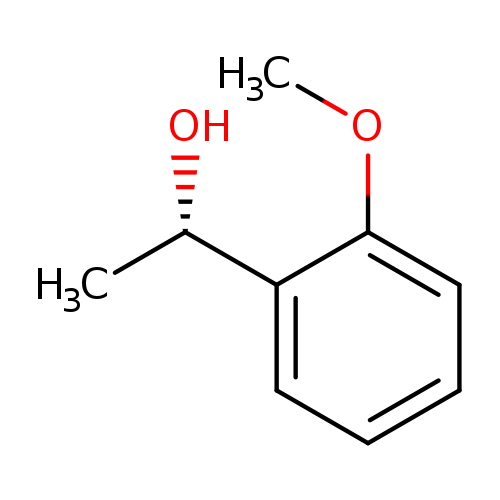
(S)-1-(2-Methoxyphenyl)ethanolCatalog No.:AA008SAQ CAS No.:108100-06-7 MDL No.:MFCD06659506 MF:C9H12O2 MW:152.1904 |
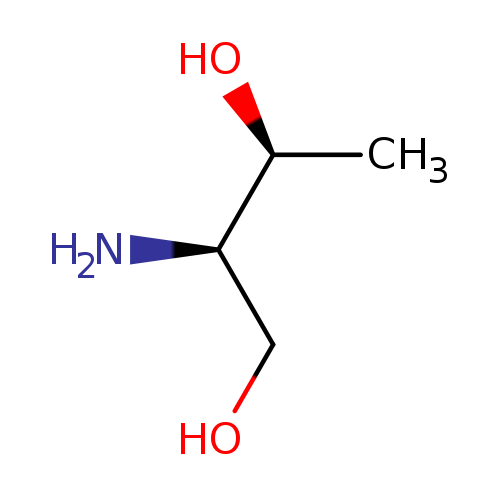
(2R,3S)-2-Aminobutane-1,3-diolCatalog No.:AA0083V7 CAS No.:108102-48-3 MDL No.:MFCD00672370 MF:C4H11NO2 MW:105.1356 |
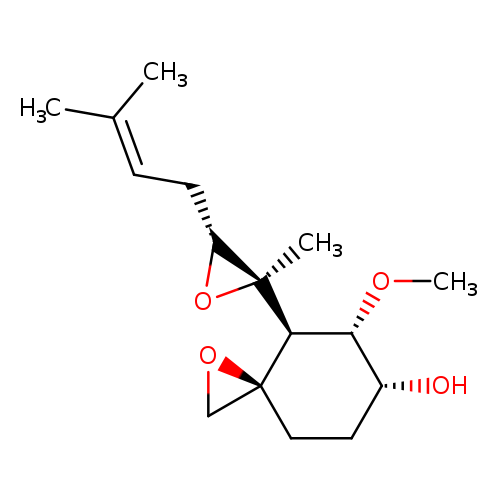
1-Oxaspiro[2.5]octan-6-ol,5-methoxy-4-[(2R,3R)-2-methyl-3-(3-methyl-2-buten-1-yl)-2-oxiranyl]-,(3R,4S,5S,6R)-Catalog No.:AA007V5A CAS No.:108102-51-8 MDL No.:MFCD04035560 MF:C16H26O4 MW:282.3752 |
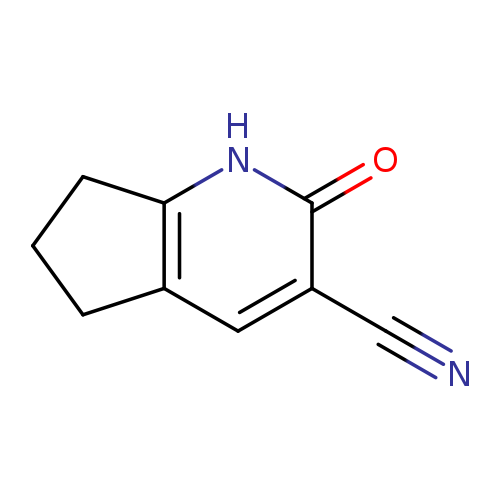
2-Oxo-1h,2h,5h,6h,7h-cyclopenta[b]pyridine-3-carbonitrileCatalog No.:AA008RG2 CAS No.:108106-97-4 MDL No.:MFCD03851982 MF:C9H8N2O MW:160.1726 |
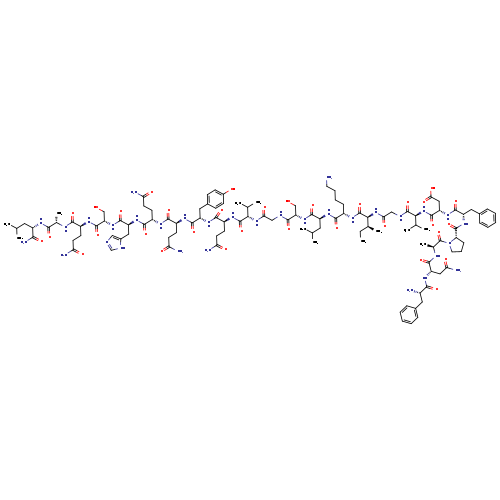
H-PHE-ASN-ALA-PRO-PHE-ASP-VAL-GLY-ILE-LYS-LEU-SER-GLY-VAL-GLN-TYR-GLN-GLN-HIS-SER-GLN-ALA-LEU-NH2Catalog No.:AA008RTX CAS No.:1081110-72-6 MDL No.:MFCD08458633 MF:C116H176N32O33 MW:2546.8332 |
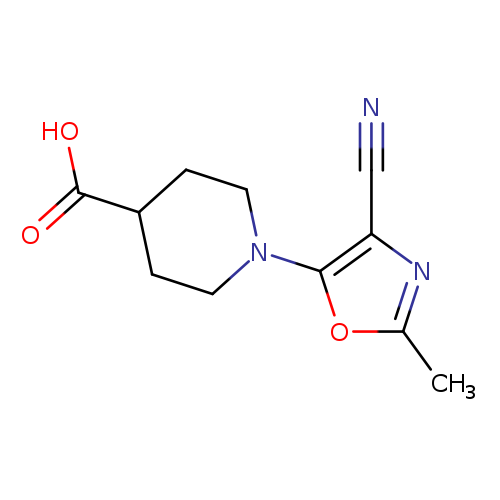
1-(4-cyano-2-methyl-1,3-oxazol-5-yl)piperidine-4-carboxylic acidCatalog No.:AA01B186 CAS No.:1081115-59-4 MDL No.:MFCD11501633 MF:C11H13N3O3 MW:235.2392 |
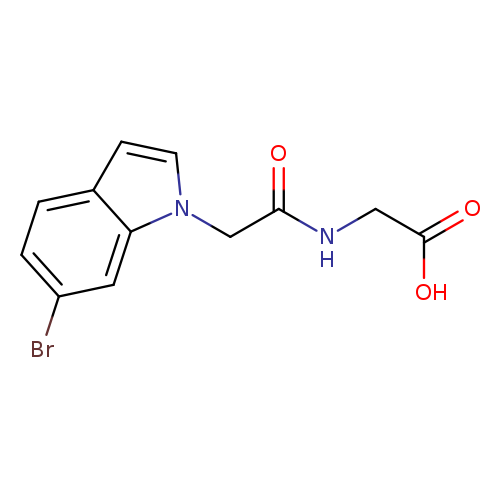
2-[2-(6-bromo-1h-indol-1-yl)acetamido]acetic acidCatalog No.:AA01B50U CAS No.:1081143-33-0 MDL No.:MFCD12206542 MF:C12H11BrN2O3 MW:311.1313 |
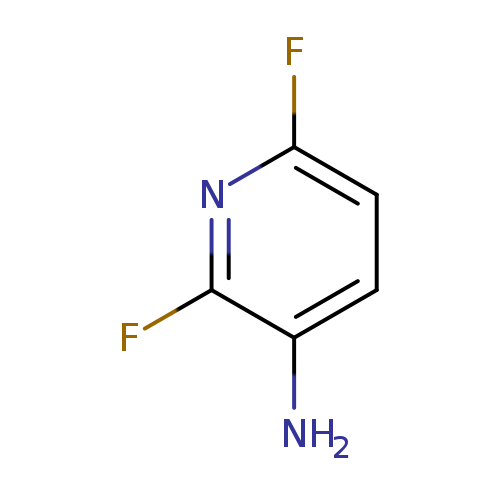
2,6-Difluoropyridin-3-amineCatalog No.:AA0093WS CAS No.:108118-69-0 MDL No.:MFCD13185331 MF:C5H4F2N2 MW:130.0955 |
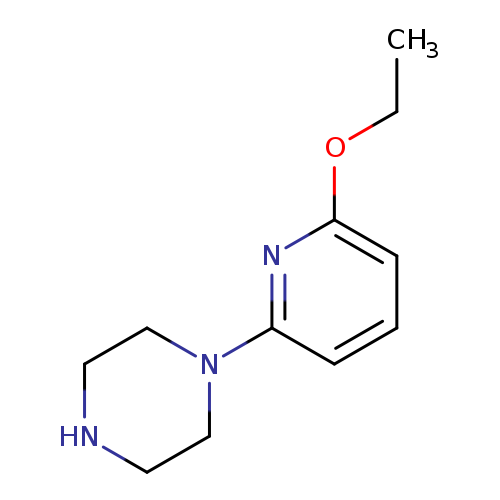
1-(6-ETHOXYPYRIDIN-2-YL)PIPERAZINECatalog No.:AA003CZ3 CAS No.:108122-25-4 MDL No.:MFCD08061051 MF:C11H17N3O MW:207.2722 |
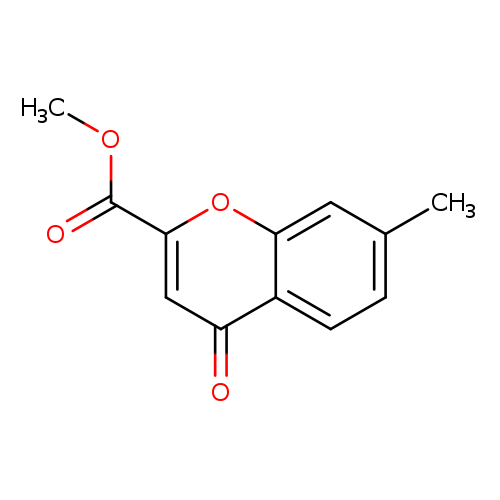
methyl 7-methyl-4-oxo-4Н-chromene-2-carboxylateCatalog No.:AA01EID0 CAS No.:108123-49-5 MDL No.:MFCD18485349 MF:C12H10O4 MW:218.2054 |
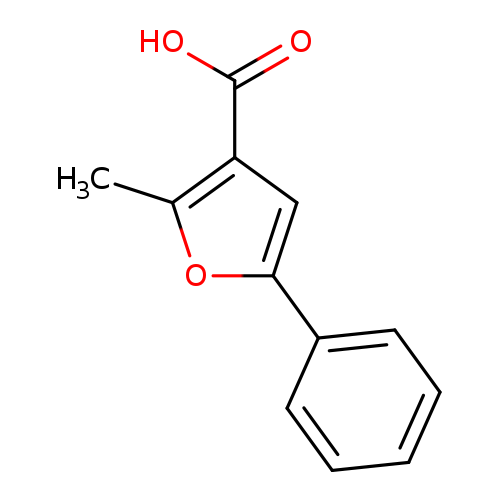
2-Methyl-5-phenylfuran-3-carboxylic acidCatalog No.:AA003HL4 CAS No.:108124-17-0 MDL No.:MFCD00221070 MF:C12H10O3 MW:202.2060 |
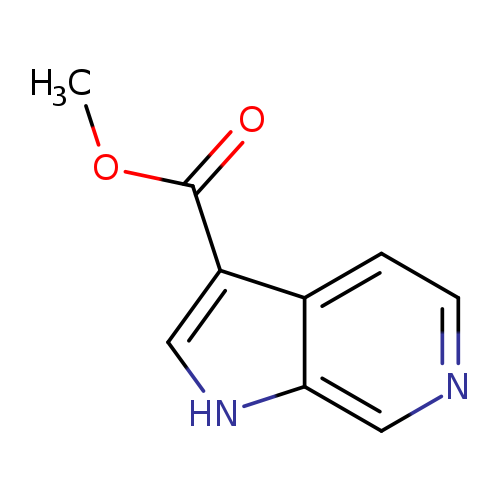
Methyl 1h-pyrrolo[2,3-c]pyridine-3-carboxylateCatalog No.:AA008XNX CAS No.:108128-12-7 MDL No.:MFCD16036494 MF:C9H8N2O2 MW:176.1720 |
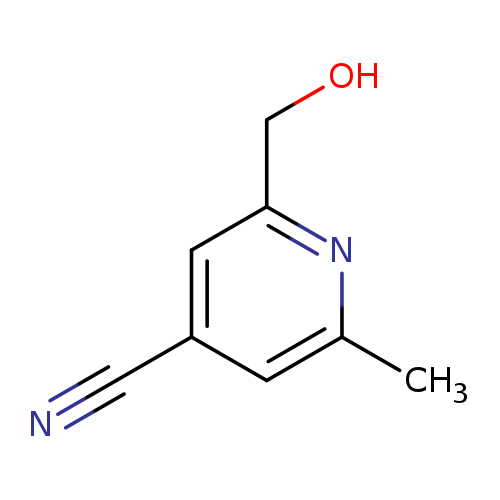
2-(Hydroxymethyl)-6-methylisonicotinonitrileCatalog No.:AA0091GB CAS No.:108129-25-5 MDL No.:MFCD18384585 MF:C8H8N2O MW:148.1619 |
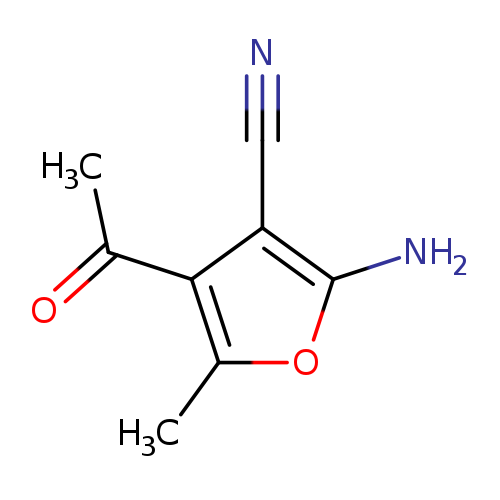
4-Acetyl-2-amino-5-methyl-3-furonitrileCatalog No.:AA0083UZ CAS No.:108129-35-7 MDL No.:MFCD06618375 MF:C8H8N2O2 MW:164.1613 |
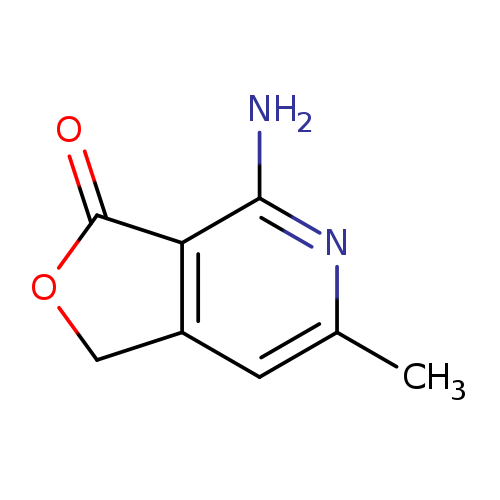
4-Amino-6-methylfuro[3,4-c]pyridin-3(1h)-oneCatalog No.:AA007V4P CAS No.:108129-37-9 MDL No.:MFCD02989821 MF:C8H8N2O2 MW:164.1613 |
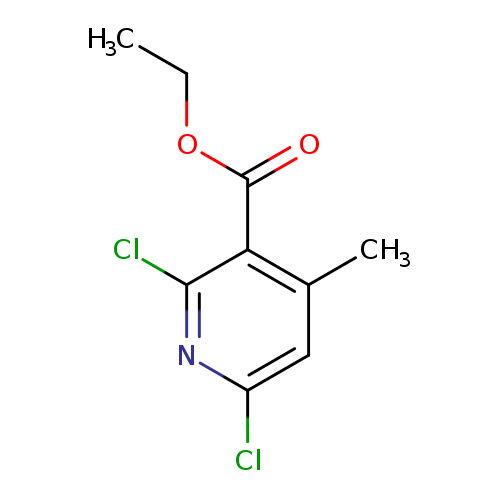
Ethyl 2,6-dichloro-4-methylnicotinateCatalog No.:AA007D8G CAS No.:108130-10-5 MDL No.:MFCD21604262 MF:C9H9Cl2NO2 MW:234.0793 |
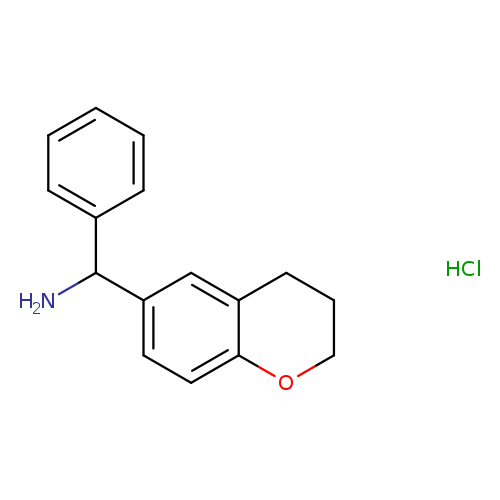
3,4-dihydro-2H-1-benzopyran-6-yl(phenyl)methanamine hydrochlorideCatalog No.:AA01AGFJ CAS No.:108132-34-9 MDL No.:MFCD01662543 MF:C16H18ClNO MW:275.7732 |
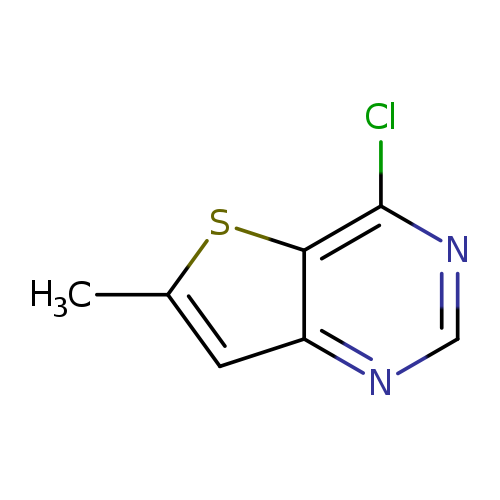
4-Chloro-6-methyl-thieno[3,2-d]pyrimidineCatalog No.:AA007D8B CAS No.:108134-22-1 MDL No.:MFCD17015877 MF:C7H5ClN2S MW:184.6460 |
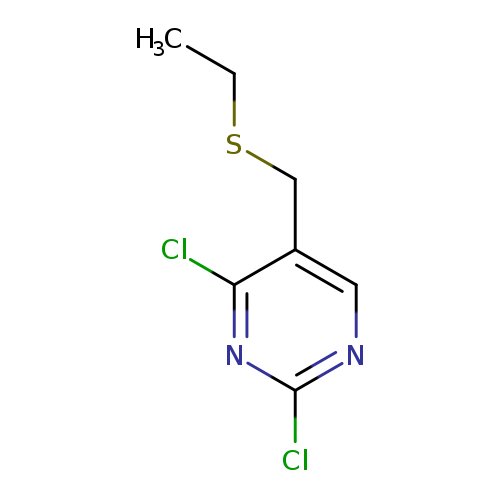
2,4-Dichloro-5-[(ethylthio)methyl]pyrimidineCatalog No.:AA008S4M CAS No.:108141-35-1 MDL No.:MFCD01631198 MF:C7H8Cl2N2S MW:223.1228 |
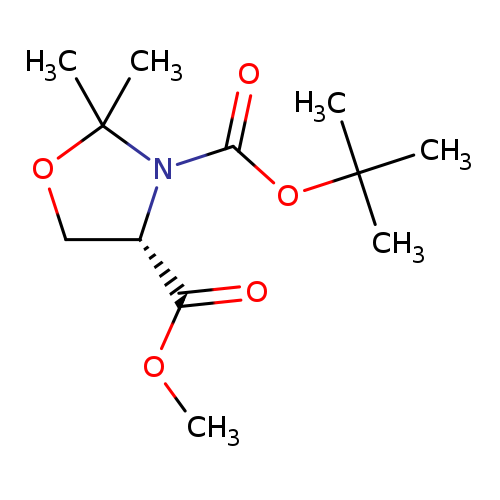
(S)-(-)-3-BOC-4-methoxycarbonyl-2,2-dimethyl-1,3-oxazolidineCatalog No.:AA007V39 CAS No.:108149-60-6 MDL No.:MFCD00192279 MF:C12H21NO5 MW:259.2988 |
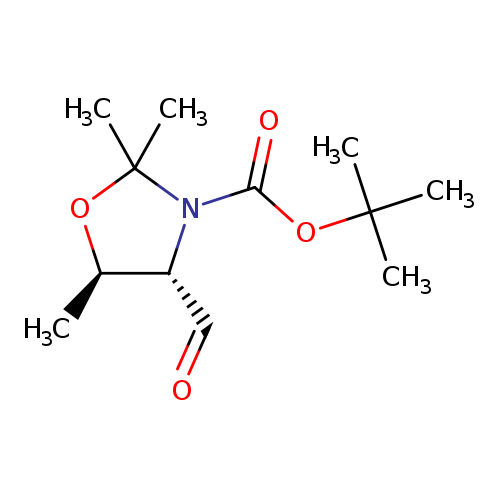
(4S,5R)-tert-Butyl 4-formyl-2,2,5-trimethyloxazolidine-3-carboxylateCatalog No.:AA008WBQ CAS No.:108149-62-8 MDL No.:MFCD16613945 MF:C12H21NO4 MW:243.2994 |
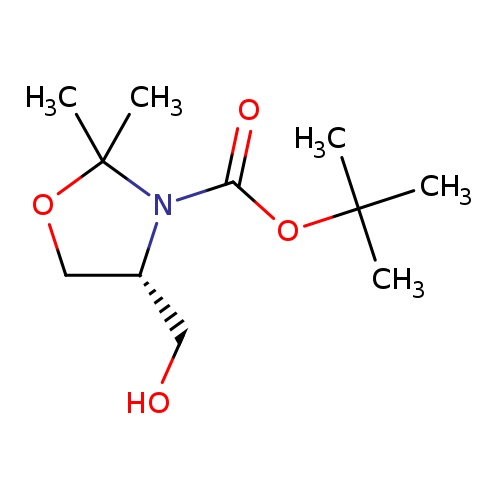
(R)-4-Hydroxymethyl-2,2-dimethyl-oxazolidine-3-carboxylic acid tert-butyl esterCatalog No.:AA003C8W CAS No.:108149-63-9 MDL No.:MFCD06202686 MF:C11H21NO4 MW:231.2887 |
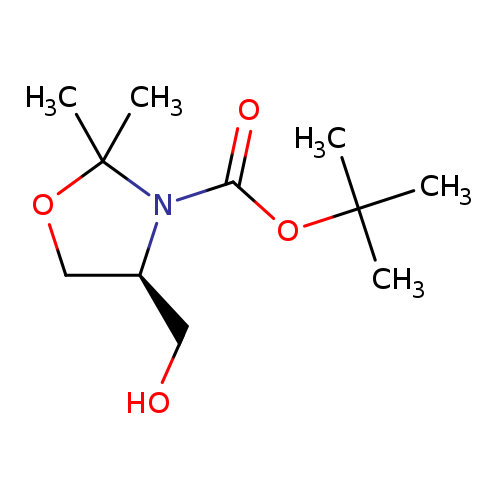
(S)-1-Boc-2,2-dimethyl-4-hydroxymethyl-oxazolidineCatalog No.:AA008UCB CAS No.:108149-65-1 MDL No.:MFCD11041410 MF:C11H21NO4 MW:231.2887 |
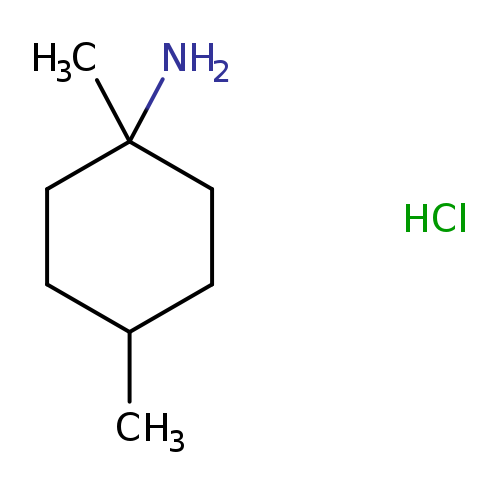
1,4-Dimethylcyclohexan-1-amine hydrochlorideCatalog No.:AA01A8KC CAS No.:1081513-98-5 MDL No.:MFCD16040119 MF:C8H18ClN MW:163.6882 |
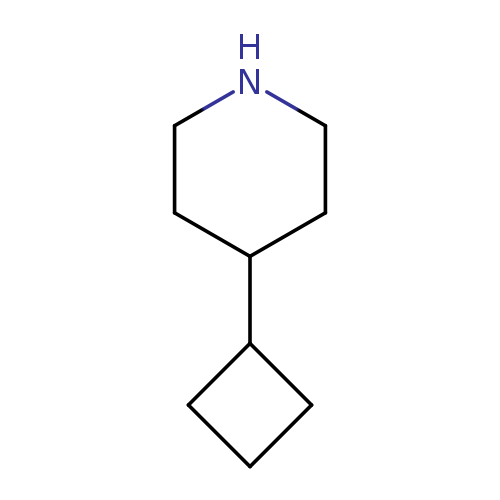
4-cyclobutylpiperidineCatalog No.:AA01B2LD CAS No.:1081514-03-5 MDL No.:MFCD23914591 MF:C9H17N MW:139.2380 |
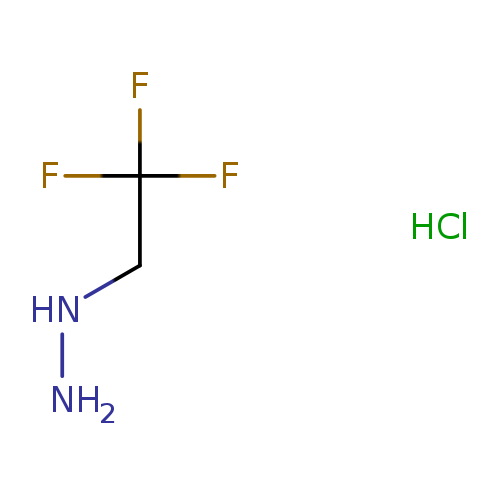
(2,2,2-trifluoroethyl)hydrazine hydrochlorideCatalog No.:AA01BTCZ CAS No.:1081515-82-3 MDL No.:MFCD27920859 MF:C2H6ClF3N2 MW:150.5306 |
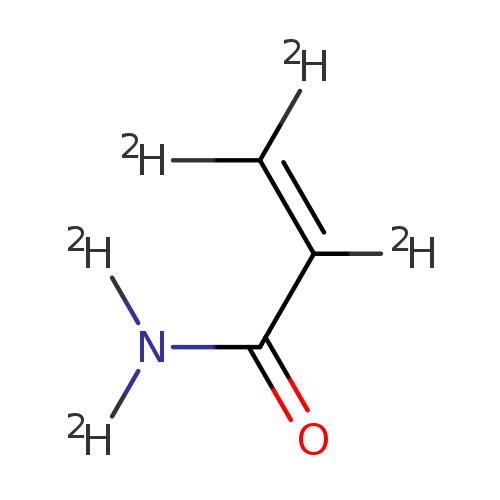
ACRYLAMIDE-D5Catalog No.:AA008SX3 CAS No.:108152-65-4 MDL No.:MFCD06658686 MF:C3D5NO MW:76.1087 |
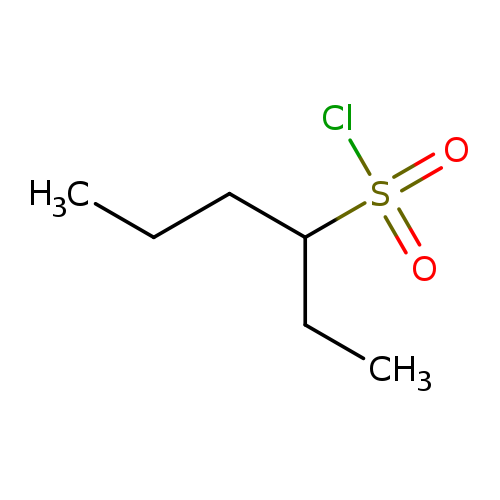
hexane-3-sulfonyl chlorideCatalog No.:AA01BHA5 CAS No.:1081524-29-9 MDL No.:MFCD19200632 MF:C6H13ClO2S MW:184.6842 |
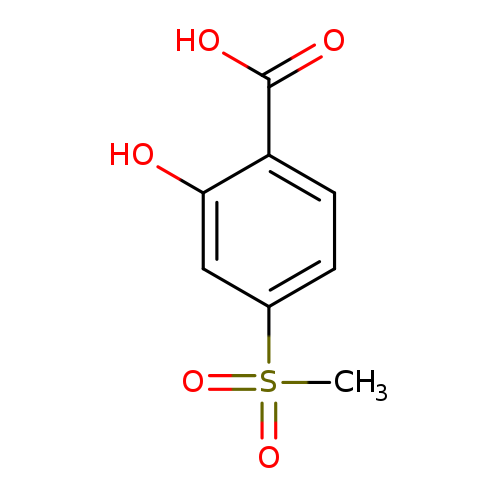
2-Hydroxy-4-methanesulfonylbenzoic acidCatalog No.:AA00HB03 CAS No.:108153-42-0 MDL No.:MFCD24483010 MF:C8H8O5S MW:216.2111 |
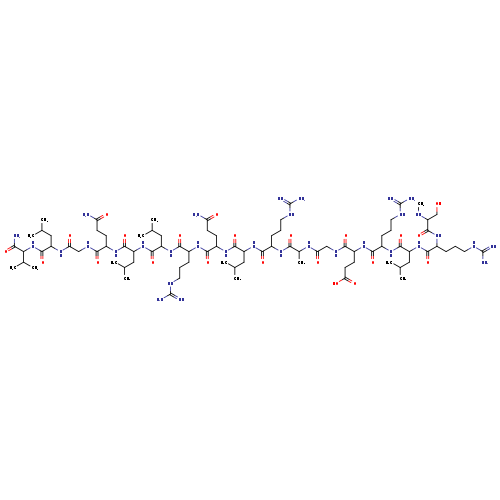
Secretin (human)Catalog No.:AA01DQ4W CAS No.:108153-74-8 MDL No.:MFCD00135670 MF:C85H156N32O22 MW:1978.3493 |
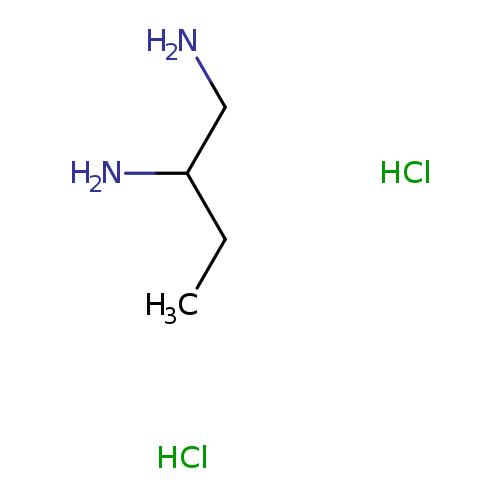
butane-1,2-diamine dihydrochlorideCatalog No.:AA01AAND CAS No.:1081531-27-2 MDL No.:MFCD17480383 MF:C4H14Cl2N2 MW:161.0734 |
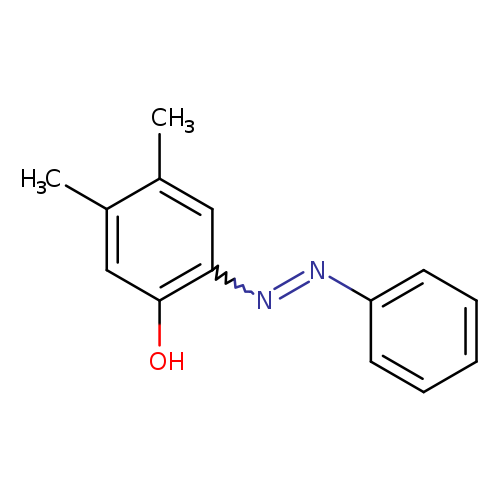
4,5-Dimethyl-2-[(E)-2-phenyldiazen-1-yl]phenolCatalog No.:AA01FG8I CAS No.:1081544-43-5 MDL No.:MFCD00086206 MF:C14H14N2O MW:226.2738 |
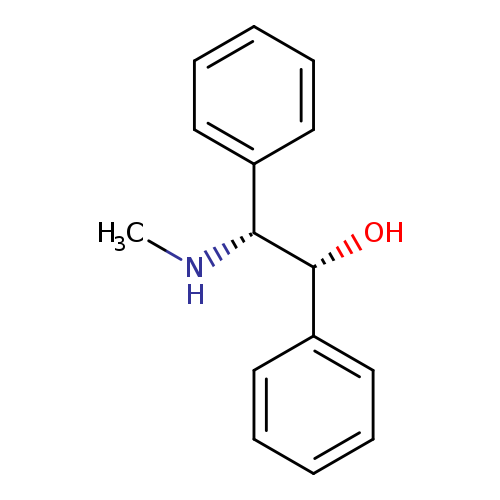
(1R,2R)-2-(Methylamino)-1,2-diphenylethanolCatalog No.:AA01DKA4 CAS No.:1081548-91-5 MDL No.:MFCD29919220 MF:C15H17NO MW:227.3016 |
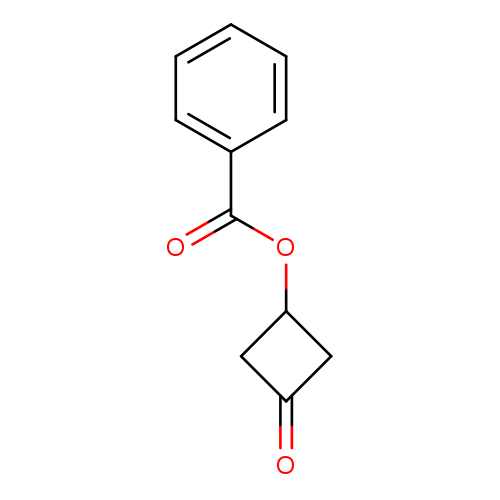
3-Oxocyclobutyl benzoateCatalog No.:AA008U1E CAS No.:1081559-36-5 MDL No.:MFCD16140376 MF:C11H10O3 MW:190.1953 |

Myristoyl-Stearoyl-3-chloropropanolCatalog No.:AA01DZGI CAS No.:1081560-89-5 MDL No.: MF:C20H18O6 MW:354.3533 |
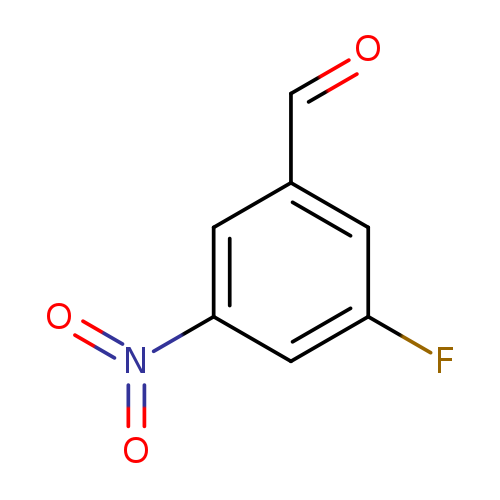
3-Fluoro-5-nitrobenzaldehydeCatalog No.:AA008UEW CAS No.:108159-96-2 MDL No.:MFCD11111000 MF:C7H4FNO3 MW:169.1100 |
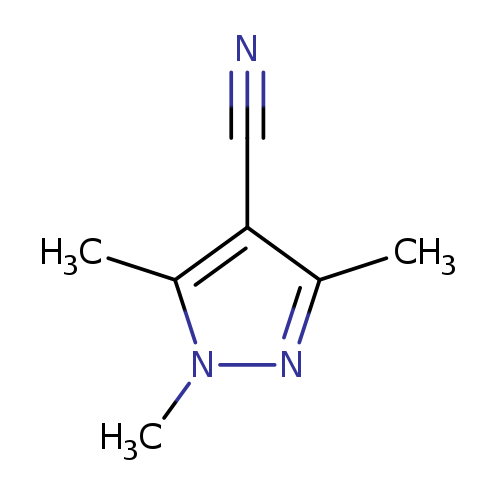
1,3,5-Trimethyl-1h-pyrazole-4-carbonitrileCatalog No.:AA008VBU CAS No.:108161-13-3 MDL No.:MFCD00173421 MF:C7H9N3 MW:135.1665 |
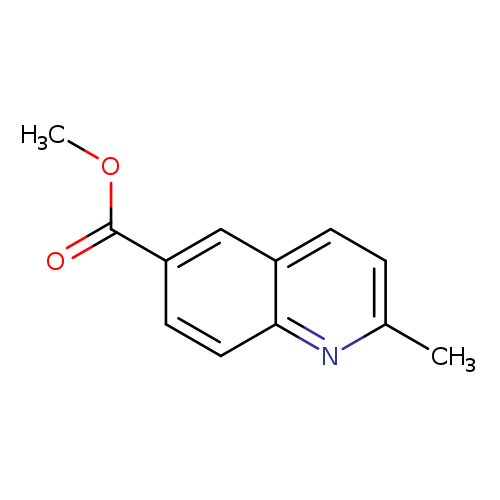
Methyl 2-methylquinoline-6-carboxylateCatalog No.:AA007D83 CAS No.:108166-01-4 MDL No.:MFCD02690144 MF:C12H11NO2 MW:201.2212 |
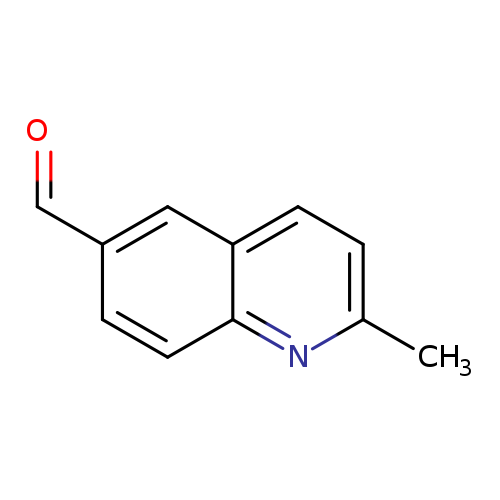
2-Methyl-6-quinolinecarbaldehydeCatalog No.:AA0083UQ CAS No.:108166-03-6 MDL No.:MFCD08060502 MF:C11H9NO MW:171.1953 |
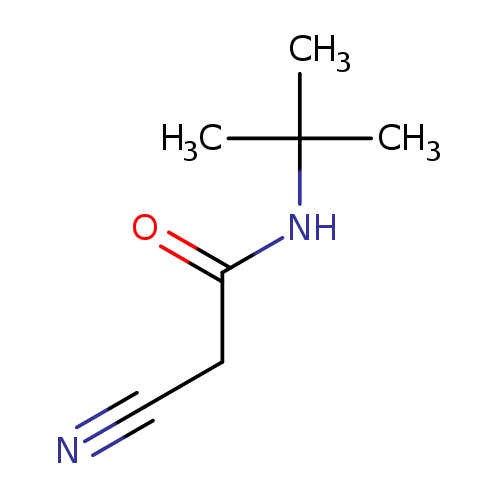
N-(tert-Butyl)-2-cyanoacetamideCatalog No.:AA0090KF CAS No.:108168-88-3 MDL No.:MFCD01351088 MF:C7H12N2O MW:140.1830 |
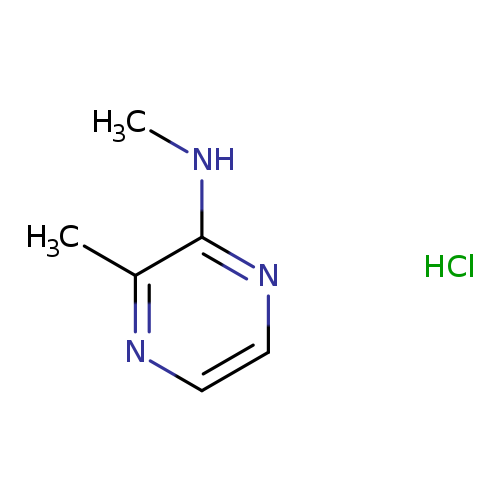
N,3-dimethylpyrazin-2-amine hydrochlorideCatalog No.:AA01E8HP CAS No.:1081759-44-5 MDL No.:MFCD30724309 MF:C6H10ClN3 MW:159.6167 |
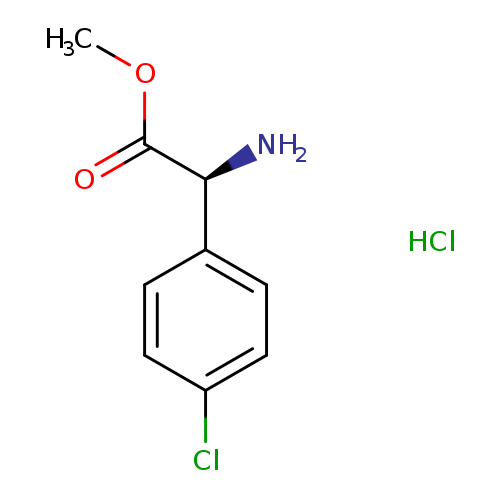
(S)-Methyl 2-amino-2-(4-chlorophenyl)acetate hydrochlorideCatalog No.:AA01B786 CAS No.:1081766-09-7 MDL No.:MFCD12910955 MF:C9H11Cl2NO2 MW:236.0951 |
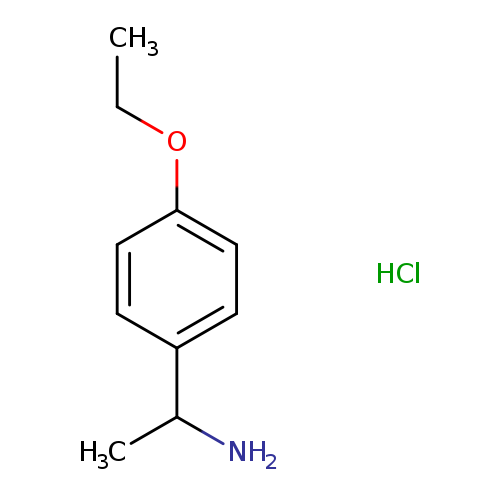
1-(4-Ethoxyphenyl)ethanamine hydrochlorideCatalog No.:AA00J0Z5 CAS No.:1081775-30-5 MDL No.:MFCD11841217 MF:C10H16ClNO MW:201.6931 |
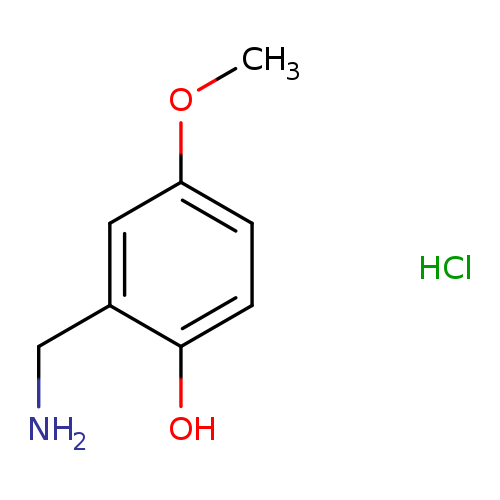
2-(aminomethyl)-4-methoxyphenol hydrochlorideCatalog No.:AA01AI9L CAS No.:1081777-51-6 MDL No.:MFCD18838996 MF:C8H12ClNO2 MW:189.6394 |
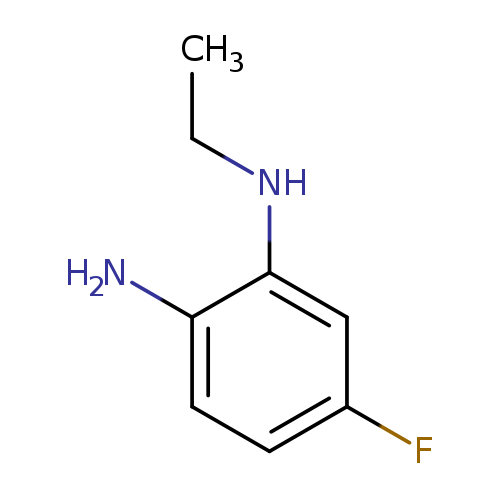
1-N-ethyl-5-fluorobenzene-1,2-diamineCatalog No.:AA01A8RD CAS No.:1081777-81-2 MDL No.:MFCD16744214 MF:C8H11FN2 MW:154.1847 |
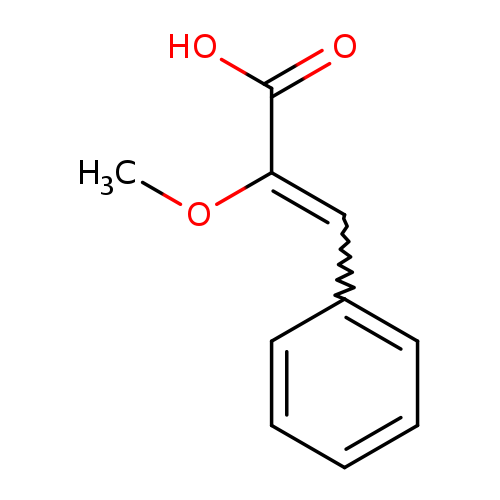
2-methoxy-3-phenylprop-2-enoic acidCatalog No.:AA01BDZG CAS No.:1081778-14-4 MDL No.:MFCD00799235 MF:C10H10O3 MW:178.1846 |
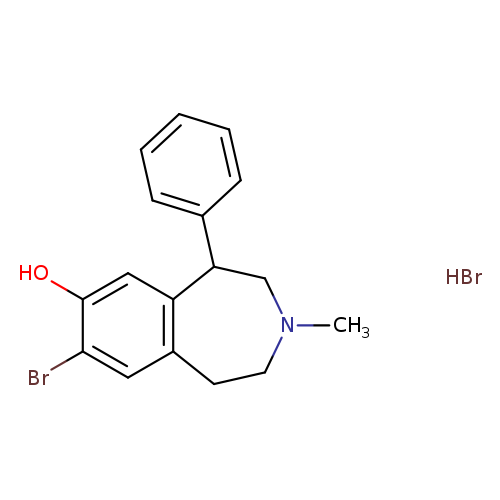
SKF 83566 hydrobromideCatalog No.:AA003U75 CAS No.:108179-91-5 MDL No.:MFCD04039787 MF:C17H19Br2NO MW:413.1469 |
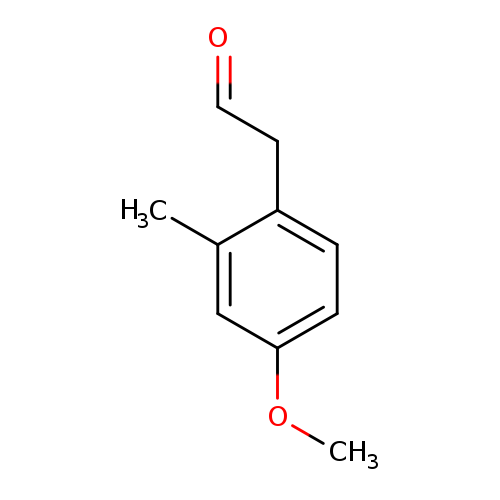
4-Methoxy-2-methylbenzeneacetaldehydeCatalog No.:AA0093WK CAS No.:1081798-35-7 MDL No.:MFCD02261771 MF:C10H12O2 MW:164.2011 |
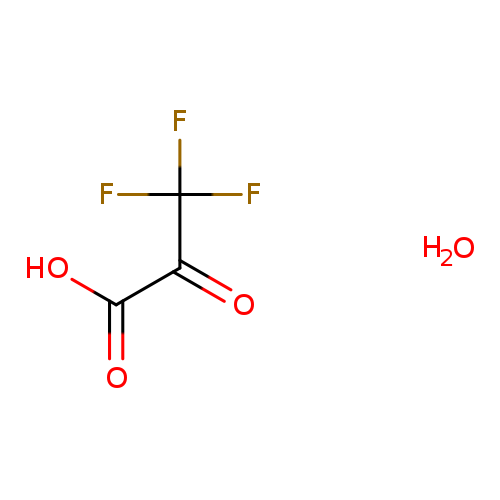
Trifluoropyruvic acid monohydrateCatalog No.:AA01BDRT CAS No.:1081801-99-1 MDL No.:MFCD04038370 MF:C3H3F3O4 MW:160.0487 |
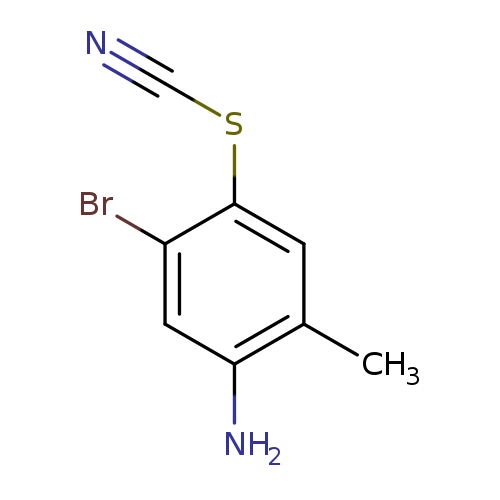
5-Bromo-2-methyl-4-thiocyanatoanilineCatalog No.:AA003MEW CAS No.:1081803-34-0 MDL No.:MFCD11855817 MF:C8H7BrN2S MW:243.1236 |

1,1,1-trifluoro-3-nitrobut-2-ene, ECatalog No.:AA019XMG CAS No.:1081822-33-4 MDL No.:MFCD09738674 MF: MW: |
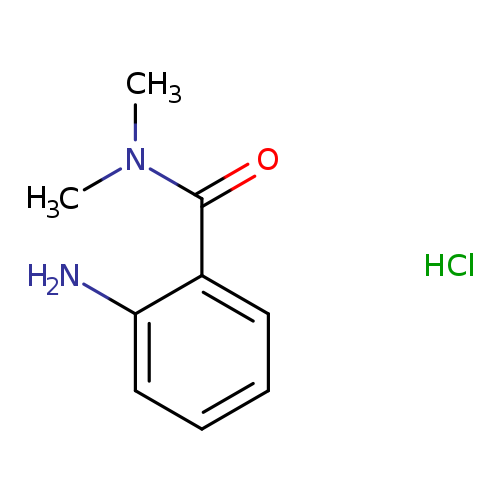
2-amino-N,N-dimethylbenzamide hydrochlorideCatalog No.:AA00J3NN CAS No.:1081834-96-9 MDL No.:MFCD08443613 MF:C9H13ClN2O MW:200.6653 |
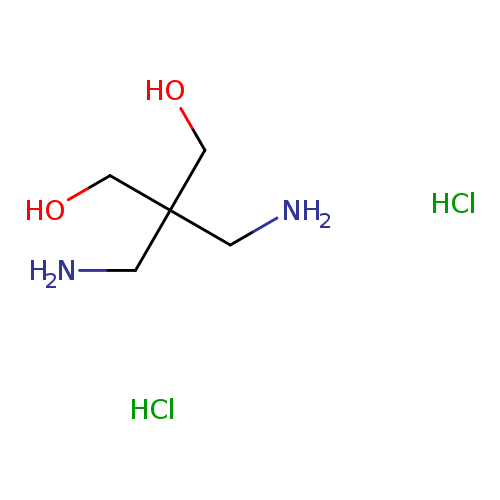
2,2-bis(aminomethyl)propane-1,3-diol dihydrochlorideCatalog No.:AA01ALGF CAS No.:1081847-32-6 MDL No.:MFCD28954381 MF:C5H16Cl2N2O2 MW:207.0987 |
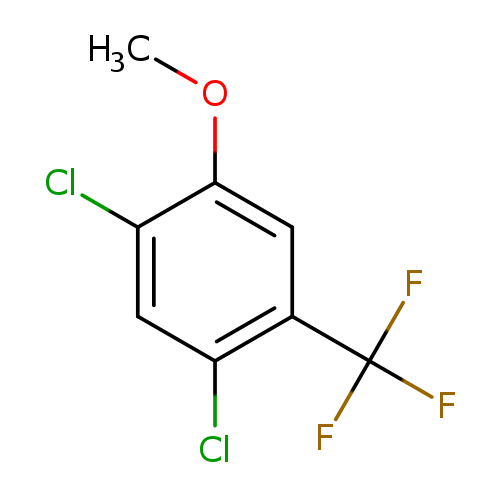
1,5-Dichloro-2-methoxy-4-(trifluoromethyl)benzeneCatalog No.:AA00MIQI CAS No.:1081849-77-5 MDL No.:MFCD28291786 MF:C8H5Cl2F3O MW:245.0259 |
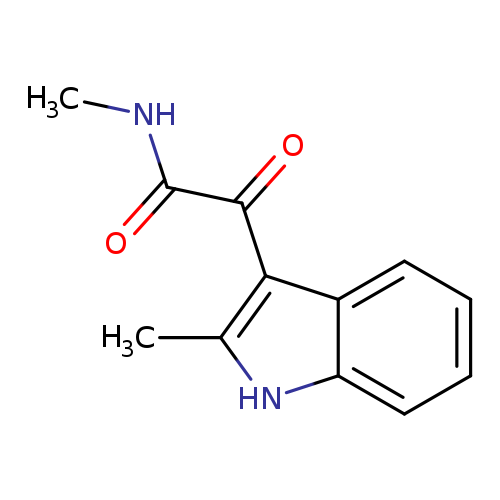
N-methyl-2-(2-methyl-1H-indol-3-yl)-2-oxoacetamideCatalog No.:AA00IXRK CAS No.:1082-42-4 MDL No.:MFCD02186718 MF:C12H12N2O2 MW:216.2359 |
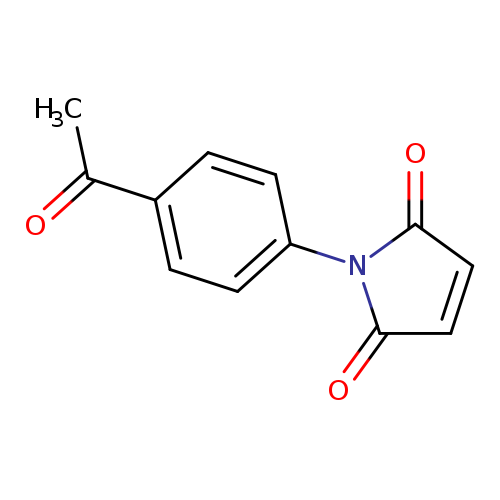
1-(4-Acetylphenyl)-1H-pyrrole-2,5-dioneCatalog No.:AA003S8S CAS No.:1082-85-5 MDL No.:MFCD00014537 MF:C12H9NO3 MW:215.2048 |
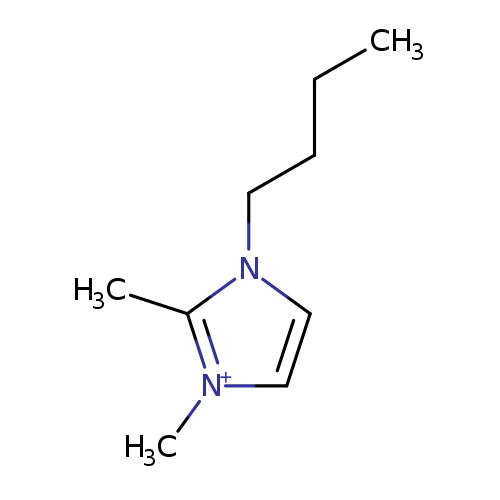
1-BUTYL-2,3-DIMETHYLIMIDAZOLIUM DIETHYLENEGLYCOLMONOMETHYLETHERSULFATE [BDIMIM] [MDEGSO4]Catalog No.:AA008WIZ CAS No.:108203-89-0 MDL No.:MFCD03788911 MF:C9H17N2+ MW:153.2447 |
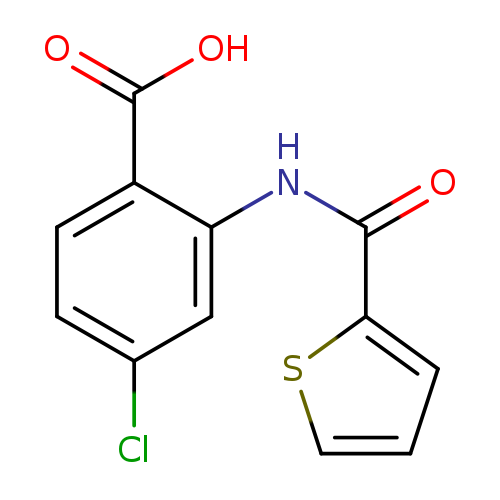
4-Chloro-2-[(thiophene-2-carbonyl)-amino]-benzoic acidCatalog No.:AA019HVM CAS No.:108204-33-7 MDL No.:MFCD01137075 MF:C12H8ClNO3S MW:281.7148 |
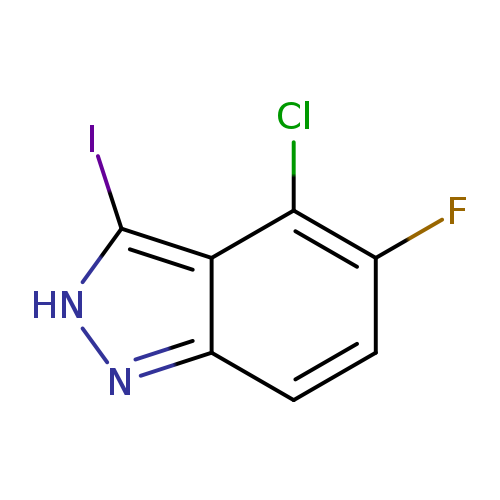
4-Chloro-5-fluoro-3-iodo-1H-indazoleCatalog No.:AA00HB0H CAS No.:1082040-26-3 MDL No.:MFCD11007990 MF:C7H3ClFIN2 MW:296.4680 |
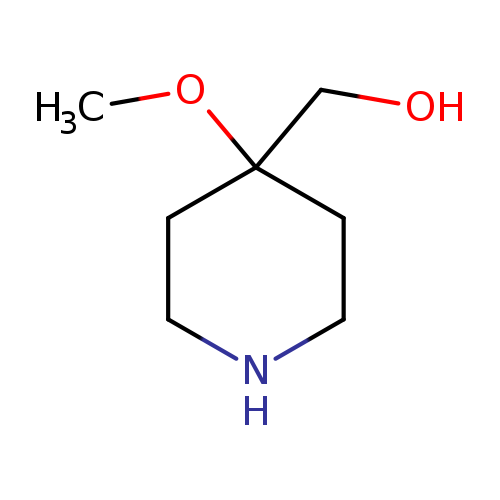
4-Methoxy-4-piperidinemethanolCatalog No.:AA008XNU CAS No.:1082040-31-0 MDL No.:MFCD11845426 MF:C7H15NO2 MW:145.1995 |
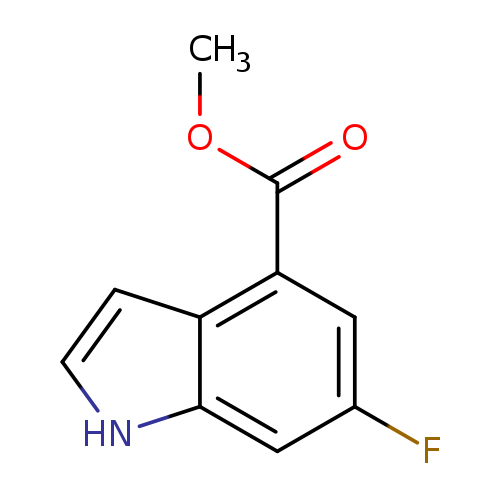
Methyl 6-fluoro-1h-indole-4-carboxylateCatalog No.:AA008SGO CAS No.:1082040-43-4 MDL No.:MFCD11845444 MF:C10H8FNO2 MW:193.1744 |
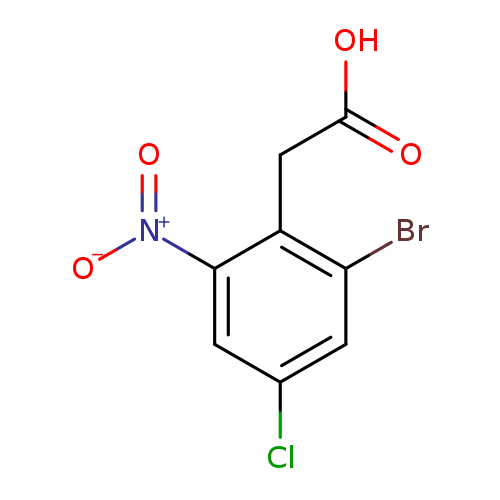
(2-Bromo-4-chloro-6-nitrophenyl)acetic acidCatalog No.:AA00HB0K CAS No.:1082040-47-8 MDL No.:MFCD11845449 MF:C8H5BrClNO4 MW:294.4866 |
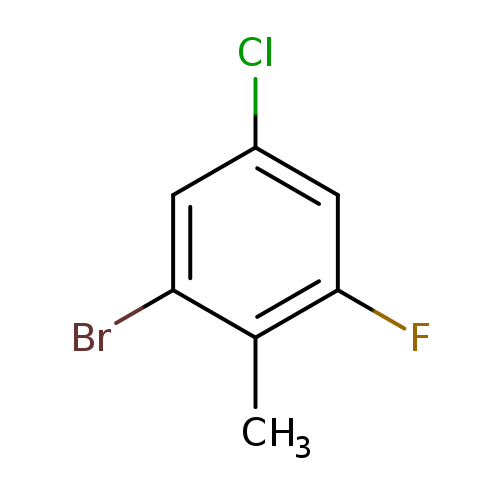
2-Bromo-4-chloro-6-fluorotolueneCatalog No.:AA01EQMM CAS No.:1082040-50-3 MDL No.:MFCD11845453 MF:C7H5BrClF MW:223.4700 |
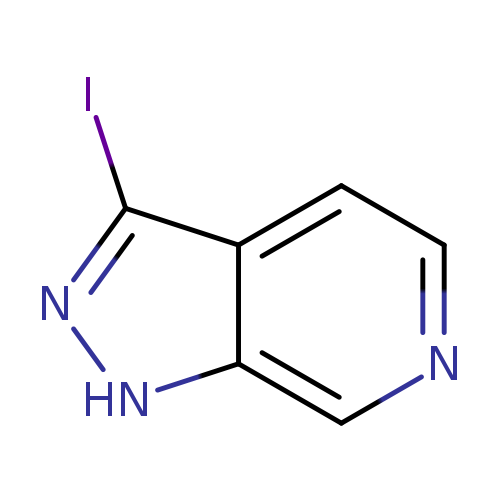
3-Iodo-1H-pyrazolo[3,4-c]pyridineCatalog No.:AA008XMV CAS No.:1082040-63-8 MDL No.:MFCD11845480 MF:C6H4IN3 MW:245.0205 |
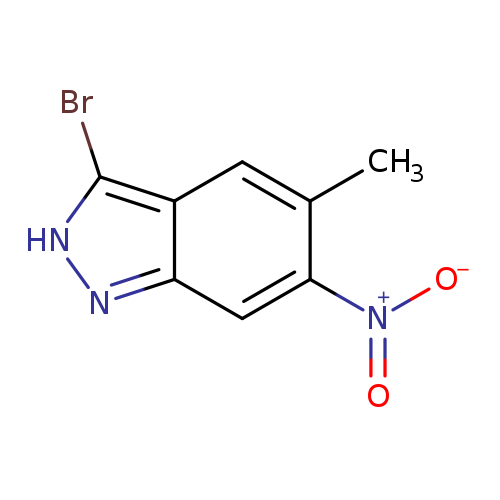
3-Bromo-5-methyl-6-nitro-1H-indazoleCatalog No.:AA009LVJ CAS No.:1082040-69-4 MDL No.:MFCD11845491 MF:C8H6BrN3O2 MW:256.0561 |
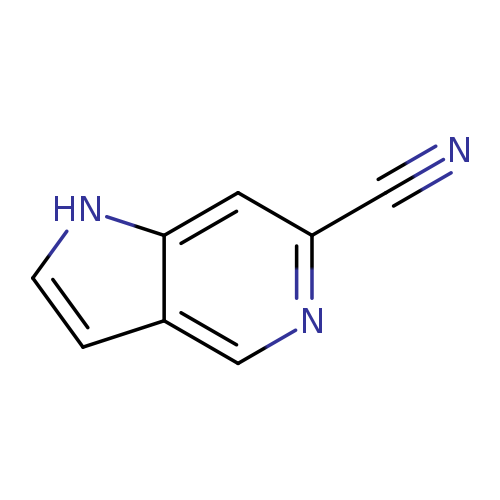
1H-Pyrrolo[3,2-c]pyridine-6-carbonitrileCatalog No.:AA00997V CAS No.:1082040-98-9 MDL No.:MFCD11845528 MF:C8H5N3 MW:143.1454 |
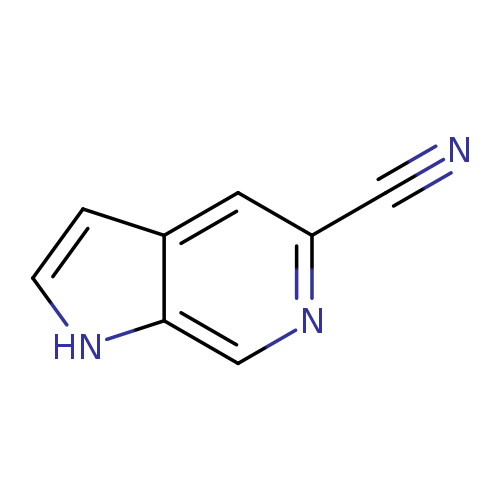
1H-Pyrrolo[2,3-c]pyridine-5-carbonitrileCatalog No.:AA007D7S CAS No.:1082041-09-5 MDL No.:MFCD11845540 MF:C8H5N3 MW:143.1454 |
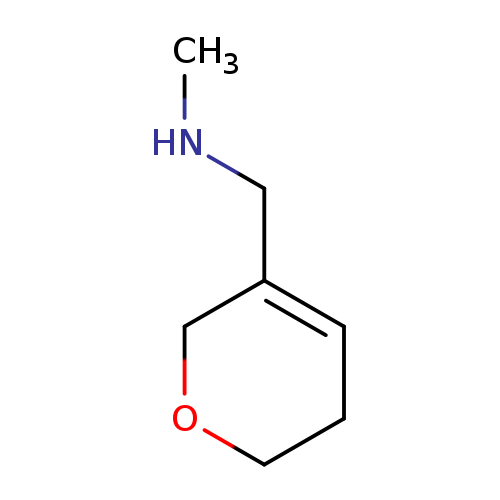
[(5,6-dihydro-2H-pyran-3-yl)methyl](methyl)amineCatalog No.:AA01B56E CAS No.:1082041-25-5 MDL No.:MFCD11007819 MF:C7H13NO MW:127.1842 |
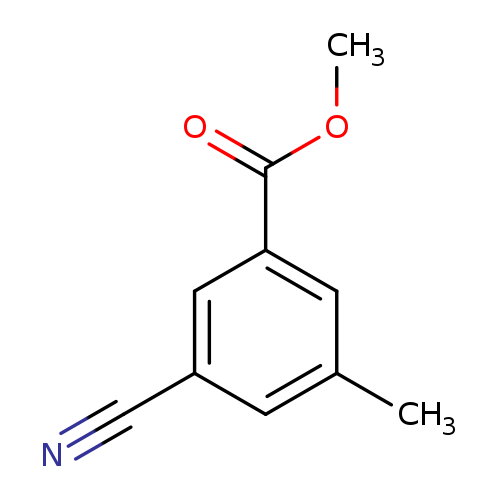
Methyl 3-cyano-5-methylbenzoateCatalog No.:AA0093XO CAS No.:1082041-26-6 MDL No.:MFCD11007826 MF:C10H9NO2 MW:175.1840 |
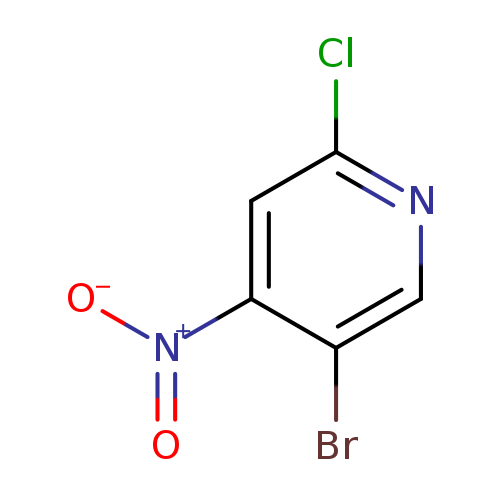
2-Chloro-4-nitro-5-bromopyridineCatalog No.:AA009LU2 CAS No.:1082041-27-7 MDL No.:MFCD11007830 MF:C5H2BrClN2O2 MW:237.4386 |
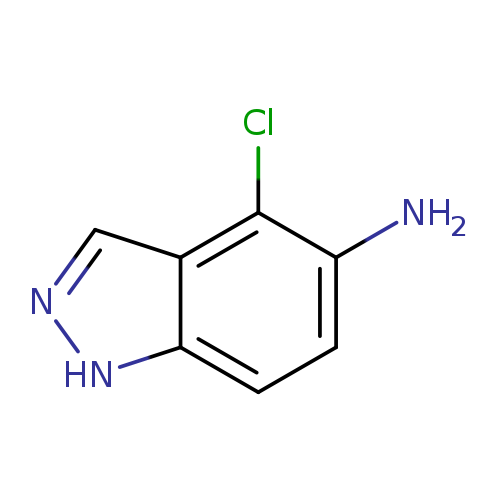
4-Chloro-1h-indazol-5-amineCatalog No.:AA008Z0V CAS No.:1082041-33-5 MDL No.:MFCD11007840 MF:C7H6ClN3 MW:167.5956 |
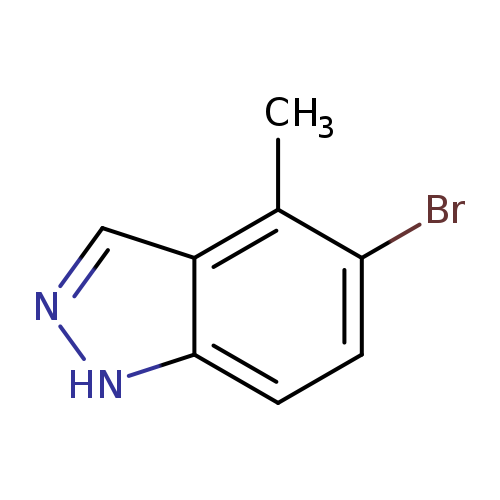
5-Bromo-4-methyl-1h-indazoleCatalog No.:AA003MGP CAS No.:1082041-34-6 MDL No.:MFCD00192129 MF:C8H7BrN2 MW:211.0586 |
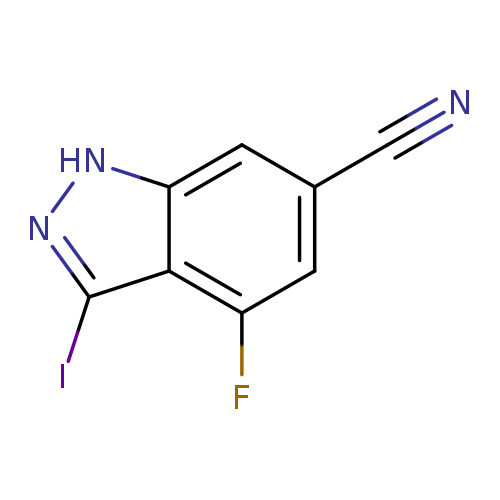
6-Cyano-4-fluoro-3-iodo (1H)indazoleCatalog No.:AA00996A CAS No.:1082041-46-0 MDL No.:MFCD11007863 MF:C8H3FIN3 MW:287.0324 |
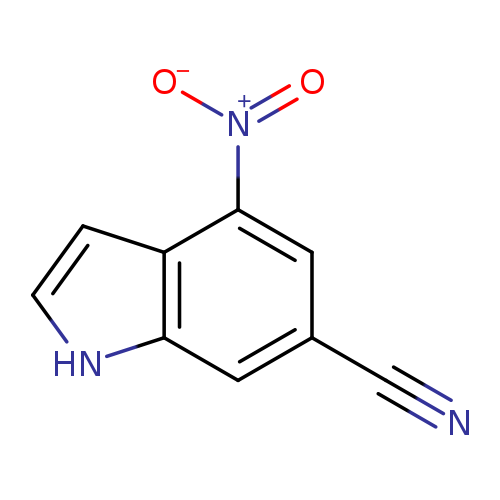
4-Nitro-1H-indole-6-carbonitrileCatalog No.:AA00996B CAS No.:1082041-51-7 MDL No.:MFCD11007868 MF:C9H5N3O2 MW:187.1549 |
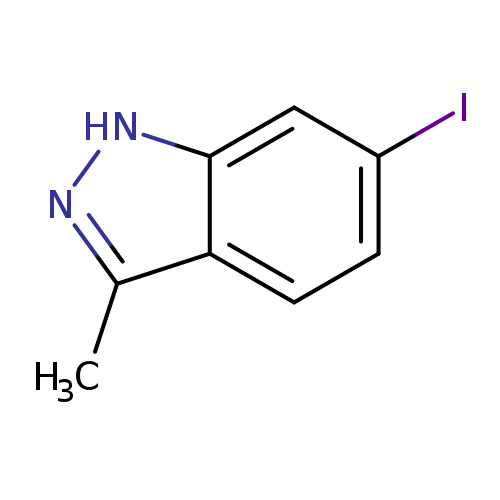
6-Iodo-3-methyl-1h-indazoleCatalog No.:AA00996C CAS No.:1082041-53-9 MDL No.:MFCD11007870 MF:C8H7IN2 MW:258.0590 |
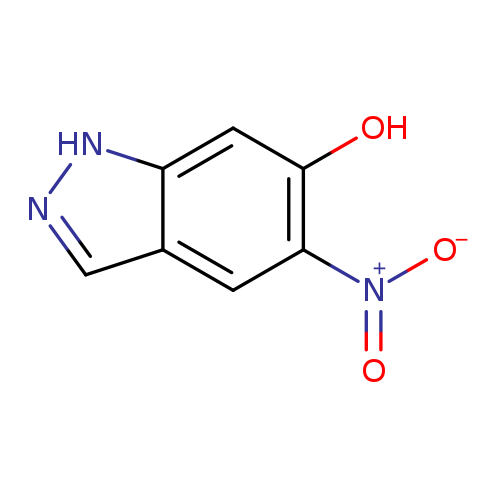
5-Nitro-1h-indazol-6-olCatalog No.:AA0095XU CAS No.:1082041-56-2 MDL No.:MFCD17676129 MF:C7H5N3O3 MW:179.1329 |
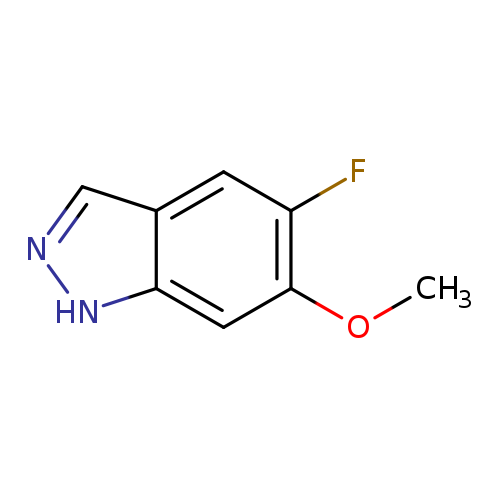
5-Fluoro-6-methoxy (1h)indazoleCatalog No.:AA00HB0L CAS No.:1082041-57-3 MDL No.:MFCD06796768 MF:C8H7FN2O MW:166.1524 |
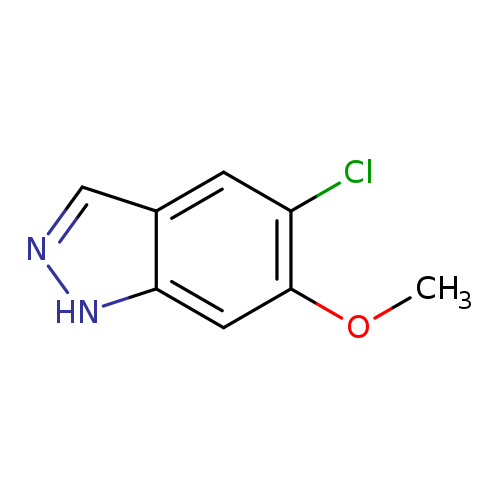
5-Chloro-6-methoxy-1h-indazoleCatalog No.:AA00996D CAS No.:1082041-58-4 MDL No.:MFCD11007885 MF:C8H7ClN2O MW:182.6070 |
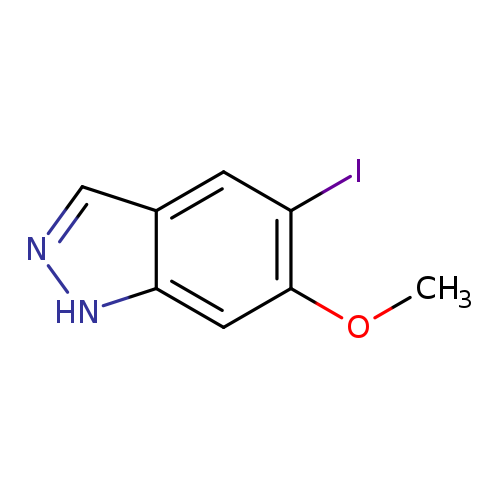
5-Iodo-6-methoxy-1h-indazoleCatalog No.:AA008XNO CAS No.:1082041-59-5 MDL No.:MFCD11007886 MF:C8H7IN2O MW:274.0584 |
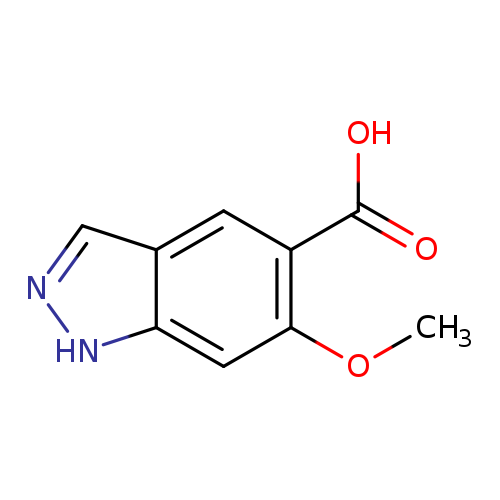
6-Methoxy-1h-indazole-5-carboxylic acidCatalog No.:AA0092I8 CAS No.:1082041-60-8 MDL No.:MFCD11007888 MF:C9H8N2O3 MW:192.1714 |
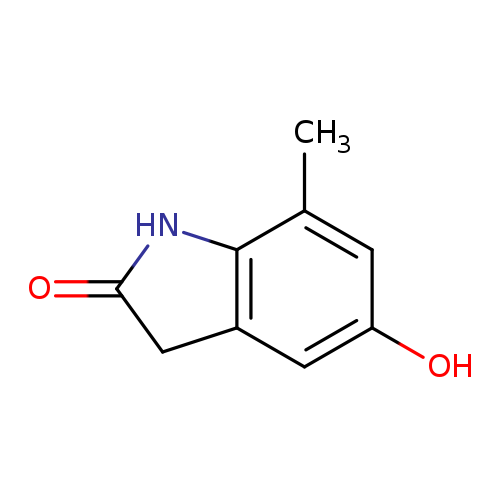
7-Methyl-5-hydroxy-2-oxindoleCatalog No.:AA00HB0M CAS No.:1082041-62-0 MDL No.:MFCD11007891 MF:C9H9NO2 MW:163.1733 |
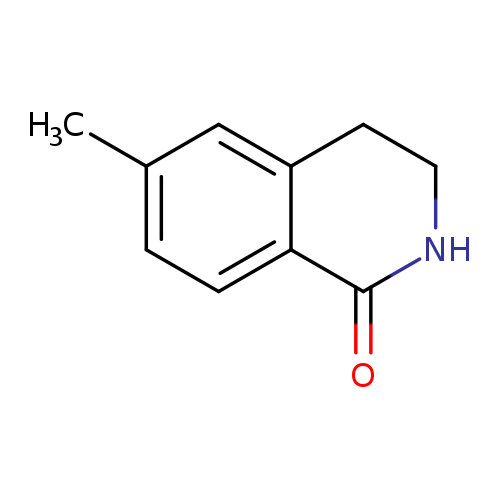
6-Methyl-3,4-dihydroisoquinolin-1(2h)-oneCatalog No.:AA00HB0O CAS No.:1082041-78-8 MDL No.:MFCD09838960 MF:C10H11NO MW:161.2004 |
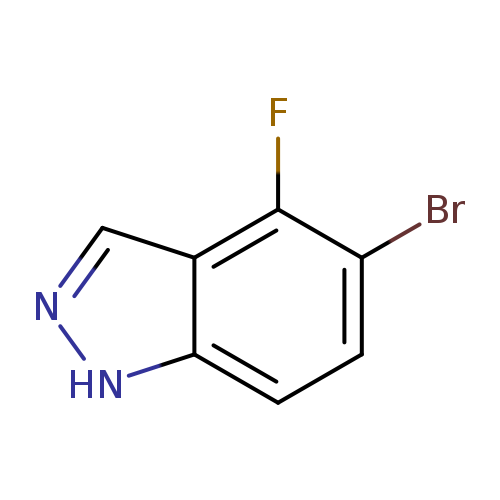
5-Bromo-4-fluoro-1H-indazoleCatalog No.:AA0035VE CAS No.:1082041-85-7 MDL No.:MFCD11007927 MF:C7H4BrFN2 MW:215.0225 |
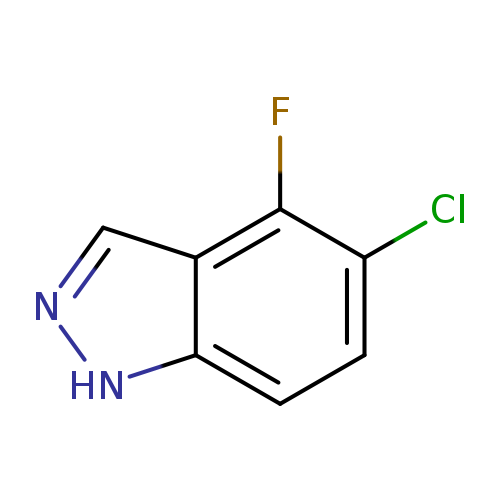
5-Chloro-4-fluoro-1H-indazoleCatalog No.:AA00IMMQ CAS No.:1082041-86-8 MDL No.:MFCD11007928 MF:C7H4ClFN2 MW:170.5715 |
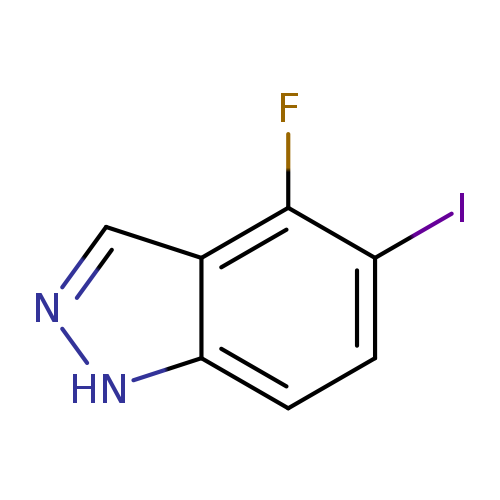
4-Fluoro-5-iodo-1h-indazoleCatalog No.:AA008TL1 CAS No.:1082041-87-9 MDL No.:MFCD11007929 MF:C7H4FIN2 MW:262.0229 |
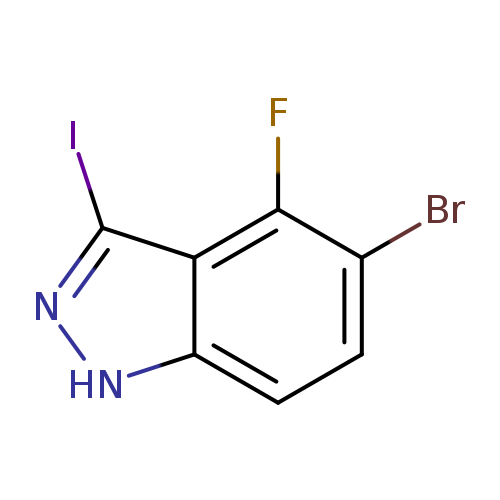
5-Bromo-4-fluoro-3-iodo-1h-indazoleCatalog No.:AA0083T6 CAS No.:1082041-88-0 MDL No.:MFCD11007931 MF:C7H3BrFIN2 MW:340.9190 |
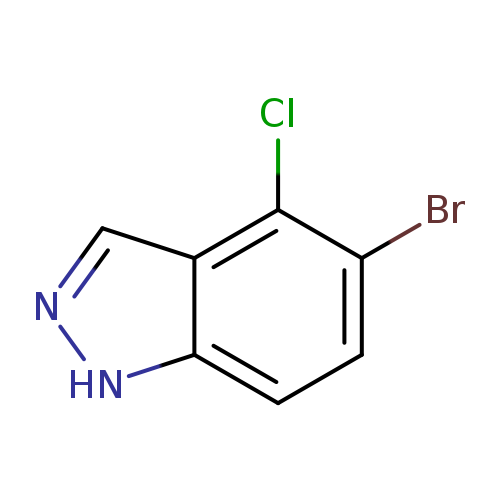
5-bromo-4-chloro-1H-indazoleCatalog No.:AA0037X9 CAS No.:1082041-90-4 MDL No.:MFCD11007933 MF:C7H4BrClN2 MW:231.4771 |
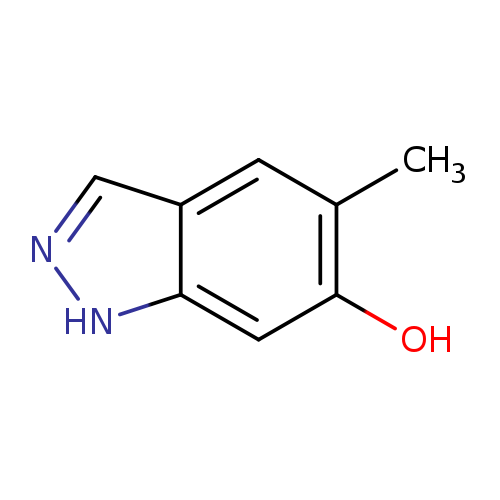
5-Methyl-1h-indazol-6-olCatalog No.:AA00996H CAS No.:1082042-15-6 MDL No.:MFCD11007971 MF:C8H8N2O MW:148.1619 |
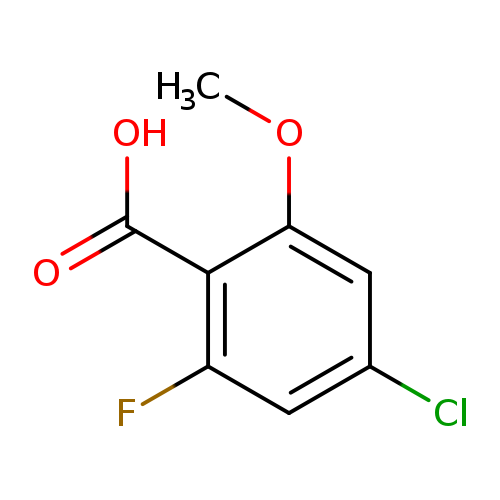
4-Chloro-2-fluoro-6-methoxybenzoic acidCatalog No.:AA00HB0P CAS No.:1082042-25-8 MDL No.:MFCD11845551 MF:C8H6ClFO3 MW:204.5828 |
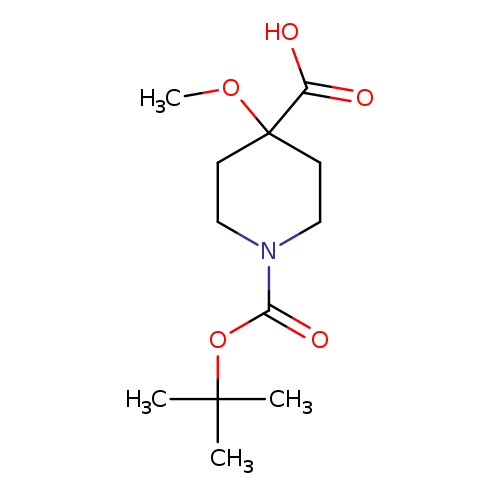
1-[(tert-Butoxy)carbonyl]-4-methoxypiperidine-4-carboxylic acidCatalog No.:AA00IKXE CAS No.:1082042-29-2 MDL No.:MFCD31381489 MF:C12H21NO5 MW:259.2988 |
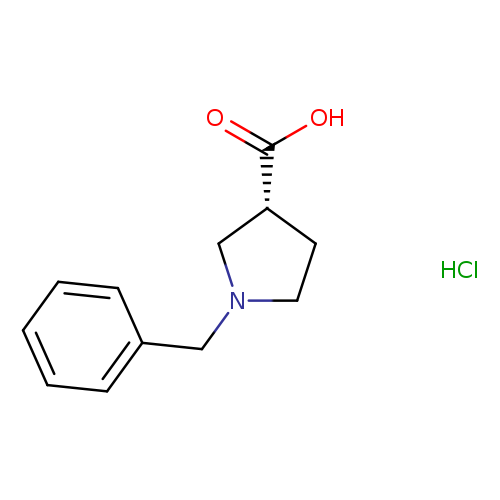
(R)-1-Benzyl-pyrrolidine-3-carboxylic acid, HClCatalog No.:AA008S4T CAS No.:1082055-65-9 MDL No.:MFCD11007680 MF:C12H16ClNO2 MW:241.7139 |
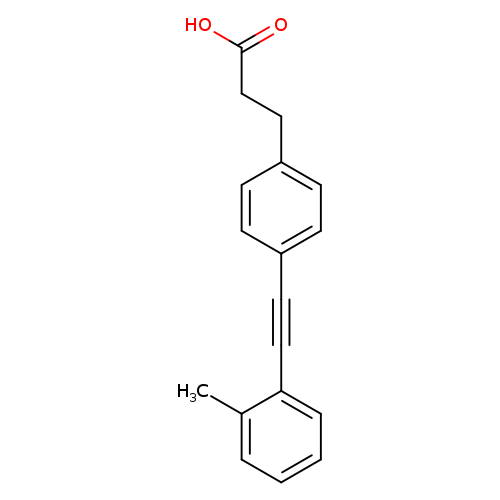
3-(4-(o-Tolylethynyl)phenyl)propanoic acidCatalog No.:AA007D68 CAS No.:1082058-99-8 MDL No.:MFCD12912449 MF:C18H16O2 MW:264.3184 |
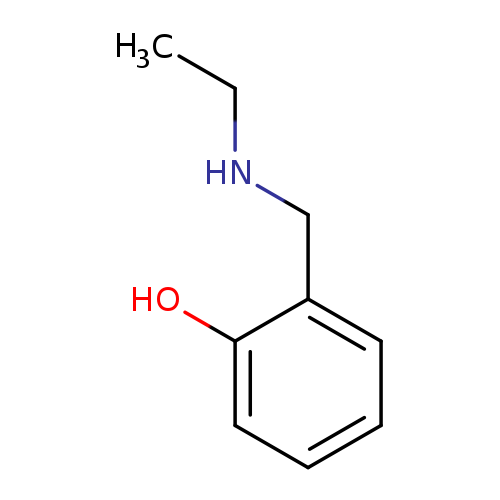
2-[(Ethylamino)methyl]phenolCatalog No.:AA00941L CAS No.:108206-05-9 MDL No.:MFCD09929100 MF:C9H13NO MW:151.2056 |
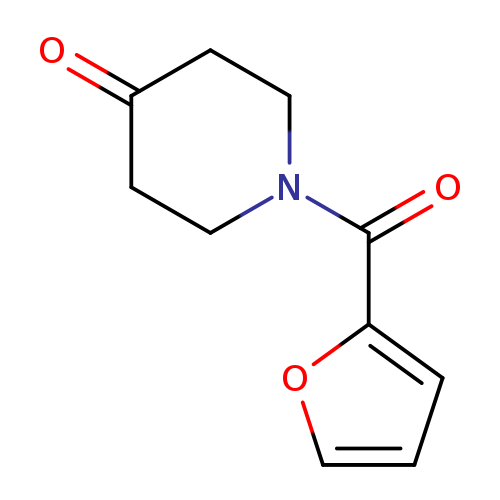
1-(Furan-2-carbonyl)piperidin-4-oneCatalog No.:AA007V2S CAS No.:108206-24-2 MDL No.:MFCD09937454 MF:C10H11NO3 MW:193.1992 |
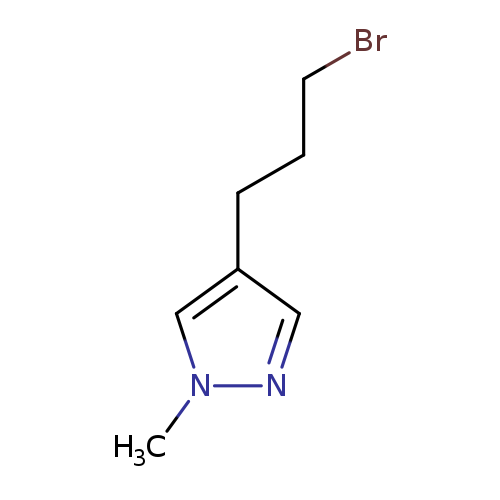
4-(3-bromopropyl)-1-methyl-1H-pyrazoleCatalog No.:AA019V5B CAS No.:1082065-79-9 MDL No.:MFCD12197058 MF:C7H11BrN2 MW:203.0796 |
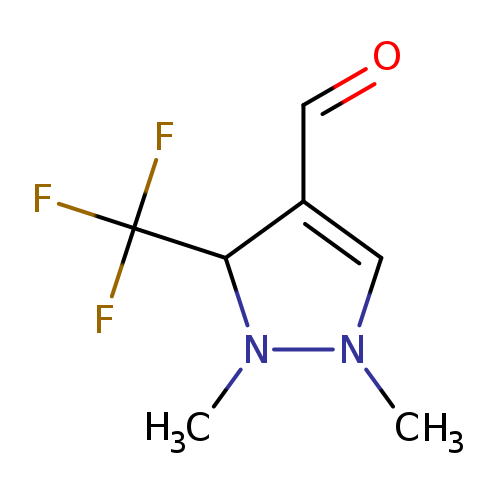
1,5-Dimethyl-3-(trifluoromethyl)-1H-pyrazole-4-carbaldehydeCatalog No.:AA007D67 CAS No.:1082065-80-2 MDL No.:MFCD09991765 MF:C7H9F3N2O MW:194.1544 |
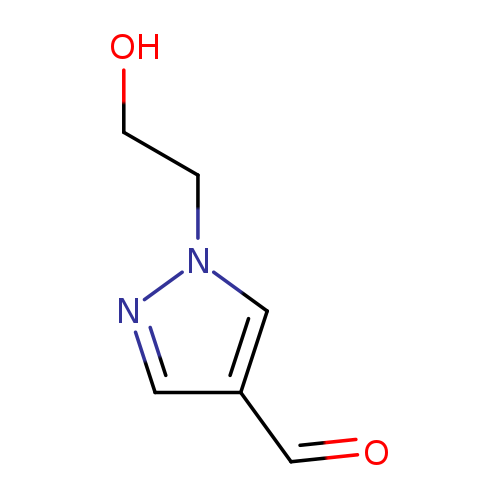
1-(2-Hydroxyethyl)-1H-pyrazole-4-carbaldehydeCatalog No.:AA0091ZJ CAS No.:1082065-98-2 MDL No.:MFCD09941789 MF:C6H8N2O2 MW:140.1399 |
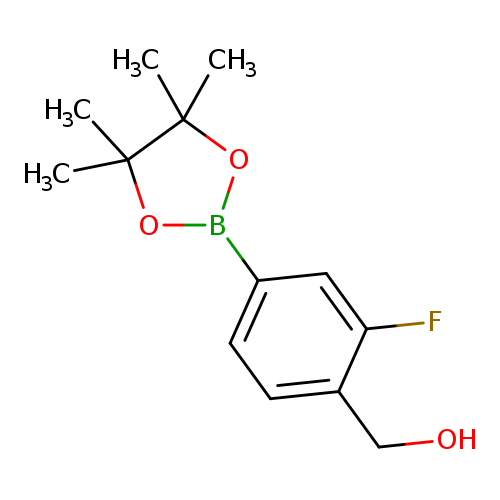
[2-fluoro-4-(tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]methanolCatalog No.:AA00HB0Q CAS No.:1082066-29-2 MDL No.:MFCD18732617 MF:C13H18BFO3 MW:252.0896 |
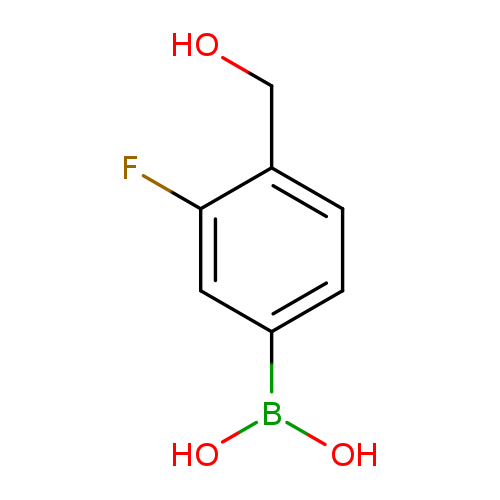
3-Fluoro-4-hydroxymethylbenzeneboronic acidCatalog No.:AA007V2R CAS No.:1082066-52-1 MDL No.:MFCD13181609 MF:C7H8BFO3 MW:169.9460 |
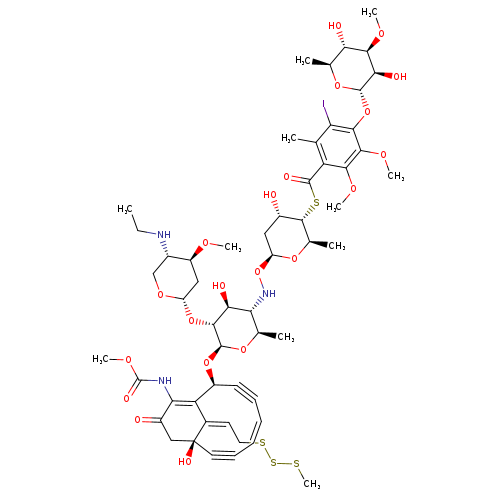
calicheamicin gamma(1)ICatalog No.:AA008TIQ CAS No.:108212-75-5 MDL No.:MFCD30478834 MF:C55H74IN3O21S4 MW:1368.3480 |
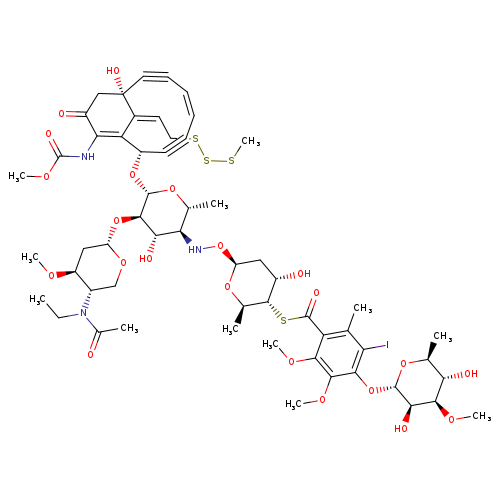
N-Acetyl-CalicheamicinCatalog No.:AA008TMG CAS No.:108212-76-6 MDL No.:MFCD29924715 MF:C57H76IN3O22S4 MW:1410.3847 |
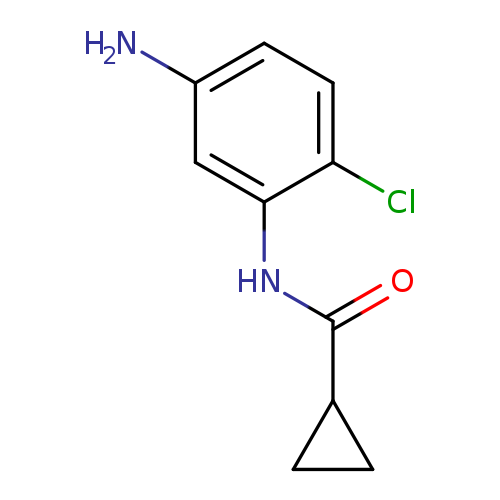
N-(5-Amino-2-chlorophenyl)cyclopropanecarboxamideCatalog No.:AA01BIMB CAS No.:1082128-59-3 MDL No.:MFCD11568374 MF:C10H11ClN2O MW:210.6601 |
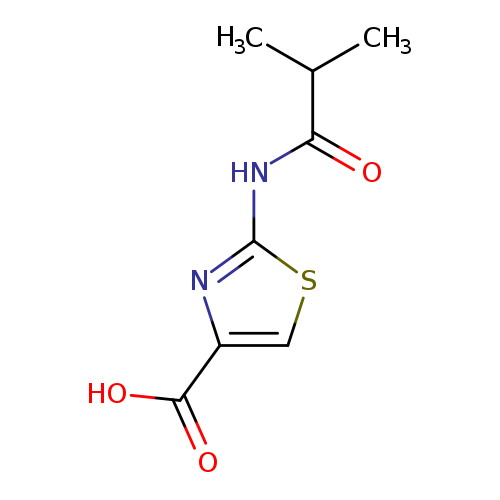
2-Isobutyrylamino-thiazole-4-carboxylic acidCatalog No.:AA009263 CAS No.:1082130-43-5 MDL No.:MFCD11564857 MF:C8H10N2O3S MW:214.2416 |
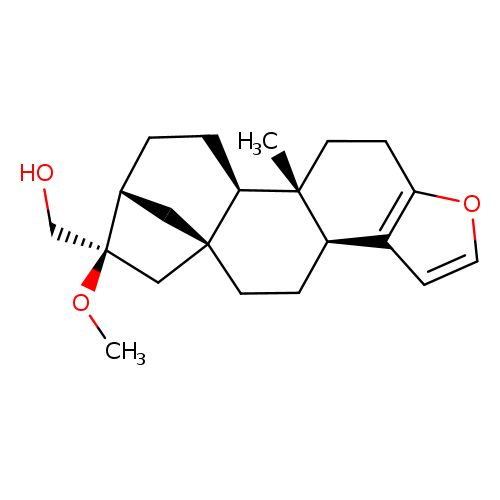
16-O-METHYL-CAFESTOLCatalog No.:AA0094BD CAS No.:108214-28-4 MDL No.:MFCD16660342 MF:C21H30O3 MW:330.4611 |
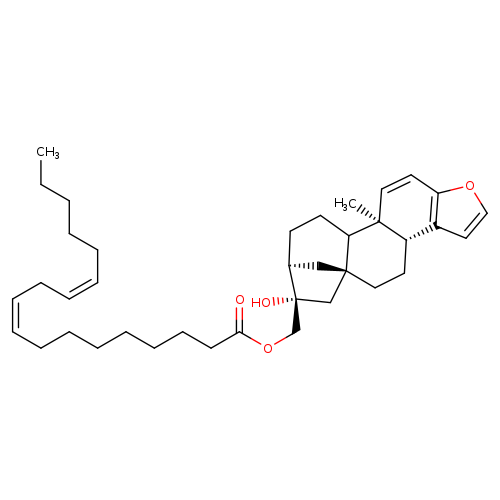
KAHWEOL LINOLEATECatalog No.:AA008RAQ CAS No.:108214-29-5 MDL No.: MF:C38H56O4 MW:576.8488 |
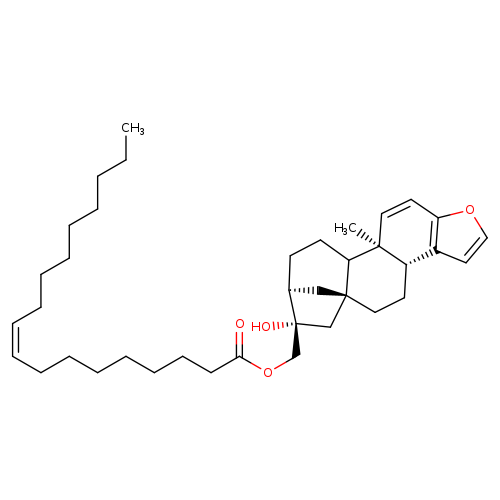
KAHWEOL OLEATECatalog No.:AA008RAR CAS No.:108214-30-8 MDL No.:MFCD02093277 MF:C38H58O4 MW:578.8647 |
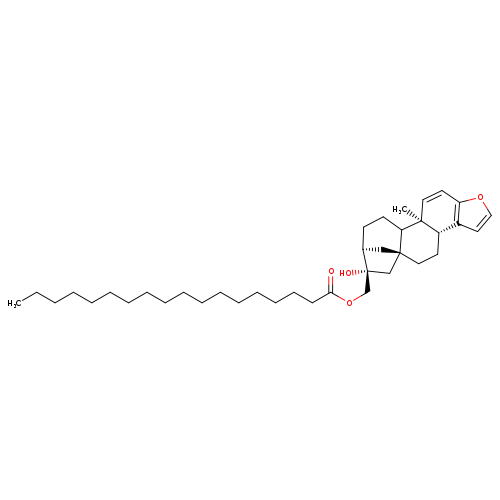
KAHWEOL STEARATECatalog No.:AA008RB6 CAS No.:108214-31-9 MDL No.:MFCD02093278 MF:C38H60O4 MW:580.8806 |
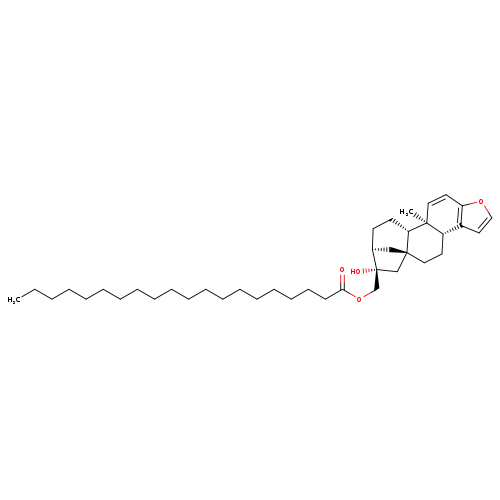
KAHWEOL EICOSANATECatalog No.:AA008RBA CAS No.:108214-32-0 MDL No.:MFCD02093275 MF:C40H64O4 MW:608.9338 |
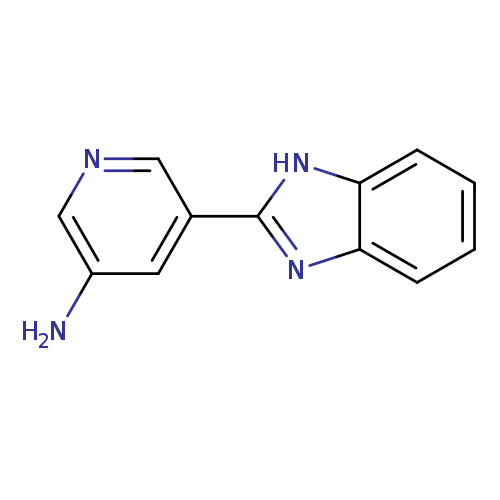
5-(1H-1,3-Benzodiazol-2-yl)pyridin-3-amineCatalog No.:AA01BR0R CAS No.:1082159-77-0 MDL No.:MFCD11107321 MF:C12H10N4 MW:210.2346 |
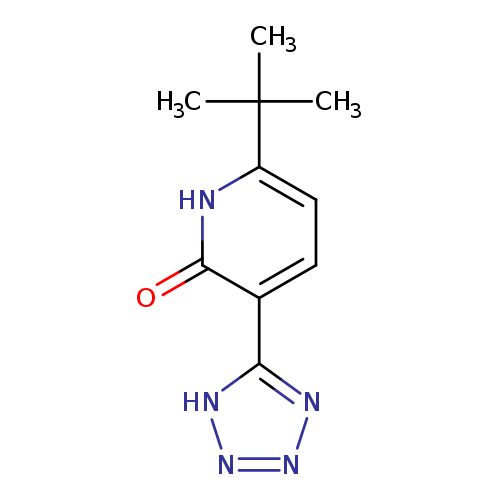
6-tert-butyl-3-(1H-1,2,3,4-tetrazol-5-yl)-1,2-dihydropyridin-2-oneCatalog No.:AA01B2IS CAS No.:1082159-82-7 MDL No.:MFCD11107325 MF:C10H13N5O MW:219.2431 |
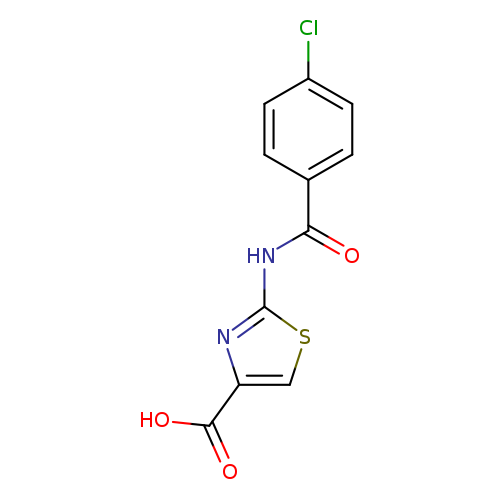
2-[(4-Chlorobenzoyl)amino]-1,3-thiazole-4-carboxylic acidCatalog No.:AA01ARGB CAS No.:1082168-15-7 MDL No.:MFCD11564904 MF:C11H7ClN2O3S MW:282.7029 |
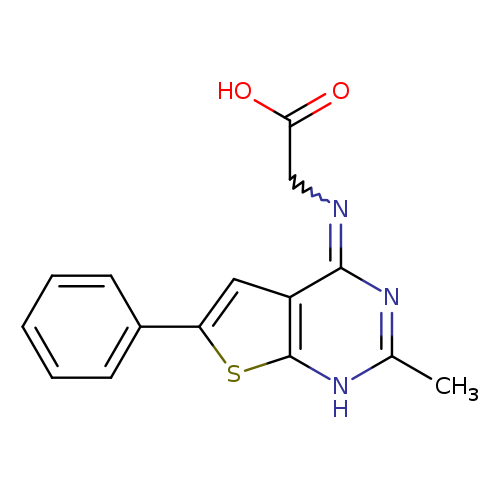
2-({2-methyl-6-phenyl-1H,4H-thieno[2,3-d]pyrimidin-4-ylidene}amino)acetic acidCatalog No.:AA01A180 CAS No.:1082176-31-5 MDL No.:MFCD22392231 MF:C15H13N3O2S MW:299.3476 |
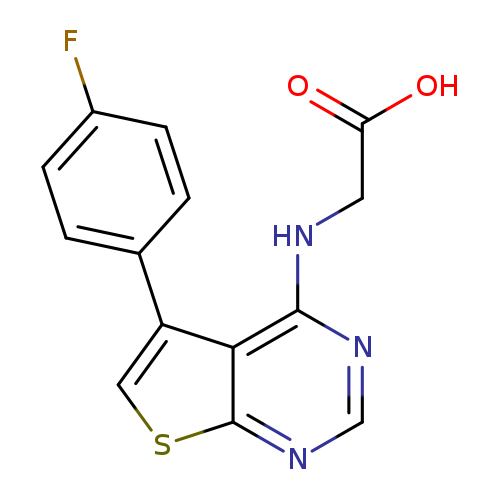
2-{[5-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl]amino}acetic acidCatalog No.:AA01AIFM CAS No.:1082187-03-8 MDL No.:MFCD11561679 MF:C14H10FN3O2S MW:303.3115 |
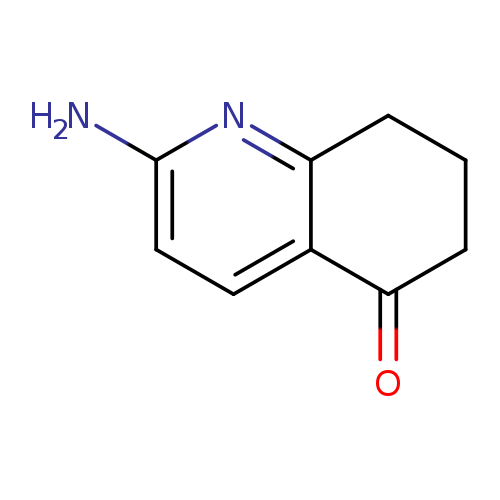
2-Amino-7,8-dihydroquinolin-5(6h)-oneCatalog No.:AA01C1H3 CAS No.:1082192-52-6 MDL No.:MFCD11865246 MF:C9H10N2O MW:162.1885 |
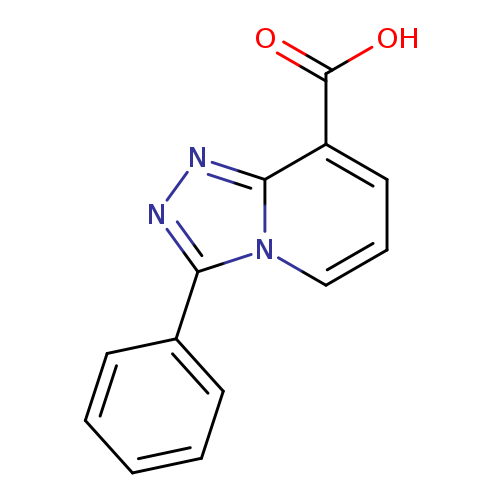
3-Phenyl[1,2,4]triazolo[4,3-a]pyridine-8-carboxylic acidCatalog No.:AA008V2H CAS No.:1082193-69-8 MDL No.:MFCD07376951 MF:C13H9N3O2 MW:239.2295 |
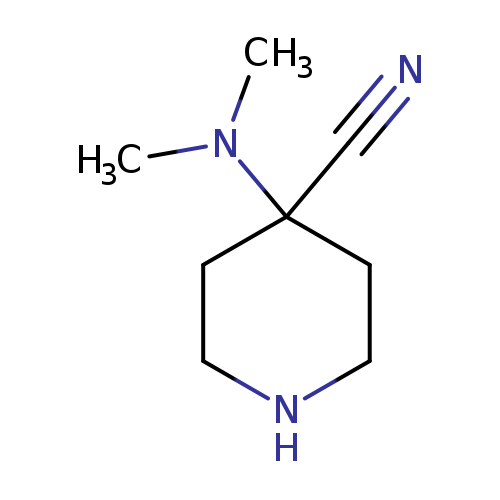
4-(dimethylamino)piperidine-4-carbonitrileCatalog No.:AA01AF8U CAS No.:1082240-13-8 MDL No.:MFCD11585208 MF:C8H15N3 MW:153.2248 |
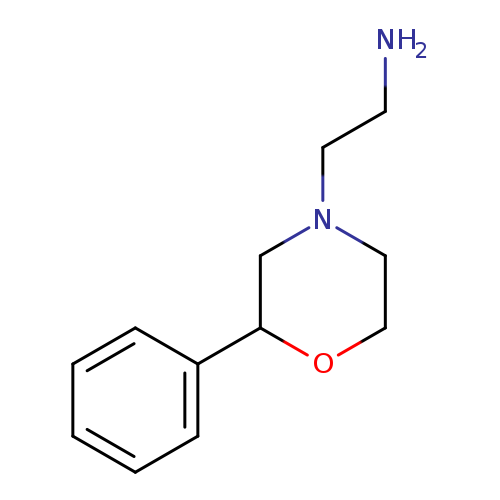
[2-(2-PHENYLMORPHOLIN-4-YL)ETHYL]AMINECatalog No.:AA01ARGY CAS No.:1082248-83-6 MDL No.:MFCD11592818 MF:C12H18N2O MW:206.2841 |
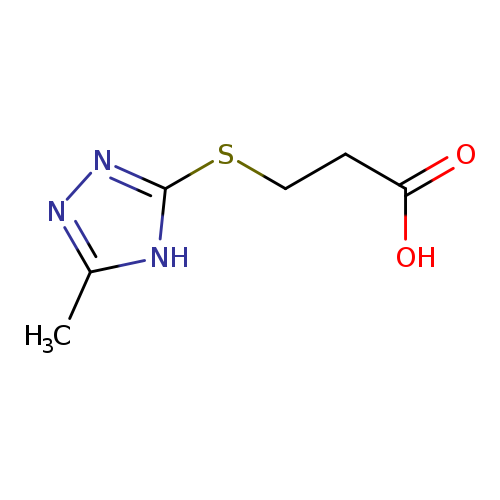
3-[(5-methyl-4H-1,2,4-triazol-3-yl)sulfanyl]propanoic acidCatalog No.:AA019SRO CAS No.:1082259-53-7 MDL No.:MFCD11576372 MF:C6H9N3O2S MW:187.2196 |
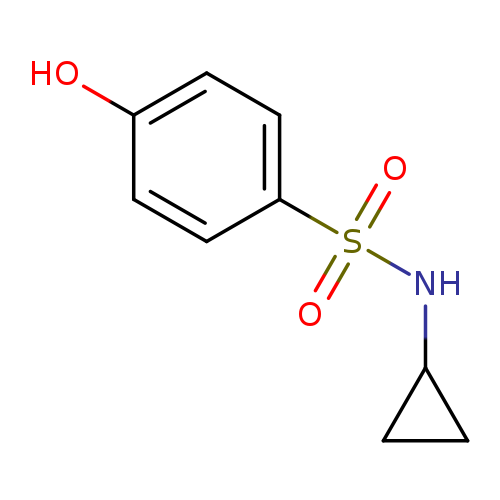
N-Cyclopropyl-4-hydroxybenzene-1-sulfonamideCatalog No.:AA00HB12 CAS No.:1082262-36-9 MDL No.:MFCD11164635 MF:C9H11NO3S MW:213.2535 |
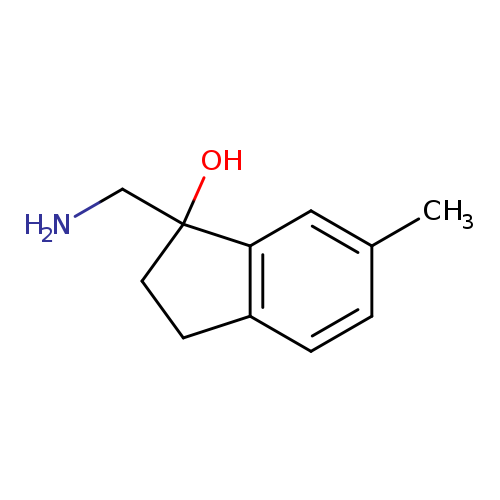
1-(Aminomethyl)-6-methyl-2,3-dihydro-1h-inden-1-olCatalog No.:AA01FMG1 CAS No.:1082268-36-7 MDL No.:MFCD11591012 MF:C11H15NO MW:177.2429 |
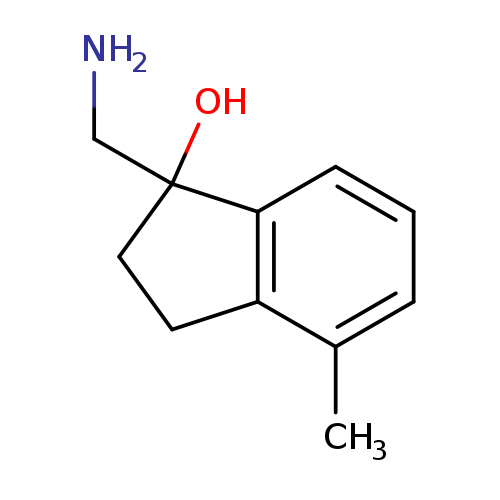
1-(Aminomethyl)-4-methyl-2,3-dihydro-1h-inden-1-olCatalog No.:AA01FMG4 CAS No.:1082268-41-4 MDL No.:MFCD11591020 MF:C11H15NO MW:177.2429 |
1-[(4-Methylpiperazin-1-yl)methyl]cyclopentan-1-olCatalog No.:AA01FP83 CAS No.:1082268-62-9 MDL No.:MFCD11591086 MF:C11H22N2O MW:198.3052 |
N,4-Dimethyl-3-nitrobenzenesulfonamideCatalog No.:AA009496 CAS No.:108227-52-7 MDL No.:MFCD01004320 MF:C8H10N2O4S MW:230.2410 |
N-Butyl-4-methyl-3-nitrobenzenesulfonamideCatalog No.:AA009498 CAS No.:108227-53-8 MDL No.:MFCD00577535 MF:C11H16N2O4S MW:272.3207 |
4-(aminomethyl)-1-propylpyrrolidin-2-oneCatalog No.:AA01AMKR CAS No.:1082278-90-7 MDL No.:MFCD11579918 MF:C8H16N2O MW:156.2254 |
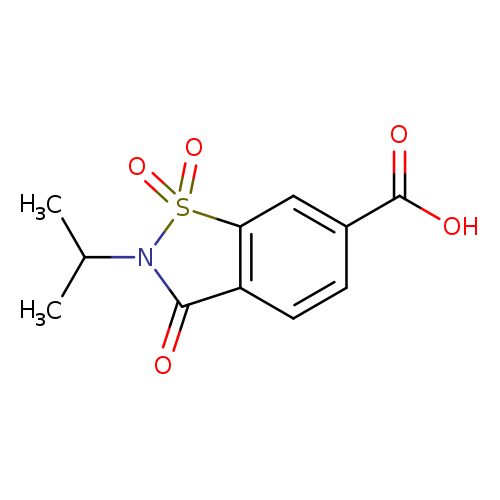
1,1,3-trioxo-2-(propan-2-yl)-2,3-dihydro-1,2-benzothiazole-6-carboxylic acidCatalog No.:AA019VT6 CAS No.:1082279-37-5 MDL No.:MFCD10689376 MF:C11H11NO5S MW:269.2737 |
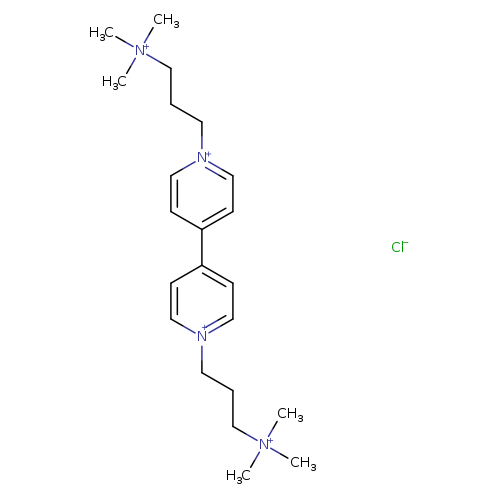
4,4'-Bipyridinium, 1,1'-bis[3-(trimethylammonio)propyl]-, tetrachlorideCatalog No.:AA00IMBW CAS No.:108228-37-1 MDL No.:MFCD31618118 MF:C22H38ClN4+++ MW:394.0169 |
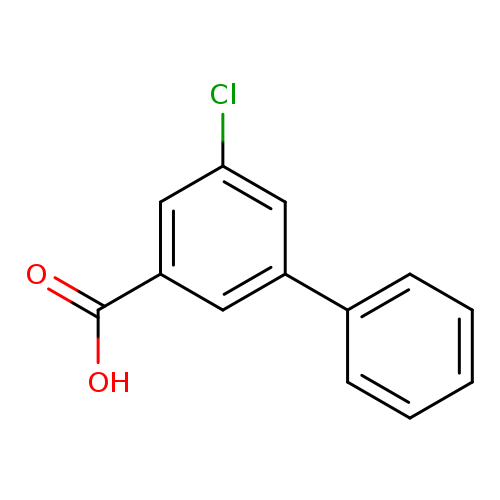
5-Chloro-3-phenylbenzoic acidCatalog No.:AA0083SZ CAS No.:1082283-43-9 MDL No.:MFCD11577111 MF:C13H9ClO2 MW:232.6624 |
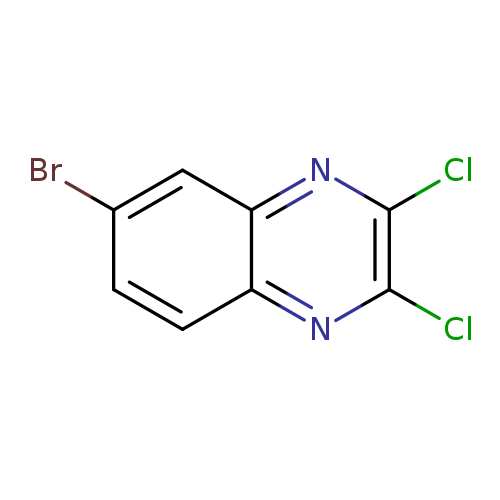
6-Bromo-2,3-dichloroquinoxalineCatalog No.:AA007V2O CAS No.:108229-82-9 MDL No.:MFCD09842518 MF:C8H3BrCl2N2 MW:277.9328 |
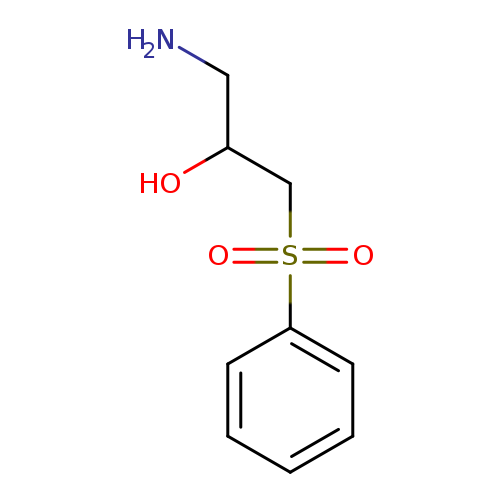
1-amino-3-(benzenesulfonyl)propan-2-olCatalog No.:AA01EMFT CAS No.:1082293-16-0 MDL No.:MFCD11585517 MF:C9H13NO3S MW:215.2694 |
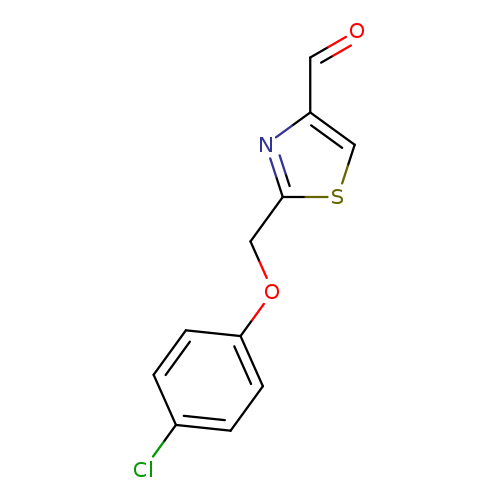
2-(4-chlorophenoxymethyl)-1,3-thiazole-4-carbaldehydeCatalog No.:AA019WGV CAS No.:1082312-23-9 MDL No.:MFCD11587925 MF:C11H8ClNO2S MW:253.7047 |
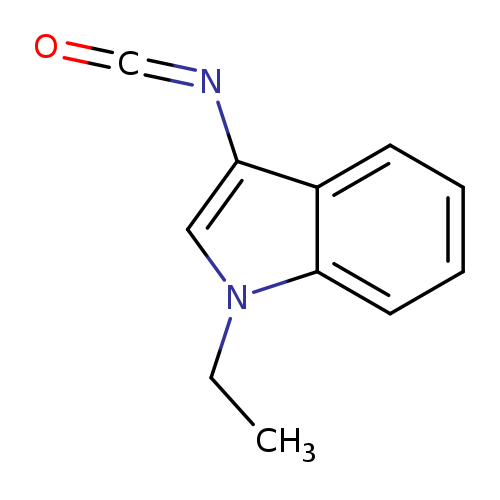
1-Ethyl-3-isocyanato-1h-indoleCatalog No.:AA01APET CAS No.:1082325-97-0 MDL No.:MFCD11588130 MF:C11H10N2O MW:186.2099 |
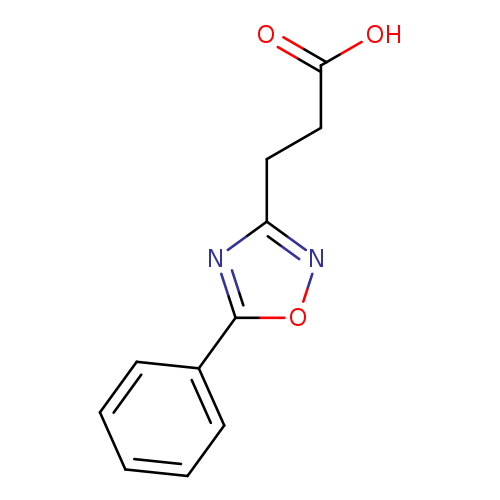
3-(5-Phenyl-1,2,4-oxadiazol-3-yl)propanoic acidCatalog No.:AA0039YB CAS No.:1082329-59-6 MDL No.:MFCD11584300 MF:C11H10N2O3 MW:218.2087 |
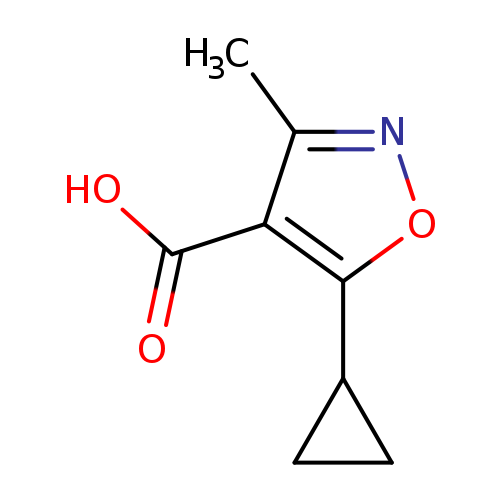
5-cyclopropyl-3-methyl-1,2-oxazole-4-carboxylic acidCatalog No.:AA01A66X CAS No.:1082329-71-2 MDL No.:MFCD11584321 MF:C8H9NO3 MW:167.1620 |
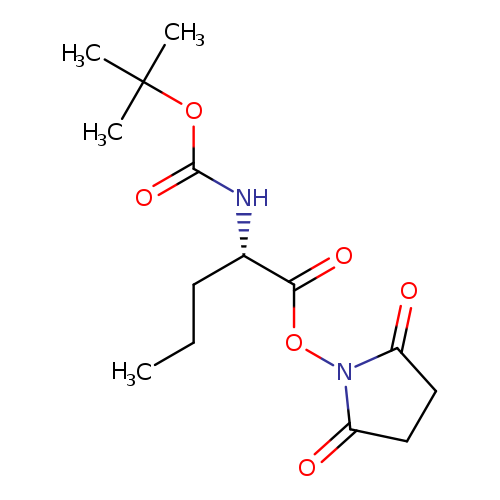
Boc-nva-osuCatalog No.:AA00HB19 CAS No.:108233-37-0 MDL No.:MFCD01862291 MF:C14H22N2O6 MW:314.3343 |
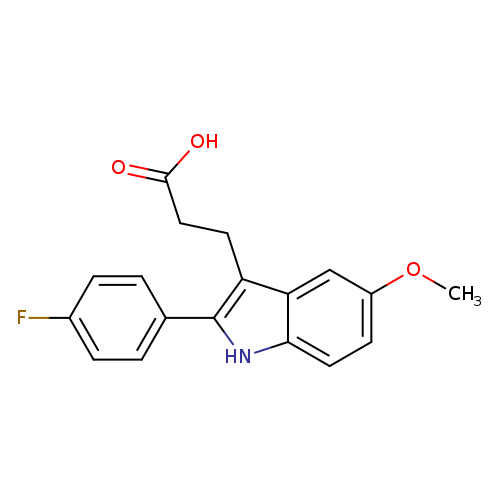
3-[2-(4-Fluorophenyl)-5-methoxy-1h-indol-3-yl]propanoic acidCatalog No.:AA01E8EN CAS No.:1082334-06-2 MDL No.:MFCD11589918 MF:C18H16FNO3 MW:313.3229 |
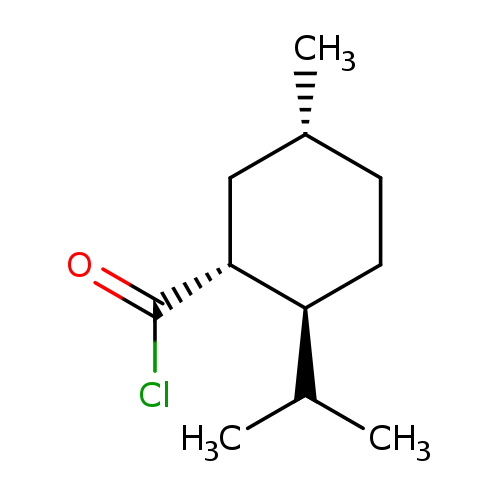
Cyclohexanecarbonyl chloride, 5-methyl-2-(1-methylethyl)-, (1R,2S,5R)-Catalog No.:AA0083SV CAS No.:108235-80-9 MDL No.:MFCD13174459 MF:C11H19ClO MW:202.7210 |
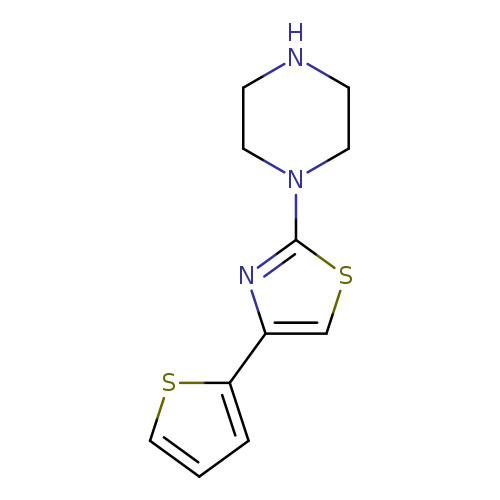
1-[4-(thiophen-2-yl)-1,3-thiazol-2-yl]piperazineCatalog No.:AA01AAUQ CAS No.:1082363-07-2 MDL No.:MFCD11592088 MF:C11H13N3S2 MW:251.3710 |
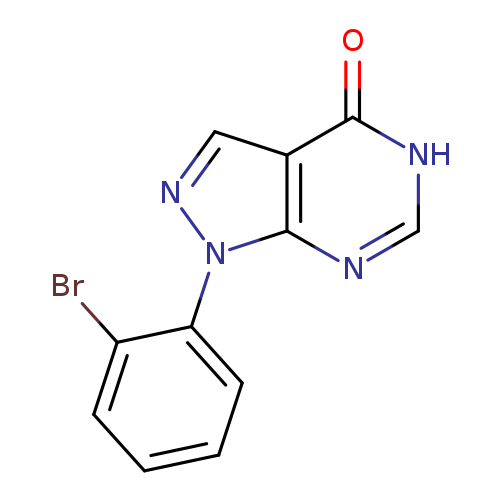
1-(2-Bromophenyl)-1H-pyrazolo[3,4-d]pyrimidin-4(5H)-oneCatalog No.:AA01FFO0 CAS No.:1082366-75-3 MDL No.:MFCD11588467 MF:C11H7BrN4O MW:291.1035 |
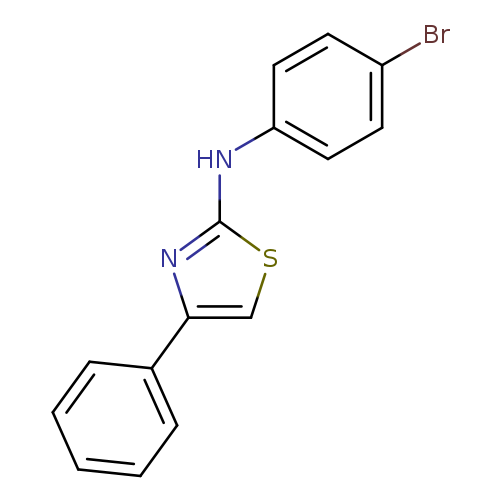
N-(4-Bromophenyl)-4-phenylthiazol-2-amineCatalog No.:AA01EOHM CAS No.:108237-91-8 MDL No.:MFCD01123615 MF:C15H11BrN2S MW:331.2302 |